Abstract
Background:
The application of orthobiologics at the time of arthroscopic rotator cuff repair (RCR) has received an increasing amount of clinical interest despite a relative scarcity of human clinical studies on their efficacy.
Purpose:
To utilize a national administrative database to determine the association of bone marrow aspirate concentrate (BMAC) and platelet-rich plasma (PRP) applied at the time of RCR with revision surgery rates.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
The Mariner data set from the PearlDiver patient records repository was utilized to identify patients undergoing RCR using Current Procedural Terminology (CPT) code 29827. Patients receiving BMAC or PRP at the time of RCR were then identified using CPT coding. For comparison purposes, a matched cohort was created consisting of patients who underwent RCR without biologic augmentation in a 5:1 fashion for each biologic separately. Cases were matched according to age, sex, tobacco use, biceps tenodesis, distal clavicle excision, and subacromial decompression. All groups were then queried for revision RCR or conversion to reverse shoulder arthroplasty. Revision rates were compared utilizing a multivariate binomial logistic regression analysis. Adjusted odds ratios (ORs) and 95% CIs were calculated.
Results:
A total of 760 patients who underwent biologic augmentation during RCR were identified, including 646 patients in the PRP group and 114 patients in the BMAC group. They were compared with 3800 matched controls without documented biologic application at the time of surgery. Compared with matched controls, patients who received BMAC at the time of surgery experienced a significantly lower incidence of revision surgery at 2 years (OR, 0.36; 95% CI, 0.15-0.82; P = .015). There was no significant difference in revision rates between PRP and matched controls (OR, 0.87; 95% CI, 0.62-1.23; P = .183).
Conclusion:
The application of BMAC at the time of RCR was associated with a significant decrease in the incidence of revision surgery. There was no apparent effect of PRP on the incidence of revision surgery after primary RCR. Higher-level clinical studies considering surgical factors are needed to more clearly define the role of biologic adjuvants in RCR.
Keywords
Arthroscopic rotator cuff repair (RCR) remains the gold standard for acute full-thickness rotator cuff tears and chronic tears that fail nonoperative management. However, failures secondary to tendon nonhealing or retear remain high, ranging from 11% to 94%. 12,13,26 The high failure rate persists despite advancements in techniques and implants. 18,23 –25 Risk factors for retear have been well described and include advanced age, 10,22 recent corticosteroid injection, 8 increased critical shoulder angle, 28 increased tear size, 1 and fatty degeneration. 17,27 Despite the high radiographic failure rate, revision surgery is less common. In a study of more than 39,000 patients undergoing isolated outpatient arthroscopic RCR, Mahure et al 30 reported a reoperation rate of 6%. Of these, 57% were revision RCRs. Revision surgery was more commonly performed in younger patients, tobacco users, and those receiving workers’ compensation. 30
Patients with documented tendon-to-bone healing after RCR report improved strength and satisfaction. Accordingly, healing of the tendon is the anatomic goal of the procedure. There is near-universal agreement that optimizing the biologic environment of the tendon-to-bone interface is a key to improving cuff repair healing. Thus, surgeons have begun to explore biologic augmentation of RCRs in order to improve primary healing rates. Small randomized controlled trials (RCTs) evaluating platelet-rich plasma (PRP) administered at the time of RCR have shown increased healing rates. 19,20,34 However, similar studies have shown conflicting results. 11,31,39 Malavolta et al 31 evaluated 44 shoulders after arthroscopic single-row RCR for small to medium supraspinatus tears, in which half of the patients were treated with concurrent application of PRP to the tendon-to-bone interface at the time of surgery. They found that at 60 months after surgery, magnetic resonance imaging (MRI) evaluation showed no difference in the retear rate. Other authors have evaluated the effect of bone marrow aspirate concentrate (BMAC) on the function and healing of patients undergoing arthroscopic RCR. Cole et al 9 reported on 62 patients randomized to concurrent treatment with BMAC (n = 28) compared with a blinded control group (n = 34) and reported improved tendon quality on postoperative MRI in patients who received additional BMAC at the time of RCR. Similar improvements have been shown with local microfracture of the greater tuberosity at the time of RCR, which allows venting of bone marrow stem cells, growth factors, and other proteins to the repair site. 32 Studies have demonstrated both improved healing and diminished retears in patients undergoing RCR with microfracture compared with patients undergoing isolated RCR. 32,35
Studies on both PRP and BMAC are limited by small numbers and are at risk of data fragility and being underpowered given the low incidence of retearing in small cohorts. Limitations of these types of studies are common given the cost of RCTs. Given these limitations, larger cohort studies utilizing administrative databases offer unique strengths and an increased power to examine the effectiveness of biologic augmentation at the time of RCR.
The primary purpose of this study was to use a national administrative database to determine the association of BMAC and PRP applied at the time of RCR with revision surgery rates compared with matched controls. We hypothesized that the application of PRP and BMAC at the time of surgery would be associated with a significant reduction in revision shoulder surgery rates at 2 years compared with matched controls.
Methods
Database
This study was deemed exempt from institutional review board approval. A retrospective case-control study utilizing a national database of deidentified patient information (PearlDiver Patient Records Database; www.pearldiverinc.com) was performed. The Mariner shoulder arthroscopy data set within PearlDiver was used for the current study, as it contains all patients with a shoulder arthroscopy procedure and contains patient records from several private insurers, including 100% of Medicare patients, as well as Medicaid patients. Similar to the broader PearlDiver data set, this database provides procedural volumes and patient characteristics for patients with International Classification of Diseases, 9th Revision (ICD-9) and 10th Revision (ICD-10) diagnoses and procedures or with Current Procedural Terminology (CPT) codes. Patients are tracked across all locations of care as long as the same insurance is used for the encounter.
Study and Control Cohorts
To identify the study population of patients who received BMAC or PRP at the time of primary RCR, the database was first queried for all patients who underwent primary RCR between 2015 and 2018 using CPT code 29827. Patients undergoing open RCR were excluded. Patients who received BMAC at the time of surgery were identified using CPT code 38220, and patients who received PRP, with CPT code 0232 T. All primary RCRs were required to have been performed 2 years before data collection (2020), representing a minimum 2-year follow-up. Patients who underwent a primary RCR without BMAC or PRP augmentation were then identified and matched 5:1 by age, sex, tobacco use, and concurrent procedures (biceps tenodesis, distal clavicle excision, and subacromial decompression) individually for each of the BMAC and PRP study groups.
Outcomes Evaluated
The primary outcome studied was revision shoulder surgery in the form of revision RCR or reverse shoulder arthroplasty at any time after the primary surgery. The procedures were identified using CPT code 29827 for the ipsilateral shoulder after the index procedure for revision RCR and CPT code 23472, ICD-9 code 81.88, and ICD-10 codes 0RRJ00Z and 0RRK00 for reverse shoulder arthroplasty. To ensure that procedures occurred on the ipsilateral shoulder, CPT code laterality modifiers were verified.
Data and Statistical Analysis
Patient characteristics of the study and control cohorts were recorded as provided by the database, or as defined by relevant ICD-9 and ICD-10 codes after matching, and compared. These included age group, sex, obesity, morbid obesity, tobacco use, alcohol use, concomitant procedures, diabetes mellitus status, hyperlipidemia, hypertension, peripheral vascular disease, congestive heart failure, coronary artery disease, chronic kidney disease, chronic lung disease, chronic liver disease, thyroid disease, and depression. Each of these categorical comparisons was performed using a chi-square test. Statistical comparisons of revision shoulder surgery between cohorts were completed using a multivariate binomial logistic regression analysis controlling for the previously mentioned covariates; adjusted odds ratios (ORs) with respective 95% CIs were calculated for all comparisons. For all statistical comparisons, P < .05 was considered significant. The embedded statistical software within PearlDiver was used for all statistical comparisons, which is based on the open-source R program (R Foundation for Statistical Computing; www.r-project.org).
Results
We identified 760 patients who underwent biologic augmentation during primary RCR, including 646 patients in the PRP group and 114 patients in the BMAC group. The PRP cases were performed by 167 surgeons with a mean follow-up of 2.2 ± 0.9 years. BMAC-augmented cases were performed by 39 surgeons with a mean follow-up of 2.3 ± 0.7 years. Each group was matched to a similar group of control patients undergoing RCR without documented biologic application at the time of surgery (3230 matched to the PRP group; mean follow-up, 2.7 ± 1.1 years; and 570 matched to the BMAC group; mean follow-up, 2.7 ± 1 years). There were no significant differences in the reported characteristics, concomitant procedures, or comorbidities between the PRP group and the matched controls (P > .05) (Table 1). Similarly, there were no significant differences in the reported characteristics or concomitant procedures between the BMAC group and the matched controls. There were also no significant differences in the comorbidities studied between the BMAC group and matched controls with the exception of a significantly higher rate of hypertension in controls (P = .034) (Table 2).
Patient and Surgery Characteristics of the PRP Group Versus Matched Controls a

a Values are presented as n (SD). BMI, body mass index; PRP, platelet-rich plasma.
Patient and Surgery Characteristics of the BMAC Group Versus Matched Controls a
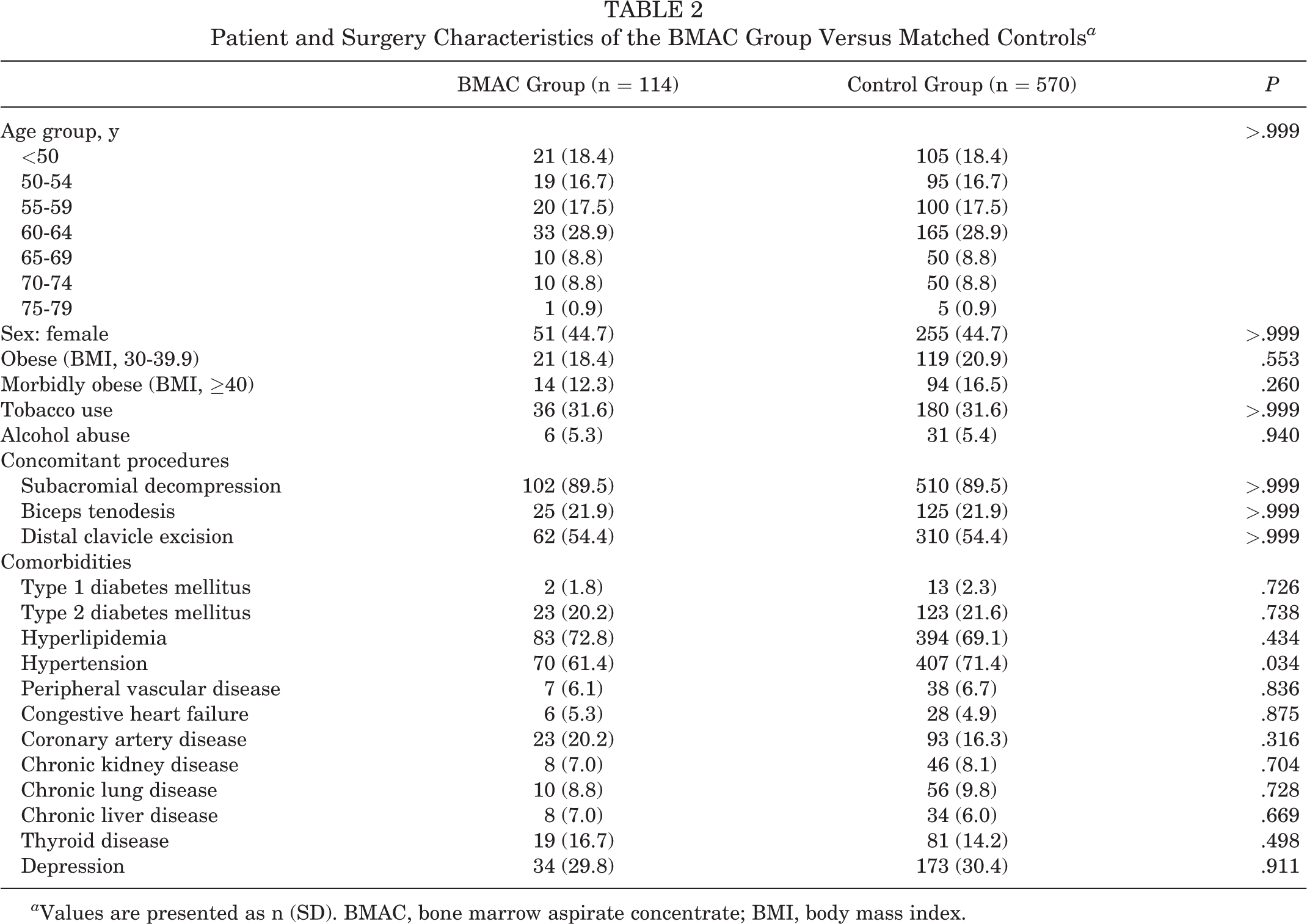
a Values are presented as n (SD). BMAC, bone marrow aspirate concentrate; BMI, body mass index.
Two shoulders undergoing RCR with BMAC (1.75%) underwent revision RCR. Compared with matched controls, patients who received BMAC at the time of surgery experienced a significantly lower incidence of revision surgery (1.75% vs 3.51%; OR, 0.36; 95% CI, 0.15-0.82; P = .015). This represents a 2.8 times increased likelihood of having revision shoulder surgery in controls compared with those receiving BMAC at the time of RCR. In total, 21 patients treated with RCR and PRP (3.25%) underwent revision RCR. There was no significant difference in revision rates between the PRP study group and matched controls (3.25% vs 3.56%; OR, 0.87; 95% CI, 0.62-1.23; P = .183).
Discussion
In the past decade, orthobiologics have gained increasing clinical interest as adjuncts during arthroscopic RCR to potentiate tendon healing and reduce the risk of clinical failure. 14,38 The current study adds to a growing body of literature on this topic and suggests that BMAC application at the time of RCR was associated with a significantly lower risk of revision surgery. A similar comparison of PRP applied at the time of surgery did not show a similar association, with no observable difference seen in revision rates in patients who received PRP and those who did not. Further prospective studies on the effect of BMAC administered at the time of RCR are needed to confirm these findings.
BMAC has rapidly emerged as a popular biologic therapy to augment tendon-to-bone healing during RCR. In addition to possessing several of the same favorable concentrations of growth factors found in PRP, BMAC also contains progenitor cells, which have been shown to facilitate tissue healing. 29,33,36 While multiple animal studies have demonstrated BMAC’s ability to augment tendon repair by improving enthesis regeneration and repair quality, high-level human clinical studies are scarce. 29,38 Despite being scarce, the available clinical investigations have shown promise. 38 In one of the first clinical studies, Hernigou et al 15 investigated 90 matched patients who received a single-row RCR for small tears, half of whom received BMAC at the completion of the repair and the other half who did not. At 6 months, all 45 patients who received BMAC had evidence of healed repairs on MRI compared with only 67% of patients who did not receive BMAC. Furthermore, at 10 years after surgery, 87% of the BMAC cohort maintained intact repairs compared with only 44% of patients who did not receive BMAC. 15
More recently, Cole et al 9 performed a prospective randomized trial of 62 patients undergoing repair of a full-thickness isolated supraspinatus tear. When comparing the 28 patients in the BMAC group with the 34 control patients, patients who received BMAC at the time of surgery exhibited significantly improved Sugaya scores on postoperative MRI at 1-year follow-up (3.25 vs 2.6; P = .0012). Thus far, human clinical studies have primarily focused on the impact that BMAC has on the MRI appearance of tendon-to-bone healing, and this is the first report on a difference in revision rates. 38 While it is important to report the effect that BMAC has on tendon healing as seen by MRI, it is well known that structural failure may not often manifest clinically. 21 The difference in revision rates shown in this study suggests that the differences seen on MRI may be a marker for higher failure rates and further support the consideration of insurer-covered cost of BMAC augmentation. In the current study, we were able to study the largest number of patients to date undergoing RCR with BMAC augmentation to demonstrate a nearly 3-fold difference in revision rates in patients who received BMAC compared with those who did not.
Overall, the majority of the clinical literature examining the application of biologics at the time of RCR has focused on PRP. 38 And although individual studies have produced promising results, pooled data from these studies have remained inconsistent. 7,16,37 Hurley et al 16 recently performed a meta-analysis of 18 RCTs and 1147 patients and reported that patients receiving PRP experienced significantly lower rates of incomplete healing, lower pain levels, and improved functional outcomes. Similarly, Chen et al 7 focused exclusively on level 1 RCTs examining the effects of PRP on RCR and reported a significant decrease in long-term retear rates in PRP-treated patients. In contrast, Saltzman et al 37 performed a systematic review and meta-analysis that included 3193 patients and reported that PRP use at the time of RCR did not universally improve retear rates or affect clinical outcome scores. One factor that has contributed to the inconsistency seen in PRP application at the time of RCR is the significant heterogeneity in both the preparation of PRP and the reporting of PRP characteristics between studies in the published literature. 6 This, coupled with differing injection protocols, has limited the ability to effectively compare results between studies and deliver a consistent message about its utility. Furthermore, similar to the BMAC literature, few studies have used revision surgery as an outcome measure. In the current study, there was no significant difference in revision surgery rates between patients receiving PRP at the time of surgery and those who did not.
The findings of the current study indicated a significantly lower revision rate in patients who received BMAC at the time of surgery, but not when they received PRP. To our knowledge, few human clinical studies have sought to compare these 2 adjunctive therapies. 14,38 Liu et al 29 performed an in vivo animal study evaluating the influence of BMAC and PRP on tendon-to-bone healing in a rabbit chronic rotator cuff model. The group reported that rabbits treated with BMAC exhibited enhanced mechanical strength of the tendon-to-bone junction and histologically improved biologic healing compared with rabbits treated with PRP at the time of repair. In addition, collagen fiber continuity at the enthesis was similar between rabbits treated with PRP and those treated with saline. The group postulated that isolated mesenchymal stem cells or PRP alone may be insufficient to support the tissue repair and regenerative process needed to support tendon-to-bone healing and that an environment encouraging differentiation and recruiting growth factors, possibly provided by BMAC, is necessary to see such an effect. 29
Whereas basic science studies investigating the effect of PRP on tendon healing have reported the most significant improvements in early, acute tendon healing, differences between acute and chronic tendon injury should be kept in mind when considering why PRP failed to show consistent success in chronic RCR applications and in the same rabbit model. 29 While the biological effects of PRP on RCR healing are reasonably well studied, BMAC has been less so. Nevertheless, studies have demonstrated the ability of BMAC to concentrate theoretically important factors at an efficient level. Additionally, differences have been demonstrated between the 2 products in vitro. 5,41 Most notably, BMAC has a much higher concentration of interleukin receptor 1 antagonist protein, among others. While it is unclear how the differences in growth factor quantities between the 2 products may affect in vivo healing, these differences in the biological makeup are but 1 of several hypotheses for why there may or may not be a true difference in the effect of PRP versus BMAC when applied to RCR. Ultimately, more basic science, animal, and human studies are needed to fully understand the impact of these biologics on RCR outcomes.
Limitations
The results of the current study must be viewed within the context of the study’s limitations, many of which are inherent to administrative database research. 4,40 First, the strength of our analysis and reliability of our results were dependent on the quality of the data and the accuracy with which the selected procedures and diagnoses were coded. Thus, miscoding, noncoding, and direct cash payments to providers are potential sources of error. A major limitation of utilizing an administrative database to study biologics is the inability to determine the technique of procurement, preparation, and administration of the biologic utilized, much less the characteristics or concentration of that being used from patient to patient. Furthermore, we were only able to assess failure as defined by revision surgery. Patients with poor clinical outcomes and structural failure without revision would not have been captured as having structural or clinical failures using this database.
A second limitation was that, although we performed a matching algorithm to limit the effects of patient-related covariates that have been shown to increase RCR failure rates, we were unable to control for multiple other variables that affect RCR failure (eg, tear size, fatty infiltration, and repair technique). Third, clinically significant endpoints in administrative data sets are identified using CPT and ICD coding. The use of both ICD-9 and ICD-10 and the coding changes that resulted from changing from ICD-9 to ICD-10 may potentially have affected our results. Thus, we were unable to identify the structural failures identified on MRI or clinical failures based on outcome scores from the database. Last, we were unable to evaluate the techniques used for the preparation and placement of both PRP and BMAC. There are currently large variations in preparation techniques that are known to affect cell counts and quality. Despite these limitations, we believe that an administrative database is a useful and powerful method to examine outcomes associated with biologic applications because of its ability to study a large number of patients across a representative population.
Despite the limited number of cases listed in the PearlDiver database, there remains some relative value to the differences in BMAC outcomes. As mandated by the 21st Century Cures Act, which was passed by Congress and was signed into law in late 2016, the US Food and Drug Administration has stated that it will consider real-world evidence (RWE) and real-world outcomes (RWOs) when reviewing information on regenerative medicine treatments. 2,3 RWE and RWOs are gathered outside of conventional clinical trials and include data obtained from patient charts, laboratory reports, patient registries, pragmatic clinical trials, surveys, and mobile health devices. RWE and RWOs complement evidence obtained from RCTs and provide information about the long-term safety and effectiveness in large populations in a more natural setting. Furthermore, RWE and RWOs allow stakeholders and health insurance companies to assess the risk-benefit and economic value of medical interventions.
Conclusion
The application of BMAC at the time of RCR appears to be associated with a significant reduction in the incidence of revision ipsilateral shoulder surgery. The difference in RCR revision rates for BMAC-treated shoulders represents early RWE in favor of biologic augmentation of RCR that should be considered alongside the outcomes of prospectively designed trials.
Footnotes
Final revision submitted May 16, 2022; accepted July 5, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: B.S.S. has received education payments from Arthrex, consulting fees from Exactech, and honoraria from Encore Medical. B.C.W. has received education payments, consulting fees, and speaking fees from Arthrex and hospitality payments from Integra LifeSciences. C.C. has received nonconsulting fees from Arthrex. P.C. has received education payments from Active Medical, consulting fees from DePuy/Medical Device Business Services, and nonconsulting fees from Arthrex. J.C. has received research support from Arthrex, education payments from Arthrex and Smith & Nephew, and hospitality payments from Medical Device Business Services and Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
