Abstract
Background:
Despite improved outcomes, failure or nonhealing of graft materials has been reported after superior capsular reconstruction (SCR) for massive irreparable rotator cuff tears.
Purpose:
To evaluate the short-term clinical and radiological outcomes of a novel technique for SCR using an Achilles tendon–bone allograft.
Study Design:
Case series; Level of evidence, 4.
Methods:
We performed a retrospective review of patients who underwent SCR using an Achilles tendon–bone allograft with the modified keyhole technique and who had a minimum follow-up of 2 years. The visual analog scale score for pain, American Shoulder and Elbow Surgeons score, and Constant score were evaluated as subjective outcomes, while range of motion of the shoulder joint and isokinetic strength were evaluated as objective outcomes. The acromiohumeral interval (AHI), bone-to-bone healing of the allograft and humeral head on computed tomography, and graft integrity on magnetic resonance imaging were evaluated as radiological outcomes.
Results:
This study included 32 patients with a mean age of 56.8 ± 4.2 years and a mean follow-up of 28.4 ± 6.2 months. A significant improvement from preoperatively to the last follow-up was seen in the mean visual analog scale score for pain (from 6.7 to 1.8), American Shoulder and Elbow Surgeons score (from 42.7 to 83.8), Constant score (from 47.2 to 78.5), and AHI (from 4.8 to 8.2 mm) (P < .001 for all) as well as range of motion in forward elevation and internal rotation (P < .001 for both). Medial-to-lateral graft integrity was good in all patients. Nonunion at the fitting zone of the keyhole on the greater tuberosity was diagnosed in 1 case (3.1%), and failure of incorporation between the allograft and remnant tendon at the site of posterior margin convergence was observed in 4 cases (12.5%).
Conclusion:
The outcomes after SCR using an Achilles tendon–bone allograft and the keyhole technique improved, with an increased AHI and excellent integrity in the medial and lateral directions compared with preoperatively. This technique is a reasonable option for the surgical treatment of irreparable rotator cuff tears.
Keywords
Since Mihata 21 described superior capsular reconstruction (SCR) of the shoulder using a fascia lata autograft, it has become a popular treatment method for irreparable rotator cuff tears. Although previous surgical options for irreparable rotator cuff tears have included arthroscopic debridement, partial repair, margin convergence, patch graft, tendon transfer, and reverse total shoulder arthroplasty, SCR provides a static restraint to proximal migration of the humeral head in superior rotator cuff–deficient shoulders with early encouraging outcomes. 1,11,26,36 Despite the recent success of this operative procedure, up to 55% of SCR procedures have been reported to fail, result in retears, or have nonhealing of the graft. 2,3,5,9,37 Additionally, 5% of patients undergoing SCR require revision surgery, and 20% of patients undergoing this procedure report being dissatisfied. 2,3,9
Graft integrity after SCR is an important independent factor for improving postoperative functional outcomes. 8,21,24 At present, graft failure rates reportedly vary between 0% and 55% and appear to be comparable among various graft types. Most of these failures have been observed at the fixation point of the graft on the greater tuberosity. 19 Basic science studies have emphasized the importance of bone-tendon interface regeneration at the greater tuberosity to reduce the rate of rotator cuff repair failure. 20,31,32 Results from animal studies have shown that homogeneous tissue healing (bone to bone) is superior to heterogeneous tissue healing (bone to tendon) in terms of healing time and tissue integration with respect to the tensile properties of the healing complex. 18,35 The advantage of an Achilles tendon–bone allograft with respect to anterior cruciate ligament reconstruction is an intact native bone–tendon interface that allows for homogeneous tissue healing. 4,34
In 2020, the senior author (J.-W.K.) introduced the technique of performing SCR with an Achilles tendon–bone allograft to provide stronger fixation at the greater tuberosity. 15 The purpose of this study was to evaluate the clinical and radiological outcomes of this technique. We hypothesized that this technique would produce satisfactory outcomes with good graft integrity at a minimum 2-year follow-up.
Methods
Patient Selection
This study was a retrospective review of patients with irreparable rotator cuff tears who underwent SCR with an Achilles tendon–bone allograft between 2018 and 2020. The research protocol was approved by an institutional review board, and all included patients provided written informed consent. Included were patients with (1) chronic irreparable rotator cuff tears (massive or 2-tendon rotator cuff tears), (2) glenohumeral cartilage changes not more severe than Hamada type 3, 12 (3) an intact or repairable subscapularis tendon, (4) an age ≤70 years, and (5) ≥2 years of follow-up data. The exclusion criteria were (1) a rotator cuff reducible to the lateral footprint during arthroscopic surgery (no interposition needed) and (2) the inability to participate in the postoperative rehabilitation protocol. A total of 32 patients were included in the analysis (Figure 1).
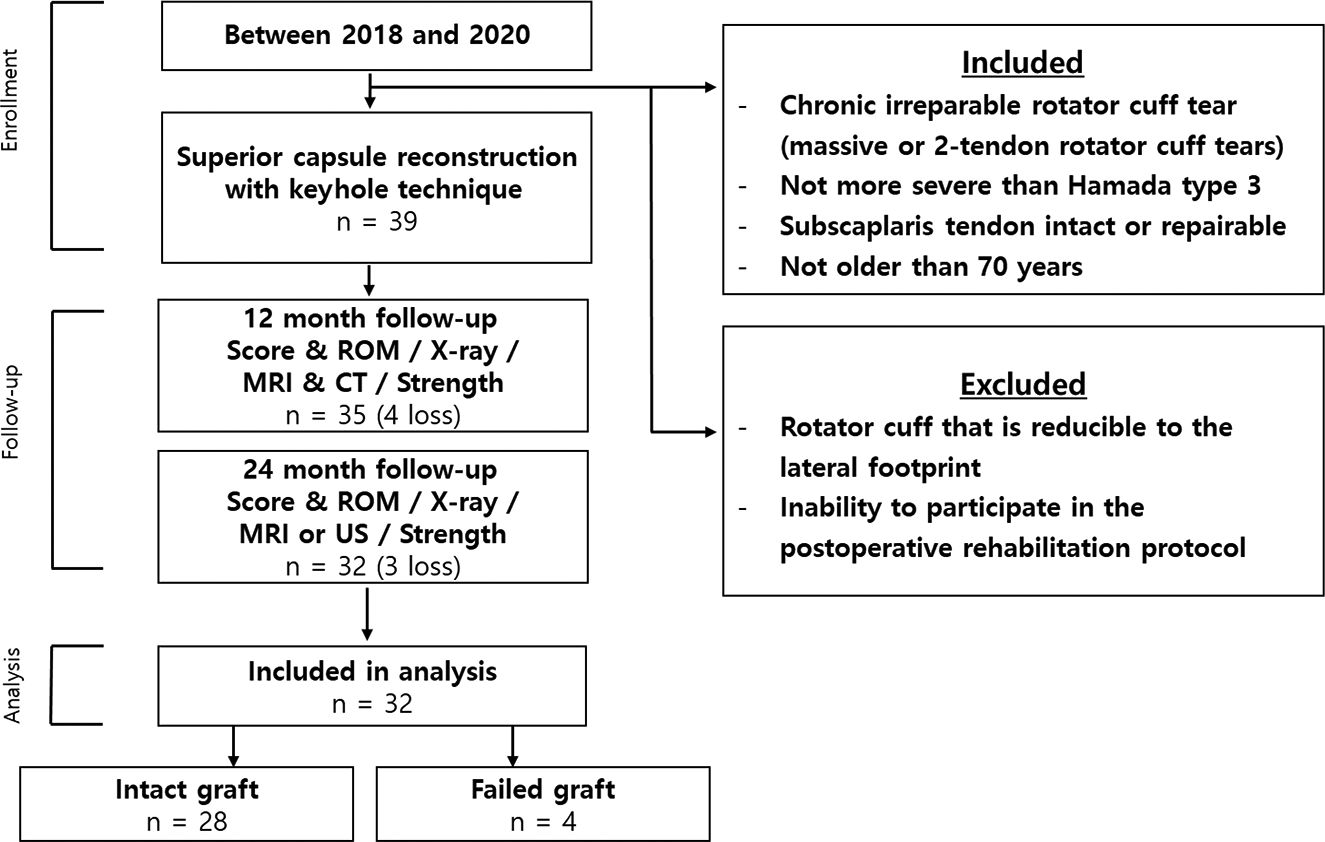
Study flow diagram. CT, computed tomography; MRI, magnetic resonance imaging; ROM, range of motion; US, ultrasound.
Surgical Technique and Rehabilitation
The surgical technique and rehabilitation protocol have previously been described in detail. 15 The patient was placed in a standard beach-chair position, and diagnostic arthroscopic surgery was performed according to the standard protocol. SCR was considered if there was an irreparable full-thickness tear of the supraspinatus and infraspinatus that could not be reduced to the native footprint.
An Achilles tendon–bone allograft was prepared intraoperatively. The tendon-bone diameter was checked through a graft sizing block to ensure that it was 10 mm, which was the size needed to insert it into the keyhole of the humeral head. After the allograft was thawed, a bur, microsaw, rasp, and rongeur were used to create a 10-mm transverse bone plug from the calcaneus, along with the attached tendon. The appropriate graft length (5.5 cm for male patients and 5.0 cm for female patients) was measured by using the scale of the probe for each patient. Once the size was confirmed, the length of the Achilles tendon that was to be folded into 2 layers using multiple No. 2 Ethibond sutures (Ethicon) was marked to obtain ≥8 mm–thick graft through the Krackow suture technique. Then, a No. 2 Ethibond suture was used to make 4 shuttle relay stitches on the opposite side of the bone (Figure 2).

Preparing the Achilles tendon–bone allograft. (A) The diameter of the calcaneus attached to the allograft was confirmed to be ≥10 mm. (B) After the allograft was prepared for the 10-mm transverse bone plug from the calcaneus with the attached tendon, the Achilles tendon was folded into 2 layers using the Krackow suture technique to obtain a minimum 8 mm–thick graft. (C) The appropriate graft length was measured by using the scale of the probe for each patient. This was usually 5.5 cm for male patients and 5.0 cm for female patients.
The anchors (Y-Knot RC All-Suture Anchor with 2 No. 2 Hi-Fi Sutures; ConMed Linvatec) were placed as far anteriorly and posteriorly as possible to provide adequate spread and coverage for medial graft fixation on the glenoid; 2 anchors were placed in the region between the 10- and 2-o’clock positions.
After glenoid preparation through arthroscopic surgery, the skin incision was extended about 5 cm from the anterior portal along the anterolateral approach lines. The entry point of the tunneling guide pin was 1 cm below the humeral upper margin in the biceps groove. Also, the direction of the pin was parallel to the articular margin of the humeral head as viewed from the top. A 10-mm reamer was used to create a tunnel for bone fitting. Because penetrating the far cortex with the reamer in this process may cause fractures of the greater tubercle, it was important to avoid reaming up to the far cortex. After cutting the bone using a microsaw, the Achilles tendon–bone allograft was extracted from the superior margin between the humeral head and greater tubercle and was then inserted into the humeral hole using a bone tamp. Stability was obtained by fixing the graft using the suture bridge technique. The anchor threads from the glenoid were taken separately to the mini-open site through the arthroscope and passed through the shuttle relay from the deep to superficial side of the Achilles tendon. The Achilles tendon was then inserted back into the joint through the mini-open site and tied. At this point, the remnant portion of the rotator cuff and capsule near the glenoid was augmented with the Achilles tendon to be used as scaffolding. Side-to-side margin convergence sutures were placed to secure the graft to the intact rotator cuff. Typically, 2 or 3 side-to-side sutures were used posteriorly to connect the graft to the intact part of the infraspinatus or teres minor (Figure 3).
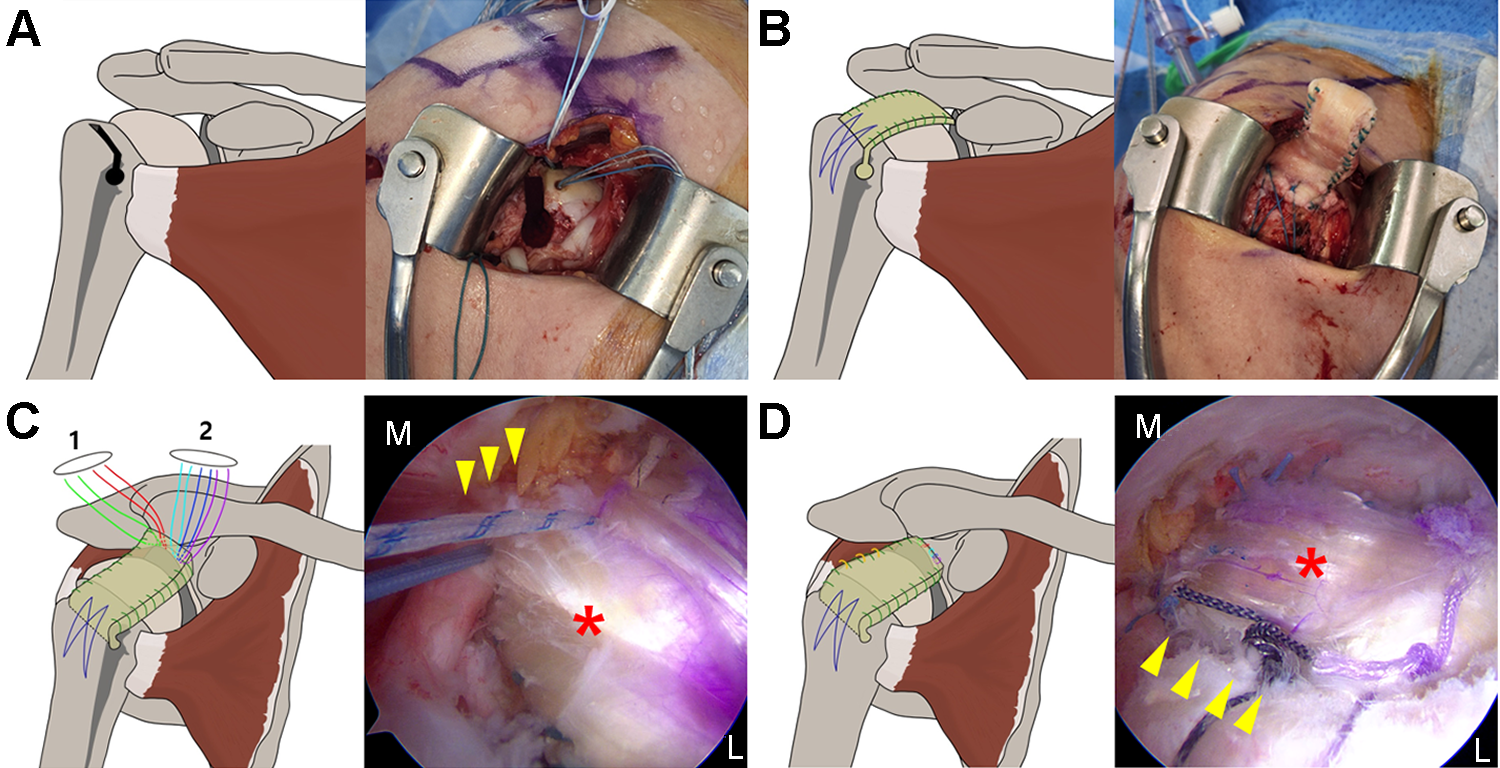
Diagrams of the surgical technique. (A) Tunneling using a 10-mm reamer into the biceps groove at 1 cm below the humeral head, avoiding reaming up to the far cortex. (B) A prepared Achilles tendon–bone graft was inserted into the humeral head and fixed using the suture bridge technique. (C) The anchor threads from the glenoid were taken separately to the mini-open site and passed through the shuttle relay from the deep to the superficial side of the Achilles tendon and were used to suture the medial remnant portion of the rotator cuff and capsule (arrowhead) and allograft (asterisk) on the glenoid side. 1 = posterior portal; 2 = Neviaser portal. (D) The Achilles tendon was inserted back into the joint through the mini-open site and tied. Then, 2 or 3 side-to-side sutures were used posteriorly to incorporate the allograft (asterisk) to the posterior remnant of the infraspinatus or teres minor (arrowhead). L, lateral; M, medial.
Postoperatively, the patient used a sling with an abduction brace and was allowed only elbow, wrist, and hand exercises for 4 weeks. Progressive passive motion, such as forward elevation (FE), external rotation (ER), and internal rotation (IR), began at 4 weeks postoperatively, and full active motion was allowed after 8 weeks. Strengthening progressed at 12 weeks, and return to activities requiring overhead lifting was allowed only after 16 weeks.
Patient Data
Demographic data including age, sex, smoking status, and body mass index were evaluated. Based on preoperative magnetic resonance imaging (MRI), the grade of the rotator cuff tear, fatty degeneration according to the modified Goutallier classification, 10 muscle atrophy according to the Warner classification, and the arthritic condition according to the Hamada classification were evaluated.
Subjective and Objective Assessments
All patients completed the visual analog scale for pain, American Shoulder and Elbow Surgeons (ASES) score, and Constant score preoperatively, at 1 year after surgery, and at the last follow-up.
Range of motion (ROM; FE, ER, and IR) was measured using a handheld goniometer preoperatively and at each follow-up point. For IR, the most proximal point at which the tip of the thumb touched the spinous process was scored based on contiguously numbered groups: T1-T12, 1-12; L1-L5, 13-17; buttock, 18; and greater tubercle of the proximal femur, 19. 28 Isometric muscle strength of the rotator cuff was measured using the PrimusRS dynamometer (BTE Technologies) preoperatively, at 1 year after surgery, and at the last follow-up.
The positions described by Kelly et al 13 were applied to measure muscle strength in FE, IR, and ER. Strength in FE was evaluated in a standing position with the shoulder in 90° of abduction in the scapular plane (a plane angled 30° anterior to the coronal plane) and 90° of ER of the humerus (“full can” position).
Strength in ER was evaluated in a sitting position with the shoulder in full adduction and 45° of IR of the humerus. The elbow was flexed to 90°. Patients were asked to keep their elbows at their side during testing, and an assistant stabilized the patient’s elbow against the patient’s waist to help maintain the position. Strength in IR was evaluated in a sitting position with the shoulder in full adduction and neutral rotation. The elbow was flexed to 90°. Stabilization of the patient’s elbow was performed in an identical manner to that in ER.
Radiological Assessment
The acromiohumeral interval (AHI) was analyzed using simple radiography (true anteroposterior view) at each follow-up visit. Union between the keyhole area of the greater tuberosity and bone of the Achilles tendon–bone allograft was evaluated using computed tomography. When adequate healing of ≥2 of the 3 cortical surfaces was observed, it was defined as union; signs of adequate healing consisted of reactive sclerosis, blurring of the tunnel margin, and the presence of bone within the tunnel, whereas signs of inadequate healing were sclerosis of the donor fragment in comparison with the host fragment, fragmentation, and a cystic lesion 27,33 (Figure 4). MRI at 1-year follow-up was performed in all 32 patients, and at 2-year follow-up, 14 of 32 patients were evaluated by MRI and 18 patients by ultrasonography to determine graft healing .

(A) Adequate healing and (B) nonhealing between the bone fragment of the allograft and the greater tuberosity.
The integrity of the graft was confirmed using MRI. MRI was used at 1-year follow-up, whereas either MRI or ultrasonography was used at the last follow-up, to check graft integrity. Graft integrity was classified on sagittal and coronal sections of T2-weighted images according to a 5-point scale of the Sugaya classification 30 as follows: type 1, sufficient thickness and homogeneous low intensity on each image; type 2, sufficient thickness associated with a partial high-intensity area; type 3, insufficient thickness without discontinuity; type 4, presence of minor discontinuity on >1 image, suggesting a small tear; and type 5, presence of major discontinuity on each image. Type 4 or 5 was defined as graft failure.
Statistical Analysis
Statistical analyses were performed using SPSS Statistics (Version 25.0; IBM). Data were reported as means with standard deviations or ranges or as absolute values. The paired-samples t test was used for comparing changes in subjective and objective outcomes from before to after surgery, the Mann-Whitney test or Student t test was used for comparing unpaired samples according to normality, and the chi-square test was used for comparing categorical data between 2 groups. A P value <.05 indicated statistical significance.
Results
Patient Data
The study population of 32 patients consisted of 14 male and 18 female patients, with a mean age of 56.8 years (range, 51-68 years) and a mean follow-up period of 28.4 months (range, 24-32 months). The baseline characteristics of the patients are summarized in Table 1.
Characteristics of Study Population (N = 32) a
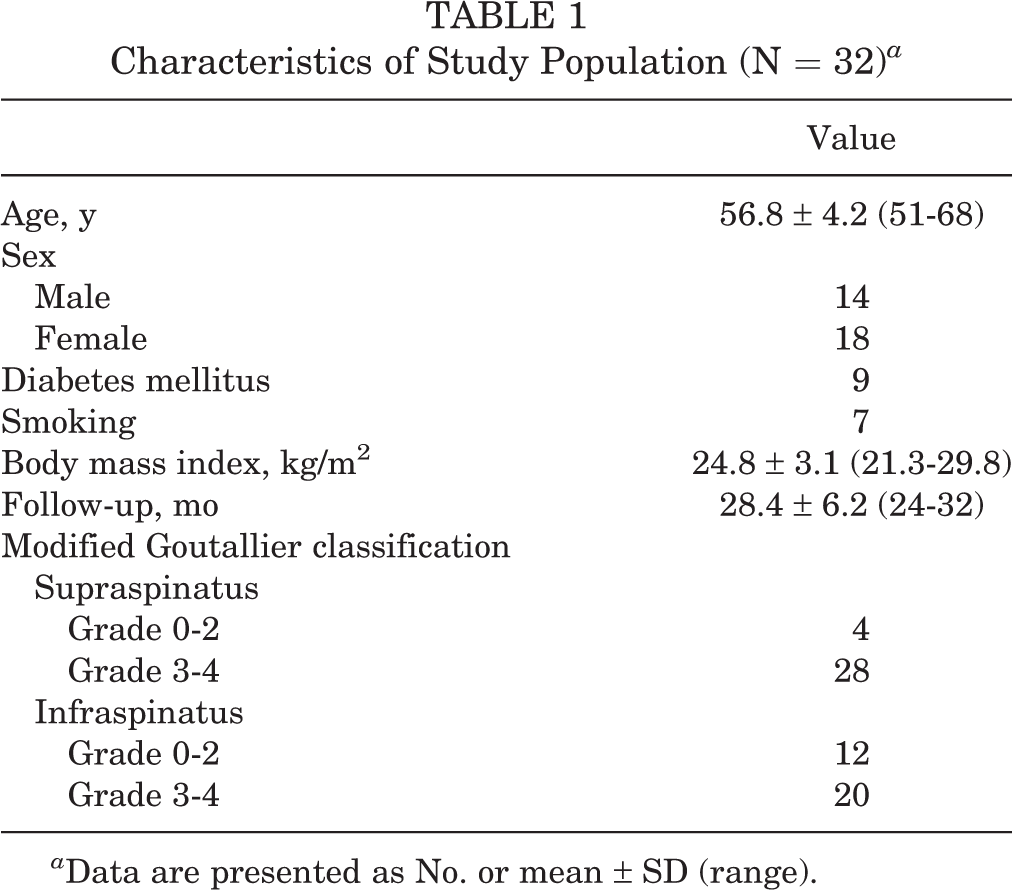
a Data are presented as No. or mean ± SD (range).
Subjective and Objective Outcomes
A statistically significant improvement was seen for the visual analog scale score for pain, the ASES score, the Constant score, ROM in FE and IR, and isometric strength at both 1-year follow-up and the last follow-up compared with preoperatively. Only ROM in ER did not improve significantly (Table 2 and Figure 5).
Preoperative and Postoperative Subjective and Objective Outcomes a
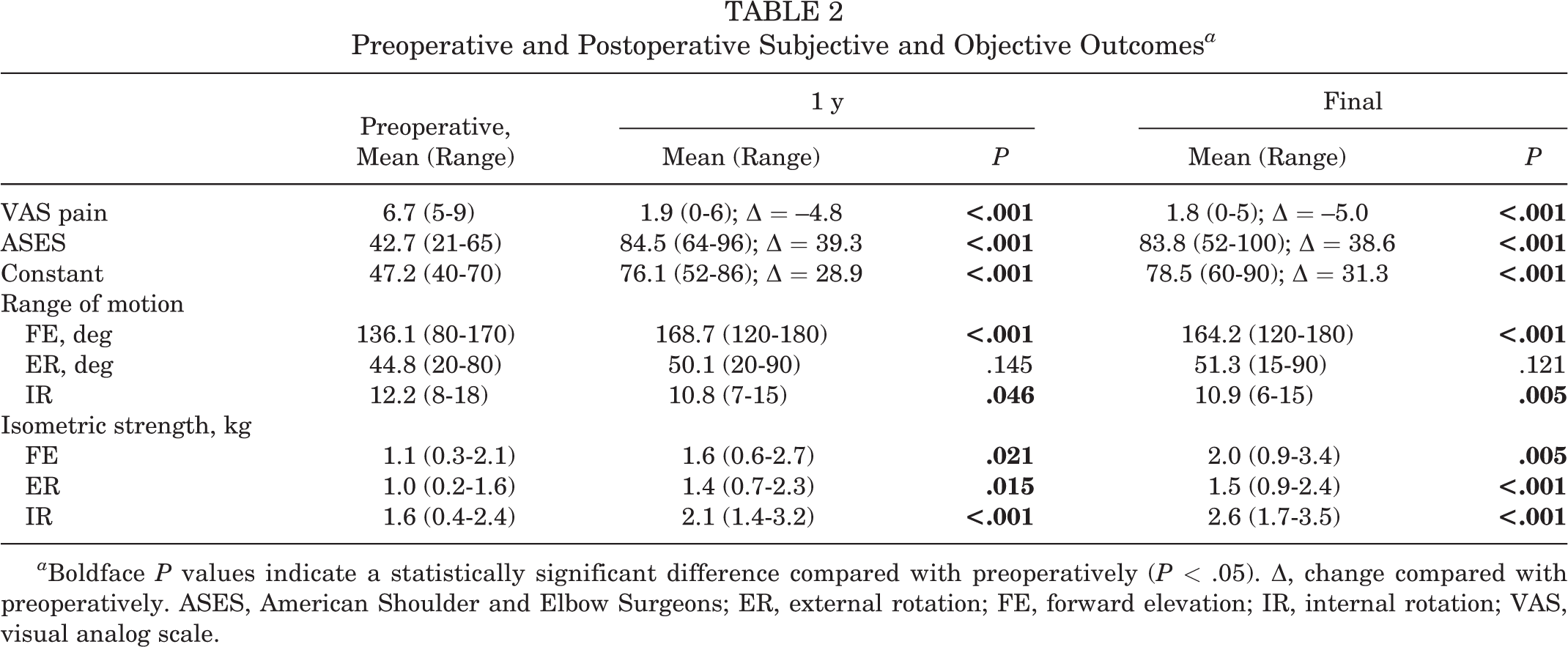
a Boldface P values indicate a statistically significant difference compared with preoperatively (P < .05). Δ, change compared with preoperatively. ASES, American Shoulder and Elbow Surgeons; ER, external rotation; FE, forward elevation; IR, internal rotation; VAS, visual analog scale.

Patient-reported outcome scores before and after surgery. Visual analog scale (VAS) scores were multiplied by 10. ASES, American Shoulder and Elbow Surgeons.
Radiological Outcomes
The mean AHI improved from 4.8 ± 1.1 mm (range, 4-8 mm) before surgery to 8.6 ± 1.6 mm (range, 7-12 mm) at 1-year follow-up and to 8.2 ± 1.2 mm (range, 7-11 mm) at the last follow-up (P < .001 for both) (Figure 6).
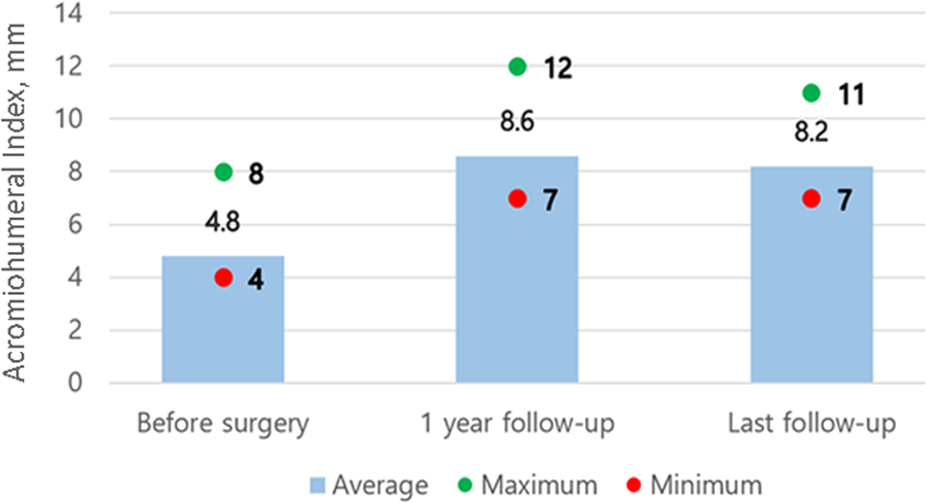
Acromiohumeral index before and after surgery.
Nonunion between the keyhole area of the greater tuberosity and bone of the Achilles tendon–bone allograft was observed in 1 patient (3.1%). However, no functional inferiority was observed in that patient.
Grafts were intact, in the medial to lateral direction, on MRI at 1 year and on MRI or ultrasonography at 2 years after surgery. However, failure of incorporation in the anteroposterior direction between the allograft and remnant tendon at the site of posterior margin convergence (PMC) was observed in 4 cases (12.5%) (Figure 7). There were significantly inferior clinical outcomes in patients who had failure of incorporation at the PMC site compared with patients who did not have failure (Table 3).

(A) Adequate healing of the graft on magnetic resonance imaging at 1-year follow-up (left: T2-weighted coronal; right: T2-weighted oblique sagittal). (B) Intact graft integrity in the mediolateral plane (left: T2-weighted coronal) and failure of engraftment at the site of posterior margin convergence (right: T2-weighted oblique sagittal).
Outcomes Between PMC Groups a

a Data are presented as mean (range). Boldface P values indicate a statistically significant difference compared with preoperatively (P < .05). AHI, acromiohumeral interval; ASES, American Shoulder and Elbow Surgeons; ER, external rotation; FE, forward elevation; IR, internal rotation; PMC, posterior margin convergence; VAS, visual analog scale.
Complications
There was a surgical complication of an avulsion fracture of the subscapularis tendon. After the avulsion fracture was detected, open reduction and screw fixation were performed. This did not significantly affect the graft, and the clinical outcomes after surgery were good.
Discussion
In the present study, SCR with an Achilles tendon–bone allograft using the modified keyhole technique 15 demonstrated significant improvements in patient-reported and clinical outcomes while maintaining an increased AHI and excellent medial-to-lateral integrity.
SCR is intended to restore superior stabilizing forces to prevent proximal migration of the humeral head caused by a deficient rotator cuff. This technique is accomplished by substituting an autograft or allograft between the superior glenoid and the humeral head. 6 Graft thickness plays a key role in maintaining glenohumeral stability after SCR. The graft failure rate can be decreased with a thick graft, which enhances the stiffness. 23 Several studies involving acellular dermal allografts reported graft elongation and persistent superior migration of the humeral head, as evidenced by a narrowed AHI. Furthermore, acellular dermal allografts were reported to elongate by 15% during biomechanical testing of shoulder ROM, glenohumeral translation, glenohumeral joint force, and subacromial contact pressure with physiological muscle forces. 22 Mihata et al 22 demonstrated that a fascia lata autograft, typically 6 to 8 mm thick, undergoes less elongation and thinning and provides better stability compared with 3 to 4 mm–thick dermal allografts. An Achilles tendon–bone allograft is an attractive choice because of its robustness, being of a thick and stiff material; it has shown success in the treatment of other ligamentous and tendinous abnormalities throughout the body. 7,8 In this study, the Achilles tendon–bone allograft had a minimum thickness of 8 mm, broadening out to appropriately cover the humeral head.
In previous studies, graft failure occurred mainly on the humeral side after SCR. Lee and Min 17 reported that 36.2% of graft tears occurred after SCR using a tensor fasciae latae (TFL) autograft: 30.6% occurred on the humeral side, and 5.6% occurred on the glenoid side. Graft tears on the humeral side were probably caused by excessive tension concentrated on the lateral portion of the graft (graft on the greater tuberosity) during active shoulder abduction and elevation. 19 An Achilles tendon–bone allograft with a native bone–tendon interface allows homogeneous tissue healing at the greater tuberosity. In the current study, 1 patient who was diagnosed with nonunion of the keyhole according to our criteria of nonunion had no pain in the graft area and no functional deficits. Although complete healing of the bone to bone itself could not be obtained, it is considered that the large contact area from the keyhole and fibrotic union provided sufficient fixation for functional movement. No graft failure was observed at the medial (glenoid) or lateral (greater tuberosity) fixation sites. Medial and lateral healing of the graft was probably associated with improvements in ROM and functional scores, as the shoulder joints were able to obtain force couples and remain functional.
In a systematic review by Lee et al, 16 the mean ASES score after SCR using a TFL autograft improved from 38.8 to 88.6, with a mean postoperative change of 49.7, and the mean ASES score using a dermal allograft also improved from 52 to 85.3, with a mean postoperative change of 34.6 from 6 studies. In our study, the mean ASES score improved from 42.7 to 83.8, and the mean postoperative change was 38.6. The degree of postoperative change was between a TFL autograft and dermal allograft in the study of Lee et al, and thus, our technique cannot be considered inferior compared with those 2 methods.
Failure of incorporation occurred at the tendinous portion of the PMC site, where side-to-side repair with the remaining rotator cuff was performed. The importance of margin convergence and posterior rotator cuff fixation has been emphasized in several studies. Oh et al 29 reported that posterior rotator cuff (infraspinatus) repair is necessary to restore abnormal glenohumeral kinematics, and margin convergence was recommended to decrease gap formation of the repaired tendon edge, which may have provided a better biomechanical environment for healing in a cadaveric model. In addition, Mihata et al 25 reported that when side-to-side suturing at the posterior portion during SCR was not performed, superior glenohumeral translation was markedly increased. Therefore, firm maintenance of PMC and its incorporation with the remaining infraspinatus are considered important for good outcomes. In the present study, failure of engraftment in the anteroposterior direction between the allograft and remnant tendon at the PMC site was observed in 12.5% of cases, and overall functional outcomes and ROM were found to be inferior compared with patients with intact PMC. Kholinne et al 14 suggested poor vascular ingrowth on histological analysis of a failed graft, which could lead to inferior healing. It is possible that the type and dose of sterilization, particularly gamma irradiation, may have contributed to the failure of engraftment. Another consideration is the poor vascularity of remaining rotator cuff tissue used for posterior repair. Regardless of the reason, during SCR using an Achilles tendon–bone allograft, it is important to perform firm and stable side-to-side reinforcement suturing to the medial and posterior remnant tendon to ensure a good prognosis.
Limitations
This study had limitations. First, this was a retrospective case series with a small sample size. Second, performance bias was possible, as the outcomes represented the experience of a single surgeon specializing in shoulder surgery. Third, there was no biomechanical examination to investigate the strength and stability of the lateral bone-to-bone fixation site of the greater tuberosity. Therefore, further biomechanical cadaveric studies that investigate the strength and stability of Achilles tendon–bone allografts after SCR using the keyhole technique should be performed.
Conclusion
The overall outcomes of SCR were improved with an Achilles tendon–bone allograft using the keyhole technique because of excellent integrity in the medial and lateral directions and maintenance of the AHI. SCR using an Achilles tendon–bone allograft was found to be a reasonable option for the surgical treatment of irreparable rotator cuff tears.
Footnotes
Final revision submitted February 15, 2023; accepted March 20, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was supported by a grant from Wonkwang University (2022). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Wonkwang University Hospital (No. 2022-04-021).
