Abstract
Background:
Carboplasty is a new minimally invasive technique for knee osteoarthritis (OA) that consists of injecting tibial marrow aspirate into the bone-cartilage interface as well as intra-articularly.
Purpose:
To compare the clinical and imaging outcomes, as well as the safety, of carboplasty for symptomatic knee OA in a placebo-controlled trial.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
The authors conducted a randomized controlled trial to compare carboplasty with placebo for the treatment of symptomatic knee OA. Patients who had failed medical treatment and had bone edema on magnetic resonance imaging (MRI) were randomized in a 1:1 ratio to carboplasty or placebo. The primary outcome of the study was the Numeric Pain Rating Scale (NPRS) for the knee at 1 year (scores range from 0 to 10, with a higher score indicating worse pain). Secondary outcomes were the Knee injury and Osteoarthritis Outcome Score (KOOS), treatment responder rate (based on achieving the minimal clinically important difference of the NPRS), MRI bone edema reduction, and treatment safety.
Results:
In total, 50 patients (25 carboplasty vs 25 placebo) were enrolled and followed up with for an average of 18 months (range, 14-24 months). The average NPRS at baseline decreased from 7.1 ± 0.9 to 2.9 ± 2.1 (P < .001) at 1 year in the carboplasty group and from 7.7 ± 0.9 to 4.9 ± 2.2 (P < .001) in the placebo group. On average, patients after carboplasty improved 60% from their initial NPRS, and patients after placebo improved 37% (P = .003). Patients had a statistically significantly greater improvement from baseline in all KOOS subscales in the carboplasty group compared with the placebo group (P < .001). The responder rates were 96% for carboplasty and 76% for placebo (P = .098). Bone edema was reduced in 72% of patients in the carboplasty group and 44% of patients in the placebo group (P = .045). Neither group had adverse events related to treatment.
Conclusion:
Carboplasty resulted in greater pain reduction, a significantly greater improvement in all KOOS subscales, and a similar safety profile compared with placebo in patients with symptomatic knee OA and bone edema.
Registration:
ISRCTN69838191 (ISRCT Registry).
Osteoarthritis (OA) is the leading cause of lower extremity disability in older adults, with approximately 40% to 47% of people developing symptomatic knee OA during their lifetime. 27 Current treatment options have been directed at improving symptoms, but limited progress has been made regarding the pathophysiology of the disease. 24 Loss of hyaline articular cartilage was interpreted as the main pathoanatomic finding of OA, although interest in the osteochondral unit as a whole (articular cartilage, calcified cartilage, and subchondral bone) has continued to grow. Studies have demonstrated that while the subchondral bone is responsible for some of the nutrition that cartilage receives, a distinct entity called the bone-cartilage interface (BCI), composed of the deep layers of the articular cartilage, maintains the integrity of the osteochondral unit. 15,18,20 All the components in the osteochondral unit have a close relationship, meaning that when one component is affected, the others are harmed.
Bone edema is a common finding in the subchondral bone of patients with knee OA. In an ongoing trial, Kolin et al found bone edema in up to 86% of patients with full-thickness cartilage loss (unpublished data, David Kolin MD, 2022). The mechanism behind this finding is not well understood, but histological evaluation of bone edema has shown microfracturing and bone remodeling accompanied by ischemic cellular changes. 39 Bone edema has been documented as an important element for knee pain and OA progression, reinforcing the idea that OA goes beyond the articular cartilage. 10,13,14,19,29 Since then, studies have suggested bone edema to be a possible treatment target and outcome measure for patients with knee OA.
Carboplasty is a novel treatment that combines the beneficial biochemical properties of bone marrow aspirate (BMA) and the mechanical properties of bone decompression using the percutaneous cartilage bone interface optimization system (PeCaBoo; Vad Scientific). The term “carboplasty” comes from “cartilage” and “bone” interface optimization for preserving, repairing, and regenerating cartilage. The main objective of this technique is to treat OA by addressing the problem directly in the BCI while decompressing bone edema. This technique involves the application of tibial BMA in the femoral and tibial BCI as well as intra-articularly. This procedure is the next step after intra-articular BMA injections in the evolution of the biologic treatment for knee OA. A previous study demonstrated the procedure to be safe and to generate clinical and magnetic resonance imaging (MRI) improvement in a small cohort of patients with a short-term follow-up, 36 but no randomized controlled trial (RCT) has been performed to evaluate the effectiveness of this technique.
The purpose of our study is to compare the clinical and imaging outcomes of carboplasty versus placebo to evaluate the effectiveness of the treatment. This RCT was performed under the hypothesis that carboplasty is safe and leads to greater pain reduction, function improvement, and bone edema reduction compared with placebo in patients with knee OA and underlying bone edema.
Methods
The study was performed according to the CONSORT (Consolidated Standards of Reporting Trials) guidelines and principles of the Declaration of Helsinki. It was approved by the local ethics committee and the internal review board at Ruby Hall Clinic, Pune, India. This trial was retrospectively registered on the ISRCTN website. Written informed consent was obtained from all patients before enrollment.
Study Design
This parallel placebo-controlled RCT was performed at a single institution (Ruby Hall Clinic); patients were recruited from January 1, 2015, to January 1, 2016. All procedures were standardized and performed by 1 of 2 fellowship-trained physicians (R.B. and V.V.). The study inclusion and exclusion criteria are described in Table 1.
Study Inclusion and Exclusion Criteria a

a AP, anteroposterior; MRI, magnetic resonance imaging; OA, osteoarthritis.
b Oral medications, rehabilitation, and orthotics.
Patients who met inclusion criteria were randomized to either carboplasty or placebo at a 1:1 ratio. The radiographic Kellgren-Lawrence system was used to grade the severity of OA using weightbearing anteroposterior, lateral, and merchant views. 23 Patient allocation was performed by a research assistant (K.M.C.) blinded to the treatment using a computerized random-number generator.
The sample-size calculation was made based on the expected difference between means of the Numeric Pain Rating Scale (NPRS) for the knee. The parameters used were the 2-tailed test, a 95% confidence level, 80% power, an expected difference of 2 points, and an SD of 2.1. An expected difference of 2 points was used since a previous study described this to be the threshold in patients with chronic musculoskeletal pain for them to consider that they felt “much better” after routine ambulatory management. 30 The standard deviation was based on a previous study using carboplasty for knee OA. 36 We obtained a minimum population of 36 (18 in each group), but accounting for probable losses, a minimum of 44 patients (22 in each group) were needed. A total of 50 patients (25 in each group) completed the 12-month follow-up (Figure 1).

CONSORT (Consolidated Standards of Reporting Trials) flowchart. The study was powered to find differences on the Numeric Pain Rating Scale for the knee, resulting in 18 patients per group (N = 36); additional patients were added in each group to compensate for probable losses. *Patients completed ≥12 months of follow-up.
Procedure Technique and Rehabilitation Protocol
All procedures were performed under sterile conditions, using intravenous sedation and fluoroscopic guidance. Patients in the placebo group had the PeCaBoo system inserted into the inferior (tibial) and superior (femoral) BCI by itself with no BMA applied either in the BCI or intra-articularly. BMA was not obtained from these patients.
For patients in the carboplasty group, 9 mL of tibial marrow aspirate was obtained from the proximal tibia using the PeCaBoo system and a syringe preloaded with 1 mL of heparin (Figure 2). Using the same syringe and the PeCaBoo system, 2 mL of the 10 mL was injected into the tibial BCI. Another 2 mL was injected into the femoral BCI of the affected side in the knee joint. The remaining 6 mL was injected intra-articularly into the knee joint using the medial approach and a 22-gauge needle.

Carboplasty technique. Left knee as seen under fluoroscopy, with bone edema localized on the lateral femoral condyle. (1) Nine milliliters of bone marrow aspirate is obtained from the tibial tuberosity using the percutaneous cartilage bone interface optimization system (PeCaBoo; Vad Scientific) and a syringe preloaded with 1 mL of heparin. (2) Two milliliters of bone marrow aspirate are injected to the tibial bone–cartilage interface (BCI) using the PeCaBoo system. (3) Two milliliters of bone marrow aspirate injected to the femoral BCI. (4) The remaining bone marrow aspirate is injected intra-articularly using a 22-gauge needle and the regular medial approach.
Patients were instructed not to exercise for 2 days after the intervention. After day 3, patients were allowed to use a stationary bike for 30 minutes every day followed by 15 minutes of cryotherapy. Complete return to activity was permitted after 1 month. Patients in both groups were allowed to continue with their regular pain medications (including nonsteroidal anti-inflammatory drugs) taken before the procedure.
Patient Evaluation
The primary outcome of this study was knee pain evaluated with the NPRS; the secondary outcomes were the Knee injury and Osteoarthritis Outcome Score (KOOS), responder rate, MRI bone edema reduction, and treatment safety.
Patient-Reported Outcome Measures
The NPRS (0 = no pain, 10 = the worst pain) and the KOOS (0 = extreme problems, 100 = no problems) with all its subscales (Symptoms [KOOS-S], Pain [KOOS-P], Activities of Daily Living [KOOS-ADL], Sport and Recreation [KOOS-SR]; and Quality of Life [KOOS-QoL]) were performed before treatment (baseline) and ≥12 months after treatment by an independent nurse blinded to the treatment.
Treatment Responder Rate
A nonprespecified exploratory subanalysis of the responder rate was performed. This was made to evaluate the clinical significance of the pain reduction and not only the statistical significance. Patients were considered responders if they achieved the minimal clinically important difference (MCID) for NPRS, our main outcome. The MCID value was calculated specifically for our studied population using a distribution-based method that consisted of half the standard deviation of our change from baseline NPRS score. 2,21 This method was used since previous studies have recommended calculating one’s own MCID instead of using preestablished values. 2,21
Magnetic Resonance Imaging
An MRI was performed before the treatment to identify the presence of subchondral bone edema and to register the precise location and size. Three months after the intervention, a subsequent MRI of the knee was obtained. Measurements of the bone edema were obtained by measuring the length of the longest axis in the sagittal and coronal views of the MRI, choosing the image with the greatest area of bone edema. Measurements from the pre- and posttreatment MRI scans were compared, and patients were grouped between “reduced” or “not reduced” if the overall size of the bone edema area had decreased. MRI studies were performed using a 1.5-Tesla clinical imaging system (GE Healthcare), using an 8-channel high-definition knee array (GE Healthcare). Standard morphologic MRI evaluation was performed using a fast spin-echo sequence in the axial, sagittal, and coronal planes. Images were acquired with a repetition time of 1800 to 1450 ms, echo time of 30 to 40 ms, echo train length of 6, and spatial resolutions of 256 mm (frequency), 256 mm (phase), and 3 mm at 2 excitations. All MRI evaluations and interpretations were performed by an independent fellowship-trained radiologist blinded to the treatment.
Treatment Safety
Adverse events related to the treatment were documented and reported at each follow-up visit. Major adverse events were defined as any life-threatening condition.
Statistical Analysis
All continuous data with a normal distribution were expressed in terms of mean ± SD; the categorical data were expressed as frequencies and percentages. The Shapiro-Wilk test was performed to assess the normality of continuous variables. All data with normal distribution were compared using the Student t test for continuous variables and chi-square test for categorical variables. The Fisher exact test was used in cases where a cell in a 2 × 2 table had an expected count <5. Nonparametric tests were used to compare data with nonnormal distribution. P values <.05 were considered statistically significant. Statistical analysis was performed by a research assistant (A.M.-I.) using SPSS Version 26 (IBM).
Results
Patient Baseline Characteristics
A total of 50 patients (24 female, 26 male) with a mean age of 65 ± 7 years were randomized and followed up to an average of 18 months (range, 14-24 months). There were no significant differences in the characteristics between groups (Table 2). All baseline KOOS scores were significantly lower in the carboplasty group compared with the placebo group (Table 3).
Characteristics of the Study Patients (N = 50) a
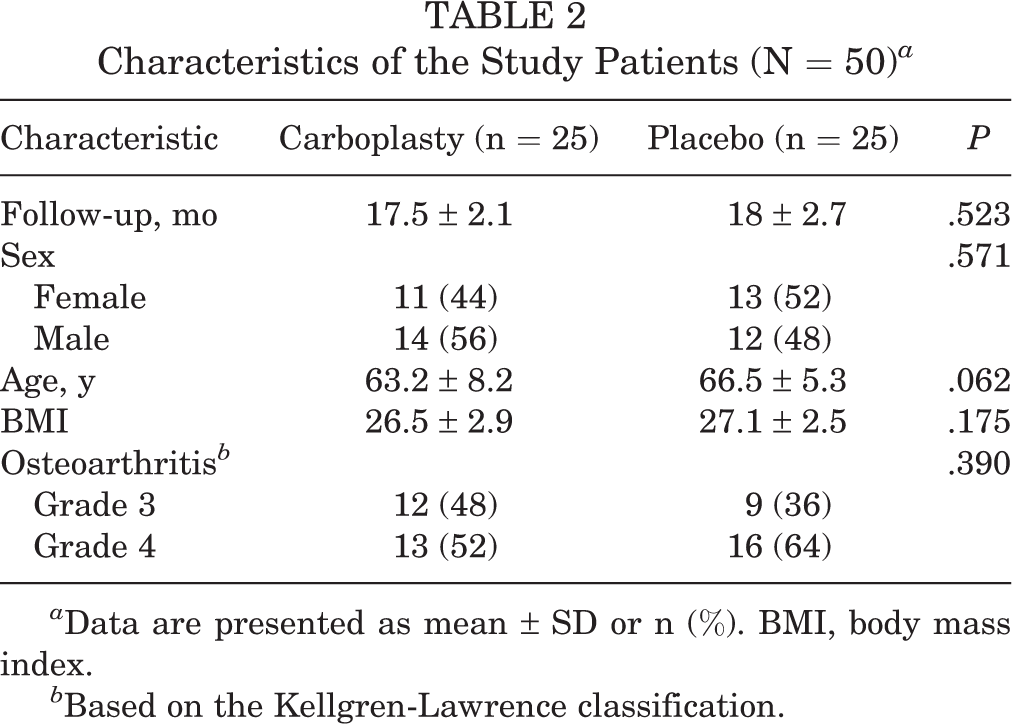
a Data are presented as mean ± SD or n (%). BMI, body mass index.
b Based on the Kellgren-Lawrence classification.
Patient-Reported Outcome Measures a
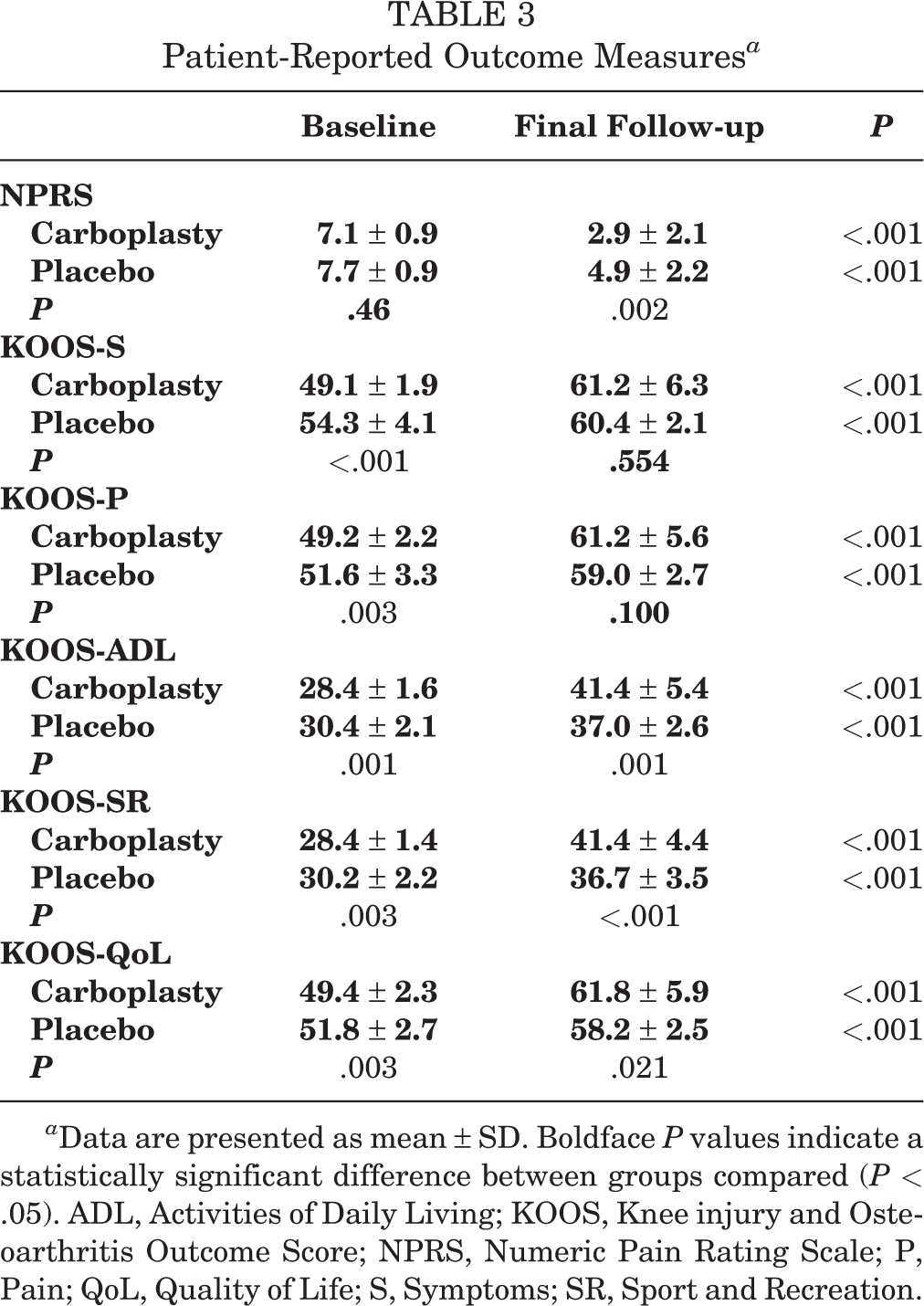
a Data are presented as mean ± SD. Boldface P values indicate a statistically significant difference between groups compared (P < .05). ADL, Activities of Daily Living; KOOS, Knee injury and Osteoarthritis Outcome Score; NPRS, Numeric Pain Rating Scale; P, Pain; QoL, Quality of Life; S, Symptoms; SR, Sport and Recreation.
Patient-Reported Outcome Measures
Both treatment groups showed a statistically significant improvement in all patient-reported outcome measures from baseline to final follow-up (Table 3). The average NPRS in the placebo group decreased from 7.7 ± 0.9 (range, 6-9) to 4.9 ± 2.2 (range, 1-9) at 1 year, and in the carboplasty group from 7.1 ± 0.9 (range, 6-9) to 2.9 ± 2.1 (range, 0-7). On average, patients in the carboplasty group had a greater decrease of the baseline NPRS compared with the placebo group after 1 year (60% carboplasty vs 37% placebo; P = .003). Final KOOS subscale scores (KOOS-S, KOOS-P, KOOS-ADL, KOOS-SR, and KOOS-QoL) were numerically higher after carboplasty compared with placebo and significantly higher only in the KOOS-ADL, KOOS-SR, and KOOS-QoL subscales (Table 3). Patients had a statistically significantly greater improvement from baseline in all KOOS subscales in the carboplasty group compared with the placebo group (P < .001) (Figure 3).

Improvement in Knee injury and Osteoarthritis Outcome Score (KOOS) from baseline. Mean (95% CI) points increased from the baseline scores. ADL, Activities of Daily Living; P, Pain; QoL, Quality of life; S, Symptoms; SR, Sport and Recreation. *Statistically significant difference for carboplasty versus placebo (P < .05).
Treatment Responder Rate
The MCID obtained for NPRS was a 1-point decrease from the baseline score. Of the 25 patients in each group, 96% of the patients in the carboplasty group and 76% of patients in the placebo group were considered responders (P = .098). Nonresponder patients (those with <1 point difference in their final follow-up NPRS score compared with their baseline score) were significantly older (mean age, 72.4 ± 3.4 years for nonresponders vs 63.7 ± 6.8 years for responders; P < .001), had a significantly greater body mass index (BMI) (mean, 28.7 ± 2 nonresponders vs 26.5 ± 2.8 responders; P = .041), and all had grade 4 OA (P = .017).
MRI Outcomes
Bone edema was reduced in 44% of patients in the placebo group and 72% of patients in the carboplasty group (P = .045).
Treatment Safety
No adverse events were reported in either group.
Subgroup Analysis
Further analysis was conducted to determine if different characteristics affected the clinical and MRI outcomes of patients in either group. Despite having a greater severity of OA, both patients with grade 3 and 4 knee OA had a significant clinical improvement from baseline in both treatment arms. Patients with grade 3 OA showed significantly better clinical scores than patients with grade 4 OA at 1 year after carboplasty (NPRS, P = .002; KOOS-S, P = .002; KOOS-P, P < .001; KOOS-ADL, P = .016; KOOS-SR, P = .001; KOOS-QoL, P = .016), but no differences were found in the clinical scores between grade 3 and 4 OA after placebo. Similarly, patients with grade 3 OA were significantly more likely to have a bone edema reduction on MRI evaluation compared with patients with grade 4 OA in both groups (carboplasty, P = .003; placebo, P < .001) Age and BMI had no statistically significant effect on clinical and imaging outcomes.
Discussion
This placebo-controlled, randomized clinical trial of carboplasty in patients with symptomatic knee OA who had failed nonoperative treatment, and with evidence of bone edema on MRI, showed that carboplasty was more effective than placebo in reducing pain at 1 year, based on the NPRS. Secondary outcomes also showed clinical and imaging superiority of carboplasty over placebo. After carboplasty, patients showed a statistically significantly greater improvement from baseline in all KOOS subscales compared with patients after placebo (Figure 3). The treatment responder rate, despite not being statistically different between both groups, was numerically higher in the carboplasty group (96%) compared with the placebo group (76%). MRI evaluation showed that the overall size of bone edema found at baseline decreased in both treatment groups, but significantly more patients had a reduction after carboplasty compared with placebo. Neither group had adverse events related to treatment. To our knowledge, this is the first RCT describing the therapeutic potential and safety of this novel technique targeting the BCI, as opposed to other techniques directed to the subchondral bone.
Previous studies comparing the intra-articular application of biologics for patients with knee OA versus subchondral plus intra-articular application of the same biologic demonstrated that the addition of subchondral injection results in better clinical and imaging outcomes. 17,35 These findings have triggered interest in intraosseous treatments for OA. Three studies have been published using BMA in the subchondral bone for knee OA. 16,17,25 Kon et al 25 used a technique similar to carboplasty but combined subchondral with intra-articular BMA. They reported a statistically significant clinical improvement at 1 year posttreatment based on the visual analog scale for knee pain, KOOS, and the International Knee Documentation Committee score, similar to our findings with carboplasty. Subchondral BMA was shown to decrease bone edema in 61% of patients after 1 year 25 and up to 40% of its original size after 12 years. 16 The reported failure rate of subchondral BMA (commonly defined as the need for surgery after BMA due to persistent symptoms) has been between 11% in the first year and 10% to 20% after 12 years. 16,17,25 Carboplasty, on the other hand, showed bone edema reduction in almost three-quarters of the patients treated, and only 1 patient with insufficient clinical improvement at 1 year (nonresponder). This greater structural improvement and lower nonresponder rate could be explained by the direct application of BMA to the BCI instead of the subchondral bone. By targeting the BCI, the tibial marrow may be acting in all 3 components of the osteochondral unit (articular cartilage, BCI, and subchondral bone) and not limited only to the subchondral bone or superficial cartilage. Hoemann et al 18 suggested that calcified cartilage (or BCI) may block cells and vessels migrating to regenerate articular cartilage, but by slightly perforating the BCI, cells and vessels could migrate freely, reaching the articular cartilage (Figure 4).

Bone-cartilage interface. Frontal view of the right tibia and fibula. On the left side, the image shows a healthy bone with an intact articular cartilage, bone-cartilage interface (calcified cartilage), and subchondral bone. On the right, the image shows an osteoarthritic bone with a significant loss of articular cartilage, a thickened bone-cartilage interface, and neoangiogenesis in the subchondral bone. New blood vessels emerge from the bone marrow trying to regenerate articular cartilage; however, because of the thickened bone-cartilage interface secondary to osteoarthritis, vessels are unable to reach articular cartilage.
Platelet-rich plasma (PRP) is one of the most utilized biologic treatments in orthopaedics, and recent studies have also suggested its application in the subchondral bone for knee OA. Its application in the subchondral bone has shown consistent clinical improvement at 6, 12, and 18 months posttreatment. 26,31,32 Lychagin et al 26 reported that patients had improvement on MRI morphologic evaluation after PRP, but no further information regarding imaging outcomes has been published to our knowledge. However, no RCT comparing BMA with PRP has been performed to determine which treatment is more effective. PRP shares many components of BMA, but the latter has additional cells and a higher concentration of growth factors and cytokines that could favor the use of BMA over PRP. 5,40 Connective tissue progenitor cells (stem cells), despite being 0.01% to 0.02% of cells present in tibial marrow aspirate, 33 have important immunomodulatory effects, trophic abilities, and chondrocyte differentiation potential. 22,37,38 These progenitor cells are primarily secretory cells capable of stimulating preexisting chondrocytes in repairing and regenerating articular cartilage. 4,28,34 Because of these important cells, BMA has been previously suggested to be a more appropriate biologic treatment for OA. 40
Commercially available, minimally invasive and arthroscopic techniques targeting the subchondral bone, such as Subchondroplasty (Zimmer) and BioPlasty (Arthrex), have shown positive short-term outcomes; however, there are some limitations regarding these procedures. 1,3,6 –9,11,12 Subchondroplasty uses calcium phosphate, a nonbiological product that may alter the microarchitecture of the bone, as suggested by Astur et al. 1 BioPlasty, despite using a fully biologic bone marrow concentrate, requires an arthroscopic surgery for its application, making this treatment costly and exposing the patients to unnecessary risks related to surgery. Carboplasty is a minimally invasive ambulatory procedure with minimal costs, downtime, and morbidities, and most importantly, it uses a 100% biologic product directly applied to the BCI. Because of these characteristics, this new technique can be easily used in almost any patient with knee OA and evidence of bone edema.
Limitations
Limitations of this study include a short and nonstandardized follow-up of 12 to 24 months. The placebo treatment used in this study was not a true placebo since patients had the PeCaBoo system inserted in the femoral and tibial BCI without injecting saline and heparin. However, this was performed as such to try to maintain the placebo as pure decompression of the bone edema without the biological component of the technique. A limited MRI evaluation made it difficult to have a clear view of the effects of carboplasty in the articular cartilage and the subchondral bone. No cell count or molecular analysis of the tibial marrow aspirate was performed. There was a possible flaw in the randomization protocol that resulted in significantly different baseline KOOS scores between both groups (see Table 3), which may limit the possibility of drawing conclusions regarding this evaluation. Last, despite being minimally invasive, carboplasty is not an office procedure since it requires sedation and sterile conditions. Future studies evaluating carboplasty with a longer follow-up and a more in-depth MRI evaluation could help us to have a better understanding of the true effectiveness of this technique.
Conclusion
Carboplasty was superior to placebo with respect to pain reduction, KOOS improvement, and bone edema reduction in patients with knee OA and evidence of bone edema on MRI. The combination of mechanical decompression and BMA is promising and worthy of further research, with the hope of having a simple and effective treatment for OA.
Footnotes
Acknowledgment
The authors thank Paul Mulhauser for designing the instruments and Alexa Müller for the drawings.
Final revision submitted August 19, 2022; accepted September 26, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was funded by the Vad Foundation. V.V. is the founder of the biotech company Vad Scientific, and R.B. is a shareholder of Vad Scientific. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Poona Medical Research Foundation.
