Abstract
Background:
Limited evidence suggests a positive correlation between tibial tubercle–trochlear groove (TT-TG) distance and the risk of native anterior cruciate ligament (ACL) tear. The relationship between TT-TG distance and the risk of ACL graft failure is unknown.
Hypothesis:
TT-TG distance is independently associated with risk of ACL graft failure.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
All patients who underwent ACL revision surgery between 2010 and 2018 at a single institution were identified. A control cohort underwent primary ACL reconstruction (ACLR) between 2006 and 2015, with no evidence of graft failure at 8.1 ± 2.5 years postoperatively. Record review included anthropometrics, graft type, and estimated Tegner activity score at ≥6 months after primary ACLR. Magnetic resonance imaging (MRI) scans after native ACL tear (controls) or graft failure (revision cohort) were assessed for (1) TT-TG distance, (2) proximal tibial slopes, (3) depth of tibial plateau concavity, and (4) tunnel position (revision cohort). Associations between ACL graft failure and MRI measurements, surgical variables, and patient characteristics were evaluated with logistic regression analyses. Sensitivity analyses, excluding patients with tunnel malposition, were performed to confirm multivariable results in patients with “ideal” tunnel placement.
Results:
Participants included 153 patients who underwent revisions and 144 controls. Controls were older than the patients who underwent revision (26.6 ± 8.8 vs 20.6 ± 7.3 years; P < .001). The mean TT-TG distance and lateral posterior tibial slope (PTS) were smaller for the control group than for the revision group (TT-TG: 9.3 ± 3.9 vs 11.2 ± 4.2 mm; P < .001; lateral PTS: 6.2° ± 3.3° vs 7.2° ± 3.6°; P = .01). TT-TG distance, lateral PTS, and age were associated with risk of ACL graft failure by multivariable analysis (OR, 1.15; 95% CI, 1.07-1.23; P < .001; OR, 1.13; 95% CI, 1.04-1.22; P = .004; and OR, 0.90; 95% CI, 0.87-0.94; P < .001, respectively). With sensitivity analyses, TT-TG distance, lateral PTS, and age at index surgery remained significantly and independently associated with ACL graft failure.
Conclusion:
Increased TT-TG distance, increased lateral PTS, and younger age are independently associated with increased odds of ACL graft failure. Patients with these characteristics may require a more comprehensive strategy to reduce the risk of ACL reinjury.
Approximately 200,000 anterior cruciate ligament (ACL) reconstructions (ACLRs) are performed each year in the United States. Despite improvements in surgical techniques and postoperative rehabilitation, graft failure is approximately 4% at 2 years postoperatively for the general population and approximately 9% at 6 years postoperatively in high school and collegiate athletes. 16 Even worse, cumulative graft failure rates ranging from 3.2% to 29.9% have been reported at a 10-year follow-up. 6 Younger patients, those of male sex, those with preoperative ligamentous laxity, and those who participate in high-risk sports are at increased risk of ACL reinjury, especially if return to sport occurs prematurely. 36 Potential causes of failed ACLR have been classically categorized into traumatic reinjury, technical error, diagnostic error, and biologic origins. Several studies have cited high rates of technical error, with estimates of tunnel malposition as high as 70% to 80%. 18,21,24 Other contributors to atraumatic ACL graft failure include use of hamstring autograft (vs bone–patellar tendon–bone) or allograft, failure of graft incorporation, and failure to address secondary causes of instability. 10,16,23,25,36
Proximal tibial geometry and lower extremity alignment have been evaluated as risk factors for native and ACL graft tears. Recent studies have focused on the relationship between posterior tibial slope (PTS) and risk of ACL tear and rerupture. 5,33,34 Mechanically, increased PTS produces greater anteriorly directed shear force and anterior tibial translation, resulting in increased ACL strain. Thus, increased PTS has been associated with an increased risk of native ACL rupture and with earlier and higher rates of ACL graft failure. 5,33,34 The role of coronal limb alignment (quadriceps angle [Q angle]) in the origin of both native ACL injury and graft failure after reconstruction is less clear. 3 In theory, a widened pelvis, as demonstrated by an increased Q angle, may increase abduction moment at the knee, predisposing individuals to lower extremity injuries including ACL tears. Systematic reviews have failed to confirm this relationship, likely because of the difficulty in obtaining accurate measurements and the influence of other anatomic landmarks such as femoral anteversion and tibiofemoral alignment on the Q angle. 27,31 Furthermore, rotational alignment at the knee likely represents an additional important factor in determining injury risk, and this is not captured by Q angle measurement.
The tibial tubercle–trochlear groove (TT-TG) distance, most commonly assessed in relation to patellar instability, serves as a surrogate for the Q angle. TT-TG distance is defined as the offset distance between the center of the distal trochlear groove and the center of the patellar tendon insertion on the tibial tuberosity. 8 Although correlation between the 2 measures is imperfect, TT-TG distance is more precise than Q angle assessments and may therefore provide a better gauge of ACL injury risk. 4,11 Limited data in the current literature suggest that greater TT-TG distances may be associated with an increased risk of native ACL tear. 28,29 In addition, increased TT-TG distance has been shown to correlate with increased rotational laxity and anterolateral ligament (ALL) rupture in the ACL-injured knee. 27 These findings suggest a potential association between increased TT-TG distance and risk of ACL graft failure, but we are aware of no studies examining this relationship.
In the current study, we aimed to evaluate the role of TT-TG distance in predicting risk of ACL graft failure. We evaluated TT-TG measurements, tibial slopes, and tunnel placement parameters using magnetic resonance imaging (MRI), comparing a group of patients who underwent ACL graft revision after failure of primary ACLR versus a control group who underwent ACLR but had no evidence of graft tear at >2-year follow-up. We hypothesized that TT-TG distance would predict risk of ACL graft failure, independent of tibial slopes and tunnel placement.
Methods
Patient Population
This retrospective study was performed at a single academic center after institutional review board approval was received for the study protocol. A data query identified all patients who had undergone ACL revision surgery between 2010 and 2018 at our institution. Many of these (28%) had undergone primary ACLR at a different institution. For the purposes of this study, all patients who underwent ACLR without concomitant ligament surgery, such as multiligament or medial patellofemoral ligament reconstruction, were included. The patient’s electronic health record was reviewed, and patients were excluded for prior ACL revision surgery, age at index surgery unknown, and use of a physeal-sparing technique at the index surgery.
A control cohort was included for comparison with the revision group to assess whether MRI parameters (TT-TG distance, tibial slope), surgical variables (graft type, meniscal tear), and patient characteristics (age, sex, body mass index [BMI], activity level) were associated with ACLR graft failure. The control cohort underwent primary ACLR at our institution between 2006 and 2015. Included patients had ACLR without concomitant ligament surgery, such as multiligament or medial patellofemoral ligament reconstruction. Data for these patients had been prospectively collected via enrollment in a study evaluating ACLR outcomes; patients were included in the current study if they had also responded to a query at 2 to 13 years postoperatively. This query requested information regarding any post-ACLR surgery and solicited patient-reported outcome (PRO) measures, including the International Knee Documentation Committee (IKDC) and Single Assessment Numeric Evaluation (SANE) scores. Patients with an age at surgery that fell outside the age range of the revision cohort at the time of the primary ACLR were excluded, as were patients who reported having undergone a subsequent revision ACLR or had known graft rupture.
After excluding patients based on study criteria, 153 revision and 144 control patients were included in the data analyses. A flowchart detailing inclusion and exclusion for both groups is presented in Figure 1.

Flowchart demonstrating selection of patients and exclusion criteria. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; MRI, magnetic resonance imaging.
Patients in both study cohorts underwent imaging in the injured state (ie, after native ACL tear in the control group and after graft failure in the revision group). Because many patients in the revision group had undergone their initial surgery at an outside institution, we did not have access to MRI scans after the original ACL tear in every patient. For this reason, we relied on the assumption that imaging parameters for the revision group were the same after graft failure as they would have been after tear of the native ACL. To test this assumption, we compared TT-TG distance on scans obtained after the original injury and after ACL graft tear in all patients for whom they were available (roughly two-thirds of the revision group). Patients with unusable or unavailable MRI scans were excluded.
Data Collection
Descriptive data as collected by EHR review included birth date, sex, height, and weight at the time of initial ACLR; BMI was calculated from these data. Operative and clinical notes were reviewed for the presence of varus or valgus instability, patellar instability, and posterolateral laxity to avoid including patients with concomitant sources of instability. Surgical side, graft type, history of prior ipsilateral knee surgery, and presence of concomitant meniscal tear at the time of primary ACLR were also collected from operative and preoperative notes.
To minimize the possibility that differential activity by control versus revision groups after primary ACLR might confound examination of our primary outcome variables, Tegner activity scores were estimated from activity documented in the EHR as a potential predictor of graft failure in regression analyses. 35 All assessments were performed by a single investigator (K.J.C.). For patients who underwent revision, we estimated a Tegner score based on patient self-reported activity after primary ACLR, just before ACLR graft injury. Patients in the control cohort received estimated Tegner scores based on self-reported activity documented at their final follow-up clinic visit after ACLR (range, 6-24 months postoperatively). A small number of control patients who responded to PRO surveys but did not attend follow-up evaluation at a minimum of 6 months postoperatively did not receive an estimated Tegner score.
MRI Measurements
Preoperative MRI scans were evaluated for all 297 study participants, using T1-weighted and proton-density sequences to measure study parameters, as described below. Measurements were made by a single investigator (E.J.L.), an orthopaedic surgery resident trained by musculoskeletal radiologists to follow a standardized protocol for each measurement. TT-TG distance, tibial slopes in coronal and sagittal planes, and medial tibial plateau concavity were measured for all included patients. 13 For the revision group, the MRI scans obtained closest to but before the date of revision surgery were analyzed, and measurements included assessment of tunnel position from the primary surgery. In addition, 28 participants (19%) from the control group underwent a postoperative MRI scan for indications other than graft failure; these scans were also analyzed for tunnel position and used in sensitivity analyses as described in the Statistical Analysis section. 12,13 All measurements were made using annotation tools on the digital picture archiving and communication system (Change Healthcare Radiology Solutions 14.0; Change Healthcare). MRI scans were obtained with the knee at or near full extension, and for all scans obtained within our health care system, a standardized protocol was used.
For all scans, the longitudinal tibial axis was assessed in the sagittal plane, determining the midpoint between the anterior and posterior cortices at 2 points; one point was at the distal-most portion of the image and the other was 4 to 5 cm proximal. A line connecting the midpoints was deemed the longitudinal axis of the tibia in the sagittal plane. 13 TT-TG distance was measured on axial MRI scans using a method similar to that of Schöttle et al 30 (Figure 2). Medial and lateral PTSs and the coronal tibial slope were measured using sagittal and coronal MRI scans according to the method of Matsuda et al 22 (Figure 3). Depth of concavity of the medial tibial plateau was measured as follows: the center of the medial plateau in the sagittal plane was identified; a perpendicular distance was measured from the medial plateau slope line to the deepest part of the medial plateau concavity.
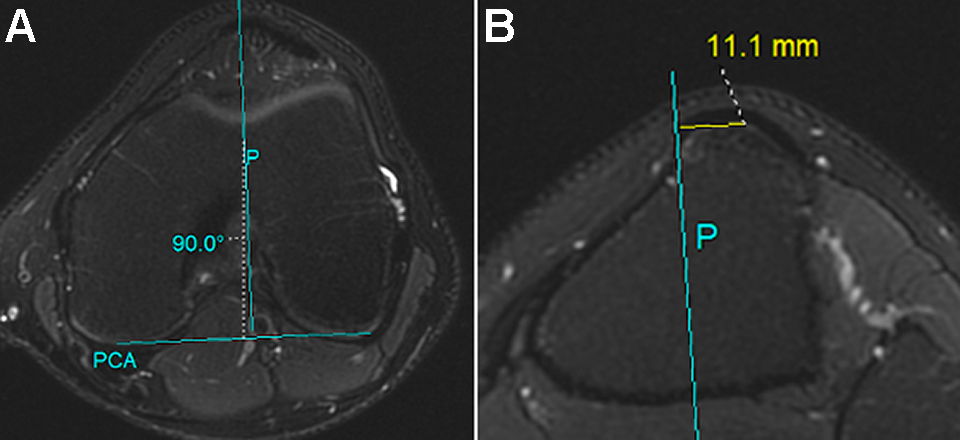
Method for determining tibial tubercle–trochlear groove (TT-TG) distance. (A) The axial slice containing the deepest trochlear groove was identified, and a line was drawn denoting the posterior femoral condylar axis (PCA). A perpendicular line was drawn that bisected the deepest portion of the trochlear groove (line P). (B) The tibial tubercle was located at the level in which the patellar tendon was still in contact with the tubercle. The perpendicular distance from line P to the midpoint of the patellar tendon insertion was calculated to determine the TT-TG distance.

Method for determining tibial plateau slope in the (A) coronal and (B) sagittal planes. (A) On the coronal image, the axial plane through the top of the tibial plateau was drawn (line ML). The diaphyseal axis of the tibial shaft was identified by drawing a line down the middle of the shaft (line A). A Cobb angle measurement was used to determine the slope between the lateral (L) and medial (M) peaks of the tibial plateau. If the medial peak of the plateau was distal to the lateral peak, the angle measurement was denoted as positive. If the medial peak was proximal to the lateral peak, the angle measurement was negative. (B) On an axial cut of the tibial plateau, the midline of the medial (M) and lateral (L) plateaus was identified, and the sagittal images corresponding to these cuts were used for slope measurements: (C) corresponding image for the medial cut and (D) corresponding image for the lateral cut.
For the revision cohort and the 28 control patients with postoperative MRI scans, tunnel position and graft slope were assessed in both sagittal and coronal planes, as outlined by Grassi et al. 12 To measure the position of the tibial tunnels, the sagittal slice where the tibial tunnel entrance was best visualized was identified. The diaphyseal axis of the tibia was drawn on this slice, and a line perpendicular to the diaphyseal axis was drawn between the anterior and posterior margins of the tibia to measure the anteroposterior (AP) distance. The midpoint of the tibial tunnel was calculated as the midpoint between the anterior and posterior tunnel borders. The distance between the anterior and posterior borders of the tibial tunnel and the anterior margin of the tibia was also measured. These values were also calculated as a percentage distance along the AP diameter of the proximal tibia based on total AP distance.
To measure coronal graft inclination, the proximal tibial plateau axis was determined by joining a line between the medial and lateral peaks of the tibial plateau. The angle between the tibial plateau axis and the line which best defined the intra-articular portion of the graft was measured. The sagittal graft inclination was calculated by measuring the angle between a line perpendicular to the tibial-diaphyseal axis and a line that best defined the course of the intra-articular portion of the graft. In cases in which the ruptured fibers made graft orientation difficult to determine, the inclination angle was measured between the femoral and tibial tunnels at the location closest to the knee joint. In these scenarios, the center points of the femoral and tibial tunnels were chosen, and a line was drawn connecting them to represent the graft course.
Statistical Analysis
Standard descriptive statistics were used to describe the study population overall and by revision status. Means ± SDs are reported for continuous variables; frequencies, and percentages are reported for categorical variables. For the revision group, paired t tests were used to evaluate the difference between TT-TG distance measured on scans obtained at the time of initial ACL tear versus the time of ACL graft tear. Univariable logistic regression was used to identify associations with ACL graft failure, evaluating TT-TG distance, proximal tibial slopes, depth of medial plateau, age, sex, BMI, graft type, the presence of concomitant meniscal tears, and Tegner activity score as predictors of ACL graft failure. A multivariable logistic regression model was built by including all variables with a univariable association P < .10. Odds ratios (ORs) with their 95% CIs were reported.
To account for poor tunnel placement as a predominant cause of ACL graft failure, a sensitivity analysis was performed using postoperative MRI scans from 28 control patients, at a mean of 3.1 ± 3.2 years after ACLR. MRI indications included reinjury (n = 19), new onset of pain and/or mechanical symptoms (n = 8), and persistent pain (n = 1). Results from these scans demonstrated meniscal tears (n = 16), isolated chondral injury (n = 3), loose body (n = 1), or cyclops lesion (n = 2). Eleven patients subsequently underwent arthroscopy for meniscectomy (n = 9) or cyclops debridement (n = 2). ACL grafts for these patients were visibly intact, and examination under anesthesia demonstrated negative Lachman and pivot-shift tests. None of the 28 controls had gross tunnel malposition by radiographic criteria. 12,15,32 These MRI scans were used to represent “ideal” tunnel position for comparison with the revision cohort. Tibial tunnel position, coronal graft inclination, and sagittal graft inclination were measured, yielding mean and cutoff values for 1, 2, and 3 SDs from the mean. Patients who underwent revision were divided into subgroups based on these standard deviation ranges, creating 3 groups with varying levels of homogeneity in tunnel placement (1 SD, 2 SDs, 3 SDs). Patients were only included in a group if all MRI measurements fell within the group range. Multivariable logistic regression models were built to evaluate associations between ACLR graft failure and the predictors of interest in each revision standard deviation subgroup, where the subgroup of 1 SD within the mean tibial tunnel position of the controls provided the most homogeneity between comparison groups, followed by 2 SD and 3 SD, respectively.
To evaluate interrater agreement, Tegner scores were assigned by a second investigator (T.A.S.) in a randomly selected group of 30 patients, and weighted kappa (κ) values for the ordinal data were calculated. Agreement was interpreted as none to slight (0.01-0.20), fair (0.21-0.40), moderate (0.41-0.60), substantial (0.61-0.80), or almost perfect (0.81-1.00). 20 Similarly, MRI measurements for a randomly selected sample of 30 scans were repeated by the same investigator who performed the other measurements, and intraclass correlation coefficients (ICCs) were calculated to assess intrarater agreement.
Results
The descriptive and clinical characteristics of the 2 study populations are shown in Table 1. Allograft was used for primary ACLR in 5 control patients (3%) and 17 revision patients (11%). For the revision cohort, the time from primary to revision ACLR was 4.0 ± 4.7 years (range, 0.4-24.8 years). The final follow-up was obtained from the control cohort at 8.1 ± 2.5 years (range, 2.4-13.9 years) after ACLR, yielding mean IKDC and SANE scores of 87.8 ± 13.2 and 90.0 ± 11.8, respectively. For the subset of 28 control participants with postoperative MRI scans used for sensitivity analyses, the IKDC and SANE score means were 85.0 ± 15.1 and 86.7 ± 15.8, respectively, at a mean follow-up of 8.3 ± 1.9 years after ACLR. Tegner scores were not included for 4 patients who underwent revision with no activity records at the time of graft reinjury or for 10 control patients with <6 months of clinical follow-up after primary ACLR. For 99 patients (65%) in the revision cohort, scans were available at the time of both primary ACL tear and graft tear; the mean difference in TT-TG distance for paired scans was 0.02 ± 2.90 mm (P = .96; paired t test).
Descriptive and Clinical Characteristics of Study Population by Revision Status and Overall a

a Data are reported as mean ± SD or n (%) except for Tegner score, which is reported as median [IQR]. ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; TT-TG, tibial tubercle–trochlear groove.
b n = 149; 4 patients who underwent revision had no record of athletic participation at the time of graft reinjury.
c n = 134; 10 control patients had <6 months of clinical follow-up after primary ACLR.
d n = 283.
Univariable and multivariable logistic regression results are depicted in Table 2. By univariable analysis, TT-TG distance, lateral PTS, Tegner score, and allograft were all positively associated with ACL revision (P < .001 to P < .02), while age at surgery was negatively associated with revision surgery (P < .001). By multivariable analysis, adjusting for all other variables in the model, increased TT-TG distance and lateral PTS remained positively associated with ACL revision (OR, 1.15; 95% CI, 1.07-1.23; P < .001, and OR, 1.13; 95% CI, 1.04-1.22; P = .004, respectively). Allograft use was also associated with increased odds of ACL revision (OR, 7.14; 95% CI, 2.10-24.25; P = .002), while increased age was associated with decreased odds of revision surgery (OR, 0.90; 95% CI, 0.87-0.94; P < .001). No association was identified between ACL revision and Tegner score by multivariable analyses.
Univariable and Multivariable Logistic Regression (n = 282) a
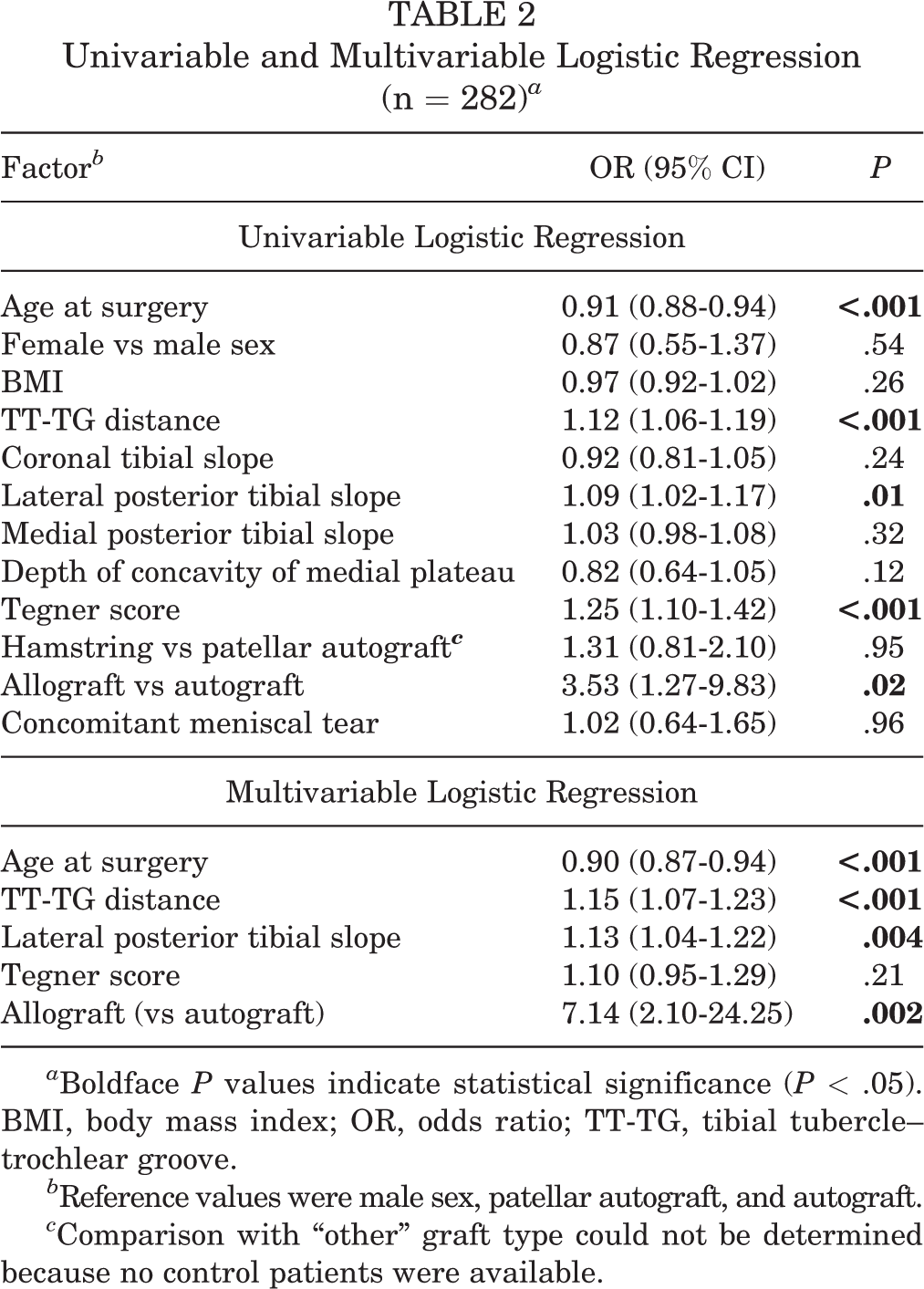
a Boldface P values indicate statistical significance (P < .05). BMI, body mass index; OR, odds ratio; TT-TG, tibial tubercle–trochlear groove.
b Reference values were male sex, patellar autograft, and autograft.
c Comparison with “other” graft type could not be determined because no control patients were available.
The results of sensitivity analyses, evaluating predictors of ACL revision in participants with tunnel position within 1 SD, 2 SD, and 3 SD of the 28 intact control participants, are shown in Table 3. Even under the most restrictive setting (SD = 1), age, TT-TG distance, and lateral PTS remained significantly associated with ACL revision because of graft failure in a multivariable model (Table 4).
Sensitivity Analysis a

a Boldface P values indicate statistical significance (P < .05). BMI, body mass index; OR, odds ratio; TT-TG, tibial tubercle–trochlear groove.
b Reference values were male sex, patellar autograft, and autograft.
Sensitivity Analysis: Multivariable Model for Most Restrictive Cases (SD = 1) a
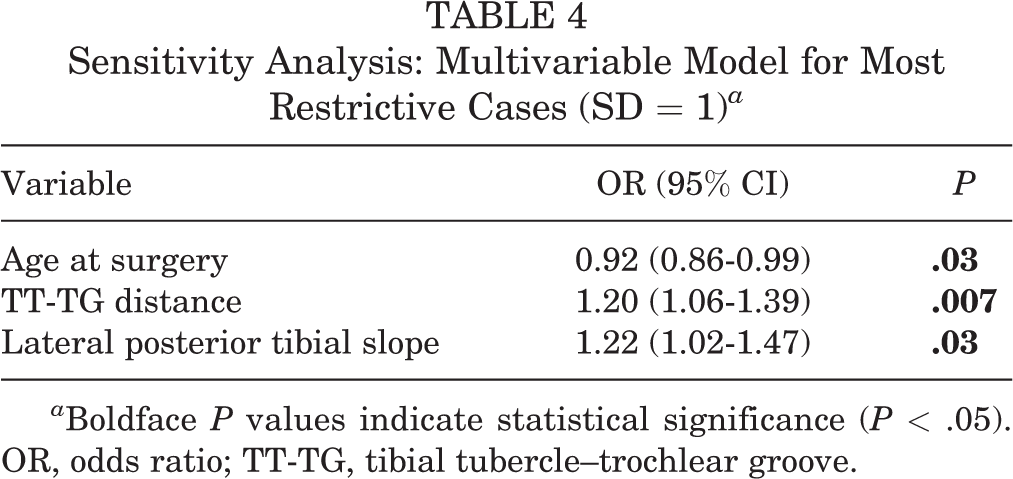
a Boldface P values indicate statistical significance (P < .05). OR, odds ratio; TT-TG, tibial tubercle–trochlear groove.
The interrater reliability for Tegner score estimation was considered substantial (κ = 0.73; Z = 4.15; P < .001). The intrarater agreement for the MRI measurements was high for TT-TG distance (ICC, 0.992; 95% CI, 0.983-0.996; P < .001) as well as lateral tibial slope (ICC, 0.984; 95% CI, 0.966-0.992; P < .001).
Discussion
In this relatively large, single-institution study, increased TT-TG distance was independently associated with ACL graft failure. After adjusting for the effects of age, lateral PTS, allograft use, and activity score, the odds of ACL graft failure increased by 15% for each 1-mm increase in TT-TG distance. As shown in previous studies, lateral PTS was independently associated with risk of ACL graft failure, with likelihood of failure increasing 13% for each increase in degree of posterior slope. 5,25,33 Risk of graft failure was also independently associated with age, as noted by prior authors, decreasing 10% for each year of increased age at the time of primary ACLR. 1,16 Sensitivity analyses, incorporating the most stringent definition of appropriate tunnel placement, substantiate these findings.
Our results are the first published report identifying an association between ACL graft failure and TT-TG distance. We surmise that increased dynamic rotational forces at the knee, represented by greater TT-TG distance, predispose one to ACL graft injury. The results of our paired scan analysis for the revision cohort did not identify a difference in TT-TG distance at the time of the initial injury versus the revision surgery, indicating that TT-TG distance was greater in the revision cohort at least since the initial injury. Because we did not have pre-ACL tear measurements, we could not determine whether greater TT-TG distance represents an anatomic, pre-existing, pre-ACL tear condition for the patients in the revision group, or whether greater TT-TG distance is a result of the initial injury. It is certainly possible that additional soft tissue injury at the time of the initial ACL tear may account for the increased TT-TG distance observed in the revision cohort.
Prior studies have demonstrated an increased TT-TG distance in ACL-injured knees and equalization of preoperative side-to-side differences in TT-TG distance after ACLR. 19,28,29 Furthermore, Polat et al 28 identified an even greater TT-TG distance in combined ACL and ALL injuries than in isolated ACL injury. The latter finding is surprising, as ALL injury is associated with increased anterolateral rotary instability and TT-TG distance would be expected to increase with anteromedial rotary instability. 25 Polat et al 28 suggested that although a combined ACL and ALL injury would theoretically increase internal rotation of the tibia, combined injury produces a “more rotationally unstable” knee and leads “to an increase in TT-TG distance in knee MRs taken in full extension and a neutral position.” Our measurements, all obtained in the injured state, identify increased TT-TG distance in a group with ACL graft failure versus a group that ultimately maintained long-term graft integrity. Whether this is because of an underlying predisposition to ACL tears via rotational offset or additional soft tissue injury at the time of the primary ACL tear, our results identify increased TT-TG distance as a marker for risk of ACL graft failure.
As prior studies have shown, we also identified an independent relationship between increased lateral PTS and increased risk of ACL graft failure. 5,34 In a case-control study of 152 skeletally immature knees, lateral PTS was significantly increased in ACL-injured patients compared with controls. 7 Additionally, several studies have investigated the relationship between PTS and subsequent injury after ACLR surgery. In a series of 330 patients after primary ACLR, Napier et al 26 identified a significant relationship between increased lateral PTS and recurrent ACL injury. Similarly, Christensen et al 5 found that increased lateral PTS was a risk factor for early graft failure after ACLR, regardless of graft type. In our study, the risk of ACL graft failure increased 13% for each degree increase in lateral PTS, regardless of TT-TG distance, age at surgery, allograft, and activity score. These results add to the growing body of literature implicating PTS as a risk factor for ACL graft failure.
Age at surgery was inversely related to likelihood of graft failure in our cohort, with the odds of failure decreasing 10% for each 1-year increase in age. This independent relationship was supported by sensitivity analyses, identifying age as a significant predictor of graft failure even under the most stringent exclusion of patients with malpositioned tunnels. A younger age at index surgery is a known risk factor for revision, as evidenced by a number of large studies. 1,16 Risk may be related to increased activity levels in younger patients, higher involvement in pivoting sports, and patient noncompliance with rehabilitation. Notably, a study by King et al 17 examined risk factors for ACL graft rupture exclusively in athletes who played high-intensity, high-risk sports. In this select patient population, there was only a weak correlation between younger age and ACL reinjuries, suggesting that other factors (eg, graft type) may also play a role in this relationship. We attempted to account for the likelihood that young age and increased activity are correlated and interrelated with regard to the prediction of ACLR failure. Although calculated Tegner activity scores were associated with ACL graft failure by univariable analysis, this relationship was not significant in multivariable analysis. It is possible, as surmised by King et al, that the predictive value of activity was bound up in age and thus not significant in multivariable analyses. The lack of significance may also be related to our methodology, which included retrospective calculation of Tegner scores.
Tunnel malposition is implicated as a leading cause of graft failure. Depending on placement, improper tunnel positioning can cause excessive strain in flexion or extension and resultant graft elongation. 10 In our revision cohort, 75 patients (49%) had tunnels that fell ≥2 SDs outside of the ideal position as determined by our subset of 28 control patients with intact grafts. This number is similar to that reported by the large Multicenter ACL Revision Study, which cited femoral tunnel malposition as the specific technical cause of failure in almost 50% of revision cases. 21,22 The results of our sensitivity analysis, sequentially removing individuals with poor tunnel placement, and evaluating those with ideal tunnel placement provide added confidence that TT-TG distance, lateral tibial slope, and young age are all associated with ACL graft failure independent of tunnel placement.
Acknowledging the role of structural anatomy in predicting ACL graft failure may have important implications for patient outcomes. Specific alterations in surgical technique or rehabilitation might allow better graft survival despite these predisposing anatomic factors. For example, patients with increased TT-TG distance, potentially signifying increased rotatory instability, may benefit from the addition of extra-articular augmentation at the time of ACLR. Further investigations should evaluate whether extra-articular augmentation, osteotomy, or delayed rehabilitation programs and/or slower return to sport could mitigate the predisposition to graft failure in this cohort.
Limitations
This study was limited by its retrospective nature, dependent on information available in the EHR. For example, Tegner scores were estimated based on patient-reported activity in the EHR and are likely less accurate than if a survey had been completed directly by the patient. It is likely that anatomic factors are even more important in predicting noncontact or atraumatic tears, but our cohort includes both contact and noncontact injuries, and we could not distinguish between them. We note that the primary surgery in our revision group was done by many different surgeons at many sites. Aside from MRI assessment of tunnel placement, we could not assess the potential role of other technical factors. Furthermore, it is possible that tunnels in our subset of patients with intact grafts were imperfect and did not represent the best standard for tunnel position, as we assumed for sensitivity analyses. The mere existence of the postoperative MRI scans, ordered to evaluate the cause of pain and/or outcome of reinjury, suggests the possibility of selection bias, although this is mitigated by the fact that these grafts were actually intact.
It has been shown that TT-TG distance varies based on patient positioning in the scanner. 9 The majority of scans were performed within our health care system, using reproducible methods including a specific knee coil, thus standardizing the position during scanning. The large number of patients in our study also diminishes the likelihood that scanner positioning affected imaging measurements. Furthermore, all patients were evaluated using MRI, avoiding potential for error that would be introduced by intermingling computed tomography and MRI modalities. 14 All MRI measurements were made by a single, trained observer, and although we identified high intrarater reliability (ICC, 0.992), we did not assess accuracy using interrater comparisons to other observers. The standard error of measurement (SEM), calculating using the ICC (SEM = SD × √[1 – ICC]) to estimate the expected variation in the measurement, was 0.37 mm, considerably smaller than the observed difference in mean TT-TG distance between cohorts (1.91 mm), indicating that observed differences between cohorts were not likely because of measurement error.
Finally, evidence that ACL grafts were intact in the control cohort was based on self-report and PRO measures. Although our control cohort reported mean IKDC scores (87.8 ± 13.2) at the final follow-up (8.1 ± 2.5 years) that are consistent with previous reports of IKDC scores in patients with functionally intact ACL grafts, it is possible that a portion of this cohort had an incompetent or failed graft. 2 However, even if some patients in the control cohort had failed grafts, incorrectly including these patients’ data in the control cohort would have diminished the noted differences between control and revision cohorts, not magnified it.
Conclusion
Increased TT-TG distance, increased lateral PTS, and young age at primary ACLR were independent risk factors for ACL graft failure even under the most restrictive criteria for ideal tunnel position. These findings suggest an important role for proximal tibial geometry and alignment in ACL graft survival. Future studies should explore the possibility that extra-articular augmentation, osteotomies, or other factors may mitigate the risk of ACL graft failure in the setting of increased TT-TG distance and/or increased PTS.
Footnotes
Final revision submitted January 28, 2023; accepted March 9, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.A.S. has received consulting fees from Medical Device Business Services. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Wisconsin–Madison (2015-0566-CR001).
