Abstract
Background:
Failure of a subscapularis repair construct after anatomic total shoulder arthroplasty can result in difficulty with internal rotation and an increased likelihood of dislocation. Although suture tape has been demonstrated to be an efficacious augment for tendonous repairs elsewhere in the body, it has not been investigated as a method for augmenting subscapularis peel repairs.
Purpose:
To determine the biomechanical efficacy of suture tape augmentation for the repair of a subscapularis peel.
Study Design:
Controlled laboratory study.
Methods:
Twelve human cadaveric shoulders underwent a subscapularis peel. Specimens were randomly split into 2 groups: 6 specimens underwent repair using a transosseous bone tunnel technique with 3 high-strength sutures placed with a Mason-Allen configuration (control group), and 6 specimens underwent the control repair using augmentation with 2 suture tapes placed in an inverted mattress fashion and secured to the proximal humerus using a suture anchor (augmentation group). Shoulders underwent biomechanical testing to compare repair displacement with cyclic loading, load at ultimate failure, and construct stiffness.
Results:
There were no significant between-group differences in displacement after cyclic loading at the superior (P = .87), middle (P = .47), or inferior (P = .77) portions of the subscapularis tendon. Load to failure was significantly greater in the augmentation group (585.1 ± 97.4 N) than in the control group (358.5 ± 81.8 N) (P = .001). Stiffness was also greater in the augmentation group (71.8 ± 13.7 N/mm) when compared with the control group (48.7 ± 5.7 N/mm) (P = .003).
Conclusion:
Subscapularis peel repair with augmentation via 2 inverted mattress suture tapes secured with an anchor in the proximal humerus conferred significantly greater load at ultimate failure and construct stiffness when compared with a traditional repair using 3 Mason-Allen sutures. There was no difference in repair displacement with cyclic loading between the repair groups.
Clinical Relevance:
Suture tape augmentation of subscapularis peel repairs after shoulder arthroplasty provides an effective segment to the strength of the repair.
Repair of the subscapularis after anatomic total shoulder arthroplasty (aTSA) is important to restore glenohumeral kinematics, prevent dislocation, and provide adequate internal rotation. 14 While subscapularis repair after primary aTSA is often successful, repair loosening and failure are documented complications. 2,12 –14 Patients with subscapularis failure or dysfunction after aTSA may have difficulty with liftoff and belly-press testing and may struggle when performing daily activities. 14 Should dysfunction or failure occur, surgical intervention for revision repair with or without augmentation of the tendon is indicated. 6,8,13 –15
Multiple studies have been conducted to understand the optimal technique to prevent subscapularis repair failure after aTSA. 11,19,21 Krishnan et al 11 described a limited lesser tuberosity osteotomy (LTO) technique that demonstrated superior results when compared with a side-to-side repair. Van Thiel et al 19 reported inconclusive biomechanical evidence to support any method over another. Recently, there has been an increased interest in using suture anchors for subscapularis repair. Werner et al 21 reported that an all-suture anchor-based (FiberTak DR; Arthrex) repair is superior to a side-to-side tendon repair. Denard et al 4 demonstrated superior load to failure (LTF) of lesser tuberosity osteotomies repaired with 2 tensionable high-strength tapes compared with high-strength standard sutures. While the study by Denard et al was successful regarding lesser tuberosity osteotomies, there have been no studies evaluating the efficacy of a suture tape augmentation for the repair of a subscapularis peel.
The purpose of this study was to determine the effects of augmentation of a transosseous subscapularis repair using 2 suture tapes placed into the subscapularis tendon in an inverted mattress fashion and then secured to the proximal humerus with a single suture anchor. We hypothesized that this augmentation would result in a decrease in repair displacement after cyclic loading and an increase in load at ultimate failure when compared with a standard subscapularis repair.
Methods
Specimen Preparation
This study was determined to be exempt from institutional review board approval at our institution. Twelve fresh-frozen human shoulders were obtained from MedCure. Specimens were 75 years old and without a history of shoulder arthritis or other shoulder pathology. Before the day of testing, specimens were removed from the freezer and allowed to thaw at 21°C for 8 hours. After the removal of the skin and deltoid, the rotator cuff was inspected to ensure the tendons were intact. Starting medial and moving lateral, the subscapularis was elevated off the scapula to preserve the muscle. The glenohumeral joint was disarticulated to free the subscapularis and humerus from the scapula. Bone density measurements were obtained for each specimen at the lesser tuberosity using dual-energy x-ray absorptiometry. The humeri were each potted to allow for attachment to the testing apparatus. The humeri were potted in 3.8 × 7.6–cm polyvinyl chloride (PVC) pipes. The diaphysis was placed in the potting so that the superior edge of the PVC pipe was 5 cm distal to the inferior insertion of the subscapularis. Specimens were then randomly allocated to either the control repair or the tape-augmented groups using a random number generator.
Group 1: Single-Row Bone Tunnel Repair
Six specimens underwent repair using a transosseous bone tunnel construct (control group). This technique was chosen as the control because it is the authors’ preferred method for subscapularis repair and because the use of high-strength sutures placed in a Mason-Allen configuration has been commonly utilized in the literature. 3,9,18 First, a subscapularis peel was performed by sharply dissecting the subscapularis tendon from the lesser tuberosity using a No. 15 blade. The long head of the biceps tendon was then tenotomized and discarded. With the subscapularis and biceps tendon removed, an anatomic humeral head osteotomy was performed to match the specimen’s native head inclination and version. The subscapularis footprint was then divided into 4 sections of equal length, and 2-mm drill holes were then placed between these 4 segments, resulting in 3 evenly spaced bone tunnels. The superior and middle tunnels were made on the medial edge of the bicipital groove, while the inferior tunnel was made 1 cm medial to the medial edge of the groove. This position of the inferior tunnel was chosen, as it decreased the distance between the bone tunnel and the cut edge of the neck.
A suture passer was used to shuttle a high-strength suture (No. 2 FiberWire; Arthrex) through each of the bone tunnels. A stemless humeral head implant (Eclipse; Arthrex) was then sized to match each specimen’s anatomic head size and secured in place. The subscapularis tendon was then positioned over the lesser tuberosity. To maintain the positioning of the subscapularis, a figure-of-8 stitch with a high-strength suture was used to secure the superior aspect of the subscapularis to the inferior edge of the coracohumeral ligament. The 3 bone tunnel sutures were then sequentially sutured to the subscapularis tendon, 1 cm from the cut tendon edge, using a Mason-Allen technique with even spacing along the tendon. These sutures were tightened and then secured using 6 square knots (Figure 1A).

(A) Single-row bone tunnel repair (control), in which 3 Mason-Allen sutures are passed through bone tunnels along the bicipital groove. (B) Repair with suture tape/anchor augmentation, in which the control repair is augmented with 2 inverted mattress sutures that are secured with a suture anchor placed in the proximal humerus.
Group 2: Tape/Suture Anchor Augmentation
Six specimens underwent a subscapularis repair using the control technique with augmentation using 2 tape sutures affixed to the proximal humerus with a suture anchor (augmentation group) (Figure 1B). Each specimen underwent a subscapularis peel, humeral head cut, implant placement, and bone tunnel–based subscapularis repair in the same fashion as the control group. After completion of the standard repair, 2 No. 2 SutureTape (Arthrex) sutures were passed through the subscapularis tendon just medial to the previous Mason-Allen stitches in an inverted mattress fashion, with equal spacing along the tendon (Figure 2). A position on the proximal humerus was marked 2 cm distal to the tip of the greater tuberosity, and 1 cm lateral to the lateral aspect of the bicipital groove. A punch for a 4.75-mm suture anchor (Swivelock C; Arthrex) was gently malleted into the marked area of the humerus to create an entry point. The ends of the 2 tape sutures were then passed through a suture anchor (polyether ether ketone Swivelock C; 4.75 × 19.1–mm; Arthrex), and the suture anchor was then inserted into the humerus.
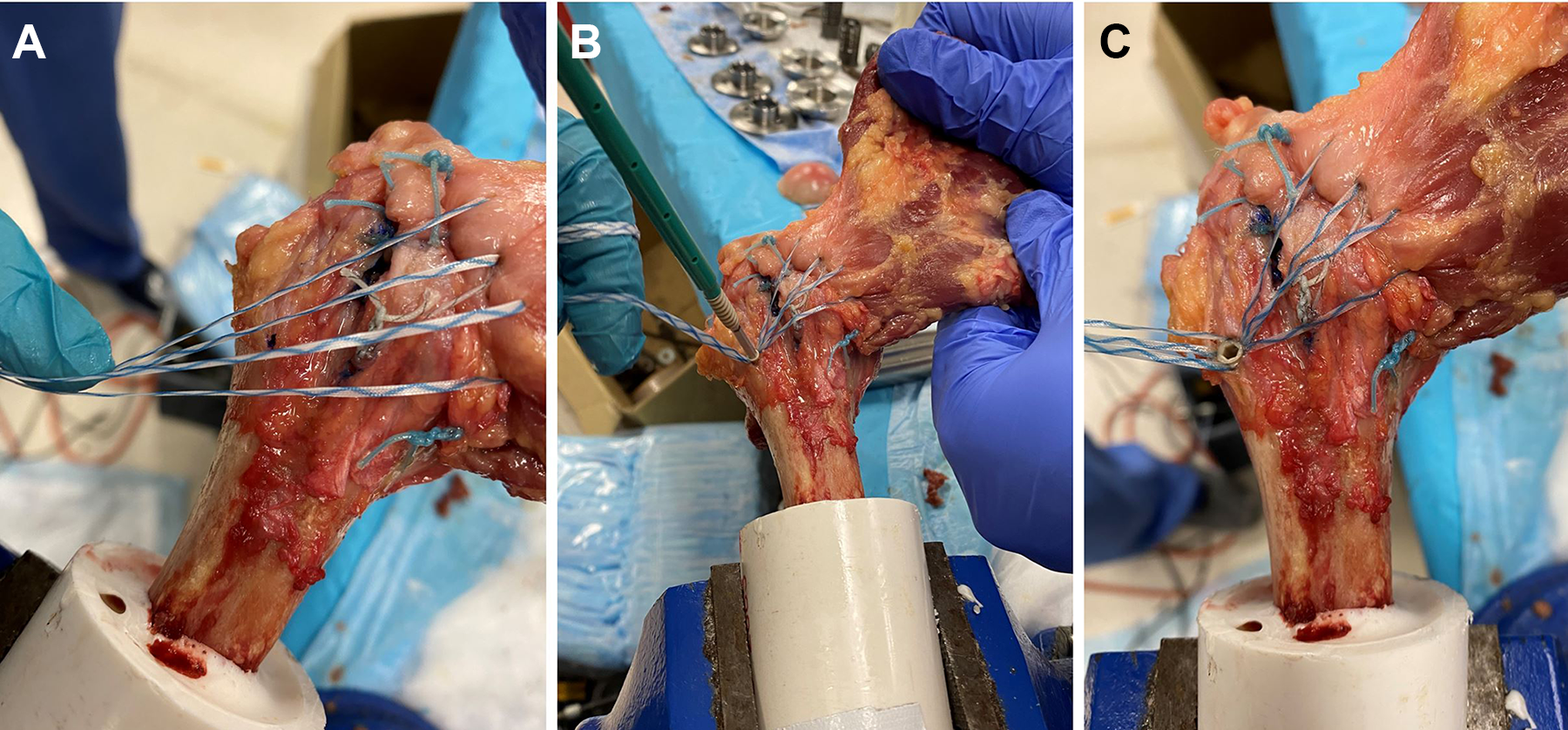
(A) Two inverted mattress sutures were placed in the subscapularis tendon just medial to the Mason-Allen sutures in the control repair. (B) A hole was punched 1 cm lateral to the bicipital groove and 2 cm distal to the tip of the greater tuberosity, and the sutures were secured here using a polyether ether ketone suture anchor. (C) The finished augmented repair.
Biomechanical Testing
Testing was performed using a servohydraulic testing system (MTS 858 Bionix; MTS Systems) with a 2.5-kN load cell. First, the PVC potting was affixed to the base of the testing system using a custom mounting apparatus. 16 The mounting system was adjusted so that the axis of the humerus was 135° from the axis of the subscapularis to replicate the natural line of pull. The subscapularis tendon was then affixed to a sigmoidal clamp (Figure 3). The force of the machine was zeroed with the tendon in no tension to account for the weight of the tendon and the sigmoidal clamp.

A control repair specimen is mounted in the testing system. Optical tracking markers are evenly spaced along each side of the repair to determine superior, middle, and inferior displacement during cyclic loading.
To determine the displacement of the repair construct at the superior, middle, and inferior portions of the subscapularis insertion, 3 pairs of optical tracking markers were placed in the specimen. One row of 3 markers was placed lateral to the repair in the humerus. The second row of 3 markers was placed in line with their respective first-row markers in the musculotendinous junction of the subscapularis.
Once the testing apparatus was appropriately positioned and secured, a preload of 10 N was applied for 10 seconds. Specimens were then cyclically loaded between 10 and 100 N for 500 cycles at 1 Hz. 4 The cyclic loading phase was recorded using a high-resolution video camera (HC-V770 Full HD Camcorder; Panasonic). The video recordings were digitized using MaxTRAQ 2D (Innovision Systems), and the cyclic displacements of the superior, middle, and inferior repair were calculated. Cyclic displacement equaled the displacement of the 500th cycle minus the displacement of the first cycle.
LTF testing was performed after cyclic loading at a tensile loading rate of 1 mm/s until catastrophic failure of the repair construct occurred. 1,19,20 The mechanism of failure and ultimate LTF (N) of each specimen were recorded. Using the slope of the load versus displacement curve of the LTF testing, the stiffness (N/mm) was calculated.
Statistical Analysis
An a priori power analysis was performed before the initiation of this study. Based on a standard deviation of a previously published study (±71.2 N), to determine a 150-N difference between the 2 groups, we determined that a minimum of 4 specimens per group was required 4,21 for a power of 0.80 at an α of .05.
Descriptive statistics were compared between the control and augmentation groups using the Student t test for continuous data and the Fisher exact test for categorical data. Differences in displacement with cyclic loading, LTF, and stiffness between the 2 groups were calculated using the Student t test. All statistics were performed using SPSS statistical software (Version 28; IBM).
Results
There was no difference in specimen age, sex, body mass index, laterality, or bone mineral density between the control and augmentation groups (Table 1). After cyclic loading, there was no significant difference between the groups in the displacement of the superior tendon repair (2.1 ± 0.4 mm vs 2.1 ± 0.7 mm, respectively), nor were there significant differences between the groups in middle tendon displacement (3 ± 1.6 mm vs 2.3 ± 1.4 mm, respectively) or inferior tendon displacement (3.2 ± 1.2 mm vs 2.9 ± 1.3 mm, respectively) (Figure 4).
Specimen Characteristics a

a Data are presented as mean ± SD unless otherwise indicated. BMI, body mass index.
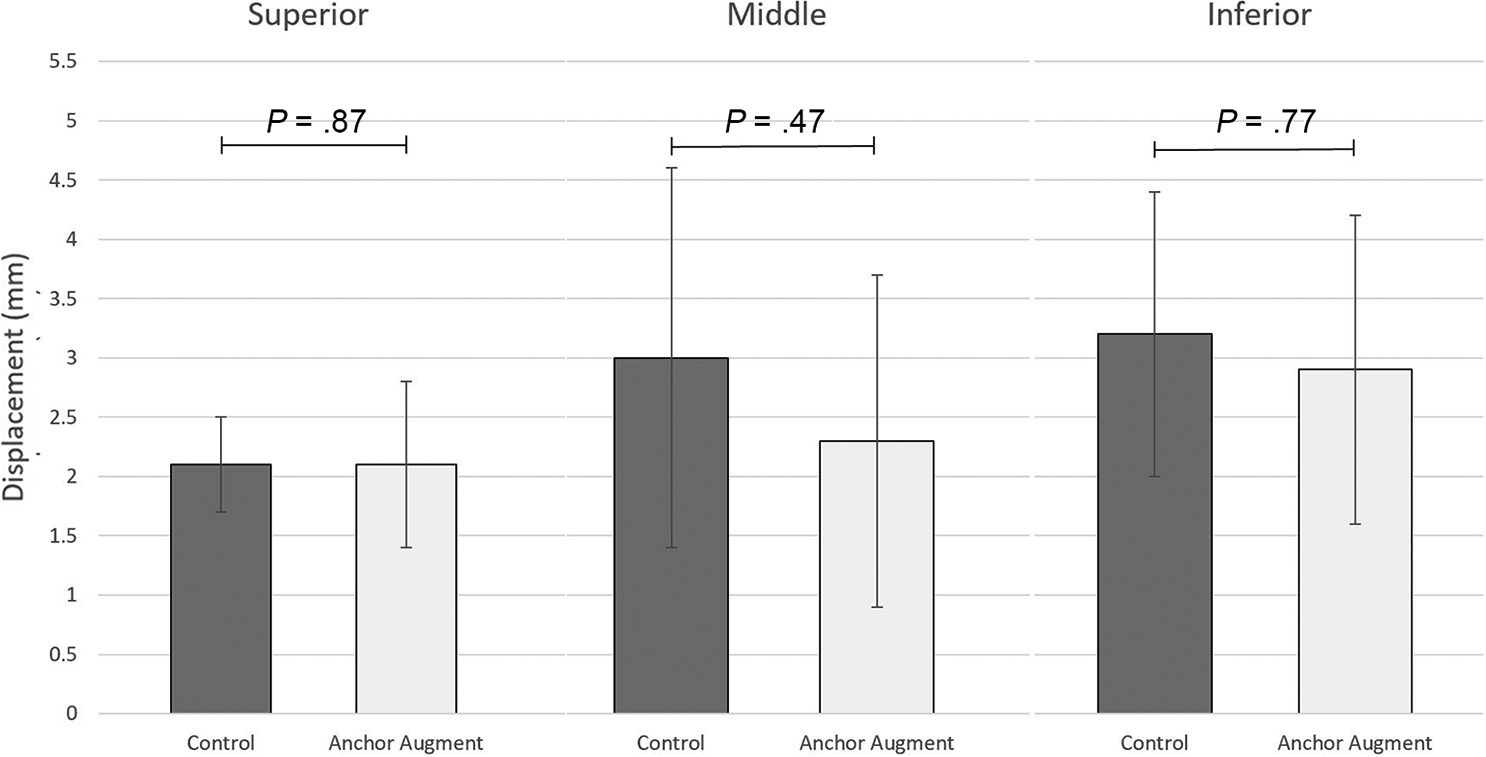
Repair displacement after cyclic loading of the control repair and repair with suture anchor augmentation at the superior, middle, and inferior portions of the subscapularis tendon. There was no statistically significant difference in displacement for any of the 3 locations. *Statistically significant difference (P < .05).
The anchor augmentation group demonstrated a significantly greater load at ultimate construct failure when compared with the control group (585.1 ± 97.4 N vs 358.5 ± 81.8 N,respectively; P = .001) (Figure 5A). Additionally, the augmentation group demonstrated a significantly larger stiffness when compared with the control group (71.8 ± 13.7 N/mm vs 48.7 ± 5.7 N/mm, respectively; P = .003) (Figure 5B). In the control group, the most common mode of failure was attenuation of the tendon at the repair site (gradual thinning of the tendon at the interface between the repair sutures and the tendon), with 4 specimens failing in this manner. One specimen failed via suture pullout from the bone tunnels, and 1 specimen failed via breakage of the suture away from the knot, likely from friction at the bone tunnel interface. In the anchor augmentation group, 1 specimen failed via tendon attenuation at the repair site, while 5 failed via pullout of the anchor from the bone. When pullout of the anchor occurred in these 5 specimens, they proceeded to fail via tendon attenuation (Figure 6).
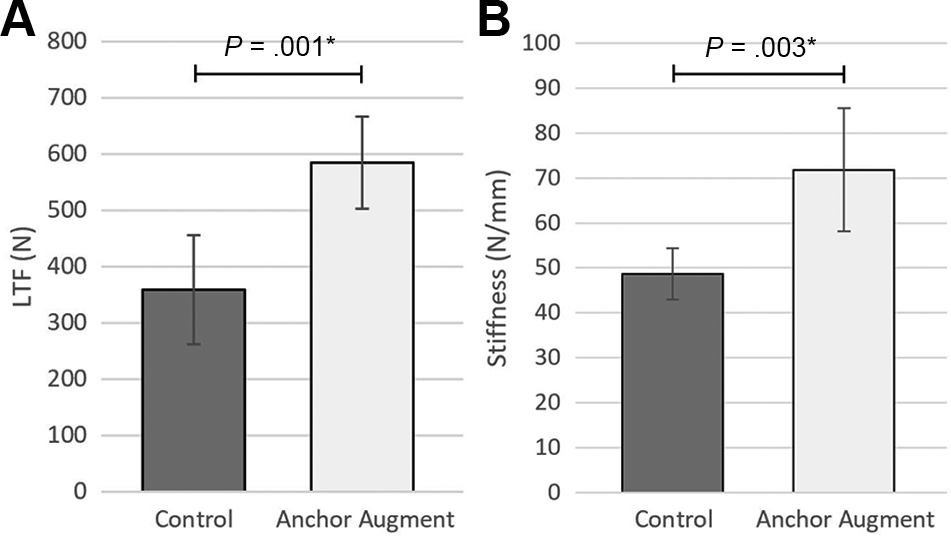
(A) Load to failure (LTF) and (B) stiffness for the control repair and suture augmented repair groups. *Statistically significant difference (P < .05).

(A) Failure of control repair via tendon attenuation and suture pullout. (B) Failure of suture anchor augmented repair via anchor pullout and tendon attenuation.
Discussion
The findings of this study indicated that augmentation of a subscapularis peel repair with 2 inverted mattress sutures secured with a suture anchor significantly increased construct stiffness and load at ultimate failure when compared with a standard transosseous repair. Additionally, there was no significant difference in repair displacement with cyclic loading between these 2 methods of repair.
Subscapularis failure after shoulder arthroplasty is tolerated by some patients but may be a devastating problem for others requiring revision surgery. 6,13,15 Accordingly, many studies have attempted to identify the optimal method to repair the subscapularis tendon after aTSA. Recent attention has been turned to LTO, as it has been shown in biomechanical studies to have superior repair strength compared with traditional peel and tenotomy repairs. 18 Previous studies have reported the LTF of LTO repairs as between 301.3 N and 632.3 N for various repair techniques. 5,7,10,17,19 Our study demonstrated a mean ultimate LTF of 585.1 N for the anchor augmented group.
These findings suggest that the augmentation method addressed in this study offers similar repair strength to LTO repairs while avoiding some of the potential downsides of LTO, such as a possible fracture or a decreased metaphyseal fit of press-fit implants. However, it is challenging to compare biomechanical results across studies. 18 Additionally, as opposed to LTO, a subscapularis peel may have the benefit of allowing for some tendon lengthening in patients with restricted external rotation.
Interestingly, while the anchor augmentation increased LTF and construct stiffness, it did not affect displacement with cyclic loading. The displacement seen in both groups was minimal (<3.5 mm for both groups), suggesting that transosseous Mason-Allen sutures with a rotator interval repair is quite durable. Given the limited gap formation, it is unlikely that augmentation would significantly reduce displacement. Additionally, whenever multiple suture-tendon interfaces are present, each suture may undergo a differential amount of loading. When some sutures are subjected to more force than others, the repair may lengthen until the force is evenly distributed among all the sutures. We attempt to combat this tendency with preloading. However, the force needed to counteract this phenomenon may be above the amount exerted during the preload phase, and as such this level of displacement would not occur until cyclic loading. Given the greater number of sutures utilized in augmented repair, this technique is more susceptible to such a mode of repair lengthening.
While the suture anchor construct did confer greater LTF when compared with the control repair, it is unclear to what degree this increased strength would affect different clinical scenarios. It is likely that the control repair confers enough strength to withstand most in vivo loading scenarios. However, the suture augmentation may provide additional security for high-load situations such as catching a falling object or sustaining a fall onto an outstretched extremity.
One significant advantage of this augmentation is its technical simplicity. Only 2 inverted mattress sutures are required, secured with a single bone tunnel. This anchor is easily inserted when using a stemless device. While it is likely that this technique can be used to augment other methods of subscapularis peel or tenotomy repair, further research is warranted to determine whether this is the case. Additionally, further research is necessary to determine whether other kinds of suture anchors or different-sized anchors would affect the strength of the repair.
Although the results of this study are favorable for the use of the suture anchor construct, it is important to discuss the modes of failure for each of these groups. The control group most often failed via attenuation of the tendon at the repair site; however, the anchor augmented group often failed via anchor pullout. This latter mode of failure can be concerning, as it could result in both failure of repair as well as loose hardware stuck in the anterior shoulder. However, failure of the control group could also require revision surgery in vivo; thus, the augmented repair at minimum offers some increased protection from loading forces to prevent failure from occurring in the first place.
Limitations
As with any cadaveric biomechanical study, assessment of these repairs reflects the immediate postoperative period and does not represent the strength of the repair after a period of healing. Additionally, this study was conducted with stemless implants. While the effects on repair strength would likely translate to arthroplasty using a stemmed implant, it is possible that anchor placement would be made more difficult with the presence of a stem. With a stemmed implant, the direction of the anchor might require alteration to fit around the stem, which could affect the overall construct strength. A suture anchor also adds additional cost to the surgery. Finally, as previously mentioned, the findings of this study may not reflect the efficacy of this augmentation technique when applied to other kinds of subscapularis repairs.
Conclusion
Suture anchor augmentation for subscapularis peel repair after TSA confers a greater stiffness and LTF when compared with a single-row transosseous repair alone. There was no effect of the suture anchor augmentation on repair lengthening during cyclic loading. Given the technical simplicity of this repair and superior biomechanical strength, augmentation with this technique may be useful for orthopaedic surgeons performing these procedures.
Footnotes
Acknowledgment
The authors thank Geneva Hargis, PhD, and Christopher Bonin, PhD, for their creation of the medical illustrations.
Final revision submitted December 6, 2022; accepted January 19, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.D.M. has received consulting fees from Arthrex and Astellas Pharma, nonconsulting fees from Arthrex and Kairos Surgical, and honoraria from Arthrosurface. P.M.S. has received consulting fees from Arthrex, Catalyst OrthoScience, Medical Device Business Services, and Pacira Pharma; nonconsulting fees from Arthrex and Pacira Pharma; royalties from Arthrex; and hospitality payments from Exactech, Stryker, and Wright Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by the University of Connecticut.
