Abstract
Background:
Latissimus dorsi tendon transfer (LDTT) is increasingly performed with arthroscopic assistance, requiring an open axillary incision, which could increase risks of infection, hematoma, and lymphoedema. Technological advancements now enable LDTT to be fully arthroscopic, but its benefits and safety have not yet been confirmed.
Purpose:
To compare the clinical outcomes and complication rates of arthroscopic-assisted versus full-arthroscopic LDTT for irreparable posterosuperior massive rotator cuff tears in shoulders with no surgical antecedents.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
The study included 90 patients who had undergone LDTT over 4 consecutive years by the same surgeon and did not have prior surgery. During the first 2 study years, all procedures were arthroscopically assisted (n = 52), while during the last 2 years, all procedures were fully arthroscopic (n = 38). Procedure duration and all complications were recorded, as well as clinical scores and range of motion at minimum 24-month follow-up. To enable direct comparison between the techniques, propensity score matching was used to obtain 2 groups with equivalent age, sex, and follow-up.
Results:
From the initial cohort of 52 patients who underwent arthroscopic-assisted LDTT, 8 had complications (15.4%), of which 3 (5.7%) required conversion to reverse shoulder arthroplasty and 2 (3.8%) required drainage or lavage. From the initial cohort of 38 patients who had full-arthroscopic LDTT, 5 had complications (13.2%), of which 2 (5.2%) required conversion to reverse shoulder arthroplasty but no patients (0%) required other procedures. Propensity score matching resulted in 2 groups, each comprising 31 patients, with similar outcomes in terms of clinical scores and range of motion. The procedure time was about 18 minutes shorter for full-arthroscopic LDTT, which had different complications (2 axillary nerve pareses) as compared with arthroscopic-assisted LDTT (1 hematoma and 2 infections).
Conclusion:
Equivalent outcomes at minimum 24-month follow-up were found for arthroscopic-assisted and full-arthroscopic LDTT in terms of complications rates (15.4% and 13.2%, respectively), conversion to reverse shoulder arthroplasty (5.7% and 5.2%), clinical scores, and range of motion.
Keywords
Management of irreparable massive rotator cuff tears (mRCTs) is often challenging, as nonoperative treatments provide little or no improvement in the long term, while surgical treatments are technically demanding and associated with risks of complications and inadequate recovery. Posterior latissimus dorsi tendon transfer (LDTT) has demonstrated satisfactory outcomes to treat posterosuperior mRCTs regardless of patient age. 15,18,20
At present, LDTT is sometimes performed with arthroscopic assistance, 11,14,22 which requires an open axillary incision, increasing risks of infection, hematoma, delayed skin healing, and lymphoedema. 10 Technological advancements now render LDTT feasible using full-arthroscopic techniques, 16 although their benefits over arthroscopic-assisted techniques have not yet been investigated. While full-arthroscopic LDTT might be more technically demanding, it could improve healing and reduce complications as compared with arthroscopic-assisted LDTT.
The purpose of the present study was to compare clinical outcomes and complication rates of arthroscopic-assisted versus full-arthroscopic LDTT for the treatment of irreparable posterosuperior mRCTs in shoulders that had no prior surgery. The hypothesis was that at a minimum follow-up of 24 months, full-arthroscopic LDTT would grant equivalent or better outcomes with fewer complications than arthroscopic-assisted LDTT.
Methods
The study protocol received ethics committee approval, and all patients provided informed consent for the use of their data and images for research and publishing purposes. We retrieved the records of a consecutive series of 165 patients who underwent LDTT for irreparable mRCTs by the same senior surgeon (J.K.) between 2016 and 2019. A total of 75 patients had undergone previous shoulder surgery, leaving 90 patients who had no surgical antecedents. All procedures performed in 2016 to 2017 were arthroscopically assisted (n = 52), while all procedures performed in 2018 to 2019 were fully arthroscopic (n = 38). The senior surgeon had performed 59 arthroscopic-assisted LDTT procedures in 2014 to 2015 and was experienced with the procedure at the start of the inclusion period; therefore, there were no learning curve effects regarding the arthroscopic-assisted technique while there may have been for the full-arthroscopic technique.
Indications and Contraindications
The indications for LDTT were as follows: a massive irreparable tear of the posterosuperior rotator cuff with fatty infiltration grade ≥3 in at least 2 muscles, ≥2 tendons retracted to the glenoid, and persistent pain after failed nonoperative treatment. The contraindications were as follows: concomitant irreparable tear of the subscapularis, cuff tear arthropathy with glenohumeral arthritis (Hamada grade ≥4), complete and permanent axillary nerve palsy, shoulder pseudoparalysis (active forward elevation <45° despite 3 months of physiotherapy), 4 or shoulder stiffness (limitation of passive movements in forward elevation, external rotation, and internal rotation despite 3 months of physiotherapy). 16
Preoperative Assessment
Before surgery, we collected demographic data, and all patients underwent radiographic evaluation to assess the subacromial space and grade of glenohumeral arthritis according to the Hamada classification 12 on standard anteroposterior radiographs. All patients also underwent preoperative magnetic resonance imaging to assess tear pattern, 5 tendon retraction, and fatty infiltration according to Goutallier et al 9 in each rotator cuff muscle. Finally, an independent observer (S.M.) collected range-of-motion data, including active forward elevation, abduction, and external rotation (elbow at the side and at 90° of abduction), as well as the Constant score. 6
Surgical Treatment
Arthroscopic-assisted LDTT was performed in the lateral decubitus position under general anesthesia and an interscalene nerve block. A 5-cm incision was made along the anterior (axillary) border of the scapula (Figure 1). The latissimus dorsi was separated from the teres major, and its neurovascular bundle was identified. Once the muscle belly was released from its surrounding structures, the aponeurotic band leading to the latissimus dorsi tendon was identified and followed until its humeral insertion. The latissimus dorsi tendon was then cut on its axillary attachments and detached from the humerus. The tendon was left flat and harvested using two No. 2 nonresorbable sutures (Vims). The long head of the triceps was identified, and the dissection was pursued under the posterior deltoid toward the subacromial space to prepare the most direct route for the transfer. Arthroscopic debridement of the subacromial space was performed (without acromioplasty to prevent superior escape of the humeral head) with tenotomy of the biceps, if present. 3 Adjuvant subscapularis repair was performed in shoulders with Collin type C tears. The free sutures of the flat latissimus dorsi tendon were retrieved arthroscopically and fixed onto the junction between the footprints of the supraspinatus and infraspinatus using 2 knotless anchors (Versalok; DePuy Mitek), which could also tighten the posterior cuff remnants.
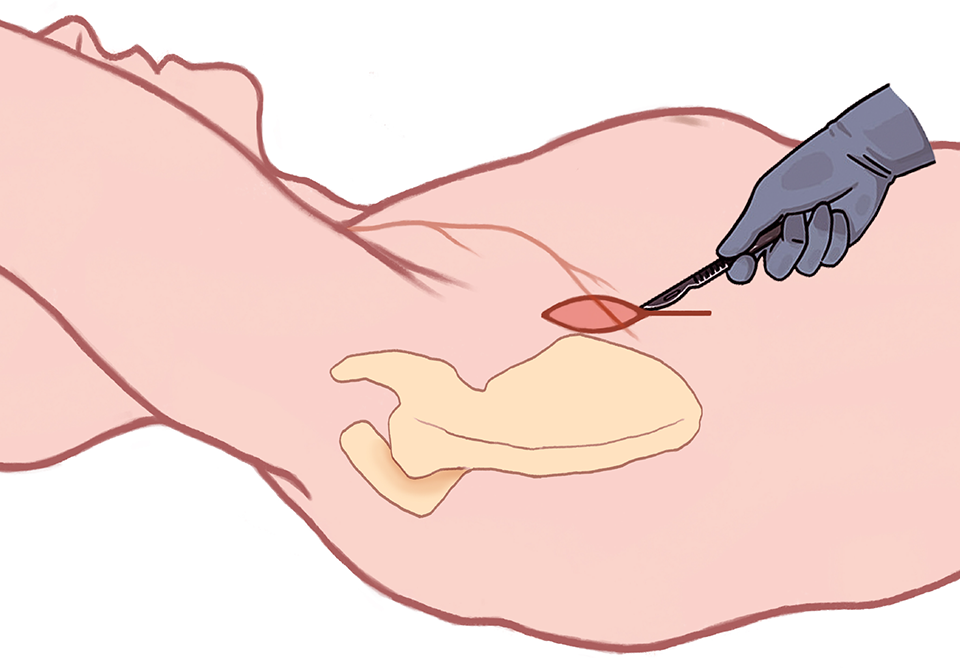
Arthroscopic-assisted latissimus dorsi tendon transfer was performed in the lateral decubitus position. A 5-cm incision was made along the anterior (axillary) border of the scapula.
Full-arthroscopic LDTT was performed under general anesthesia in the beach-chair position with the arm in a pneumatic holder (Spider; Smith & Nephew) without axillary portals (Figure 2A). Arthroscopic debridement of the subacromial space was performed (without acromioplasty to prevent superior escape of the humeral head) with tenotomy of the biceps, if present. 3 Adjuvant subscapularis repair was performed in shoulders with Collin type C tears. Dissection of the posterior space was performed using an extra-large radiofrequency probe (Turbo XL 90°; Smith & Nephew) after identification of the scapular spine. Further dissection was performed in an inferomedial direction between the distal deltoid aponeurosis and the remnants of infraspinatus and teres minor. The vertical fibers of the long head of the triceps were visualized to prevent any injury to the axillary nerve crossing through the quadrilateral space (lateral to the long head of the triceps).

Full-arthroscopic latissimus dorsi tendon transfer. (A) Eight portals, represented in the figure by crosses, were created. The dashed line is the continuation of the line to help position the portals. Solid lines, acromion; oval lines, coracoid process. (B) Posterior view of the shoulder. Soft tissues were released medial to the long head of the triceps to access the triangular space (delimited by the long head of the triceps laterally, the teres minor superiorly, and the latissimus dorsi/teres major distally). (C) Anterior view of the shoulder. The scope was placed in an anterolateral portal, following the long head of the biceps tendon to reach the lateral edge of the conjoint tendon and the upper border of the pectoralis major, which was partially released to facilitate exposure to the latissimus dorsi tendon.
Next, soft tissues were released medial to the long head of the triceps to access the triangular space (delimited by the long head of the triceps laterally, the teres minor superiorly, and the latissimus dorsi/teres major distally) (Figure 2B). The scope was switched to an anterolateral portal (Figure 2C), following the long head of the biceps tendon to reach the lateral edge of the conjoint tendon and the upper border of the pectoralis major, which was partially released to facilitate exposure. The “3 sisters” (terminal branches of the circumflex vessels) were identified, marking the inferior border of the subscapularis tendon and the upper border of the latissimus dorsi tendon. The dissection followed the upper edge and anterior surface of the latissimus dorsi. The teres major is located posterior to the latissimus dorsi, with several anatomic connections. Great care was taken to prevent any injury to the axillary nerve, the circumflex vessels, and the radial nerve that crosses the superficial surface of the latissimus dorsi, 3 to 4 cm medial to its humeral insertion.
Two “triple Krakow” sutures were made 3 cm along the common insertion of the latissimus dorsi and teres major tendons, which were then detached to perform a “double transfer” through the triangular space toward the back of the humeral head. Reattachment of the latissimus dorsi and teres major double transfer was achieved using the same anchors at the same locations as the arthroscopic-assisted technique (Figure 3).

Posterior view of the shoulder. The “double transfer” of the latissimus dorsi and teres major was fixed onto the junction between the footprints of the supraspinatus and infraspinatus using 2 knotless anchors.
Postoperative Rehabilitation
All patients were immobilized using a 30° abduction pillow in neutral rotation for 4 weeks, and self-assisted passive exercises in the supine position were started immediately. At 4 weeks, the goal was to restore passive flexion, and gentle aquatic therapy was recommended. After 3 months, strengthening exercises were started.
Outcomes Assessment
Operative time was recorded, and after surgery, all complications were noted, specifying ruptures and whether patients required conversion to reverse shoulder arthroplasty (RSA). At a minimum follow-up of 24 months, 2 independent observers recorded the range of motion (active forward elevation, abduction, and external rotation [elbow at the side and at 90° of abduction]) as well as clinical scores, including the Constant score, 6 the Subjective Shoulder Value, 8 Simple Shoulder Test, 21 the Activities of Daily Living Requiring Active External Rotation score, 2 American Shoulder and Elbow Surgeons scores, 19 and pain on visual analog scale.
Statistical Analysis
A sample size calculation indicated that 28 patients per group were needed to determine a significant minimal clinically important difference in Constant score of 10.4 points between the groups, 17 assuming an equal standard deviation of 13.3 and a statistical power of 0.80.
Descriptive statistics was used to summarize the demographic data, clinical scores, and radiographic measurements. For categorical variables, comparisons between groups were performed using the Fisher test or chi-square test for binary and nonbinary variables, respectively. Normality of continuous variables was assessed through Shapiro-Wilk tests. For continuous variables, comparisons between groups were performed using Wilcoxon signed rank tests, as none of the variables were normally distributed. To enable comparison of outcomes of arthroscopic-assisted versus full-arthroscopic LDTT, propensity scores were estimated for each patient using a logistic regression model to obtain 2 similar groups in terms of age, sex, and follow-up. A 1:1 nearest-neighbor algorithm with a caliper of 1.1 was applied to match patients using their corresponding propensity scores. Statistical analyses were conducted using R Version 4.1 (R Foundation for Statistical Computing). P values <.05 were considered statistically significant.
Results
From the initial cohort of 52 patients who had arthroscopic-assisted LDTT, 3 were lost to follow-up and 2 died for reasons unrelated to shoulder surgery, leaving 47 patients in the final cohort. From the initial cohort of 38 patients who had full-arthroscopic LDTT, 1 was lost to follow-up and 2 died for reasons unrelated to shoulder surgery, leaving 35 patients for analysis. Propensity score matching led to the inclusion of 31 patients in each group (Figure 4).

Patient enrollment and matching. LDTT, latissimus dorsi tendon transfer; mRCT, massive rotator cuff tear.
Complications After Arthroscopic-Assisted LDTT
Of the 52 patients who underwent arthroscopic-assisted LDTT, 8 (15.4%) had complications:
Patient 1: axillary hematoma that required drainage at 23 days
Patient 2: infection (Finegoldia magna) that required lavage at 23 days, followed by septic arthritis (Hamada 4) that required conversion to RSA at 7 months
Patient 3: traumatic latissimus dorsi rupture at 31 days, treated nonoperatively
Patient 4: infection (Propionibacterium acnes and Staphylococcus lugdunensis) that required lavage at 85 days
Patient 5: atraumatic latissimus dorsi rupture at 42 days, treated nonoperatively
Patient 6: asymptomatic granuloma of the greater tuberosity at 95 days, not treated
Patient 7: progression of glenohumeral arthritis (Hamada 4) at 97 days, scheduled for conversion to RSA
Patient 8: persistent pain and inadequate function since 5 years, recommended conversion to RSA
Of the 8 patients, 3 required conversion to RSA (5.7%), and 2 required drainage or lavage (3.8%).
Complications After Full-Arthroscopic LDTT
Complications were seen in 5 (13.2%) of the 38 patients after full-arthroscopic LDTT:
Patient 1: axillary nerve paresis immediately after surgery, resolved without treatment
Patient 2: axillary nerve paresis immediately after surgery that led to hypotrophy of the anterior deltoid at 3 months, not treated; electromyography showed axillary nerve denervation that was in the recovery phase, and deltoid function recovered fully after 3 months.
Patient 3: asymptomatic anchor migration seen on radiographs at 30 days, not treated
Patient 4: retear of the subscapularis tendon at 3 months in a shoulder with Collin type C tear, which required conversion to RSA at 18 months owing to progression of glenohumeral arthritis (Hamada grade 4)
Patient 5: persistent pain and inadequate function for 4 years, recommended conversion to RSA
Two patients (5.2%) required conversion to RSA, but no patients required other procedures. Axillary nerve paresis in patients 1 and 2 was observed in shoulders operated toward the beginning of the inclusion period of full-arthroscopic LDTT (during the first 5 months) and occurred owing to transfer of the latissimus dorsi through the quadrilateral space (lateral from the long head of the triceps next to the axillary nerve) instead of the triangular space (medial from the long head of the triceps where there is no nerve). Nonetheless, both these pareses were transitory and resolved without treatment.
Comparison Between Matched Cohorts
Propensity score matching resulted in 2 groups with comparable patient demographics and follow-up times (Table 1). Results of the comparison of preoperative radiologic characteristics between groups is shown in Table 2. The full-arthroscopic group had a significantly higher rate of nonfunctional teres minor as compared with the arthroscopic-assisted group (P = .02).
Characteristics of the Matched Cohorts a
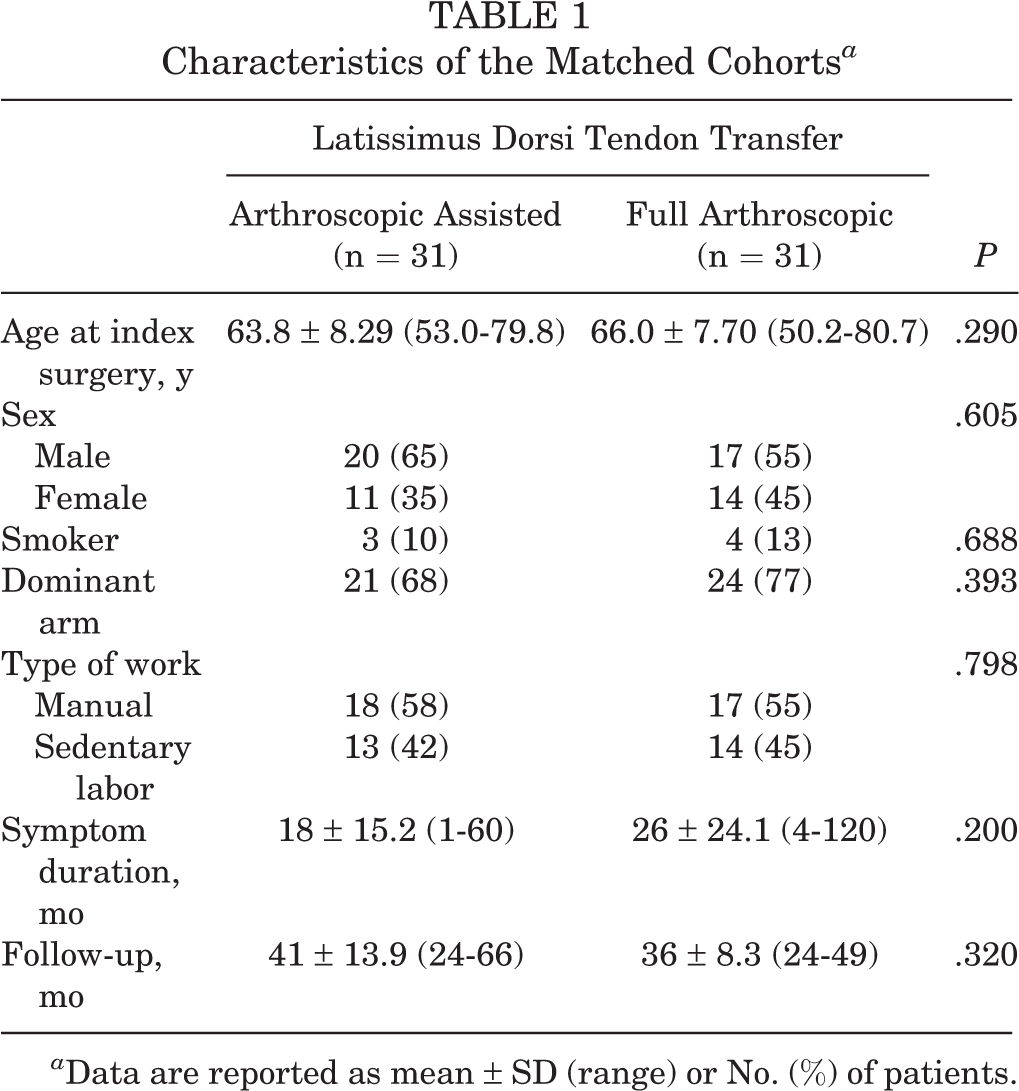
a Data are reported as mean ± SD (range) or No. (%) of patients.
Comparison of Preoperative Radiologic Characteristics Between the Matched Cohorts a

a Data are reported as No. (%) of patients.
b Bold P value indicates statistically significant difference between groups (P < .05).
The operative time was shorter by a mean 18 minutes (P = .001) during the full-arthroscopic LDTT (22 procedures lasted ≤90 minutes) versus arthroscopic-assisted LDTT (10 procedures lasted ≤90 minutes) (Table 3).
Comparison of Intraoperative Data a

a Data are reported as mean ± SD (range) or No. (%) of patients. SSC, subscapularis.
b Bold P value indicates statistically significant difference between groups (P < .05).
Range of Motion and Clinical Scores
Despite propensity score matching, patients who underwent full-arthroscopic LDTT had significantly worse preoperative active forward elevation (P < .001) and activity component of the Constant score (P = .047) as compared with patients who underwent arthroscopic-assisted LDTT (Tables 4 and 5). There were no significant differences between the groups in terms of postoperative range of motion or clinical scores.
Pre- and Postoperative Range of Motion
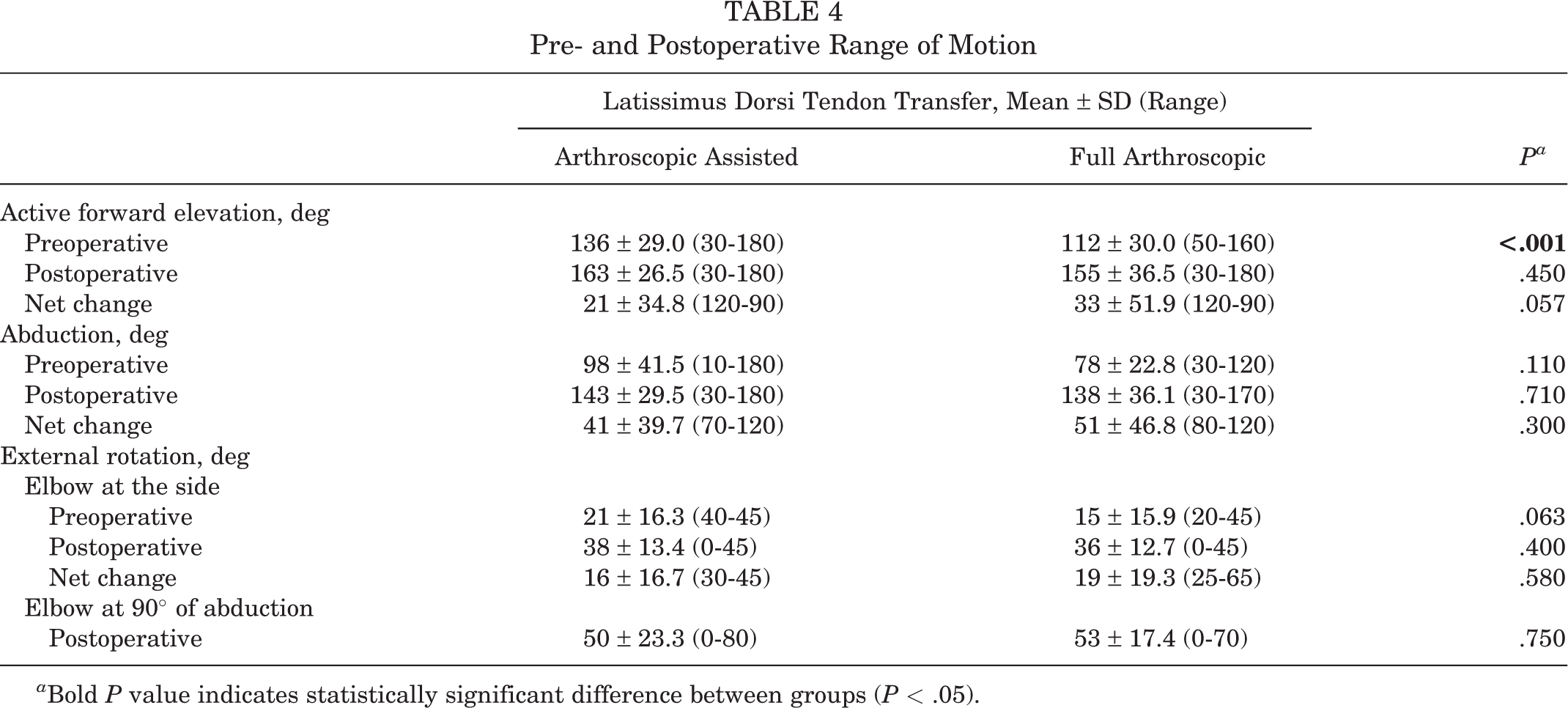
a Bold P value indicates statistically significant difference between groups (P < .05).
Pre- and Postoperative Clinical Scores a

a ADLER, Activities of Daily Living Requiring Active External Rotation; ASES, American Shoulder and Elbow Surgeons; SST, Simple Shoulder Test; SSV, Subjective Shoulder Value; VAS, visual analog scale.
b Bold P value indicates statistically significant difference between groups (P < .05).
c Adjusted for age and sex.
d Acromiohumeral distance.
Discussion
The most important finding of this study was that, for the treatment of irreparable posterosuperior mRCTs in shoulders that had no surgical antecedents, arthroscopic-assisted and full-arthroscopic LDTT resulted in similar range of motion, clinical scores, and subacromial space at a minimum follow-up of 24 months. Furthermore, the operative time for full-arthroscopic LDTT was shorter by a mean 18 minutes, but the groups had similar rates of overall complications (15.4% and 13.2%) and conversion to RSA (5.7% and 5.2%); however, arthroscopic-assisted LDTT required drainage or lavage in some cases (3.8%), while full-arthroscopic LDTT did not require other procedures (0%). Further analysis revealed different types of complications for arthroscopic-assisted LDTT (1 hematoma and 2 infections) versus full-arthroscopic LDTT (2 axillary nerve pareses). The clinical relevance of these findings is that, while full-arthroscopic LDTT seems safe and effective for the treatment of irreparable posterosuperior mRCTs with the potential to reduce surgical time, hematoma, and infections, surgeons should be aware of the risks of nerve paresis related to the approach and portals used.
Full-arthroscopic LDTT is a technically demanding shoulder procedure and has a steep learning curve. Performing full-arthroscopic LDTT instead of arthroscopic-assisted LDTT does offer benefits, as it is less invasive with reduced latissimus dorsi tendon donor-site morbidity, 7 which could improve recovery. Moreover, in the present study, the procedure time was on average 18 minutes shorter using full-arthroscopic LDTT (71% performed in ≤90 minutes) when compared with arthroscopic-assisted LDTT (32% performed in ≤90 minutes). While the clinical relevance of shortening an arthroscopic procedure by 18 minutes may not be clear, numerous studies have indicated that procedures exceeding 90 minutes are significantly associated with greater risks of complications, such as surgical site infection, pulmonary embolism, and overnight hospital stay. 1,13 because full-arthroscopic LDTT does not require opening and closing deep skin incisions. Full-arthroscopic LDTT might also be faster because performing a double transfer of the teres major and latissimus dorsi requires less excision than isolated transfer of the latissimus dorsi. Moreover, this double transfer is stronger than an isolated transfer and may therefore improve the clinical results. The potential drawbacks of full-arthroscopic LDTT include the difficulty of releasing the muscle belly from its connective tissue, as reported in a recent publication, 23 therefore making it challenging to transfer the tendon to the desired point on the greater tuberosity and exacerbating tension within the tendon-to-bone fixation. In our experience, full-arthroscopic LDTT requires extensive cadaveric training to familiarize surgeons with the approach and new portals, which could improve tendon fixation and minimize risks of radial and/or axillary nerve injuries.
In the present study, the complication rates were comparable for the 2 techniques, but the type of complications differed. Drainage or lavage for hematoma and infection was required in 3.8% after arthroscopic-assisted LDTT (0% after full-arthroscopic LDTT), while axillary nerve paresis was observed in 5.2% after full-arthroscopic LDTT (0% after arthroscopic-assisted LDTT). It is worth noting that the 2 nerve pareses occurred in shoulders operated toward the beginning of the inclusion period of full-arthroscopic LDTT (during the first 5 months) and may therefore be associated with learning curve effects. The higher infection rate after arthroscopic-assisted LDTT is due to the 5-cm incision that is made along the anterior (axillary) border of the scapula, which is a difficult area to sterilize. The complication rates of arthroscopic-assisted LDTT were comparable to the findings of a recent systematic review by Memon et al, 18 who reported that 2.3% of patients had deep infections requiring lavage and antibiotic therapy, 1.6% had hematoma, 0.4% had transient postoperative brachial plexus palsy, and 0.4% had a hematoma infection at the donor site. Furthermore, of the 90 patients who underwent LDTT in the present study, only 2 (2.2%) had postoperative ruptures of the latissimus dorsi tendon (both had arthroscopic-assisted LDTT), which was also comparable to the rate of 2.7% reported by Memon et al.
In the present study, there were no significant differences in postoperative clinical scores and range of motion between arthroscopic-assisted versus full-arthroscopic LDTT. In our series, patients achieved greater clinical scores and range of motion than reported in the systematic review by Memon et al, 18 who found a postoperative Constant score of 66, Subjective Shoulder Value of 56, active forward elevation of 155°, external rotation of 40°, and abduction of 132°. However, Memon et al included studies on patients who underwent arthroscopic-assisted LDTT with and without surgical antecedents and found that patients with surgical antecedents had inferior functional scores. In the present study, we included only patients without surgical antecedents, but the outcomes of full-arthroscopic LDTT should also be investigated in patients who had surgical antecedents.
Limitations
The results of the present study should be interpreted with the following limitations in mind. While the senior surgeon had performed a sufficient number of arthroscopic-assisted LDTT procedures before the inclusion period, this eliminated the learning curve effects regarding cases of the arthroscopic-assisted technique but not the full-arthroscopic technique, which the surgeon had not previously performed. Despite this, the outcomes might still lack generalizability owing to the steep learning curve. This is a retrospective study in which patients underwent arthroscopic-assisted LDTT from 2016 to 2017 and full-arthroscopic LDTT from 2018 to 2019. Therefore, the complications of the full-arthroscopic LDTT procedures might have been associated with a learning curve. Furthermore, differences in outcomes or complications could be due to slight variations in surgical techniques between the groups, as the arthroscopic-assisted technique transfers the latissimus dorsi tendon only, while the full-arthroscopic technique transfers the latissimus dorsi and the teres major together without intermediate dissection. In addition, the prevalence of teres minor atrophy was significantly greater among patients who had full-arthroscopic LDTT than among patients that had arthroscopic-assisted LDTT. Finally, the cohort size was small, which did not allow for regression analyses, and studies with longer follow-ups are required to confirm long-term safety.
Conclusion
In shoulders with irreparable posterosuperior mRCTs and no surgical antecedents, arthroscopic-assisted and full-arthroscopic LDTT granted equivalent outcomes at a minimum follow-up of 24 months in terms of complication rates (15.4% and 13.2%), conversion to RSA (5.7% and 5.2%), clinical scores, range of motion, and subacromial space. Notably the procedure time was about 18 minutes shorter for full-arthroscopic LDTT. Further analysis revealed different types of complications for arthroscopic-assisted LDTT (1 hematoma and 2 infections) as compared with full-arthroscopic LDTT (2 axillary nerve pareses). The clinical relevance of these findings is that, while full-arthroscopic LDTT could be effective for the treatment of irreparable posterosuperior mRCTs, with the potential to decrease surgical time and reduce hematoma and infections, it may increase risks of nerve paresis.
Footnotes
Final revision submitted November 21, 2022; accepted January 1, 2023.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Ramsay Santé (COS-RGDS-2021-09-006-KANY-J).
Acknowledgments
The authors are grateful to Alexandre Clair for preparing the illustrations, and to Ramsay Santé pour l’Enseignement et la Recherche for funding statistical analysis and manuscript preparation.
