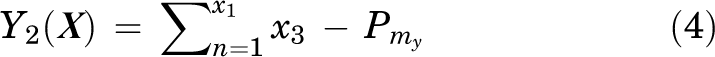Abstract
Background:
Engaging Hill-Sachs lesions (HSLs) pose a significant risk for failure of surgical repair of recurrent anterior shoulder instability. Reconstruction with fresh osteochondral allograft (OCA) has been proposed as a treatment for large HSLs.
Purpose:
To determine the optimal characteristics of talus OCA bone plugs in a computer-simulated HSL model.
Study Design:
Descriptive laboratory study; Level of evidence, 6
Methods:
Included were 132 patients with recurrent anterior instability with visible HSLs; patients who had multidirectional instability or previous shoulder surgery were excluded. Three-dimensional computed tomography models were constructed, and a custom computer optimization algorithm was generated to maximize bone plug surface area at the most superior apex (superiorization) and minimize its position relative to the most medial margin of the HSL defect (medialization). The optimal number, diameter, medialization, and superiorization of the bone plug(s) were reported. Percentages of restored glenoid track width and conversion from off- to on-track HSLs after bone plug optimization were calculated.
Results:
A total of 86 patients were included in the final analysis. Off-track lesions made up 19.7% of HSLs and, of these, the mean bone plug size was 9.9 ± 1.4 mm, with 2.2 mm ± 1.7 mm of medialization and 3.3 mm ± 2.9 mm of superiorization. The optimization identified 21% of HSLs requiring 1 bone plug, 65% requiring 2 plugs, and 14% requiring 3 plugs, with a mean overall coverage of 60%. The mean width of the restored HSLs was 68%, and all off-track HSLs (n = 17) were restored to on-track. A Jenks natural-breaks analysis calculated 3 ideal bone plug diameters of 8 mm (small), 10.4 mm (medium), and 12 mm (large) in order to convert this group of HSLs to on-track.
Conclusion:
Using a custom computer algorithm, we have demonstrated the optimal talus OCA bone plug diameters for reconstructing HSLs to successfully restore the HSL track and, on average, 60% of the HSL surface area and 68% of the HSL width.
Clinical Relevance:
Reconstructing HSLs with talus OCA is a promising treatment option with excellent fit and restoration of HSLs. This study will help guide surgeons to optimize OCA bone plugs from the humeral head, femoral head, and talus for varying sizes of HSLs.
Keywords
The Hill-Sachs lesion (HSL) is a compression fracture of the posterosuperolateral humeral head that is typically related to anterior shoulder instability or dislocation. The incidence of HSLs has been cited as high as 100% in patients with recurrent anterior shoulder dislocations. 16 A lesion is considered engaging, or “off-track,” when it is located outside the glenoid track, which is the contact zone between the glenoid and humeral head. Yamamoto et al 27 initially suggested that an engaging HSL is an indication for surgical management in 2007. 3 The engaging HSLs result in pathological contact between the humeral head and glenoid rim, creating further formation of bony defects on both the glenoid and humeral head. 16 The prevalence of the engaged lesion has been reported to range from 1.5% to 34% of all HSLs. 13 On-track and off-track HSLs may guide treatment options for a bipolar lesion. 4,19 The goal of treating an HSL is to directly or indirectly prevent engagement of the lesion along the glenoid track (off-track HSL). 3,16,27 There are many procedures to address a large, engaging HSL defect, including humeral head reconstruction (autograft or allograft), Hill-Sachs reduction, rotational osteotomy, and partial or complete humeral head arthroplasty (hemiarthroplasty). 6,7,16 The decision behind each treatment option differs based on the size of the HSL, concomitant glenoid bone loss, chronicity, glenohumeral version, age, and demand of the patients. 16,19
Humeral head reconstruction is indicated for a large HSL with or without significant glenoid bone loss. 6,16 Fresh or fresh-frozen allograft from the femoral or humeral head is commonly used to reconstruct the humeral head. 21,22,28 Fresh osteochondral allografts (OCAs) are preferred because they allow for an anatomic reconstruction of the humeral head while also addressing any associated chondral deficiencies while maintaining a lower resorption rate. 17,22 The use of fresh humeral head allografts has shown success in repairing HSLs, but concerns over the availability of donors and the risk of graft harvest contamination from the donor have justified the search for alternative allograft sites. 15 The talus has been indicated as a promising source for osteochondral graft for the humeral head. 2,9 The talar dome shows a high congruence match and offers a maximum size of 30 mm × 10 mm for a single graft. 2 In addition, the anthropometric properties of the talus are similar to the native humeral head and the radius of curvature (ROC) of the glenoid. 9
Previous literature has demonstrated that an osteochondral talus allograft is an excellent option for anatomic humeral head reconstruction. 14,20 However, inaccurate measurement and shaping of the allograft can be detrimental to the postoperative outcomes when choosing a single size-match allograft or an “orange slice” shape allograft. 14,20 Single or multiple OCA plugs were introduced for filling the large defect instead of a large graft cut to size since they are technically easier and may modify for an arthroscopic purpose in the future to avoid the morbidity of the open approach. 12,26 In addition, the location of the medial margin of HSL relative to the anterior glenoid rim is important to determine the risk of engagement. 10 It is vital to repair the most medial margin of the HSLs to convert the off-track HSL into an on-track HSL. Despite this, there has been limited information regarding the ideal size, location, or shape of the OCA bone plug to address the large and engaging HSL defect.
The purpose of this study was to determine the optimal number, diameter, medialization, and superiorization of talus OCA bone plugs in a computer-simulated HSL model. We hypothesized that reconstructing HSLs with talus OCA bone plugs would restore the surface area of most HSLs, thus converting all off-track HSLs into on-track HSLs.
Methods
Patient Selection
A retrospective review was performed of consecutive patients who were evaluated by the senior author (M.T.P.) for operative stabilization of anterior glenohumeral instability from January 2004 to January 2010. The data received institutional review board approval for use in this study. Excluded were patients who did not present with an HSL; patients younger than 16 years old; and patients with multidirectional instability, glenohumeral arthritis, a history of posterior shoulder instability, and prior ipsilateral shoulder surgery.
Diagnostic Imaging
All patients underwent standard glenohumeral CT scans using Siemens Sensation 64 (Siemens), a 64-detector scanner. Patients were placed supine with arms in an adducted and neutral position. Image data were acquired with 0.6 mm of collimation, 140 kV, and 300 mA-seconds at a slice thickness set to 2 mm. Virtual 3-dimensional (3D) geometry of intact humeri was modeled by matching the contour of the lesion’s rim to the articular surface of the ipsilateral humeral head. The geometry of the HSL was extracted by subtracting the humeral head geometry from the virtual geometry of the intact humeral head. A detailed description of the methods used to reconstruct the 3D HSL models has been described previously. 8
Anatomic Measurements
Anatomic measurements of the HSL, including position (medial, lateral, superior, and inferior), width, height, surface area, and glenoid track width, were recorded. After completion of all imaging analysis, inter- and intrarater reliability testing was performed for 3D HSL measurements. Twenty predetermined HSLs were measured by 2 trained researchers (P.G., orthopaedic resident, and A.M.P., premedical orthopaedic researcher) and compared.
Two-dimensional HSL geometry was defined for each HSL. First, local coordinate systems were created for each HSL. The most medial, most lateral, most superior, and most inferior points on the HSL geometry were recorded. The origin was defined as the closest point on the line connecting the superior and inferior points, represented in vector format as SI, to the line connecting the medial and lateral points, represented in vector format as ML. The x-axis was defined as the vector, SI, the z-axis was defined as the cross product of the x-axis and the ML vector, and the y-axis was defined as the cross product of the z-axis and x-axis. Next, a 2-dimensional HSL boundary was derived by projecting the 3D HSL point cloud onto the x-y plane and extracting the resulting boundary points.
A custom optimization algorithm was created to maximize bone plug surface area Y 1(X) and minimize its position relative to the medial margin of the HSL Y 2(X), subject to the following constraints:
where
where n is the bone plug index (n = 1 if describing the first bone plug, n = 2 if describing the second bone plug, etc), is the y-axis coordinate of the bone plug position, is the x-axis coordinate of the bone plug position, is the y-axis coordinate of the most medial point on the HSL, and is the x-axis coordinate of the most superior point on the HSL. Equality constraint h(X) (Equation 1) constrains the bone plug parameters such that the entirety of the bone plug area is located within the HSL and such that bone plugs do not overlap. Inequality constraint g(X) (Equation 2) constrains the bone plug parameters such that the bone plug diameter(s) must be greater than 8 mm, the bone plug diameter(s) must be less than 12 mm, and that no more than 3 bone plugs can be implanted.
A Hooke-Jeeves method was employed to solve the objective functions, using 2 types of moves, an exploratory move and a pattern move. 24 This process is briefly described. Initial base points, u 0, were set such that x 1 was equal to 1 bone plug, x 2 was equal to the minimum bone plug diameter (8 mm), x 3 was equal to the medial position of the most superior point of the HSL, and x 4 was equal to the position of the most superior point of the HSL (8 mm). With these initial conditions, the objective functions of the first bone plug were solved from the superior margin of the HSL, and the objective functions of the subsequent bone plugs were solved from the superior margin of the last bone plug added. Step-size parameters for each bone plug parameter were 1 for x 1, 0.1 mm for x 2, 0.1 mm for x 3, and 0.1 mm for x 4. The exploratory move of the Hooke-Jeeves method determined if an improvement could be made to the current base point along every coordinate axis equal to the step-size parameter. This process was repeated until all the search directions were used and additional improvements to the current base point failed. At this point, the step size for x 2, x 3, and x 4 was reduced by a factor of 10, and the exploratory process repeated until convergence was achieved (step size <0.001). When convergence was achieved in the exploratory move, the pattern move was applied that searched beyond the current base point, u 1, along the successful search directions to create a point u 2 = u 0 + 2(u 1 – u 0). An exploratory move was then applied at u 2, and if the current base point was improved, then u 2 was updated as the new current base point. This process was repeated until convergence was achieved (Figure 1). Validation of the Hooke-Jeeves optimization method was established by performing convergence analyses on all bone plug parameters. For all bone plug parameters, fewer than 100 iterations were necessary to reach the final parameter values (step size <0.001). This convergence rate is comparable to other methods that have used the Hooke-Jeeves method for other purposes. 24,25

Flow chart of the Hooke-Jeeves method to solve the objective functions, using 2 types of moves, an exploratory move and a pattern move, to find the optimal number, diameter, medialization, and superiorization of the bone plug(s).
The Hooke-Jeeves method yielded optimal bone plug number, bone plug diameter, bone plug medialization, and bone plug superiorization for all HSLs. Medialization was defined as the distance between the center of each bone plug to the most medial aspect of the HSL defect, while superiorization was calculated by the distance between the most superior apex of the HSL defect and the center of each bone plug (Figure 2). If the optimization problem did not converge, or if the HSLs were too small to fit the smallest bone plug size, the HSL was excluded. The resulting bone plug surface area and bone plug surface area to HSL surface area percentage were calculated. Finally, a Jenks natural breaks classification method was performed on the resulting optimal bone plug diameters to identify 3 bone plug sizes that maximized surface area coverage. The Jenks natural breaks classification method is an iterative process where calculations are repeated at different interfaces in the data set to determine which set of interfaces have the smallest in-class variance and largest variance between classes. For the purpose of this study, the data set of bone plug diameters was first ordered from largest to smallest. The method calculated the sum of squares deviations at each potential interface starting from the largest diameter (12 mm) to the smallest diameter (8 mm) and identified the interface at which the in-class variance was minimal and the between-class variance was maximal.

Hill-Sachs lesion (HSL) of a left humerus generated using 3-dimensional modeling software and showing the optimal bone plug numbers, medialization, and superiorization. Medialization (X 1 and X 2) was calculated by the distance between the centers of the bone plugs (C 1 and C 2) and the line parallel to the most medial aspect of the HSL (L 2). Superiorization (Y1 and Y2) was calculated by the distance between the line parallel to the most superior aspect of the HSL (L 1) and the centers of the bone plugs (C 1 and C 2).
Statistical Analysis
Descriptive statistics were obtained for all demographic data and anatomic measures. After completion of all imaging analysis, inter- and intrarater reliability testing was performed for the 3D HSL measurements. Twenty predetermined measures for HSL anatomic position, width, height, surface area, and glenoid track width were measured by 2 trained researchers (A.M., premedical orthopaedic researcher, and P.G., orthopaedic resident) and compared. One week later, the measurements were repeated by a researcher (A.M.) to assess the intrarater reliability. Based on the 95% confidence interval (CI) of the intraclass correlation coefficient (ICC), the reliability values were classified as excellent (ICC ≥0.9), good (ICC 0.75-0.89), fair (ICC 0.5-0.75), or poor (ICC <0.5). 11 Statistical analysis was performed using SPSS Statistics for Windows (Version 25; SPSS).
Results
A total of 132 patients were reviewed, of which 86 met the criteria for inclusion and were subsequently enrolled. The study population consisted of 53 right and 33 left shoulders (76 male, 10 female), with a mean age of 28.9 years (range, 19-43 years). Mean HSL width, height, surface area, and glenoid track width are displayed in Table 1.
HSL Parameters of Included Patients (n = 86)

HSL, Hill-Sachs lesion.
The optimization identified 18 (21%) of HSLs requiring 1 bone plug, 56 (65%) requiring 2 bone plugs, and 12 (14%) requiring 3 bone plugs (Figure 3). The mean optimal bone plug diameters were 9.62 ± 1.31, 10.02 ± 1.31, and 9.61 ± 1.46 mm for HSLs with 1, 2, and 3 bone plugs, respectively. The mean width, height, and surface area of HSLs that required 1, 2, or 3 bone plugs are displayed in Table 2. The mean restored HSLs defect was 68.3% ± 2.6%. All off-track HSLs (n = 17) were restored to on-track HSLs.

Hill-Sachs lesions (HSLs) of right humeri that were generated using 3-dimensional modeling software. The optimizations identified (A) 18 (21%) of HSLs requiring 1 bone plug, (B) 56 (65%) of HSLs requiring 2 bone plugs, and (C) 12 (14%) of HSLs requiring 3 bone plugs.
Optimal Width, Height, and Surface Area for HSLs that Required 1, 2, or 3 Bone Plugs a

a Data are shown as mean ± SD. HSL, Hill-Sachs lesion.
Mean optimal bone plug medialization (distance from the center of the bone plug to the most medial margin of the HSL defect) was 1.54 mm ± 0.95 mm, 2.45 mm ± 1.91 mm, and 2.32 mm ± 1.43 mm for HSLs with 1, 2, and 3 bone plugs, respectively. Mean optimal bone plug superiorization (distance from the center of the bone plug to the most superior margin of the HSL defect) was 6.84 mm ± 5.97 mm, 2.87 mm ± 1.90 mm, and 1.81 mm ± 1.20 mm for HSLs with 1, 2, and 3 bone plugs, respectively. The percentage of the bone plug of restored HSL surface area was 49.2% ± 14.9%, 60.0% ± 6.0%, and 60.0% ± 5.9% with 1, 2, and 3 bone plugs, respectively (Table 3). The Jenks natural-breaks classification method calculated 3 ideal bone plug diameters of 8 mm (small), 10.4 mm (medium), and 12 mm (large) (Figure 4). The ICCs for interrater agreement ranged from 0.94 to 0.98, and intrarater reliability ranged from 0.89 to 0.97, indicating good to excellent reliability. 11
Characteristics After Bone Plug Optimization for HSLs that Required 1, 2, or 3 Bone Plugs a

a Data are shown as mean ± SD. HSL, Hill-Sachs lesion.

Results of Jenks natural-breaks classification method to determine the optimal bone plug diameters that maximized bone area. (A) The algorithm identified interfaces at the 1st, 36th, and 96th percentile of the data set. (B) These interfaces corresponded to the optimal bone plug diameters at 12.0, 10.4, and 8.0 mm (red arrows).
Discussion
The principal finding of the study was that the most engaging HSLs required 2 bone plugs (65%), covering 60% of the overall surface area of the humeral defects. This data can assist surgeons with optimizing their treatment and placement of OCAs for HSLs. When using osteochondral talus allografts, approximately 68% of HSL width needed to be covered, while all off-track HSLs in this study were converted to on-track lesions. Lastly, the optimal talus OCA bone plug diameters to reconstruct HSLs are small (8 mm), medium (10.4 mm), and large (12 mm). The results of this study found optimal sizing and coverage of the bone graft for addressing off-track HSLs utilizing osteochondral talus allografts.
HSLs pose a significant risk for recurrent anterior glenohumeral instability; however, it is not clearly defined which morphological defects require bony augmentation. 6,18 Sekiya et al 23 demonstrated that the size and orientation of HSLs had a significant effect on glenohumeral joint function. They found that increasing HSL size required less anterior translation force before dislocation and decreased the stability ratio. They also found that larger defects (ie, 37.5-50%) would benefit from OCA transplantation by essentially restoring biomechanical stability and stability ratio. 23 Those authors emphasized that it is prudent to treat larger and engaging HSLs with an augmentation procedure. There is generally a paucity of clinical data regarding the use of OCA for HSLs; however, Zhuo et al 28 conducted a retrospective case series of 19 patients undergoing OCA transplantation for large HSLs with a mean follow-up of 27.8 months and found significant improvements in ROM, American Shoulder and Elbow Surgeons (ASES) score (53.2 ± 6.83 vs.96.9 ± 2.43, P < .001), Constant-Murley score (81.1 ± 5.11 vs.88.8 ± 3.48, P < .001), and Rowe score (23.6 ± 7.22 vs. 97.6 ± 2.12, P < .001) from preoperative to postoperative outcomes. However, they failed to include OCA implant parameters. Using a customized computer algorithm, we demonstrated in the present study the optimal talus OCA bone plug diameters for reconstructing HSLs in order to successfully restore the HSL track and, on average, 60% of the HSL surface area and 68% of the HSL width. We believe that the study findings can be used to guide the treatment of HSLs.
OCA transplantation is the treatment of choice for the reconstruction of large and engaging HSLs using various techniques ranging from miniopen to arthroscopic. 12,21,22,26,28 There has been significant evolution in surgical technique since Kropf and Sekiya 12 further demonstrated a single OCA plug to fill the large HSL and showed good short-term outcomes postoperatively. A biomechanical study of 9 cadaveric shoulders by Barkshi et al 1 found a perfect OCA repair had significantly less anterior translation with an anterior load than an imperfect OCA at 0° (15.3 ± 8.2 vs. 16.6 ± 9.0 mm; P = .008) and 30° of shoulder abduction (13.6 ± 7.1 vs. 14.4 ± 7.1 mm; P = .011). These findings emphasized the biomechanical importance of performing an osteoallograft repair in which the allograft closely matches the Hill-Sachs defect. 1 They emphasized that a disparity of 5% to 10% from articular congruity could severely disrupt the convexity of the humeral head, resulting in increased glenohumeral translation. However, ensuring a good press-fit of an allograft is challenging. Careful measurement and shaping of the defect (orange slice) and allograft are important to optimize good postoperative outcomes. 14
There is limited biomechanical evidence to compare single-plug and multiple-plug OCA transplantation for the treatment of HSLs. A biomechanical study in a porcine model by Dwivedi et al 5 demonstrated that there was no significant difference in pull-out strength and displacement in maximum load between the large cylindrical single plug and multiple plugs OCA transplantation. Results from their study showed good initial stability for the multiple plugs OCA; however, further in vivo study is needed to validate stability, healing process, and clinical outcomes. In the present study, we chose multiple-plug OCAs to fill the HSLs and demonstrated that the most engaging HSLs required 2 bone plugs to convert to on-track HSLs. We also demonstrated the HSL parameters that required 1, 2, or 3 bone plugs and found that width and surface area were crucial for considering multiple bone plugs for HSL reconstruction.
The present study redemonstrated that the risk of engaging HSLs is contingent upon the size of HSLs, as well as the medial margin of the HSL. 10 This is substantiated by a 3D computed tomography (CT) study of 100 patients with recurrent anterior instability by Golijanin et al 8 demonstrating that a more medialized HSL had a larger volume, greater width, more surface area loss, more inferior in the humeral head, and higher lesion angles. In addition, the study showed that more medialized HSLs are associated with a higher risk of creating off-track lesions, as well as a lower functional outcome. 8 It is vital that the surgeon not only fill the entirety of the defect but also repair the most medial margin of the HSL in order to convert the off-track lesion into an on-track lesion (stable). This is the first study to determine the optimal number, location, and size of the bone plug to repair the HSLs defect. In our study, there was no remaining off-track lesion after bone plug insertion. Consequently, humeral reconstruction with a talus OCA bone plug is an acceptable treatment option to reduce the risk of recurrent instability.
There are a wide variety of allograft types between fresh or fresh-frozen versions of the femoral head, humeral head, or talus allograft. 9,14,20 –22,28 The study by Zhu et al 28 showed a high union rate, significantly improved range of motion, and good functional outcomes at midterm follow-ups after reconstructing large HSLs with fresh-frozen humeral head allograft, although they did demonstrat a high graft resorption rate (43.1%) in their case series. Saltzman et al 22 conducted a systemic review of OCA transplants for large osteochondral defects on the humeral head and found that OCA transplantation showed improvement in range of motion and functional outcomes at 1 year postoperatively and that the use of fresh allograft tissue resulted in less resorption rate and necrosis compared to frozen grafts. 22 Thus, fresh allograft tissue may be the promising reconstructive option for HSL defects. The talus allograft offers several advantages over other graft options, such as the similar ROC between the talus and humeral head, which is consistent regardless of the donor graft size and laterality of the talar dome graft, allowing for more consistent size matching. 2 The cartilaginous surface of the talus creates more congruent articular contact with the glenoid that can provide a stable graft fixation. 20 The talus allograft can be utilized to reconstruct both humeral and glenoid bone defects in recurrent shoulder instability. However, the use of the talus allograft does come with notable disadvantages. There is limited availability of allografts in some regions of the world, the procedure may be technically difficult, and proper allograft preparation is required to reduce the track of the HSL.
Limitations
This study is not without limitations. First, this was a computer-simulated model study, where measurements of HSL and osteochondral talus allografts are inherently operator-dependent. Second, due to the study design, several risk factors for recurrent instability, such as the Hill-Sachs angles, cannot be measured or accounted for by the computer algorithm. Finally, the purpose of our study was to evaluate the morphometric properties of HSLs and their respective osteochondral talus allograft for off-track lesions, and as such, there is no comparative cohort to the other alternative graft options or outcome measures, especially the healing rate or complications. Future studies investigating outcomes relating to osteochondral talus allograft size would be warranted.
Conclusion
The optimal OCA bone plug diameters to reconstruct HSLs are small (8 mm), medium (10.4 mm), and large (12 mm), and can successfully restore HSL track and, on average, 60% of HSL surface area and 68% of the glenoid track width. Reconstructing HSLs with OCA is a promising treatment option with excellent fit and restoration of HSLs. This study will help guide surgeons to optimize OCA bone plugs from humeral head, femoral head, and talus for varying sizes of HSLs. Additional clinical work is needed to validate the procedure.
Footnotes
Acknowledgment
The authors give special thanks to the Digital Media Department of the Steadman Philippon Research Institute for its help with the illustrations.
Final revision submitted April 5, 2023; accepted May 3, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: M.T.P. has received grant support from DJO; consulting fees from Arthrex, Arthrosurface, JRF Ortho, and Zimmer Biomet; royalties from Anika Therapeutics, Arthrex, Arthrosurface, and Responsive Arthroscopy; honoraria from Flexion Therapeutics; is a governing board member for SLACK; and serves on the medical board of trustees for MTF Biologics. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Naval Medical Center San Diego (Ref No. NMCSD.2005.0038).