Abstract
Background:
Biologic healing after rotator cuff repair remains a significant challenge. Injectable biologic augmentation may improve tissue quality at the suture-tendon interface.
Purpose:
To investigate the effect of injectable biologic supplementation in rotator cuff repair and to assess the quality and adherence to evolving reporting standards.
Study Design:
Systematic review; Level of evidence, 3.
Methods:
A systematic review was conducted following PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. Included were 40 studies: 29 preclinical (in vivo animal models) and 11 clinical. Each clinical study was assessed for quality, risk of bias, and adherence to relevant MIBO (Minimum Information for Studies Evaluating Biologics in Orthopaedics) guidelines. The outcomes of interest were reported load to failure, load to gap, gap size, and stiffness in the preclinical studies, and healing rate and any patient-reported outcome measures in the clinical studies.
Results:
Injectables reported included growth factors (eg, transforming growth factor–beta 3, erythropoietin), bone marrow–derived mesenchymal stem cells and adipose-derived mesenchymal stem cells (ADSCs), and other agents such as platelet-rich plasma (PRP) and hyaluronic acid. The most common findings for preclinical injectables were increased load to failure (16/29 studies; 55.2%) and improved collagen histological quality (11/29 studies; 37.9%). All 11 clinical studies (10 PRP, 1 ADSC) indicated no adverse events, with similar or improved patient-reported outcomes compared with repairs in the control groups. In 1 study utilizing an innovative delivery technique, a concentrated PRP globule with fibrin matrix was shuttled over a suture to maintain concentrated PRP at the repair site and demonstrated a significant decrease in retears (P = .03) at a 31-month follow-up. A matched-cohort study investigating augmentation with ADSCs demonstrated a significantly lower retear rate in the ADSC-augmented group than the control group at a 28-month follow-up (P < .001). On average, the clinical studies adhered to 66% of relevant MIBO reporting guidelines and had a low risk of bias.
Conclusion:
Approximately 83% of preclinical studies found a positive biomechanical or histological effect, with no studies showing an overall negative effect. Clinically, utilization of innovative delivery techniques may reduce the risk of arthroscopic washout of PRP and improve retear rates. ADSCs were shown to reduce retear rates at a 28-month follow-up.
Rotator cuff tears (RCTs) are a significant source of morbidity, often causing pain, loss of function, and difficulty with activities of daily living. Surgical management of RCTs via rotator cuff repair (RCR) is often indicated after failed nonoperative management given the potential to improve symptoms and function. The disease burden of RCTs has increased as the average population in the United States ages and remains physically active. Day et al 5 demonstrated that the frequency of RCRs has increased by 188% between 2007 and 2015. There continues to be a wide discrepancy in retear rates despite improvement in surgical techniques, with retear rates varying between 11% and 94% depending on tear size. 14,36 Mechanical failure of the RCR is most common within the 3 to 4 months after the initial repair. 26,45 Early postoperative failures suggest that biologic tendon healing may be slower than predicted, and that slow tendon healing may be linked to a failed healing response and decreased vascularity. 30 It has been shown that a subpar anabolic response is more common as preoperative tear size increases. 44 Failed RCR can lead to altered glenohumeral joint kinematics resulting in superior humeral translation, articular wear, and ultimately cuff tear arthropathy. Given the association of failed healing response and decreased vascularity with increased retear rate, methods to stimulate the biological milieu are crucial to decrease the morbidity of early RCR retears. 16
Novel suture materials and anchors, in combination with new surgical techniques such as the double-row transosseous equivalent repair, have enhanced the time-zero mechanical strength of RCRs. 4,62 However, the suture-tendon interface of the RCR continues to have the greatest susceptibility to failure. 51 A consequence of poor tissue quality at the suture-tendon interface is suture pullout and eventual retear. Biologic healing remains a significant challenge as tendon tear recurrence is associated with clinical deterioration in the long-term period. 28,66 Injectable biologic augmentation has been hypothesized to improve tissue quality at the suture-tendon interface.
In an effort to improve biological healing, various anabolic pathways have been targeted using a variety of injectables such as growth factors, platelet-rich plasma (PRP), and bone marrow–derived mesenchymal stem cells (BMSCs) or adipose-derived mesenchymal stem cells (ADSCs). Growth factors such as transforming growth factor–beta (TGF-β) have been shown to promote tenocyte differentiation and improve collagen production. 58 PRP is an amalgamation of growth factors and immunomodulators to ultimately accelerate tissue regeneration. 43 Mesenchymal stem cells also utilize growth factor paracrine signaling to improve wound healing and tissue quality. Additionally, stem cells may differentiate into the damaged tissue to improve the biologic microenvironment and promote healing. 13
Biologic injectables have been used in the treatment of a variety of orthopaedic pathologies, including osteoarthritis, tendinopathies, degenerative disk disease, and fractures. 32 Biologics stimulate the release of growth factor and anti-inflammatory cytokines in order to promote healing, improve tissue quality, and alleviate symptoms. There is considerable variability in the types of biologics and their preparations (eg, hydrogels, saline suspensions, and electrospun matrixes), as well as the reporting of processing details. 37 The United States Food and Drug Administration has created expedited pathways to help support the exploration and utilization of regenerative products, including injectable biologics. 63 This has sparked an improved understanding and implementation of the technologies.
The purpose of this study was to both investigate the effect of injectable biologic supplementation in RCR and assess the quality and adherence to evolving reporting standards.
Methods
A systematic review was performed using PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 46 The search utilized the PubMed central database in January 2022. Two independent researchers performed the search, with the senior author mediating any disputes. Broad augmentation search terms were initially used. The search terms used were ((((microfracture) OR (vented anchors)) OR (platelet rich plasma)) OR (amnion)) OR (adipose tissue)) OR (bone marrow aspiration concentrate)) OR (nandrolone)) OR (augmentation)) OR (biologics)) OR (growth factors)) OR (gene therapy)) OR (stem cells)) OR (tissue engineering)) AND (((rotator cuff repair) OR (single-row repair)) OR (double-row repair)) with filters as follows: date range of January 1, 2010, to January 1, 2022; English language.
The initial search resulted in 1159 titles, to which the following criteria were applied. Inclusion criteria were titles that specified RCR and augmentation techniques in peer-reviewed journals. The exclusion criteria were review articles, systematic reviews, editorials, technique articles, those without reported patient outcomes or the appointed outcome surrogates, case reports, and articles focused on superior capsular reconstruction/not focused on RCR. The abstracts of the articles were then reviewed, excluding duplicates and articles that were case series with <15 patients; review articles not previously eliminated; articles not related to RCR; studies focused on augmentation that did not fall within the 3 categories of injectable, marrow stimulation, or graft/scaffold augmentation; clinical studies with a minimum average follow-up of <24 months; or those without abstract for review. There was no minimum follow-up time for the preclinical studies. This process eliminated 968 articles, leaving 191 for full-text review. Full-text inclusion criteria were clinical studies with a clearly reported healing rate or any patient-reported outcome measure, and preclinical studies with in vivo animal models and clearly reported load to failure, load to gap, gap size, or stiffness. Any remaining studies that were not focused on injectable augmentation were excluded. Based on this criterion, 151 studies were removed. The full-text review resulted in the inclusion of 40 studies (11 clinical and 29 preclinical). A flowchart of search methodology is shown in Figure 1.

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flowchart of the study inclusion process. RCR, rotator cuff repair.
Secondary review of the clinical studies included assessment of adherence to MIBO (Minimum Information for Studies Evaluating Biologics in Orthopaedics) guidelines and assessment of study quality and risk of bias. 37 The methodologic quality of the included primary studies was evaluated using the Cochrane risk-of-bias tool 24 for randomized controlled trials and the Newcastle-Ottawa Scale (NOS) 71 for nonrandomized studies. The NOS assesses the risk of bias in observational studies by employing 7 questions across 3 domains: selection of participants, comparability among studied groups, and outcomes assessment. The studies were then further categorized as having poor, fair, or good quality via recommendation from the Agency for Healthcare Research and Quality, dependent on each study’s NOS or Cochrane risk-of-bias tool scores. Appendix Tables 1 and 2 demonstrate the score determinations for each included study according to the Cochrane risk-of-bias tool and the NOS, respectively.
Results
Preclinical Studies
Twenty-nine preclinical studies ¶ related to injectable biologics were identified. There were 16 studies # (55%) performed in a rat model, 11 studies ** (38%) conducted in rabbits, and 2 studies 9,70 (7%) conducted in mice. Notably, no injectable was found to have a negative effect on outcomes in the preclinical setting.
A variety of growth factors (Table 1) were investigated in the preclinical setting, with 4 of the 8 studies involving TGF-β subtypes. All growth factors, except erythropoietin, were delivered in a gel/scaffold. There was a significant increase in load to failure for all growth factors when compared with the control group (P < .05), except TGF-β3 + ADSC and erythropoietin. 48,57 Interestingly, Reifenrath et al 54 found that TGF-β3 alone led to a load to failure similar to native tissue in their rat model. Another growth factor investigated that has had positive results is granulocyte colony-stimulating factor (G-CSF). Buchmann and colleagues 3 found that sustained release of G-CSF from vesicular phospholipid gels resulted in not only higher load to failure (P = .02), but also a significantly higher collagen 3 content and collagen 1/3 ratio (P < .05).
Growth Factors in Preclinical Studies a

a ADSC, adipose-derived mesenchymal stem cell; FGF-2, fibroblast growth factor 2; G-CSF, granulocyte colony-stimulating factor; TGF-β1, transforming growth factor–beta 1; TGF-β3, transforming growth factor–beta 2.
BMSCs and ADSCs demonstrated both histological and mechanical benefits, including greater load to failure and reduced fatty infiltration (Table 2). Common methods of delivery include gel and saline suspensions. Liu et al 39 investigated the application of bone marrow aspirate concentrate (BMAC) after RCR in a rat model and demonstrated that the addition of BMAC resulted in significantly higher stiffness and load to failure compared with the control group (P < .05), as well as better collagen at the tendon-bone interface. Using a rabbit model, Wang et al 68 showed that adipose stem cell–derived exosomes delivered via phosphate-buffered solution result in significantly higher stiffness and load to failure (P < .05), lower fatty infiltration (P < .001), and more newly regenerated fibrocartilage at the repair site compared with the control group. Overall, stem cell–based biologics both were the most commonly used and demonstrated the most promising results, such as increased load to failure, stiffness, and cellular infiltration.
BMSCs, ADSCs, and Derivatives in Preclinical Studies a
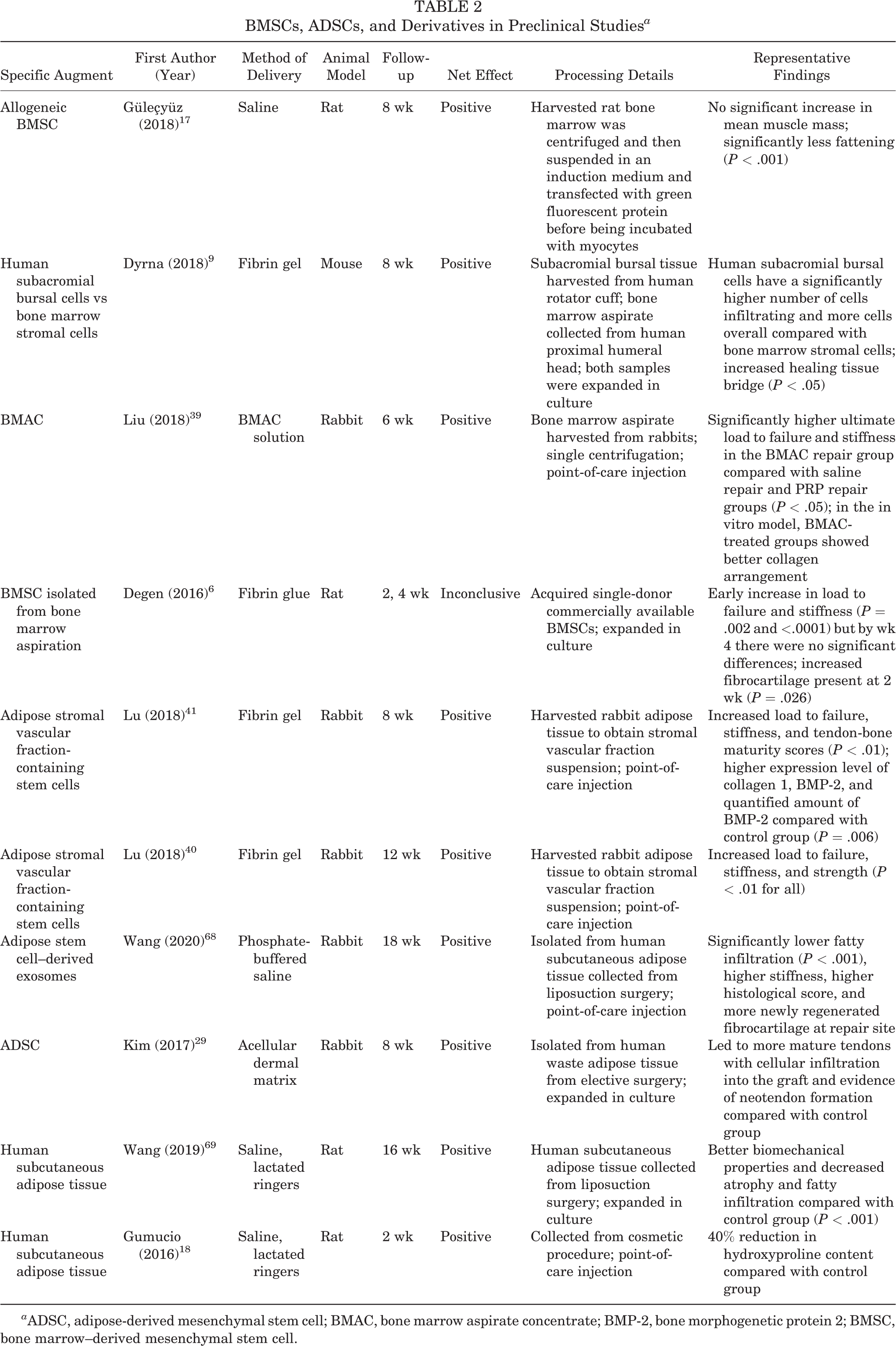
a ADSC, adipose-derived mesenchymal stem cell; BMAC, bone marrow aspirate concentrate; BMP-2, bone morphogenetic protein 2; BMSC, bone marrow–derived mesenchymal stem cell.
A wide range of other injectables have been investigated, including melatonin, hyaluronic acid, and recombinant human parathyroid hormone (Table 3). Gurger et al 19 demonstrated that PRP augmentation can be supplemented with gaseous ozone to improve both biomechanical and histopathological findings in a rat RCR model. The combination of PRP and ozone gas was shown to be a more effective intervention than either PRP or ozone alone. 19 Kwon et al 34 determined that RCR augmentation with human umbilical stem cells improved functional outcomes (eg, walking distance, walking speed) in rabbits. Of note, this was the only preclinical study that investigated functional outcomes in an animal model.
Other Biologic Injectables in Preclinical Studies a
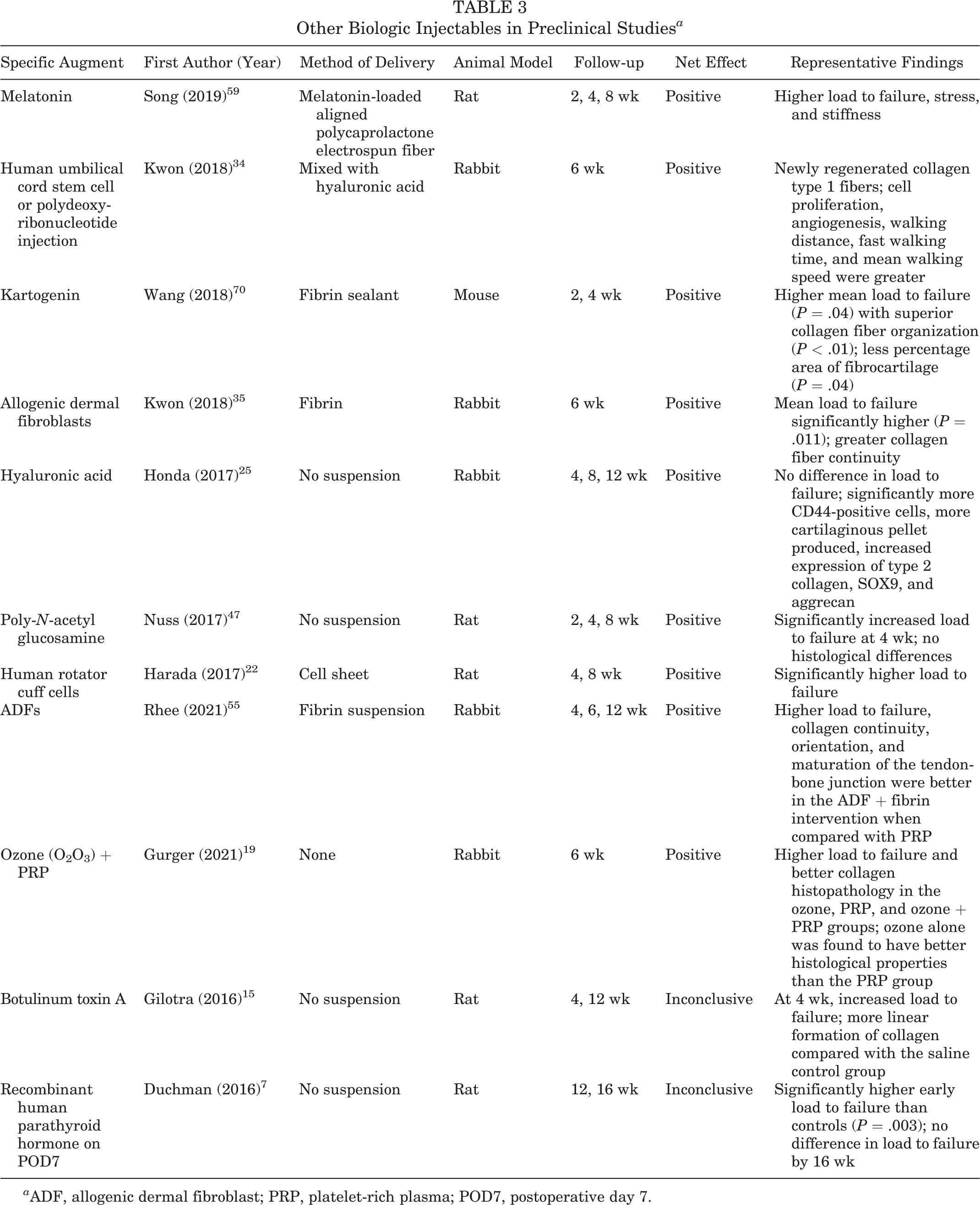
a ADF, allogenic dermal fibroblast; PRP, platelet-rich plasma; POD7, postoperative day 7.
Overall, increased load to failure was the most common biomechanical improvement, with 55.2% of all preclinical injectables demonstrating this finding. Histologically, 37.9% of the studies demonstrated improved collagen quality and/or quantity with the use of injectables. Notably, none of the BMSCs provided lasting collagenous improvements. Five substances (botulinum toxin A, recombinant human parathyroid hormone, TGF-β3 + ADSC, and BMSC from bone marrow aspiration) did not demonstrate any significant improvement in outcomes between the control and intervention groups. 6,7,15,57
Clinical Studies
Eleven clinical studies (10 PRP, †† 1 ADSC 31 ) were identified in total (Table 4). Clinical exploration of injectable biologics in RCR has focused on 2 derivatives, PRP and ADSCs. Intraoperative images demonstrating the injection of a biologic augment into the site of RCR are displayed in Figure 2. Analysis of risk of bias and reporting of relevant MIBO guidelines were performed and the results are shown in Figure 3. The studies were found to have a low risk of bias per NOS and Cochrane assessments. On average, 66% of the MIBO guidelines were adhered to (Table 4).
Injectable Biologics in Clinical Studies a

a ADSC, adipose-derived mesenchymal stem cell; ASES, American Shoulder and Elbow Surgeons; LOE, level of evidence; MRI, magnetic resonance imaging; postop, postoperative; PROM, patient-reported outcome measure; PRP, platelet-rich plasma; SANE, single-assessment numeric evaluation; Ssp, supraspinatus; SST, Simple Shoulder Test; UCLA, University of California, Los Angeles; US, ultrasound; VAS, visual analog scale; WORC, Western Ontario Rotator Cuff.
b This was both a level-1 randomized controlled trial and a level-3 cohort study.

Images of intraoperative injection of biologic augment after rotator cuff repair. A: The rotator cuff repair site visualized by arthroscope. B: Needle inserted into the site of the repair. C: Injection of the biologic augment via the needle at the site of the repair.

Heatmap of adherence to MIBO (Minimum Information for Studies Evaluating Biologics in Orthopaedics) reporting guidelines for the clinical studies reviewed. ‡‡
PRP Studies
PRP is created by rapidly spinning a sample of a patient’s whole blood in a centrifuge to isolate the plasma. In doing so, platelets and other growth factors can be concentrated within the plasma. PRP can be injected intraoperatively to augment healing with autologous growth factors such as insulin-like growth factor–1 (IGF-1), TGF-β, epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), and others. 10 The process of PRP collection and injection is shown in Figure 4. Significant heterogeneity exists in the literature, with multiple trials failing to demonstrate any significant improvement in clinical or radiographic outcomes. 12,20,42,67 Dukan et al 8 in a comparative cohort study of 69 patients investigated the effect of autologous PRP gel, which pointed to a short-term clinical benefit at 3 months that did not persist until the 2-year follow-up. In a randomized controlled trial with 102 patients, Pandey et al 49 demonstrated lower visual analog scale (VAS) pain scores in the PRP group at 1, 3, and 6 months postoperatively, but not at 12 or 24 months. The PRP group demonstrated significantly improved Constant and University of California, Los Angeles (UCLA) scores (P = .001), but not an improved American Shoulder and Elbow Surgeons (ASES) score (P = .131), at 24 months in the PRP group. The retear rate was significantly lower in the PRP group for large tears (P = .014), but not for medium tears (P > .05). 49 The autologous PRP was collected, processed, analyzed, and stored the day before the surgery.

A: Depiction of injection of biologic into the site of rotator cuff repair. B: Process of platelet-rich plasma collection and injection into the repair site releasing growth factors that stimulate tenocyte differentiation and improve collagen quality and quantity. EGF, epidermal growth factor; IGF, insulin-like growth factor; TGF-β, transforming growth factor–beta; VEGF, vascular endothelial growth factor.
To evaluate the effect of repeated PRP application on RCR, Gwinner et al 20 performed a randomized controlled trial of 36 patients with 2-year follow-up, which compared ultrasound-guided injection immediately after the repair and 7 days postoperatively to a control group who did not receive PRP. All patient-reported outcome measures (PROMs) were statistically insignificant, and MRI evaluation of retear rates did not differ between groups at final follow-up (P > .05). 20 Recently, Liu et al, 38 in a 2-part study consisting of a randomized controlled trial and comparative cohort study, investigated the effect of administration of PRP intraoperatively in addition to a 2-week postoperative PRP booster. While patients who received any PRP augmentation had a significantly improved retear rate compared with the control group (12.5% vs 29.2%; P = .038), there were no differences in healing failure rates between the intraoperative PRP-only and the PRP-booster groups at a 1-year follow-up (P = .383). Interestingly, when separated into subgroups based on tear size, only in those with tears >2 cm was the risk of retear significantly reduced in the overall PRP group compared with the control group (P = .04). Additionally, the overall PRP group demonstrated improved VAS pain (P = .016) and VAS satisfaction (P = .023) scores compared with the control group, yet there was no difference between intraoperative PRP-only and PRP-booster groups in range of motion, functional outcomes, or VAS for pain and satisfaction scores at a 2-year follow-up. 38
In a matched case-control study with a 31-month follow-up, Barber et al 2 concentrated a PRP globule onto a fibrin matrix and shuttled the globule over a suture in an attempt to maintain concentrated PRP at the repair site over time. The autologous, leukocyte-poor PRP was prepared with double centrifugation and calcium chloride in a process that resulted in a PRP construct that was sutured into the repair. There was a statistically significant decrease in retears in the PRP group (30% vs 60% in the control group; P = .03) as seen on MRI evaluation. PROMs did not differ between groups with the exception of the Rowe score favoring the intervention group. 2
Randelli et al 52,53 have published both 2- and 10-year outcomes of a cohort of patients who had PRP augmentation of RCR. The PRP was collected using double centrifugation and was delivered at the end of the arthroscopic repair in its activated form. At 24 months, patients had significantly lower VAS scores compared with 3, 6, and 12 months postoperatively. There were initial Constant shoulder score benefits 3 and 12 months postoperatively, but these benefits did not persist at the 2-year mark. Additionally, there was a decrease in retear rates of smaller tears (grades 1 and 2) compared with the control group at the 2-year mark. 52 At the 10-year mark, the PRP group had significantly improved single-assessment numeric evaluation (SANE) and ASES scores compared with the control group. However, there was no difference in retear rates or other clinical assessment outcomes at a 10-year follow-up. 53
ADSC Studies
Kim et al 31 explored the implantation of ADSCs loaded in fibrin glue as a means of RCR augmentation. The adipose tissue was harvested 1 day before surgery, and cells were subsequently cultured. A mean of 4.46 × 106 ADSCs were isolated and loaded into the fibrin glue scaffold. This matched-cohort study demonstrated that the retear rate was significantly lower in the intervention group (14.3%) compared with the control group (28.5%) at a 28-month follow-up (P < .001). However, there were no significant differences in range of motion or patient-reported outcomes between groups. 31
Discussion
The results of this review indicated that preclinically, a large variety of substances have been investigated, the majority of which led to improved repair constructs. Notably, ADSCs, BMSCs, and growth factors including TGF-β and FGF had positive effects. PRP and ADSCs have been researched in the clinical setting with various technique improvements leading to improved patient outcomes.
The MIBO guidelines were published to decrease unknown heterogeneity of compositions in orthopaedic clinical studies utilizing PRP and mesenchymal stem cells. 37 Clinical studies included in this systematic review were found to, on average, follow 66% of the relevant MIBO reporting guidelines. Most commonly absent were the details of the exact PRP composition injected. This likely was because of intraoperative PRP harvesting and processing in many trials. Further adherence to MIBO guidelines in the future may lead to improved identification of specific variables that improve outcomes. Preclinically, no such reporting guidelines have yet been developed.
Injectables reported included growth factors (such as TGF-β3 and erythropoietin), ADSCs, BMSCs, and other agents (namely PRP and hyaluronic acid). Approximately 83% of preclinical experiments demonstrated a positive biomechanical or histological effect. Importantly, there was no overall negative effect seen by biologic injectable augmentation, demonstrating the considerable potential for clinical research translation.
Among growth factors investigated, nearly all showed a net positive effect. In the preclinical setting, the most frequent positive effect was increased ultimate load to failure. TGF-β1 and TGF-β3 currently show the most promise. Both share the same receptor complex and have been proven to play a crucial role in fibrosis and wound healing. 65 Reifenrath et al 54 found that incorporation of TGF-β3 into an electrospun chitosan-coated polycaprolactone fiber scaffold led to biomechanical maximum force values that were similar to those of the native tendon (P ≤ .01). Surprisingly, Reifenrath et al 54 found no improved histological findings with the biomechanical improvements. This is in contrast to the findings of Han et al 21 of improved histology with no biochemical improvements when delivered in tendon-specific cross-linked gelatin. This highlights the importance of testing not only various biologic injectable compounds, but also the vehicle active compound delivery. With further advances in biomaterials, such as utilization of nanocarriers to deliver TGF-β, the expansion of delivery methods shows promise in advancing preclinical development of growth factor delivery. 33
While significant heterogeneity exists in the literature regarding PRP with multiple trials failing to demonstrate any significant improvement in clinical or radiographic outcomes, a recent advancement in the preclinical setting that may spark intrigue is the role of ozone augmentation of PRP injections in RCR. Gurger et al 19 showed that ozone therapy has been investigated in many acute and chronic tendinopathies but has yet to be investigated in RCR. It has been found to increase the release of various platelet growth factors from PRP including PDGF, TGF-β1, and IL-8. Gurger et al found that the combination of PRP and ozone lead to histopathology that resembled a healthy rabbit; additionally, it resulted in the highest biomechanical findings.
Furthermore, delivery of PRP may play an essential role. Both improving PRP preparation and utilizing various modes of PRP delivery such as fibrin gels show potential in improving RCR biologic injectable augmentation and should be further explored as methods to enhance patient outcomes. In 1 study, a concentrated PRP globule with fibrin matrix was shuttled over a suture in an attempt to maintain concentrated PRP at the repair site over time. There was a statistically significant decrease in retears in the PRP group (30% vs 60% in the control group; P = .03) as seen on MRI evaluation. 2 Walsh et al 67 prepared leukocyte-poor PRP through double centrifugation followed by the use of calcium chloride for activation. Randelli et al 52,53 demonstrated that PRP administration utilizing double centrifugation and activation with calcium chloride may lead to increases in ASES and SANE scores at a 10-year follow-up. Both the method of PRP preparation and growth factors within the PRP being released over a longer time period (approximately 30 days) likely contributed to the lower retear rate. Other methods such as sonication, light, and electrical activation should also be investigated to further enhance the platelets’ anabolic properties. 11 Beyond utilizing optimal preparation methods, new biomaterial vehicles of delivery are being developed, such as low-molecular-weight fucoidan-functionalized poly(ester-urea-urethane) scaffolds, which may improve bioactivation of both PRP and the surrounding native tissue. 56 Additionally, tear size may also play a role in deciding whether to augment repair with PRP, as Randelli et al 52 found that only low-grade tears that did not fully retract to the glenoid articular surface benefited from PRP injection at the 2-year mark. Utilization of these innovative delivery techniques may reduce the risk of arthroscopic washout of PRP and has been shown to potentially improve retear rates.
BMSCs and ADSCs have demonstrated an overall positive effect in the preclinical setting. Increased ultimate load to failure and improved histological findings, such as increased fibrocartilage quantity and quality, were found in both BMSCs and ADSCs. BMSCs have shown benefit not only in the short-term recovery period, but also in the long term. Hernigou et al 23 found that at a 10-year follow-up, 87% of the BMSC augmentation group had intact rotator cuffs, whereas only 44% in the control group did. Regarding translation to clinical practice, ADSCs are easier to harvest, may provide fewer complications, and possess higher MSC proliferative capacity 27 ; however, BMSCs have higher chondrogenic capacity. ADSCs are thought to upregulate tendon-related genes through immune modulation and differentiation. 64 Furthermore, a matched-cohort study demonstrated that the retear rate was significantly lower in the ADSC-augmented group (14.3%) than in the control repair group (28.5%) at a 28-month follow-up (P < .001). 31 The combination makes ADSCs a promising injectable augment for RCR. Further work is warranted in determining optimal methods of delivery of ADSCs, as initial results show potential to improve patient outcomes.
Limitations
While our review was comprehensive upon injectables studied for repair, select injectables that do not have strong evidence investigating RCRs but have been shown to affect the supraspinatus tendon were excluded from our review namely for lack of 2-year follow-up or combination protocols; however, they may still warrant attention. Additionally, there is large variability in the preparation and source of injectable biologics coupled with a small sample size of clinical studies that may contribute to result heterogeneity. 61 Regarding randomized controlled trials of PRP, a recent meta-analysis performed by Parisien et al 50 found that the majority of PRP in RCR randomized controlled trials had a fragility index of 4. This implies that reversal of only a small amount of patient outcomes is significant enough to alter trial significance. As suggested, future trials reporting a fragility index and fragility quotient in addition to the P value may lead to a clearer picture of PRP effectiveness in RCR. Despite this relatively limited evidence, RCR augmentation with injectable biologics remains a promising avenue of improving patient outcomes.
Conclusion
Preclinically, a wide range of injectables have been investigated, with approximately 83% of studies demonstrating a positive biomechanical or histological effect and no studies showing an overall negative effect. Clinically, while there remain scant data at long-term follow-up in favor of PRP, utilization of innovative delivery techniques may reduce the risk of arthroscopic washout of PRP and has been shown to potentially improve retear rates. Further, ADSCs have been shown to reduce retear rates at a 28-month follow-up.
Footnotes
Notes
Final revision submitted September 12, 2022; accepted October 21, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: J.F. has received grant support from Arthrex, education payments from Kairos Surgical and Smith & Nephew, and hospitality payments from Stryker. K.M. has received consulting fees and honoraria from Vericel. A.R. has received education payments from Arthrex. G.R. has received education payments from Kairos Surgical, consulting fees from Stryker, and hospitality payments from Arthrex and Wright Medical. S.S.S. has received education payments from Arthrex and consulting fees from Exactech. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
APPENDIX
Cochrane Risk-of-Bias Tool for Randomized Controlled Trials a

| Cochrane Risk-of-Bias Item |
||||||||
|---|---|---|---|---|---|---|---|---|
|
|
1 | 2 | 3 | 4 | 5 | 6 | 7 | AHRQ Standard |
| Malavolta (2018) 42 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
| Walsh (2018) 67 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
| Flury (2016) 12 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
| Pandey (2016) 49 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
| Randelli (2011) 52 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
| Randelli (2022) 53 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
| Liu (2021) 38 | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Low risk | Good quality |
a AHRQ, Agency for Healthcare Research and Quality.
b Items: 1 = random sequence generation; 2 = allocation concealment; 3 = selective reporting; 4 = blinding of participants and personnel; 5 = blinding of outcome assessment; 6 = incomplete outcome data; 7 = other bias.
