Abstract
Background:
Superior capsular reconstruction (SCR) is an option for patients with massive or irreparable rotator cuff tears.
Purpose:
To describe the literature on rehabilitation protocols after SCR of rotator cuff tears, with emphasis on the timing of the introduction of motion.
Study Design:
Scoping review; Level of evidence, 4.
Methods:
We conducted a scoping review of articles published on PubMed, Ovid, Embase, and the Cochrane Library from inception to October 2020. The methodological index for non-randomized studies (MINORS) was used to assess the individual studies. For each article, we summarized the study characteristics, patient demographics, and rehabilitation protocols after SCR: duration of immobilization, initiation of passive range of motion (ROM), active-assisted ROM, active ROM, strengthening, and return to activities. In a subgroup narrative analysis, rehabilitation protocols were stratified by graft type: autograft versus nonautograft (xenograft, allograft, and synthetic).
Results:
A total of 21 studies met the search criteria. Six studies had level 3 evidence and 15 had level 4 evidence; 16 studies were considered high quality according to the MINORS score. After SCR, an abduction immobilizer was recommended for a duration of 3 to 6 weeks. Of the 21 studies, 7 (33%) started passive ROM during the first week, and 5 reported strict immobilization without motion for up to 6 weeks. All 8 studies that reported return-to-sports timing recommended delaying return until at least 6 months postoperatively. Passive ROM was recommended earlier for patients with nonautograft versus autograft (χ2 = 225; P < .001). There was a high level of heterogeneity in the rehabilitation protocols after SCR.
Conclusion:
The majority of published protocols were descriptive. At present, there is little agreement on the published rehabilitation guidelines after SCR, precluding specific clinical best practice suggestions. Although there was a tendency of recommending early motion in nonautograft cases, the optimal protocols based on graft healing and functional outcomes require further verification based on the clinical outcomes from high-quality studies.
The treatment of irreparable rotator cuff (RC) tears has long been considered a challenge. Since Mihata et al 24 reported that early superior capsular reconstruction (SCR) using a fascia lata autograft achieved good early results, many studies have cited successful clinical outcomes for SCR in large to massive RC tears. 6,10
Based on previous studies, tendon healing after RC repair is affected by patient age, comorbidities, quality of RC tissue, repair technique, tear size, activity level, rehabilitation, and other factors. 3,22 In terms of surgical technique, SCR is similar to augmented repair with an allograft or autograft. 30 Thus, tendon healing and clinical outcomes after SCR are also likely affected by postoperative immobilization and rehabilitation. Currently, most SCR surgical procedures are applied in irreparable RC tears or the revision of massive RC tears. 8,35 This means that patients undergoing this procedure usually have an RC with a worse condition or quality when compared with those undergoing a common repair. It remains unclear whether the rehabilitation protocol for RC repair, with or without the augmented procedure, equally applies for patients with an irreparable RC undergoing SCR, especially when the graft type is considered. 9 Thus, rehabilitation protocols after SCR should be reevaluated per the present literature in the area of SCR.
Although there are multiple published rehabilitation protocols, including those based on thorough biomechanical rationales for SCR, 12,23 there is little agreement on the types and timeline of physical therapy interventions. 13 Given the rising prevalence of SCRs within the health care setting, it may be useful to perform a scoping review of the literature to determine the extent of the consensus and level of evidence for postoperative rehabilitation.
Methods
The review of the literature was performed according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 26
Literature Search
The articles were searched from the PubMed, Embase, Ovid, and Cochrane Database. The final search was performed on September 16, 2020, using the following terms: (superior AND (capsule OR capsular) AND reconstruction). We also reviewed the reference lists of relevant studies to identify those that may not have been found during our literature search. Initially, the title and abstract of each study were reviewed. Clinical studies that assessed the outcomes of SCR for “rotator cuff tear” were reviewed more thoroughly by 2 independent reviewers (K.Z. and Q.X.). Any disagreement during the selection process was resolved through group discussion or intervention by a third reviewer (S.L.).
Eligibility Criteria
The inclusion criteria were SCR treatment, a description of the postoperative rehabilitation protocols after SCR, a full-text article written in English, and publication in a peer-reviewed journal. The exclusion criteria were (1) case reports, reviews, technique descriptions, editorial letters, expert opinions, or cadaveric studies; (2) studies without a thorough report of the postoperative rehabilitation protocols; and (3) articles written in a language other than English.
Data Extraction
After the assessment for the study selection, 2 independent reviewers (K.Z. and Q.X.) extracted the data from the selected articles using Microsoft Excel (Version 2016). The collected data were as follows: first author, year, country, journal, level of evidence, sample size, age, graft source and types, and rehabilitation program (rehabilitation timing, precautions, and venue).
Quality Assessment
The risk of bias for each study was independently assessed by 2 authors (K.Z. and Q.X.) using the methodological index for non-randomized studies (MINORS) score. The MINORS evaluation tool consists of 8 common items plus 4 additional items for research with a comparative group. Each item can have a score of 0, 1, or 2, with a maximum score of 16 points for noncomparative studies and 24 points for comparative studies. A study that achieves >60% of the total possible score (15/24 for comparative studies or 10/16 for noncomparative studies) indicates that it is of high quality and has a low risk of bias.
Data Analysis
Given the heterogeneity and nonuniformity of the data in the studies, the results were descriptively summarized. The descriptive statistics, including the means and ranges, were calculated using Microsoft Excel (Version 2016). In addition, we compared the initiation of passive range of motion (pROM) according to graft type, stratified into autograft and nonautograft (ie, allograft, xenograft, synthetic graft). We focused on the initiation of pROM rather than active-assisted range of motion (aaROM), active range of motion (aROM), or strengthening, because the progression of the subsequent procedures was generally directly or indirectly influenced by the initiation and duration of pROM. Furthermore, this indication was the most reported in the area of rehabilitation after SCR and RC repair.
The comparison of categorical variables was conducted using chi-square test or Fisher exact test. P < .05 was considered statistically significant.
Results
Study Selection
The literature search conducted on the PubMed, Ovid, Embase and Cochrane library, with the query of major orthopaedic journals, revealed 965 individual titles and abstracts, including duplicates. After the initial screening of the titles and abstracts and the removal of duplicates, 878 studies were excluded, leaving 89 articles for full-text review. After a thorough review of these articles and their citations with a repeat search of the literature, 21 studies were included in the review. § Figure 1 summarizes the process for the study selection, and the characteristics of the studies are presented in Table 1.

Flowchart of the study selection process.
Characteristics of the Included Studies a

a LHBT, long head of biceps tendon; LOE, level of evidence; MA, mesh augmentation; MINORS, methodological index for non-randomized studies; TFL, tensor fasciae latae.
b High-quality study with MINORS score exceeding 60% of the total: 10 of 16 points or 15 of 24 points.
c The data were presented as mean, mean ± SD, mean (range) OR mean ± SD (range).
Risk of Bias
The MINORS scores of the studies are presented in Table 1. Six comparative studies 4,15,16,23,28,34 were assessed as having level 3 evidence, while the remaining studies were case series with an evidence level of 4. The median MINORS score for the 21 studies was 10, with 16 studies ∥ considered high-quality studies. However, none of these studies performed prospective collection of data or had an unbiased evaluation of endpoints.
Rehabilitation Protocol
All but 1 article 19 described the immobilization protocol after SCR, and only 1 article 12 delineated the rehabilitation strategies in detail by definite phases of recovery. Furthermore, we collated and summarized the information according to the common phase of recovery and rehabilitation in SCR, which was based on immobilization and timing of initiation, as well as duration of range of motion (pROM, aaROM, aROM), strengthening, and return to sports or activity. The summary of rehabilitation protocols for all studies is presented in Appendix Table A1.
A sling was utilized in 12 articles, ¶ followed by a pillow in 4 articles, 4,7,16,18 a brace in 2 articles, 15,36 and other device types in 2 articles. 25,27 Ten studies # used an immobilizer at abduction, which was maintained at 20° to 60°. 4,7,15,34 The immobilizer was used for 3 to 6 weeks postoperatively: 3 weeks in 1 study, 2 4 weeks in 4 studies, 7,23,28,29 and 6 weeks in 15 studies. **
The initiation of postoperative range of motion varied widely. The immobilization without pROM ranged from 0 weeks (immediate pROM) to 6 weeks postoperatively. Seven articles 5,7,12,21,28,29,34 reported the pROM that started within the first week at post-SCR as tolerated or comfortable. The immobilization without pROM was 2 to 4 weeks in 6 articles 10,15,25,27,31,32 and >4 weeks in 8 articles 2,4,11,16,18,19,23,36 (>6 weeks in 5 articles 11,16,18,19,36 ).
We found that aaROM was usually indicated with pROM or aROM as a transition in the late period of pROM or early period of aROM. Ten articles †† explicitly reported aaROM, which started after the period of pROM at 4 to 6 weeks. Five studies 4,12,18,29,34 recommended taking aaROM as tolerated, during the same period of pROM, to progress to aROM.
The start of the aROM phase ranged from postoperative 3 weeks to postoperative 12 weeks. Six articles started aROM by postoperative week 8 (3-6 weeks in 2 studies 10,28 and 6 weeks in 4 studies 5,12,21,25 ), while 7 studies did not recommend aROM by week 8 (week 8 in 3 studies, 27,29,34 10-12 weeks in 2 studies, 2,18 and 12 weeks in 2 studies 11,19 ).
The start of the strengthening phase ranged from postoperative 8 weeks to postoperative 6 months. Furthermore, strengthening started by week 12 in 5 studies, 2,7,16,18,23 at 12 weeks in 8 studies, 11,15,21,25,27,31,32,36 and up to 4 to 6 months in 4 studies. 4,10,12,34
Eight studies 10,11,12,21,31,32,34,36 cited the timing of return to sports, and these all recommended delaying it until at least 6 months post-SCR.
Rehabilitation Protocols Based on Type of Graft Used
Four types of grafts were utilized in the studies, which we stratified into autografts and nonautografts (allografts, xenografts, and synthetic grafts). Considering that autografts and allografts were utilized in studies conducted by Yoon et al 36 and Lee and Min, 19 these were excluded in the comparison. Nine studies utilized autografts, including tensor fasciae latae in 6 studies 10,15,23,27,32,34 and long head of biceps tendon in 3 studies, 4,7,16 while 10 studies used a nonautograft, including dermal allograft in 7 studies, 2,5,11,18,21,25,29 xenograft in 2 studies, 12,31 and synthetic graft in 1 study. 28 The summary of rehabilitation based on the grafts is presented in Table 2.
Comparison of Rehabilitation Protocols Based on Type of Graft a
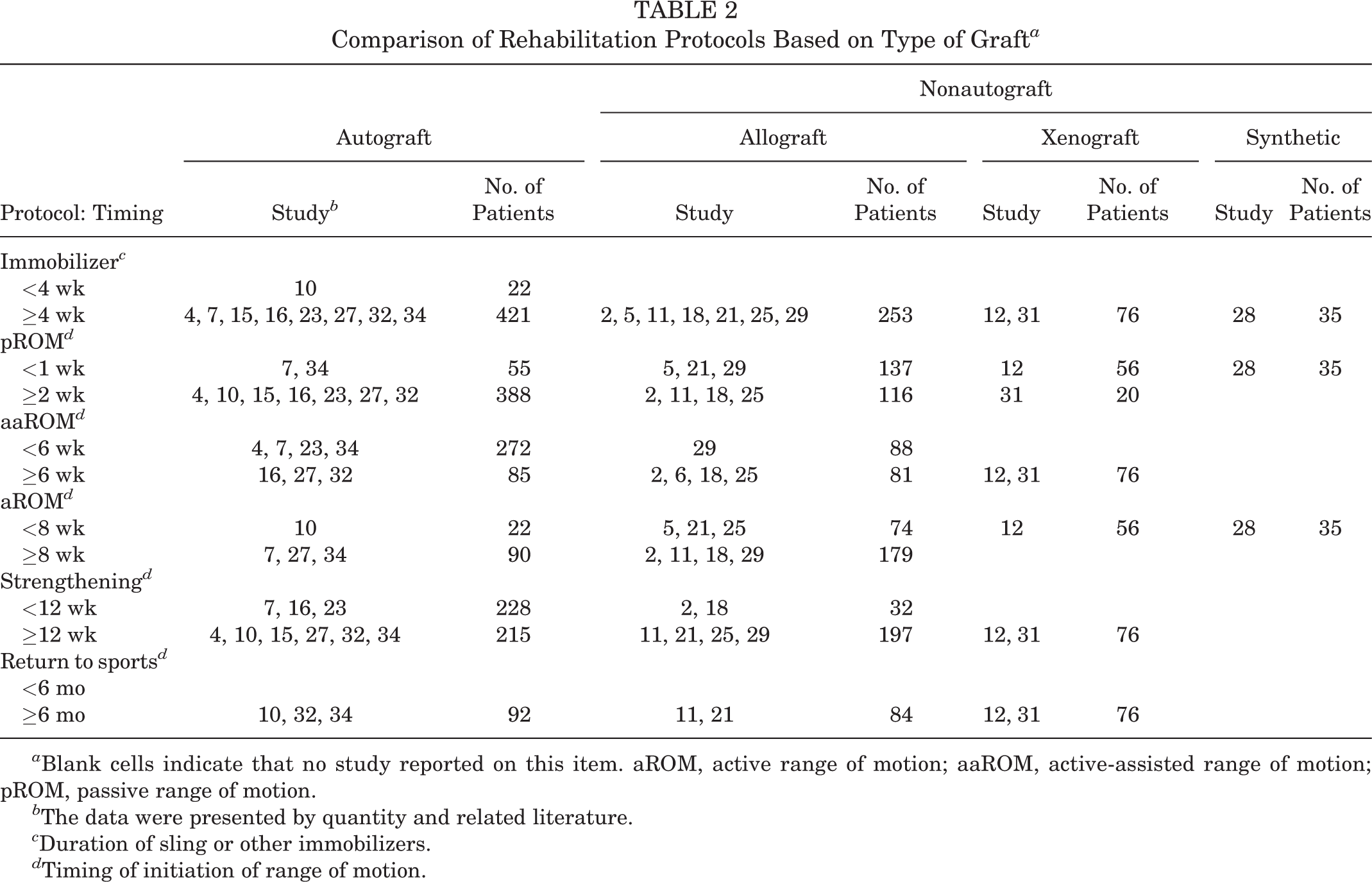
a Blank cells indicate that no study reported on this item. aROM, active range of motion; aaROM, active-assisted range of motion; pROM, passive range of motion.
b The data were presented by quantity and related literature.
c Duration of sling or other immobilizers.
d Timing of initiation of range of motion.
Of 10 studies in the nonautograft group, 5 recommended early pROM within the first week post-SCR, 5,12,21,28,29 while merely 2 of 9 recommended this in the autograft group. 7,34 Furthermore, there were significantly more patients starting early pROM in the nonautograft group than in the autograft group, and the difference was statistically significant (χ2 = 225; P < .001). Even when the autograft and allograft groups were separately compared, the difference for patients with early pROM was statistically significant (χ2 = 143; P < .001).
Discussion
At present, there is significant diversity in postoperative rehabilitation protocols, specifically when motions are initiated. Furthermore, there is an unproven tendency for surgeons to prefer and recommend early pROM for patients after SCR with nonautografts. Present rehabilitation guidelines following SCR are based on some studies with a relatively low level of evidence, and there are no randomized controlled trials. Hence, surgeons can rely on only level 3 and 4 evidence studies to evaluate the present status in this area.
Conventionally, it has been widely understood for RC repair that the initiation of pROM plays an important role in the entire phase of postoperative rehabilitation. On one hand, this influences the whole progression, including the subsequent aaROM, aROM, and strengthening. 33 On the other, there is a controversy on the early or delayed initiation of pROM in the period of immobilization after RC repair. 4 In terms of RC repair, surgeons may take many factors into consideration when recommending rehabilitation protocols—mainly, the location of the tear and the number of tendons involved, the amount of tendon retraction, tissue degeneration/fatty infiltrate, preoperative stiffness, tissue quality (affected by age, smoking, diabetes, and chronicity of tear), surgeon preference, tissue healing, and so on. 14,17,37
The present review suggests that current protocols for SCR are still a continuation of that for RC repair, without considering the difference of the 2 procedures between RC repair and SCR. Pogorzelski et al 30 recommended delayed pROM until postoperative 6 weeks after SCR, considering the similarly severe tear size between irreparable and massive RC tears. Indeed, the SCR is similar to a kind of repair procedure for graft bridging (GB), in which different types of grafts are utilized. Although the lateral margin of the graft is fixed on the greater tuberosity in GB and SCR, the medial end of the graft is sutured to the RC remnant in GB, while this is attached to the superior glenoid in SCR. 1 Theoretically, SCR reconstructs the “normal” superior capsule anatomy, while GB reconstructs a tension-free RC. 20
Few studies have exclusively given concern on the rehabilitation post-SCR, according to the present review. 12,30 Based on the available publications that indicated the rehabilitation after SCR, the present study observed that no universal protocol was applied for all conditions. The initiation timing of mobilization was mainly based on the experience and preference of surgeons and physical therapists. In addition, the type of graft may be a factor that influences the choice of recommendation. For example, Mihata et al 23 and Okamura et al 28 separately reported the early and delayed mobilization in 2 groups of patients with tensor fasciae latae and Teflon grafts, although both researchers were from the same medical team. This indicates the preference for recommending early mobilization for patients with nonautograft SCR. This was verified by the comparison in Table 2, which revealed that 5 of 10 studies in the nonautograft group recommended early pROM, while only 2 of 9 in the autograft group recommended this. Furthermore, it is noteworthy that although there was a difference in the initiation of pROM, studies that cited the timing of return to activity all recommended that patients can progressively do this at 6 months. Although the timing of return to sports appeared to be consistent in all studies, it was still unclear whether the ultimate occurrence of retear and functional outcomes, including shoulder stiffness, were potentially influenced by the different initiation timings of pROM. Finally, it remains to be explored and verified whether a surgeon’s preference in graft types has some influence on the clinical efficacy after SCR.
Limitations
The present analysis has several limitations. First, the present study included only level 3 and 4 case series and was unable to control for their heterogeneity. Second, surgery techniques related to the suture, fixation, and margin convergence of the grafts are potential factors that might influence the rehabilitation decision. Hence, the heterogeneity within and among studies prevented further analysis, we could conduct a qualitative and descriptive analysis without performing quantitative analysis based on the clinical outcomes. This decreased the research depth of the present study to some extent.
Implications of Future Research
Studies with a high level of evidence related to postoperative rehabilitation are necessary to obtain strong evidence and to compare results between early and delayed motion; as such, level 1 and 2 studies should be conducted. The present study implies that there were some different preferences among surgeons and physical therapists from varied countries. Future research should explore the effect of varying rehabilitation approaches on clinical outcomes and investigate the necessity of applying individual rehabilitation protocols while considering other potential factors that influence the clinical outcomes. Multicenter research may be necessary to obtain enough cases for a quantitative analysis to be conducted based on more homogeneous studies and to compare the different clinical outcomes of early and delayed rehabilitation or among different types of grafts.
Conclusion
At present, there is little agreement among published rehabilitation guidelines after SCR, precluding specific clinical best practice suggestions. The only consensus is that therapy is considered to have an important role in optimizing patient outcomes and that there is a need for high-quality prospective research. Although there is a tendency to recommend early motion in nonautograft cases, optimal protocols based on graft healing and functional outcomes need further verification based on the clinical outcomes of high-quality studies.
Footnotes
Final revision submitted April 22, 2022; accepted May 17, 2022.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
APPENDIX
Rehabilitation Protocols After Superior Capsular Reconstruction a

| Study | Graft Type | Duration (device) | pROM | aaROM | aROM | Strengthening | Return to Sports |
|---|---|---|---|---|---|---|---|
| Badman (2020) 2 | Allograft | 0-6 wk (sling) | 4-8 wk | 8-10 wk | >10 wk | >10 wk: graduated strengthening that focused on the deltoid, periscapular, and posterior rotator cuff musculature | NR |
| Campbell (2020) 5 | Allograft | 0-6 wk (sling) | 0-6 wk: pendulum exercises | >6 wk | >6 wk: progressed | NR | NR |
| Denard (2018) 11 | Allograft | 0-6 wk (sling) | ▪ >6 wk: FF and ER ▪ 3-4 mo: IR |
NR | 3-4 mo: FF | 3-4 mo: begin strengthening | 6-12 mo: return to full activity without restriction |
| Lacheta (2020) 18 | Allograft | 0-6 wk (abduction pillow) | >6 wk: as tolerated | >6 wk: as tolerated | 10-12 wk: full aROM | 10-12 wk: begin strengthening | NR |
| Pennington (2018) 29 | Allograft | 0-4 wk (sling) | 0-4 wk: pROM for comfort | 4-8 wk: aaROM with progression to aROM | >8 wk | NR | NR |
| Makki (2020) 21 | Allograft | 0-6 wk (sling) | ▪ >0 wk: passive assisted flexion (≤90°) and external rotation (≤30°) ▪ >6 wk: full pROM |
NR | 6 wk–3 mo: progressed aROM | >3 mo: begin strengthening with progression to full aROM | >6 mo: full activity without restriction |
| Mirzayan (2019) 25 | Allograft | 0-6 wk (padded shoulder immobilizer) | 2-6 wk: gentle, daily pendulum exercises | >6 wk | >6 wk | >12 wk: gradual strengthening | NR |
| Mihata (2020) 23 | Autograft | 0-4 wk (abduction sling) | >5 wk | >5 wk | NR | >8 wk | NR |
| Chillemi (2018) 7 | Autograft | 0-4 wk (abduction pillow at 20°) | >1 d: passive shoulder mobilization | >4 wk: active-assisted shoulder exercises | >1 d: active hand, wrist, and elbow exercises | >8 wk: strengthening exercises of the deltoid | NR |
| Barth (2020) 4 | Autograft | 0-6 wk (abduction pillow at 60°) | >4 wk | >4 wk: physical therapy, hydrotherapy, and progressive withdrawal of the abduction pillow | 0-4 wk: hand, wrist, and elbow exercises | >6 mo: begin strengthening or resistance exercises | NR |
| de Campos Azevedo (2018) 10 | Autograft | 0-3 wk (sling) | 3-6 wk: progressive | NR | 3-6 wk: progressed shoulder elevation and elbow flexion exercises | >6 wk: resistant elbow exercises | >6 mo: resistant shoulder exercises progressive return to full activity |
| Kholinne (2020) 15 | Autograft | 0-6 wk (abduction brace in 30°-45°) | >3 wk: pendulum exercise | NR | NR | >3 mo: strengthening exercise for periscapular muscles and rotator cuff | NR |
| Kocaoglu (2020) 16 | Autograft | 0-6 wk (abduction pillow) | >6 wk | >6 wk | NR | >8 wk: strengthen the rotator cuff and scapular stabilizers | NR |
| Ohta (2020) 27 | Autograft | 0-6 wk (abduction orthosis) | ▪ >3 wk: elevation in the scapular plane ▪ > 4 wk: IR and ER |
6-7 wk | 8-9 wk | >12 wk | NR |
| Polacek (2020) 32 | Autograft | 0-6 wk (sling) | >2 wk | >6 wk | NR | >12 wk: exercises strengthening the deltoid and periscapular muscles | 6-12 mo: return to full activities |
| Takayama (2021) 34 | Autograft | 0-6 wk (abduction sling at 45°) | >4 d: passive shoulder exercise with patients in the supine position | >5 wk: active-assisted exercise with patients in the supine position | ▪ >1 d: active exercise of the hand and wrist ▪ >8 wk: active exercises with the patients in the supine and sitting positions |
>5 mo: begin strengthening | >6 mo: participation in sports activities |
| Lee (2018) 19 | Autograft / allograft | NR | >6 wk | NR | 12 wk | NR | NR |
| Yoon (2018) 36 | Autograft / allograft | 0-6 wk (abduction brace) | >6 wk | >6 wk | >0 wk: shrugging, protraction, and retraction of shoulder girdles; intermittent exercise of the elbow, wrist, and hand; and ER of the arm to neutral with the brace | >3 mo: begin strengthening | 6-9 mo: full return to sports |
| Okamura (2021) 28 | Synthetic | 0-4 wk (abduction sling) | ▪ >1 d: passive elevation exercise ▪ >7 d: passive ER |
NR | >3 wk: active elevation exercise | NR | NR |
| Ferrando (2021) 12 | Xenograft | 0-6 wk (sling) | 0-6 wk: pROM may commence if the patient is showing signs of postoperative stiffness | 6 wk–4 mo: start with aaROM and progress to aROM through range with attention to scapular control | 6 wk–4 mo: glenohumeral joint and scapular mobilizations | 4-6 mo: progress strengthening program with free weights, TheraBand, and body weight | >6 mo: return to full functional activities |
| Polacek (2019) 31 | Xenograft | 0-6 wk (sling) | >2 wk | >6 wk | NR | >12 wk: exercises strengthening the deltoid and periscapular muscles | 6-12 mo: return to full activities |
a aaROM, active-assisted range of motion; aROM, active range of motion; ER, external rotation; FF, forward flexion; IR, internal rotation; NR, not reported; pROM, passive range of motion.
