Abstract
Background:
There is a broad and diverse range of symptoms after a concussion, from irritability to nausea. This heterogeneity of symptoms is a challenge for clinicians managing the different presentations among injuries. Prior research has investigated the structure of postconcussive symptoms to determine if they can be grouped into clusters of related symptoms.
Purpose/Hypothesis:
The purpose of this study was to identify symptom clusters during the acute phase after a sports-related concussion using exploratory factor analysis and to understand the relationship between risk factors for postconcussion symptoms (ie, demographics, injury characteristics, mental health, and sleep qualities) and different symptom clusters. We hypothesized that certain factors would be predictive of specific symptom clusters.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
Collegiate athletes (N = 1104) from the Concussion, Assessment, Research, and Education (CARE) Consortium completed the Sport Concussion Assessment Tool–Third Edition symptom assessment tool 24 to 48 hours after concussion. Exploratory factor analysis was conducted on the symptom evaluation to determine symptom clusters 24 to 48 hours after concussion. Regression analysis was used to examine the effects of pre- and postinjury characteristics.
Results:
Exploratory factor analysis revealed a 4-cluster structure for acute postconcussive symptoms that explained 62% of the variance in symptom reporting: vestibular-cognitive, migrainous, cognitive fatigue, and affective. Delayed reporting, less sleep before assessment, female sex, and being hurt outside of competition (during practice/training) was correlated with increased symptoms for 4 symptom clusters. Depression predicted higher vestibular-cognitive and affective symptoms. Amnesia was correlated with higher vestibular-cognitive and migrainous symptoms, whereas migraine history was associated with more migrainous and affective symptoms.
Conclusion:
Symptoms can be grouped into 1 of 4 distinct clusters. Certain variables were associated with increased symptoms across multiple clusters and may be indicative of greater injury severity. Other factors (ie, migraine history, depression, amnesia) were associated with a more specific symptom presentation and may be mechanistically related to concussion outcomes and biological markers.
An estimated 1.6 to 3.8 million sports-related concussions occur each year, making sports a major contributor to the prevalence of traumatic brain injuries. 29 After concussion, symptoms span cognitive, physical, and affective domains, such as concentration difficulty, headaches, and anxiety. However, sports-related concussion symptoms vary considerably from patient to patient, presenting a challenge for clinicians managing the injury. There is growing recognition that treatment approaches based on an individual’s symptom cluster may be preferable to a “one size fits all” approach. For example, a patient presenting with primarily affective symptoms may warrant a different treatment plan than a patient presenting with primarily vestibular symptoms. 14,23,25,45 As such, it may be useful for clinicians to conceptualize symptoms into clusters, or groups of related symptoms that are often experienced together. These symptom clusters could be useful for understanding and assessing symptoms of concussion toward the goal of facilitating more targeted treatment.
The potential utility of a symptom cluster approach has led researchers to investigate the underlying structure of various concussion symptom checklists, such as the Post-concussion Symptom Scale, Head Injury Scale, Graded Symptom Checklist, and the Sport Concussion Assessment Tool (SCAT). 1,20,21,24,42 In a study aimed at determining the structure of the Head Injury Scale in National Collegiate Athletic Association (NCAA) athletes, confirmatory factor analysis (CFA) revealed that 3 symptom clusters demonstrated good fit for the assessment: migraines, mental energy/concentration, and affect/sleep. However, the sample that was used to test this model was relatively small (n = 33) and predominantly male (75%). 42 Another study used exploratory factor analysis (EFA) and CFA to examine the structure of the SCAT–Third Edition (SCAT-3) in high school and college athletes in the acute phase (<48 hours) after an sports-related concussion. The authors found that a bifactor model showed the best fit with 4 symptom clusters—emotional, torpor, sensory sensitivities, and headache—all loading onto a general second-order factor. 40 A follow-up validation study using data from the Concussion, Assessment, Research, and Education (CARE) Consortium assessed measurement invariance of symptom clusters throughout the recovery process. It stated that 7 symptom clusters (6 specific factors and 1 general bifactor) showed good model fit and that these symptom clusters were mostly comparable immediately and 24 to 48 hours after injury. 7 A similar structure of the SCAT–Fifth Edition was identified in adolescents. 1
While prior work has provided important insights into the structure of concussion symptoms, several limitations are important to note. Previous studies had small sample sizes, potentially producing a biased and overfit model. 15 Furthermore, inclusion of predominantly male samples limits the generalizability of these results. It has been reported that female athletes experience disproportionately higher rates of concussion-related symptoms, 9,20,24,48 underscoring the need to include women in studies of concussion outcomes. Another limitation is that few studies have examined primary risk factors (eg, medical history) or secondary ones (eg, amnesia) on symptom reporting. Knowing which risk factors might predispose an individual to a given symptom cluster could improve the specificity and timeliness of intervention. A final limitation is variability in the time from concussion to when the symptoms were assessed. As concussion symptoms change over time, it is possible that clusters identified in these studies were skewed owing to differences in time after concussion.
In the present multisite study involving the NCAA and the US Department of Defense (DoD), we sought to determine the underlying structure of the SCAT-3 symptom evaluation during the acute phase of a concussion (24-48 hours) in a large sample of male and female NCAA athletes and US service academy cadets from the CARE Consortium and to examine primary and secondary risk factors for concussion symptom clusters. We first conducted an EFA to examine the latent structure of symptom reporting in the acute phase of a concussion to identify symptom clusters. We subsequently examined the association between factors such as sex, concussion history, psychiatric history, and loss of consciousness (LOC) and symptom reporting within the symptom clusters. We hypothesized that certain risk factors would be predictive of specific symptom clusters. This prognostic approach has been shown to add clinical value to understanding concussion symptoms. 48
Methods
Participants
The NCAA-DoD Grand Alliance CARE Consortium is a multisite study involving 26 civilian academic universities and 4 US service academies aimed at studying concussions and subsequent recovery. 10 The study protocol was approved by the local institutional review board at each site and the US Army Medical Research and Materiel Command Human Research Protection Office. Participants provided informed consent before participation. All participants were enrolled between 2014 and 2017. All participants were involved in intercollegiate sports and/or were military cadets.
Participants completed an annual preseason assessment to collect demographic information and self-reported medical history, as well as baseline neurocognitive and neurologic function, balance, and concussion-related symptoms. After the identification of a concussion according to the guidelines of the Fourth International Conference on Concussion in Sport, 11,31 participants completed follow-up assessments within 6 hours of the injury, at 24 to 48 hours postinjury, when they began their return-to-play protocol, when they were cleared for unrestricted return to play, and at 6 months postinjury. Of note, each site’s medical staff were responsible for return-to-play decisions. After concussion, injury-specific information was collected, such as the presence of amnesia and the duration of LOC. The present study included only participants who sustained a concussion and completed the 24- to 48-hour SCAT-3 to assess concussion-related symptoms. 10 Concussions occurred during competition (n = 272), during practice/training (n = 667), or outside a sporting event (n = 165).
Data on 3322 concussions were initially available. Concussions were excluded if there was no 24- to 48-hour assessment (n = 570). To use the most up-to-date medical history, which is collected only during baseline visits, concussions were excluded if there was no corresponding baseline assessment during the same academic year as the sustained concussion (n = 465). As previous concussions may alter baseline reporting, only the first concussion during the study period was used when a participant reported >1 concussion. Subsequent concussions during the study period were removed from analyses (n = 227). Concussions were excluded if the participant indicated a history of any of the following health conditions: meningitis, seizure disorder, diabetes, autism spectrum disorder, bipolar disorder, schizophrenia, moderate or severe traumatic brain injury, brain surgery, stroke, Parkinson disease, or memory disorders such as Alzheimer disease (n = 87). Concussions were also excluded if the participant’s primary language was not English (n = 62) or the participant did not complete the SCAT-3 or Brief Symptom Inventory–18 (BSI-18) (n = 84). As recent concussions may affect symptom reporting, concussions were excluded if the participant had a concussion within the previous 6 months of the current one (n = 41). If participants were missing any of the predictors used in subsequent analyses, the concussion was removed from the sample (n = 682).
The most frequently missing variables were sleep the night before the assessment (n = 146) and immediate removal from play (n = 301). The final sample comprised data on 1104 concussions. Concussions that were included and excluded in these analyses did not show differences in SCAT-3 symptom severity score (P = .29) or number of SCAT-3 symptoms (P = .76). Figure 1 reports the final sample in these analyses.
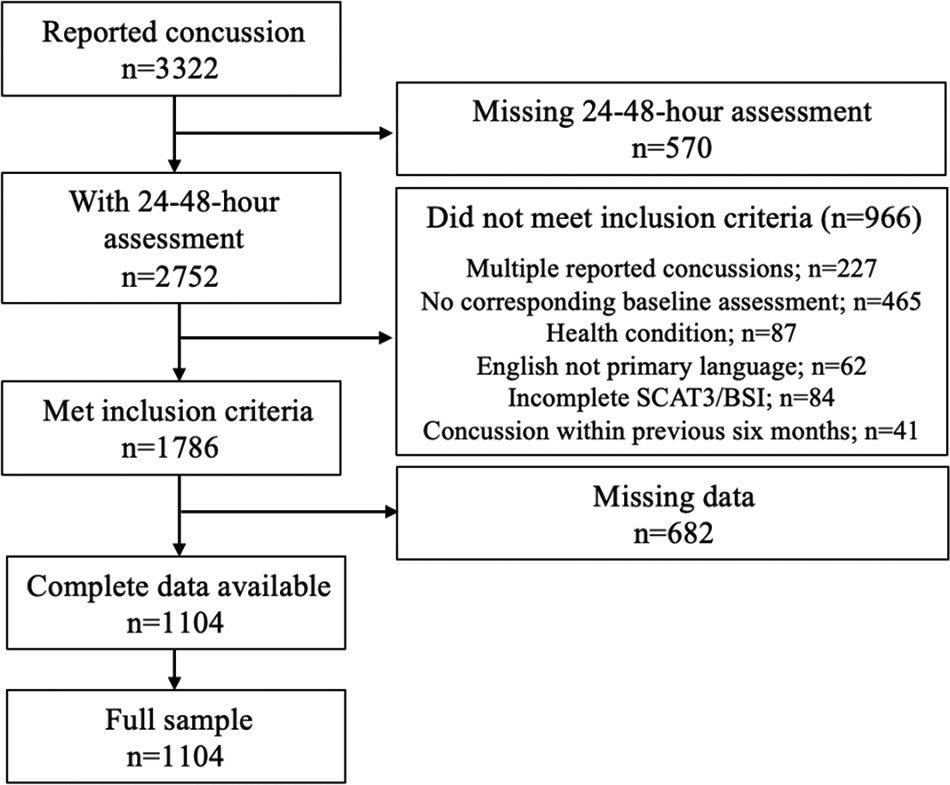
Participant inclusion and exclusion. BSI, Brief Symptom Inventory; SCAT3, Sport Concussion Assessment Tool–Third Edition.
Evaluation Measures
As part of the baseline preseason assessment, participants completed the Demographics and Personal and Family Medical History unique case report form. 10 This self-report assessment ascertains information about sport participation, concussion history, and personal and family medical history. Sport contact level was previously defined. 22 Contact sports were basketball, diving, field hockey, football, ice hockey, soccer, water polo, and wrestling. Limited-contact sports were baseball, beach volleyball, cross country/track, fencing, field events, gymnastics, softball, and volleyball. Noncontact sports were bowling, golf, rifle, rowing/crew, sailing, swimming, and tennis.
For the following analyses, history of migraine headaches or sleep disorders was each coded as a binary variable (yes/no). History of psychiatric disorders and depression was combined into a single binary variable (yes/no; originally queried separately). History of attention-deficit/hyperactivity disorder, special education classes, and learning disability was combined into a single binary variable (yes/no). Concussion history was recorded ordinally (0, 1, and ≥2), and 26 participants reported ≥2 prior concussions: 3 (n = 21), 4 (n = 3), and 6 (n = 2) concussions. During their baseline assessment, participants completed the BSI-18, 17 which includes a subscore for depression symptoms. Given the small sample size, race and ethnicity was recoded as White/non-Hispanic (n = 646), Black (n = 171), and other (n = 287). The final sample in these analyses is shown in Table 1.
After a concussion, injury-specific information was collected. These analyses used self-report data regarding LOC, amnesia, delayed reporting, and injury situation. LOC was coded as a binary variable (yes/no). Amnesia was coded as yes if participants had posttraumatic or retrograde amnesia. Delayed reporting was based on clinician report; clinicians were asked if the injury was reported immediately and if athletes were immediately removed from play. For these analyses, delayed reporting was coded as yes if clinicians responded no to either query. Information regarding time of delay was not collected and therefore not included. Injury situation consisted of competition, practice/training, or outside a sporting event.
Between 24 and 48 hours after an identified concussion, participants completed the SCAT-3. This evaluation is a 22-item self-report questionnaire that asks participants to rate symptom severity on a scale from 0 (symptom not present) to 6 (symptom is severe) and report the number of hours slept the night before the assessment. 19 Sleep can influence reporting of postconcussion symptoms, even during baseline testing. 47 As baseline-adjusted scores can lead to inflated measurement errors, particularly when using a measure with moderate test-retest reliability, 12 postinjury SCAT-3 symptom evaluation scores were used rather than baseline-adjusted scores.
Exploratory Factor Analysis
An EFA was conducted for the 22-item SCAT-3. A Kaiser-Meyer-Okin mean sampling adequacy >0.85 was confirmed before EFA. Additionally, we ensured that the case to item ratio was at least 20:1. A scree plot was used to inform the number of factors, or clusters, to include in the EFA. Clusters that occurred before the curve or elbow of the scree plot were included in analyses. To account for correlation between clusters, orthogonal varimax rotation and Kaiser normalization were employed. All clusters retained for later analyses had eigenvalues >1.0. For an item to be retained in a cluster, it had to demonstrate loading >0.5. If an item cross-loaded onto another cluster (>0.4), this item was retained in both clusters if the difference between the primary and secondary loading was >0.2 and the primary cluster loading was >0.6. This EFA did not yield any cross-loadings, and all items showed primary loading on a single cluster.
Statistical Analysis
Multivariate generalized linear models were used to examine risk factors for symptom reporting in each clinical symptom cluster identified by the EFA; the sum of severity scores for each item in the symptom cluster was used as the outcome variable. Primary risk factors (sex, race and ethnicity, BSI-18 depression subscore, concussion history, psychiatric diagnosis, migraine history, and learning disabilities) and secondary risk factors (LOC, amnesia, delayed reporting, sleep night before assessment, and injury situation) were entered as predictors within the same model. A Gaussian distribution with an identity link function, which assumes a normal distribution, and a Poisson distribution with a log link function, which assumes a distribution of integers, were considered. The distribution that had the best fit, as indicated by the lowest Akaike information criteria, was selected. A Gaussian distribution with an identity link function was shown to have the best fit for all models. All models were confirmed to meet assumptions of a linear regression. Models did not demonstrate heteroscedasticity, the residuals of the model were normally distributed, and the variables were not collinear. For these models, parameter estimates (β) represent model coefficients such that positive values indicate a positive relationship with symptom reporting and negative values indicate a negative relationship with symptom reporting. These estimates indicate the estimated change in symptom severity score for a 1-unit change in predictor. For categorical variables, this would indicate the estimated group difference in symptom severity scores. The Benjamini-Hochberg false discovery rate (FDR) procedure (q = 0.05) was used to control for FDR. Significance was defined a priori as p<0.05. All analyses were performed using R Statistical Software (v4.0.2; R Core Team).
Results
Participant Sample
The final sample consisted of 1104 participants who were diagnosed with a concussion and completed the SCAT-3 within 24 to 48 hours after concussion. The sample was 58.4% male and 58.5% White/non-Hispanic. Table 1 describes demographic characteristics of the final sample.
Demographic Information of the Participants a

a NCAA, National Collegiate Athletic Association.
The mean symptom severity scores, with skewness and kurtosis, are shown in Supplemental Table S1 (available separately). Symptom scores ranged from 0 to 6. Symptom severity scores ranged from 2.27 ± 1.54 (mean ± SD) for headaches to 0.48 ± 1.05 for nervous/anxious. Symptom scores were skewed with positive kurtosis.
Table 2 describes the distribution of primary and secondary risk factors indicated by participants for the whole sample. Most participants (62.8%) had no concussion history and were injured during practice/training (60.4%). More than half (57.0%) of participants delayed reporting their concussion.
Incidence of Primary and Secondary Risk Factors for Postconcussion Symptoms in the Participants a
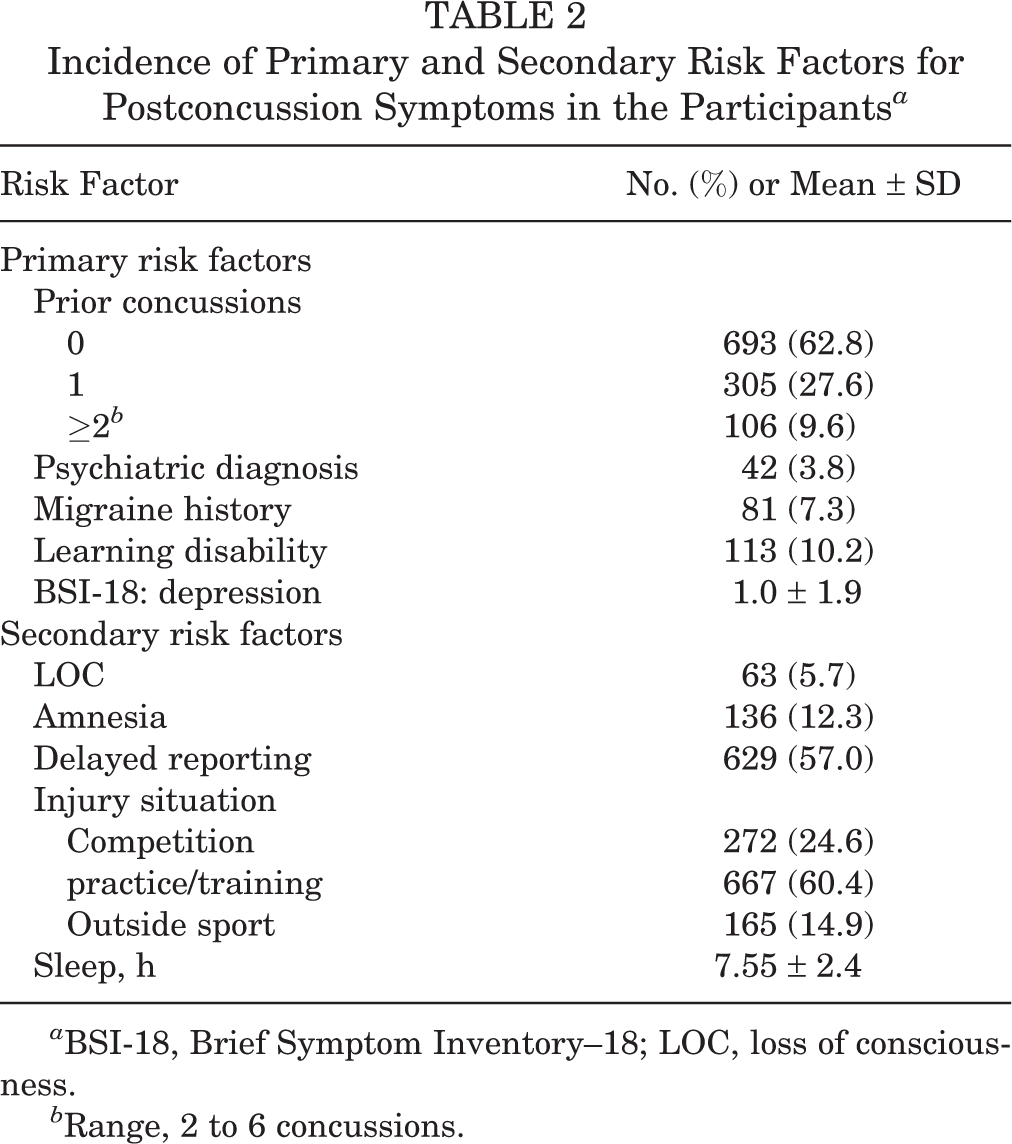
a BSI-18, Brief Symptom Inventory–18; LOC, loss of consciousness.
b Range, 2 to 6 concussions.
Exploratory Factor Analysis
The EFA demonstrated a Kaiser-Meyer-Olkin mean sampling adequacy of 0.96, suggesting that the data were well suited for factor analysis. The results supported a 4-factor, or 4-cluster, solution that accounted for 62% of the variance in symptom reporting. The structure and loadings for all 22 items are provided in Supplemental Table S2 and Figure S1. The clusters included 18 of the 22 SCAT-3 symptom evaluation items. Four items were eliminated owing to insufficient primary cluster loading (<0.5): neck pain, nausea/vomiting, difficulty concentrating, and trouble falling asleep. This may have been due to high reporting (neck pain, 46%), low reporting (nausea/vomiting, 30%), or similar loadings across multiple clusters leading to no primary loading (difficulty concentrating and trouble falling asleep). The 4 clusters were as follows: Cognitive fatigue: 5 items, 20% of variance, eigenvalue = 4.30 Migrainous: 4 items, 15% of variance, eigenvalue = 2.83 Vestibular-cognitive: 5 items, 14% of variance, eigenvalue = 3.01 Affective: 4 items, 13% of variance, eigenvalue = 2.83
The vestibular-cognitive cluster included dizziness, blurred vision, balance problems, difficulty remembering, and confusion. The migrainous cluster consisted of headache, “pressure in head,” sensitivity to light, and sensitivity to noise. The cognitive fatigue cluster comprised feeling slowed down, feeling like “in a fog,” “don’t feel right,” fatigue or low energy, and drowsiness. The affective cluster included feeling more emotional, irritability, sadness, and nervous or anxious. The same clusters were identified with principal component analysis. The loadings from principal component analysis with 4 clusters and a varimax rotation are available in Supplemental Table S3.
After identification of significant clusters, cluster scores for each were generated by summing the severity scores from each item in the cluster. These cluster scores were used for subsequent analyses.
Factors Increasing the Risk for Global Symptom Severity
Several risk factors were associated with higher symptom scores across all clusters (Table 3). Female sex was associated with increased symptom reporting in all 4 clusters: migrainous (β = 2.43), cognitive fatigue (β = 2.05), affective (β = 1.35), and vestibular-cognitive (β = 1.17).
Results From Generalized Linear Models a

a Bold P values indicate statistical significance (P < .05). Reported effect sizes are Cohen d values; dashes indicate not applicable. BSI-18, Brief Symptom Inventory–18; LOC, loss of consciousness.
bR 2 = 0.07, adjusted R 2 = 0.06, P < .001.
cR 2 = 0.12, adjusted R 2 = 0.11, P < .001.
dR 2 = 0.07, adjusted R 2 = 0.06, P < .001.
eR 2 = 0.09, adjusted R 2 = 0.08, P < .001.
f Reference group = male sex.
g Reference group = injuries occurring during a sporting game.
h Reference group = White/non-Hispanic.
i Reference group = no prior concussion.
In addition, sleep was associated with increased symptom reporting across all 4 clusters such that less sleep before the assessment was associated with greater symptom severity: cognitive fatigue (β = –0.39), affective (β = –0.28), migrainous (β = –0.26), and vestibular-cognitive (β = –0.24). Delayed concussion reporting was associated with increased symptom reporting on all 4 clusters: cognitive fatigue (β = 1.86), migrainous (β = 1.41), vestibular-cognitive (β = 1.30), and affective (β = 0.97). Finally, injuries occurring during practice/training or outside a sporting context were associated with increased symptom reporting in the migrainous cluster (β = 1.71 and 1.46, respectively), the cognitive fatigue cluster (β = 1.58, practice/training only), the vestibular-cognitive cluster (β = 0.89, practice/training only), and the affective cluster (β = 0.69, practice/training only).
All nominally significant results survived the FDR procedure.
Factors Associated With Specific Symptom Clusters
A subset of risk factors examined in this study was associated with more specific symptom clusters (Table 3) rather than an increase in symptoms broadly. Migraine history was associated with increased symptom reporting in the migrainous cluster (β = 1.19) and the affective cluster (β = 1.07). BSI-18 depression subscore was associated with increased symptom reporting in the vestibular-cognitive cluster (β = 0.12) and the affective cluster (β = 0.10). Amnesia was associated with increased symptom reporting in the migrainous cluster (β = 1.47) and the vestibular-cognitive cluster (β = 1.18). Last, other racial and ethnic minority individuals reported fewer symptoms in the cognitive fatigue cluster than their White/non-Hispanic counterparts (β = –1.12)
All nominally significant results survived FDR procedure. LOC, concussion history, psychiatric history, and learning disability were not associated with scores in any symptom cluster.
Discussion
This study examined primary and secondary risk factors associated with clusters of acute concussion symptoms in a large sample of collegiate athletes. There were 3 main findings. First, results supported a 4-cluster structure for SCAT-3 symptom evaluation: cognitive fatigue, migrainous, vestibular-cognitive, and affective symptoms. This model accounted for 62% of the variance in symptom reporting. Second, several risk factors were associated higher symptom reporting across all or most clusters—female sex, delayed concussion reporting, hours of sleep the night before assessment, and being hurt in practice/training or outside a sporting context. Third, risk factors such as migraine history and depression were uniquely associated with certain symptom clusters (ie, migrainous and vestibular-cognitive clusters, respectively). Identification of such risk factors may help inform specific treatment options after concussion.
Exploratory Factor Analysis
The factor analytic structure presented here revealed 4 clusters (in order of overall variance explained): (1) cognitive fatigue, with items related to low energy; (2) migrainous, with items associated with head pain/pressure and sensitivity; (3) vestibular-cognitive, with items associated with vestibular disturbance and confusion; (4) and affective, with items related to mood. These results provide converging evidence for the structure of concussion symptoms in male and female collegiate athletes. The differences in structure noted between the current study and previously published work are likely attributable to the various measurement tools having nonoverlapping or dissimilar symptom items, ∥ sample size differences, 40 inclusion of different age groups, 40 or use of a less condensed factor structure. 7
Global Risk
Several risk factors were associated with higher symptom scores across all clusters, suggesting that they may be related to injury severity. These risk factors were female sex, sleep the night before the assessment, delayed reporting, and injury situation.
Sex-Specific Effects
Female sex was associated with increased symptom reporting across all clusters (β = 1.17-2.43), supporting prior work demonstrating that female athletes report more symptoms than their male counterparts in at least 1 symptom cluster after concussion. 20,24,30,48 Several mechanisms have been proposed to explain greater symptom reporting: more severe injuries resulting from weaker head and neck stability, lower biomechanical thresholds for concussion, and different circulating hormones (eg, estrogen affecting pain perception and response in women). 37 However, female athletes may have greater baseline symptoms, and at least 1 review study noted that the apparent increase in symptoms among women after concussion may not be clinically significant. 8 They may also be more accurate when reporting symptoms. Thus, the mixed findings underscore the need for considering sex effects in future work.
Sleep
Less sleep before administration of the SCAT-3 was associated with increased symptom reporting in all clusters (β = –0.24 to –0.39). This is in support of growing evidence that fewer hours of sleep is associated with greater baseline symptoms, 36,47 longer recovery, 6 and more symptoms after concussion. 6,26,39 While the mechanisms influencing this relationship are likely complex, our results contribute important converging evidence on the importance of increasing sleep to reduce symptoms after a concussion.
Injury Characteristics
Delayed reporting of injury, defined as not reporting injury immediately or not being removed from play, was associated with higher symptom scores across all clusters (β = 0.97-1.86). This supports prior research associating delayed reporting with increased symptoms after injury. 3 After concussion, the brain experiences acute changes in metabolic pathways, such as an increase in free radicals and additional metabolic demands. This leads to an increase in neural vulnerability to further damage. 18 Increased vulnerability coupled with further metabolic demands and potential subsequent impacts if the athlete is not immediately removed from play may lead to slower recovery and additional symptoms.
Results from the regression analysis revealed that injuries occurring during practice/training and outside a sporting context were associated with increased symptom reporting in the migrainous cluster (β = 1.71 and 1.46, respectively), the cognitive fatigue cluster (β = 1.58, practice/training only), the vestibular-cognitive cluster (β = 0.89, practice/training only), and the affective cluster (β = 0.69, practice/training only). It is possible that injuries occurring outside a sporting context are not immediately recognized and reported, are the result of higher-impact magnitudes (ie, motor vehicle crash), and are subject to the same mechanisms as delayed reporting of injury. The presence of medical personnel may aid a quicker removal from play. This is supported by a recent study on military cadets, which found that those hurt during practice/training were more likely to delay concussion reporting than those hurt in a competition setting. 5 Of note, symptom structure was comparable when considering only sport-related concussions. Taken together, these results underscore the importance for prompt concussion reporting and removal from play, regardless of injury situation, to mitigate additional concussion symptoms.
Specific Risk Factors
A subset of risk factors examined in this study was associated with more specific symptom clusters rather than an increase in symptoms broadly.
Migraine History
Those reporting a history of migraines had higher affective (β = 1.07) and migrainous (β = 1.19) symptoms. There is limited research investigating the relationship between migraine history and postconcussive affective symptoms. One study found that preinjury migraines or headaches were associated with cognitive, sleep, and emotional symptoms 3 months after injury 49 ; emotional and somatic symptoms were still tied to migraines 6 months after injury. 13 Together, it points to migraine history as an important risk factor for the development and maintenance of affective symptoms in the acute phase of a concussion.
The relationship between migraine history and migrainous symptoms is difficult to parse, as symptoms may be either an exacerbation or a recapitulation of prior migrainous symptoms. Current research suggests that posttraumatic headache and chronic migraines may share overlapping mechanisms, specifically activation of trigeminal nociception. 46 Migrainous symptoms may also be exacerbated by a number of factors associated with head injury, such as physical exertion or neck strain, as well as unrelated factors such as sleep disturbances, affective changes, and dehydration. 16,38,46 These relationships are further complicated when considering that headaches are the most commonly reported concussion symptom, with >93% of athletes reporting headaches at some point after injury. 32 However, our findings echo prior research that has identified a history of migraines as a risk factor for greater symptom number and severity after concussion. 44
Depression
Depression severity as measured through the BSI-18 depression subscore was associated with increased symptom scores in the vestibular-cognitive cluster (β = 0.12) and the affective clusters (β = 0.10). Prior research has shown that a diagnosis of depression is associated with a greater number of endorsed symptoms after mild traumatic brain injury. 28 The results presented here extend this work and suggest that depression may be more intricately tied to certain symptom clusters. While postconcussive affective symptoms may be a recapitulation of existing internalizing symptoms, the relationship between depressive and affective symptoms still has important clinical implications.
Amnesia
Our results indicated that amnesia, either posttraumatic or retrograde, was associated with higher symptoms scores in the vestibular-cognitive (β = 1.18) and migrainous (β = 1.47) clusters. However, prior work investigating the relationship between amnesia and postinjury outcomes showed mixed results. 4,33 The vestibular-cognitive cluster here consists of symptoms such as dizziness, blurred vision, balance problems, difficulty remembering, and confusion. Given the conceptual similarities between amnesia and the vestibular-cognitive cluster identified, it may be that similar mechanisms underlie the two. Injury-driven mechanisms resulting in on-field amnesia may continue to exert effects in the acute phase of a concussion, leading to increased symptom reporting in this cluster.
Research investigating the relationship with amnesia and migrainous symptoms is limited. One study found that posttraumatic amnesia was not associated with any symptom clusters 6 months after injury. 13 While more research is needed, this study and the results presented here provide converging evidence suggesting that amnesia may be a specific indicator of subsequent migrainous symptoms.
Clinical Implications
The results presented here have important clinical implications. First, identifying migraine history as a risk factor for migrainous and affective symptoms can help inform delivery of targeted treatments such as psychotherapy, psychiatric medications, or migraine-specific treatments (ie, triptans or calcitonin gene-related peptide medications). 1,5,28,45 This could mitigate additional postconcussion symptom burden in those with a history of migraines. These results also identified amnesia as a risk factor for certain symptom clusters. Such information can help clinicians take a proactive approach to targeted injury management, especially in athletes who may have delayed symptom presentation. 41 Such risk factors can be easily assessed on the field and improve the specificity of treatment delivery. Providing appropriately specific treatment can further help reduce the deployment of unnecessary treatment, increasing the availability and cost-effectiveness of efficacious treatment options. Last, these results identified a number of risk factors that were associated with increased symptoms more broadly. Clinical consideration of these risk factors could help mitigate symptom burden after concussion. While further research is needed to understand the improvements to be gained by targeted treatment approaches, the relationships identified here provide important groundwork for this research.
Limitations
Several limitations of these analyses are worth noting. First, participants in this sample were all NCAA collegiate athletes or US service academy cadets, with the largest portion of NCAA athletes being football players. This may limit generalizability of these findings to older, younger, or less physically active populations. These results may also not generalize to symptom clusters in individuals who experience persisting symptoms: each symptom cluster explained relative low variance (13%-20%), and persistent symptom clusters may be more siloed and thus explain more variance. Additionally, the proportion of individuals reporting certain risk factors, while sufficient for analyses, is relatively low (ie, 3.8% reporting history of psychiatric diagnosis). The sample was also limited by the relatively large number of individuals excluded owing to missing data (n = 682) and exclusion criteria (n = 966). As the relationship between risk factors and symptom reporting is complex, it is difficult to parse the directionality of these relationships. For example, those with migraine history may have exacerbated migrainous symptoms, or they may be reporting increased migrainous symptoms independent of concussion. A more deeply characterized baseline profile would be needed to better clarify these relationships. However, given that symptoms may have critical implications for recovery, results described here point to important considerations for symptom presentation, which may inform subsequent treatment.
Conclusion
Several conclusions can be drawn from the results presented here. First, postconcussion symptoms can be grouped into 1 of 4 distinct clusters. Second, certain variables were associated with increased symptom scores across most clusters: delayed reporting, sleep before assessment, injury situation, and female sex. These characteristics warrant important consideration as they seem to be indicative of greater injury severity. Last, several variables were associated with higher scores in specific symptom clusters. This suggests that these variables may be mechanistically related to concussion outcomes and could therefore be linked to biological markers as well. These variables may be useful in adopting a precision medicine approach to treating concussion.
Supplemental material for this article is available at https://journals.sagepub.com/doi/full/10.1177/23259671231163581#supplementary-materials.
Supplemental Material
Supplemental Material, sj-pdf-1-ojs-10.1177_23259671231163581 - Primary and Secondary Risk Factors Associated With Concussion Symptom Clusters in Collegiate Athletes: Results From the NCAA-DoD Grand Alliance CARE Consortium
Supplemental Material, sj-pdf-1-ojs-10.1177_23259671231163581 for Primary and Secondary Risk Factors Associated With Concussion Symptom Clusters in Collegiate Athletes: Results From the NCAA-DoD Grand Alliance CARE Consortium by Kate E. Nolan, Jaclyn B. Caccese, Anthony P. Kontos, Thomas A. Buckley, Gian-Gabriel P. Garcia, Nicholas Port, Steven P. Broglio, Thomas W. McAllister, Michael McCrea, Paul F. Pasquina, Jasmeet P. Hayes and on behalf of the CARE Consortium Investigators in Orthopaedic Journal of Sports Medicine
Footnotes
Acknowledgment
The authors acknowledge the following CARE Consortium Investigators: April Hoy, MS, ATC, CSCS (Azusa Pacific University); Louise A. Kelly, PhD (California Lutheran University); John DiFiori, MD (Hospital for Special Surgery); Justus D. Ortega, PhD (Humboldt State University); Margot Putukian, MD (Major League Soccer); Dianne Langford, PhD, and Jane McDevitt, PhD, ATC, CSCS (Temple University); Christopher C. Giza, MD, and Joshua T. Goldman, MD, MBA (University of California, Los Angeles); Holly J. Benjamin, MD (University of Chicago); Thomas W. Kaminski, PhD, ATC (University of Delaware); James R. Clugston, MD, MS (University of Florida); Julianne Schmidt, PhD, ATC (University of Georgia); Patrick G. O’Donnell, MHA, COTA/L (UMass Memorial Health); Luis A. Feigenbaum, DPT, ATC (University of Miami); James T. Eckner, MD, MS (University of Michigan); Jason P. Mihalik, PhD, CAT(C), ATC (University of North Carolina at Chapel Hill); Scott Anderson, ATC (University of Oklahoma); Christina L. Master, MD (University of Pennsylvania); Michael Collins, PhD (University of Pittsburgh Medical Center); Sara P.O. Chrisman, MD, MPH (University of Washington); Jonathan C. Jackson, MD (United States Air Force Academy); Kenneth L. Cameron, PhD, MPH, ATC, and Steven J. Svoboda, MD (United States Military Academy); Adam J. Susmarski, DO (United States Naval Academy); Steve Rowson, PhD (Virginia Tech); Christopher M. Miles, MD (Wake Forest University).
Final revision submitted September 8, 2022; accepted September 26, 2022.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Delaware (No. 740790-1).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
