Abstract
Background:
Concussion is a common injury among children and adolescents, with a growing body of literature supporting a variety of diagnostic and treatment modalities. Recovery is variable and depends on multiple factors that can be evaluated through a clinic visit: a thorough history, physical examination, and use of the Post-concussion Symptom Scale (PCSS).
Purpose/Hypothesis:
The purpose of this study was to evaluate factors associated with overall recovery from concussion in children and adolescents in the clinical setting. It was hypothesized that the presence of 1 of a number of pre- or postinjury characteristics will be associated with poor concussion recovery.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
We conducted a retrospective chart review of adolescents and children aged 6 to 17 years with a diagnosis of concussion who were evaluated at a single sports medicine center between January 2015 and December 2019. Cases were categorized into recovered (PCSS <7) and poorly recovered (PCSS ≥7) cohorts based on the last PCSS scores during clinical follow-ups for concussion management.
Results:
Of the 162 charts reviewed, 110 cases met inclusion criteria. Significant statistical differences were found between the recovered and poorly recovered cohorts regarding mean days from injury to last clinic visit, previous migraine diagnosis, and emergency room (ER) visit before the first clinic visit (P < .01 for all). Binary logistic regression analysis revealed that the most predictive factors associated with poorer recovery were having an ER visit before the first clinic evaluation (P = .01) and previous migraine diagnosis (P = .04).
Conclusion:
While many factors may contribute to overall recovery from concussion in pediatric populations, our study suggested that a history of migraine and an ER visit before clinic evaluation may be associated with poor recovery of concussive symptoms.
Concussion is a common injury among adolescents participating in sports and recreational activities. In the United States, an estimated 19.5% of adolescents reported at least 1 diagnosed concussion during their lifetime. 34 The mechanism of injury and symptom presentation vary among patients, and a high index of clinical suspicion is needed to make a diagnosis. While a variety of computerized and paper-based neurocognitive tests are available in certain settings and regions, as well as novel laboratory- and image-based diagnostic modalities, these methods of evaluation are typically cost prohibitive and limited in availability. The diagnosis of concussion often remains dependent on clinical presentation. 20 To gain an accurate clinical evaluation, clinicians need to comprehensively consider the patient’s medical and social history (household environment, learning, education background, etc), which influence the response to and recovery from concussion. 8,22,23,29
Recovery from concussion ranges from a few days to a few months, 12,36 with most adolescents showing symptomatic and cognitive recovery in 5 to 15 days. 36 Having symptoms or signs of concussion beyond 28 days postinjury has been defined as prolonged recovery or postconcussion syndrome. 12,17,22 Studies have identified factors associated with prolonged recovery from concussion, including preinjury risk factors, 6,8,22,23,29 signs and symptoms, 17,21,22 and clinical measures. 1,5,6,11,18 Preinjury risk factors were identified as age, sex, prior medical conditions (ie, previous concussion, depression, headache/migraines, learning disorder [LD], and attention-deficit/hyperactivity disorder [ADHD]), and history of concussion. 6,8,22,23,29 However, the results of these studies have demonstrated mixed findings regarding the effects of the preinjury risk factors on concussion recovery. This variety in findings relating potential preinjury risk factors to concussion outcomes in youth is investigated in this study.
Signs and symptoms of concussion at the time of injury, including dizziness and amnesia, have been evaluated as possible contributors to prolonged recovery. 17,21,22 Prior studies have suggested that dizziness at the time of injury increases a patient’s likelihood of prolonged recovery. 17,21 Additionally, 1 study has suggested that amnesia increases the risk of protracted recovery, 21 while other studies did not find the same results. 17,22 Another strong predictor of prolonged concussion recovery is the number of days until first clinic visit: those who present to a clinic within 7 days are less likely to have protracted recovery. 6
Clinical measures such as the Post-concussion Symptom Scale (PCSS), the Sport Concussion Assessment Tool (SCAT), and ImPACT (Immediate Postconcussion Assessment and Cognitive Testing) have been utilized by clinicians and researchers to aid in diagnosis and symptom monitoring. 21,22,29 The PCSS and SCAT may be performed in a variety of clinical and nonclinical settings as they are available as pen-and-paper assessments. The PCSS is a component of the SCAT assessment and requires less time for administration. Studies have demonstrated the clinical validity to use the PCSS for concussion assessment and correlated higher PCSS scores at initial evaluation with prolonged recovery. 21,22,29
Other objective measures, such as near point of convergence (NPC) 1,5 and heart rate, 11,18,26,27 have been explored as potential predictors of protracted recovery. Data examining convergence insufficiency (defined as NPC >6 cm) 24 and increased recovery time after a concussion are inconsistent. 1,5,26 Low heart rate thresholds (the heart rate during exercise that patients are able to reach when their concussive symptoms are exacerbated) have been associated with longer concussion recovery times, especially thresholds <135 beats per minute. 18 In addition, a smaller change between threshold and resting heart rates has been found to be predictive of prolonged recovery. 11 However, there has yet to be a significant number of studies that have investigated heart rate at the initial office visit as a predictor of prolonged recovery.
Knowledge of the factors associated with prolonged recovery is critical to identifying children who may have prolonged recovery after concussion and ensuring timely care. The purpose of this study was to investigate the factors that may be associated with overall prognosis of concussion (ie, good vs poor recovery) in the pediatric population aged between 6 and 17 years. We hypothesized that the presence of preinjury factors, as well as postinjury symptoms (headache and dizziness) and clinical evaluation markers (NPC, heart rate variability), will be associated with overall poor concussion recovery.
Methods
Study Setting
The protocol for this study was approved by an institutional review board. Patients in the study were evaluated and treated at the sports medicine (SM) concussion clinic within the university family medicine center between 2015 and 2019 and were between the ages of 6 and 17 years at the time of initial evaluation. This was a retrospective chart review and therefore the institutional review board approved a waiver of consent from patients/parents. No consent was required or obtained. The SM concussion clinic is a partnership between the SM clinic physicians and the physical therapists within the university family medicine center. Eligible cases for the retrospective chart review and analysis were identified through use of diagnosis codes for concussion and between January 2015 and December 2019. This time frame allowed for consistency among clinical personnel and concussion evaluation practices within the SM concussion clinic. International Classification of Diseases, Tenth Revision (ICD-10) codes identified eligibility by concussion diagnosis (S06.0X0A, S06.0X0D, S06.0X0S, S06.0X1A, S06.0X1D, S06.0X1S, S06.0X9A, S06.0X9D, S06.0X9S). Cases were excluded from analysis if their recovery trajectory was beyond December 2019 or if they were >18 years old or did not show up for their follow-up clinic visit such that only 1 PCSS score was available in their records.
Patients receive a standard workup for concussion, including a physical examination with vital signs and PCSS completion. The PCSS is a symptom severity evaluation tool comprising 22 symptoms commonly associated with concussion that are ranked on a scale of 0 to 6, with larger numbers correlating with more intense symptoms. 3 Patients are asked to complete the scale according to the symptoms experienced on the day of assessment. This study employed the PCSS to define patients who recovered well (PCSS <7) or poorly (PCSS ≥7) from concussion. Iverson et al 14 reported that the mean PCSS in 56 healthy adolescents with a mean age of 17.6 years was 5.23 to 5.79, tested at 2 time points. On the basis of these data, Lau et al 17 utilized a PCSS cutoff <7 in their research, therefore accounting for up to 76% of PCSS scores in uninjured adolescents. 14 Additionally, Eagle et al 6,7 found that PCSS ≥7 differentiated athletes with sports-related concussion from controls when evaluating a cohort of 588 patients aged 13 to 25 years.
A standardized clinic-based questionnaire was employed for each patient regarding medical history and previous diagnosis of migraine, ADHD/LD, benign paroxysmal positional vertigo, and depression. All clinicians evaluating patients under suspicion for concussion are trained in measuring NPC and utilize this measurement as a component of the evaluation. NPC was assessed by having the patient focus on a target (14-point font) approximately 1 m in front of the eyes. As the target was moved slowly toward the patient’s nose, the distance from the nose was recorded when the patient reported double vision or the examiner observed convergency spasm. The result of the NPC assessment was marked N (negative) if <6 cm and P (positive) if ≥6 cm. 24
Patient Selection
Patients selected for the study were between the ages of 6 and 17 years and were evaluated within the SM concussion clinic during our study period. The initial clinic evaluation included a medical history questionnaire, PCSS, and physical examination. Once cases were selected for inclusion, a chart review was conducted to extract patient characteristics. Three reviewers performed data extraction, and if information was missing in the chart, these variables were not included. Last recorded PCSS score, defined as that at the last office visit, was used to define the recovered group (PCSS <7) and the poorly recovered group (PCSS ≥7) based on previous studies. 14,17
A total of 162 individuals with concussion visited the SM concussion clinic between January 2015 and December 2019. After the exclusion of 52 patients, 110 were retained for statistical analysis: 79 in the recovered group and 31 in the poorly recovered group. Of the patients, 31 were identified through the record from physical therapy and 131 were identified through the SM clinic through use of ICD-10 diagnosis codes. A flowchart of the patient selection process is shown in Figure 1.

Study flowchart. PCSS, Post-concussion Symptom Scale.
Statistical Analysis
Descriptive statistics were used to compare demographic data and injury information between the recovered and poorly recovered groups. Missing data points were excluded from statistical evaluation. To compare factors between groups, the Mann-Whitney U test was used for continuous variables, and the chi-square with Fisher exact test was used for categorical variables. Any variable with a statistical probability of P < .20 for a between-groups difference was included in a binary logistic regression to determine which independent variables were most predictive of poor recovery. Odds ratios associated with the poor recovery were computed. All statistical analyses were performed in SPSS (version 25; IBM Corp) with a significance level of P < .05.
Results
Table 1 summarizes the demographics and characteristics of the entire cohort as well as the recovered and poorly recovered groups. In the recovered group, there was a median 29 days from the initial to last clinic visit, while in the poorly recovered group, the median was 65 days. In the recovered group, 65 patients had sports-related concussion, 11 had concussion from motor vehicle accidents, and 3 experienced concussion attributed to a fall/accident. In the poorly recovered group, 17 patients had sports-related concussion, 6 had concussion from motor vehicle accidents, 5 had concussion because of a fall/accident, and 3 had concussion attributed to other causes. As compared with the recovered group, the poorly recovered group displayed a longer time from injury to date of last assessment, more frequent previous migraine diagnosis, a higher likelihood of seeking emergency room (ER) care, and higher PCSS scores at the first assessment (P < .01 for all).
Demographics and Characteristics for the Recovered and Poorly Recovered Groups a
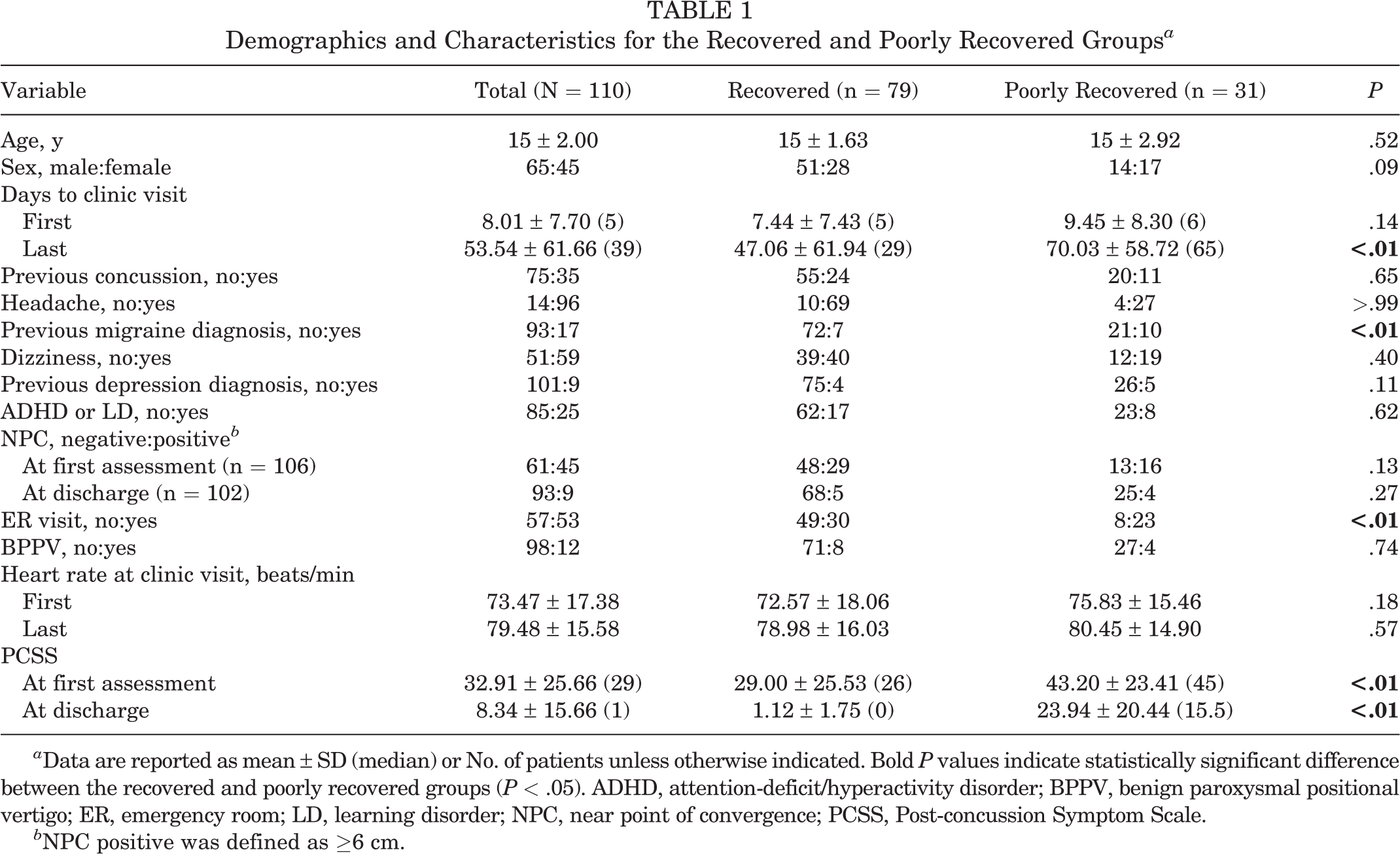
a Data are reported as mean ± SD (median) or No. of patients unless otherwise indicated. Bold P values indicate statistically significant difference between the recovered and poorly recovered groups (P < .05). ADHD, attention-deficit/hyperactivity disorder; BPPV, benign paroxysmal positional vertigo; ER, emergency room; LD, learning disorder; NPC, near point of convergence; PCSS, Post-concussion Symptom Scale.
b NPC positive was defined as ≥6 cm.
With an exploratory P < .20 level, variables were identified for inclusion in a binary logistic regression analysis to investigate the factors associated with poor concussion recovery. Independent variables were as follows: sex, mean days to the first clinic visit, mean days to the last clinic visit, previous migraine diagnosis, previous depression diagnosis, NPC at the first assessment, ER visit, heart rate at the first clinic visit, and PCSS at the first assessment. The PCSS at discharge was not included in the binary logistic regression analysis because it was used to operationally define the groups (0, 1). A significant association between the 8 independent variables and PCSS at discharge was observed (χ2[8, 110] = 24.80, P = .002). The Wald statistics, odds ratios, and 95% confidence interval for each predictor from the model are summarized in Table 2. Having an ER visit (P = .01) and previous migraine diagnosis (P = .04) were the only significant predictors of poor recovery.
Results of the Binary Logistic Regression Analysis a

a Bold P values indicate statistical significance. NPC, near point of convergence; OR, odds ratio; PCSS, Post-concussion Symptom Scale.
Discussion
The study findings demonstrated associations between the presence of a migraine history and an ER visit after injury with poor recovery from concussion in our pediatric population aged between 6 and 17 years. We found that 28% of our cases had poor recovery, with concussion symptoms extending beyond 10 weeks postinjury on average. This percentage of poorly recovered patients is somewhat higher than previous literature, where 1 study found that <7% of patients between the ages of 10 and 17 years were still symptomatic from concussion >2 months out from injury. 33 A study examining a younger population of patients (6-12 years old) recovering from concussion determined that approximately 25% were still symptomatic after 60 days. 30 Our results revealed multifactorial contributions to poor concussion recovery in children and adolescents <18 years of age. The poorly recovered group more commonly reported previous diagnosis of migraine and an ER visit for concussion symptoms before SM concussion clinic assessment. For patients experiencing an ER visit or history of migraine as compared with those without this history, the data support ≥4-fold increased odds of poor recovery.
A history of migraine was previously identified as a contributor to prognostication for duration of recovery from concussion in adolescents and young adults, 6,10,16,32 while other studies have reported no significant relationship between migraine history and duration of recovery. 22,23,29 Meehan et al 22 performed a prospective multicenter cohort study including 182 patients with a mean age of 15 years and compared many variables, such as a history of migraine to time to recovery from concussion, noting no significant influence of migraine history. Miller et al 23 arrived at the same conclusion after completing a single-center retrospective case-control study involving almost 300 participants with a mean age of 13 years. Yet, studies by Terry et al 32 and Guerriero et al 10 did demonstrate an association between migraine and prolonged recovery. Terry et al performed a retrospective cohort study on >1000 student-athletes and found significant differences in time to return to learn and sport with the presence of a migraine history in female athletes but not male athletes. Guerriero et al conducted a prospective cohort study on >500 children, showing significant associations between (1) migraine history, female sex, and age <12 years and (2) prolonged recovery. Notably, most of these studies compared time to recovery, while our study compared good and poor recovery based on the PCSS score, which should be considered when interpreting data. The results of our research support migraine history as a major contributor to poor recovery from concussion, increasing the odds of poor recovery 4.6-fold. This is important as estimates of the prevalence of a migraine diagnosis in the pediatric and adolescent population range from 1% to 11% in children 3 to 11 years old and 8% to 23% by 15 years old, 25,35 encompassing the population of interest for this study. Knowledge of a preexisting migraine diagnosis may help clinicians anticipate time to recovery. We found that 15% of our cohort reported previous migraine, and 48% of those with poor recovery cited a history of migraines, as opposed to ∼10% of the recovered group. As evidence has shown, the pathophysiology behind concussion may be similar to migraine and posttraumatic headache: ionic fluctuations, inflammatory changes, and cell energy deprivation lead to alterations in neurotransmission. 9,15,31 Patients with a history of migraine may be predisposed to a heightened neurovascular response after head injury or triggering of signaling pathways, which may prolong symptoms. 15
Our results indicated that participants with an initial ER visit for their concussion were 3.9 times more likely to have a poor recovery from concussion. Of our poorly recovered patients, 74% indicated an ER visit for concussion as opposed to only 38% in the recovered group. Bressan et al 2 found that pediatric patients presenting to the ER with concern for concussion exhibited ≥2 persistent or worsening of concussion symptoms at 1 month postinjury based on the Post-concussive Symptom Inventory. Similarly, Zemek et al 37 demonstrated that approximately one-quarter of children presenting to the ER for concussion developed postconcussion syndrome. The patients in our study who recovered poorly from concussion and had an ER visit are similar to the patients in the Zemek et al and Bressan et al studies, given the association with poor recovery; however, the total number of children and adolescents who report to our regional ER with concussion and who have poor recovery is unknown. Recovery may be prolonged in this group, owing to the increased severity of injury (utilizing presentation to an ER as a surrogate for severity) or variability in treatment plans. Our data support these findings relating an ER visit with poor recovery and extend the relationship to our pediatric population presenting in the outpatient setting.
Many clinicians evaluate concussion in adolescents without access or skills to interpret results from computerized neurocognitive testing, despite the known benefit of this testing to assist in concussion evaluation and management. 21,22 The findings of this study can be used to guide concussion recovery in children and adolescents using tools readily available in resource-poor settings without access to computerized neurocognitive testing. These tools consist of utilization of PCSS scores, a detailed history, vital signs, and physical examination. The PCSS was preferred over the SCAT given the ease of use and consistency over the numerous versions of the SCAT available throughout the course of the study. Specifically, eliciting presence of care in an emergency department before an outpatient concussion evaluation and personal history of migraine may prove beneficial in predicting recovery course.
We examined many other variables associated with concussion, but none were significantly associated with poor recovery. For nearly all cases, NPC was examined at first presentation and at discharge, given the association between prolonged NPC and concussion. 24 However, unlike previous research, we did not find any significant association between NPC abnormality and poor recovery based on a PCSS ≥7. Pearce et al 26 determined that ∼42% of the athletes had an abnormal NPC within 1 month after sports-related concussion. Our data suggested that NPC may recover after a certain period, although more studies are needed to investigate the time frame for NPC recovery and mechanisms. Moreover, it is not clear how many patients in our data had NPC abnormality before concussion. Additionally, prolonged concussion recovery has been observed in female athletes. 13,23 Yet, our results revealed no significant sex difference in PCSS at final visit. This is consistent with emerging evidence based on recovery times from date of injury in collegiate athletes, which suggests no significant difference in recovery from concussion between the sexes. 19,22 Furthermore, while research has suggested that pediatric patients who present to clinic for evaluation of concussion within 7 days of injury have a speedier recovery, 6 we did not observe differences in time to initial clinic presentation between the recovered and poorly recovered groups. This finding, though, may be subject to bias as the group of patients presenting late to clinic would not include those who had already recovered but rather those with prolonged symptoms. Finally, heart rate variability has a demonstrated association with concussion 28 but was not evaluated in this cohort. Rather, we were able to extract resting heart rate at the first and last clinic appointments, but no difference between the cohorts was observed.
Limitations
The limitations of this study include a single-site study design and challenges innate to a retrospective chart review. The study benefited from collaboration between SM physicians and physical therapists to evaluate and diagnose patients in this pediatric population. Documentation of concussion visits within the clinics follows standard templates, which include the PCSS as well as relevant medical history and concussion-specific physical examination protocols that facilitate retrospective chart review. The SM concussion clinic is among the very few facilities within the region that specialize in concussion evaluation and treatment, increasing the probability of an appropriate, region-specific sample of pediatric patients with concussion. Medical histories such as previous concussion, ADHD/LD, or migraine were obtained from documented patient reporting or electronic health record data. However, the use of medication was not included in our data analysis. Additionally, differentiation of attention-deficit disorder versus ADHD was not determined, since documentation was based on patient and parent recall and not on evaluation of the primary physician encounter providing the initial diagnosis. A recent systematic review revealed no association between ADHD and poor clinical outcomes from concussion, 4 yet more research into the effects of LDs is warranted.
Our team acknowledges other limitations to the study, such as selection bias, the small sample size, and patient self-reporting of many items included for evaluation. Patient charts were identified on the basis of ICD-10 diagnosis codes; given a large number of codes that may be used to define concussion, it is possible that the entire eligible patient population was not captured. Of note, 33 charts were excluded owing to a missing follow-up visit; it is unknown if these excluded charts were of patients equally distributed in demographics, characteristics, and concussion recovery. It is also unknown if patients missed visits because of a full recovery. The PCSS may have been challenging for younger study patients given the scaled nature of assessment, thus potentially affecting scores. Additionally, a PCSS score <7 may not accurately define recovery in all patients, as some may have a baseline score of 0 when not injured. Mechanism of injury was not accounted for in this study, which allows for greater generalizability to patients experiencing concussion from all causes. Despite these limitations and given the broad patient population and significance of study findings, results may be largely generalizable to children and adolescents.
Conclusion
Our study demonstrated that a history of migraine and/or ER visit before an outpatient clinic visit may be associated with overall poor concussion recovery, thus lending support to similar findings in the literature, particularly in the pediatric and adolescent population. However, other factors previously associated with prolonged recovery, such as initial symptoms of dizziness or headache, were not shown in this study to affect time to symptom resolution. The results of this study may be used to advance knowledge of concussion evaluation, prognosticate injury recovery, and guide injury management in the child and adolescent population.
Footnotes
Acknowledgment
Special thanks to Bishop Grant, Cumbo Joshua, Sascha Jilg, and Shelby Boatwright for their help with data collection.
Final revision submitted August 24, 2022; accepted September 16, 2022.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from East Carolina University.
