Abstract
Background:
Graft maturation is an important prognostic factor for hamstring autograft anterior cruciate ligament reconstruction (ACLR). It remains unclear whether extracorporeal shock wave therapy (ESWT) can promote graft healing after ACLR.
Purpose:
To evaluate the therapeutic and graft maturation effects of ESWT in hamstring autograft ACLR.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
Between May 18, 2019, and September 20, 2019, we randomly assigned 30 patients who met study inclusion criteria to 2 groups. Patients in the control group followed a 5-week advanced rehabilitation training program (30 minutes/session, 5 times/week) starting at 3 months postoperatively. In the ESWT group, together with the 5-week advanced rehabilitation training, radial ESWT was applied once a week for 5 weeks. Functional scores (Lysholm, International Knee Documentation Committee, and Tegner scores), KT-1000 arthrometer knee laxity measurement, and magnetic resonance imaging scans were assessed at 3 months (baseline), 6 months, and 24 months postoperatively. To evaluate graft maturation, we assessed the graft signal-to-noise quotients (SNQs) of the tibial, intra-articular, and femoral sides on magnetic resonance imaging scans. Data were compared between the ESWT and control groups.
Results:
In total, 26 patients (13 with ESWT, 13 controls) were assessed. There were no significant between-group differences on any assessment at baseline, and no significant within-group or between-group differences were found in knee laxity at any point. At 24-month follow-up, the ESWT group had significantly higher Lysholm and Tegner scores compared with the controls (P = .012 and .017, respectively). Regarding graft maturation, at 6-month follow-up, the SNQ of the tibial intraosseous graft was significantly lower in the ESWT group versus controls (P = .006), but no differences were detected at the femoral intraosseous graft (P = .321) or the intra-articular graft (P = .314). At 24-month follow-up, the SNQs of the femoral intraosseous graft and intra-articular graft were significantly lower in the ESWT group versus controls (P = .020 and .044, respectively) but no difference was found at the tibial intraosseous graft (P = .579).
Conclusion:
Both enhanced graft maturation and improved functional scores at 24-month follow-up were seen in patients who received radial ESWT during rehabilitation after hamstring autograft ACLR.
Registration:
ChiCTR1900022853 (Chinese Clinical Trial Registry).
Keywords
Anterior cruciate ligament (ACL) tears are a common injury, occurring in >50% of knee injuries. 14 ACL reconstruction (ACLR) has been recommended to restore anterior-posterior knee stability and treat rotatory knee laxity to prevent resultant damage to cartilage or menisci in young, healthy, and active patients. 21
The maturation of the intra-articular and intraosseous grafts are important for knee function after ACLR, 12 affecting the progress of postoperative rehabilitation as well as the timing of safe return to full sports activity. 3 Firm incorporation of the graft within the tunnel enables the graft to mature into a structure similar to the native ACL. 22 Nevertheless, a failure rate of up to 14% for ACLR has been reported, 29 stimulating scientific efforts to find solutions that could promote better graft maturation and healing. Many biologic agents have been introduced to promote graft healing in ACLR, including stem cells and platelet-rich plasma. 4 However, no conclusive indication can be addressed regarding the efficacy of these treatment approaches, and their applications in clinical work are still controversial.
Extracorporeal shock wave therapy (ESWT), regarded as a noninvasive alternative to conservative treatment, was initially introduced for the treatment of nephrolithiasis. Based on the delivery pathway for the propagation of acoustic energy, ESWT can be divided into 2 types: radial and focused. 23 Radial ESWT involves pneumatic waves that are generated by an air compressor through a tube on the end of an applicator. The projectile hits the applicator, by which the generated pressure wave is transmitted into the body. For focused ESWT, shock waves can be generated via 3 methods: electromagnetic, electrohydraulic, and piezoelectric. 2 Both radial and focused ESWTs are used widely to treat various soft tissue disorders, including chronic Achilles tendinopathy, lateral epicondylitis, plantar fasciitis, tendinopathy of the shoulder, and bone and skin disorders. 9,11,19,27,44
Although the underlying mechanism by which ESWT produces a therapeutic effect has not been elucidated fully, 34 it has been postulated that repeated microtrauma could stimulate the ingrowth of neovascularization associated with the upregulation of angiogenic and osteogenic growth factors. 35 Some previous evidence from animal studies showed that ESWT could enhance tendon-bone healing in ACLR and rotator cuff repair, 6,38 and biomechanical assessments suggested that either a single dose of ESWT intraoperatively or repetitive applications after the operation could result in better outcomes compared with traditional treatment. 6 These findings have raised interest in the application of ESWT to rehabilitation after ACLR; however, there is a lack of evidence for the proper treatment regimen, patient responses, or the correlation between clinical outcomes and graft maturation.
Magnetic resonance imaging (MRI) signal intensity measurements have been used in clinical studies to assess the maturity of the ACL graft in humans. 8,12,16,40 The graft signal-to-noise quotient (SNQ) has been adopted as a measurement for analyzing graft quality 10,41 ; a smaller SNQ reflects better graft maturity. 45
In this randomized controlled trial, the aim was to assess clinical outcomes and graft maturation on MRI scans in patients who received radial ESWT during rehabilitation after anatomic single-bundle ACLR with hamstring autograft. Our hypothesis was that postoperative radial ESWT would yield similar clinical outcomes compared with rehabilitation treatment alone.
Methods
The study protocol was approved by an institutional review board, and informed consent was obtained from all study patients. This study was registered on the Chinese Clinical Trial Registry (ChiCTR1900022853). Patients were recruited between May 18, 2019, and September 20, 2019, and the last follow-up was conducted on June 28, 2021.
Determination of Sample Size
Using G*Power 3.1 (HHU, Version 3.1.9.6), we calculated the sample size needed for adequate power to test our hypothesis. According to a previous study, 37 the Lysholm scores at 1 year follow-up (94 ± 4.9 in the ESWT group vs 87.3 ± 6.4 in the control group) were used to calculate the effect size. To attain a power of 80% using a 2-sided test with α = .05, the estimated number of participants needed to be at least 20 (10 in each group). With the prediction of 20% not completing follow-up, the minimal number of participants needed to be 24.
Study Population
The inclusion criteria were as follows: (1) age between 18 and 45 years, (2) unilateral ACL injury, (3) isolated ACL injury, (4) anatomic single-bundle ACLR with hamstring autograft, (4) a Tightrope (Arthrex) at the femoral side and Bio-Intrafix interference screw (DePuy Synthes Inc.) at the tibial side, and (5) receiving surgery in our hospital and agreeing to participate in the postoperative rehabilitation protocol. The exclusion criteria were (1) previous knee injury or revision ACLR; (2) graft failure during follow-up; (3) concurrent multiligament injuries; (4) receiving other conservative treatment modalities during the follow-up period; (5) postoperative joint disorders including synovitis, stiffness, or infection; (6) history of systemic diseases including rheumatic arthritis, osteoarthritis, or osteoporosis; (7) claustrophobia, unwillingness to be randomly assigned to a group or complete the full series of MRI scans, and any electrical device potentially interfering with MRI scans; and (8) other contradictions to the ESWT. After inclusion and exclusion criteria were applied, 30 patients from our institution were enrolled in this study.
Study Design
We randomly assigned the 30 patients prospectively to an ESWT group and a control group at a 1:1 ratio (Figure 1). The group assignment was determined using computer-based block randomization. Treatment assignments were placed in sequentially numbered opaque envelopes before the trial commenced, and they were assigned to the patients by the trail managers, who also arranged the facilities needed for the procedure. In the control group, patients were asked to follow a 5-week advanced rehabilitation protocol starting at 3 months postoperatively. In the ESWT group, together with the advanced rehabilitation training, radial ESWT was added after rehabilitation on a weekly basis for 5 weeks.

CONSORT (Consolidated Standards of Reporting Trials) flow diagram. ESWT, extracorporeal shock wave therapy; KT-1000, KT-1000 arthrometer knee laxity measurement; MRI, magnetic resonance imaging.
Baseline descriptive information was obtained via a self-reported preparation history, and the baseline MRI scan was performed 1 or 2 days before the trial started. The investigators (A.W. and S.Z.) who assessed the outcomes were blinded to the treatment that the patient received.
Surgical Technique
All ACLR procedures were performed by 2 surgeons (J.C. and Y.H.) from our department with >10 years of experience in arthroscopic ACLR. All patients received the same procedure of arthroscopically assisted anatomic single-bundle ACLR. 45 Associated meniscal and cartilage injuries were treated. Briefly, the ipsilateral semitendinosus and gracilis tendons were obtained to prepare 4-strand double-looped grafts. The average graft size was 8 to 9 mm in diameter and 110 to 120 mm in length. Via the standard anterior-medial portal, the medial wall of the lateral femoral condyle was viewed and prepared to expose the resident ridge and the posterior cartilage margin. The femoral tunnel location was 1 to 2 mm inferior to the resident ridge with a 4-mm distance to the posterior cartilage margin. 25 The ACL tibial stump was used as a referenced landmark for guiding tibial tunnel placement, 6 and the ACL tibial guide was set to an angle of 55° and placed medial to the center of the remaining tibial stump of the native ACL. The graft was fixed using a Tightrope (Arthrex) at the femoral side and a Bio-Intrafix interference screw (DePuy Synthes Inc) at the tibial side.
Rehabilitation Protocol
After ACLR, all patients followed the same staged rehabilitation protocol starting in the first week after the operation. During 0 to 6 weeks postoperatively, the aim of rehabilitation was to establish full range of motion, prevent muscle hypotrophy, and diminish pain and swelling. Immediate partial or full weightbearing was recommended if the patient was able to tolerate it or if the patient was referred for meniscal treatment. From 6 to 12 weeks postoperatively, patients were encouraged to perform more cardiovascular exercise, gait training, progressive strength exercises, and perturbation training.
From 3 months after ACLR, a 5-week advanced rehabilitation protocol was assigned for the patients of both the ESWT and control groups. This rehabilitation protocol included stretching exercises, progressive core muscle training (Figure 2), and balance perturbation exercises. Core muscle training was designed to progressively enhance hip joint function. Each training session lasted approximately 30 minutes and was performed 5 times per week throughout the whole program. The training program, divided into 3 stages, was upgraded 3 times during each week based on the patient’s performance. Table 1 summarizes the program at each stage.

Progressive strength muscle training.
Advanced Rehabilitation Protocol by Stage

a Letters in parentheses correspond to images in Figure 2. Bosu ball from AT Pro Company.
A single physical therapist (W.Y.) who was blinded to this study instructed patients during the rehabilitation training. All trainings were performed on site to ensure that the patients could perform the exercises properly. Afterward, the therapist followed up with patients during the entire training program to ensure every patient could complete each visit. All patients were required to visit the rehabilitation center at least once a week.
Extracorporeal Shock Wave Therapy
In addition to the rehabilitation protocol, patients in the ESWT group received radial ESWT from 3 months postoperatively. ESWT was applied once a week for 5 weeks by the same practitioner (C.L.). The radial shock wave device (EMS Swiss Dolor-Clast) was used. Before the shock wave was applied, knee radiographs were taken on anterior-posterior view, and the distances from the tibial fixation and the femoral suspension plate to the knee joint space were measured. These distances were used as references for where the device probe should be placed on the knee skin surface (Figure 3A). After applying ultrasound gel, the practitioner placed the probe on the skin surface projections of the tibial tunnel and the femoral tunnel (Figure 3, B and C). A total of 2000 impulses applied at 6 to 8 Hz with an energy density of 0.05 to 0.11 mJ/mm2 were administered during each ESWT treatment. After treatment, patients were advised to cool down, rest, and stretch to avoid ESWT side effects. Patients in the control group did not receive ESWT after advanced rehabilitation protocol.

ESWT applied to patients with ACLR. (A) Knee radiographs were taken on the anterior-posterior view, and the distances from the tibial fixation and the femoral suspension plate to the knee joint space were measured and used as knee skin surface projection references on which the ESWT probe should be placed. (B) Shock wave probe was placed on the surface projection of the femoral tunnel. (C) Shock wave probe was placed on the surface projection of the tibial tunnel. ACLR, anterior cruciate ligament reconstruction ESWT, Extracorporeal shock wave therapy; L, left side.
Magnetic Resonance Imaging
MRI examinations using a 3.0-T MR (MAGNETOM Verio) were performed on all patients at 3-, 6-, and 24-month follow-up. A senior radiologist who was blinded to the study performed the MRI examinations. A 15-channel multielement phased-array knee coil was applied with the patient lying on the examination table. Sagittal images were taken using an oblique proton density fat saturation sequence: repetition time, 3000 ms; echo time, 28 ms; flip angle, 160°; matrix, 320 × 272; slice thickness, 3 mm. Each MRI examination took approximately 5 minutes.
The signal intensity of the ACL graft was calculated at 5 sites: (1) the graft within the femoral tunnel (FG), (2) the intra-articular graft (IG), (3) the graft in or close to the tibial tunnel (TG), (4) the posterior cruciate ligament (PCL), and (5) background taken approximately 2 cm away from the patellar tendon (Figure 4).
Here, since the signal intensity of the graft within the tibial tunnel was difficult to measure because of the fixing screw on a certain slicing image, the signal intensity was taken at the intra-articular graft close to the tibial tunnel, which was within 2.0 cm of the tibial plateau. 16 A circular region of interest (ROI) with an area of 0.25 cm2 was used for each site. Within the intraosseous and intra-articular graft sites, the signal intensity was measured at 3 randomly selected ROIs, and the mean value was calculated.

Illustration of signal-to-noise quotient measurements on MRI scans. A circular ROI with an area of 0.25 cm2 was taken on the MRI scan. 1, graft within the femoral tunnel; 2, intra-articular graft; 3, graft in or close to the tibial tunnel; 4, posterior cruciate ligament; 5, background ROI taken approximately 2 cm away from the patellar tendon. MRI, magnetic resonance imaging; ROI, region of interest.
To quantify the normalized signal intensity of the ACL graft, we calculated the SNQs of the FG, IG, and TG separately using the following equation: SNQ = (ACL graft signal - PCL signal)/background signal. 18 The SNQ was calculated 3-, 6-, and 24-months postoperatively. Two authors (A.W. and S.L.) performed all the measurements after receiving training from a senior radiologist. The interval between each measurement was at least 1 week to minimize the memory effect. The same 2 authors independently measured 20 sample cases to assess interobserver reliability. After 4 weeks, 1 of the authors (A.W.) was asked to rate the same set of images again to measure the intraobserver reliability.
Study Outcomes
The study baseline was set at 3 months after ACLR. The baseline assessment was taken 1 or 2 days before the study began by investigators (A.W. and S.Z.) who were blinded to the study. Patients should not have required other therapies or medication beyond the protocol-defined allowable period. If a patient was lost to follow-up, the last available measurement was used to determine treatment success.
The primary outcome was the Lysholm score at 24-month follow-up. The Lysholm score is a patient-rated measurement for examining knee-specific functions, including mechanical locking, instability, pain, swelling, stair climbing, and squatting. The International Knee Documentation Committee (IKDC) score and Tegner activity score at 24-month follow-up were considered the secondary outcomes. The minimal clinically important difference (MCID) thresholds for the Lysholm, IKDC, and Tegner scores are 8.9, 16.7, and 1 point, respectively. 42
In addition, knee laxity was measured as the side-to-side difference (SSD) in anterior tibial translation (ATT) between the reconstructed and healthy knee of each patient. Knee laxity was evaluated using a KT-1000 arthrometer (MEDmetric Corp) at an 89-N load.
All of the subjective functional scores (Lysholm, IKDC, Tegner) were assessed at 3-, 6-, and 24-month follow-up after the operation. Patients were asked to finish their subjective functional scores online, and the results were then calculated automatically and reported to the investigators. The knee laxity measurement was also taken at the 3-, 6-, and 24-month follow-up.
Statistical Analysis
Data analyses were performed using SPSS 21.0 software (SPSS Inc), and the data were reported as the means ± standard deviations. Since the sample in this cohort was fewer than 50 patients, the Shapiro-Wilk test was used to test for normality. The normality test showed that the continuous variables were not normally distributed, and thus the Mann-Whitney U test was used for comparison of continuous variables between the ESWT and control groups. The Fisher exact test was used to compare categorical variables. The Friedman test was used to compare the results from different time points within the same group. The correlation between functional scores and SNQs at 24-month follow-up was assessed in both groups using Pearson correlation coefficient (r). For all analyses, results were considered statistically significant at P < .05.
Results
At the 6-month follow-up, 2 patients from the control group left the cohort as they were moving to another country. There were 2 patients who dropped out of the ESWT group, as 1 patient received laparoscopic cholecystectomy and 1 patient had a meniscal repair on the contralateral knee. Furthermore, 1 patient in each group did not undergo MRI examination or knee laxity assessments. Thus, at the 24-month follow-up, 12 patients in each group completed MRI scan and knee laxity assessment, and 13 patients in each group completed all functional assessment scores (Figure 1). No systemic or local complications or ESWT device-related problems were reported during follow-up.
There were no significant differences in characteristics between the ESWT and control groups (Table 2). No rerupture or other surgery-related complications were found among any of the participants.
Baseline (12-wk Postoperative) Patient Characteristics for the ESWT and Control Groups a
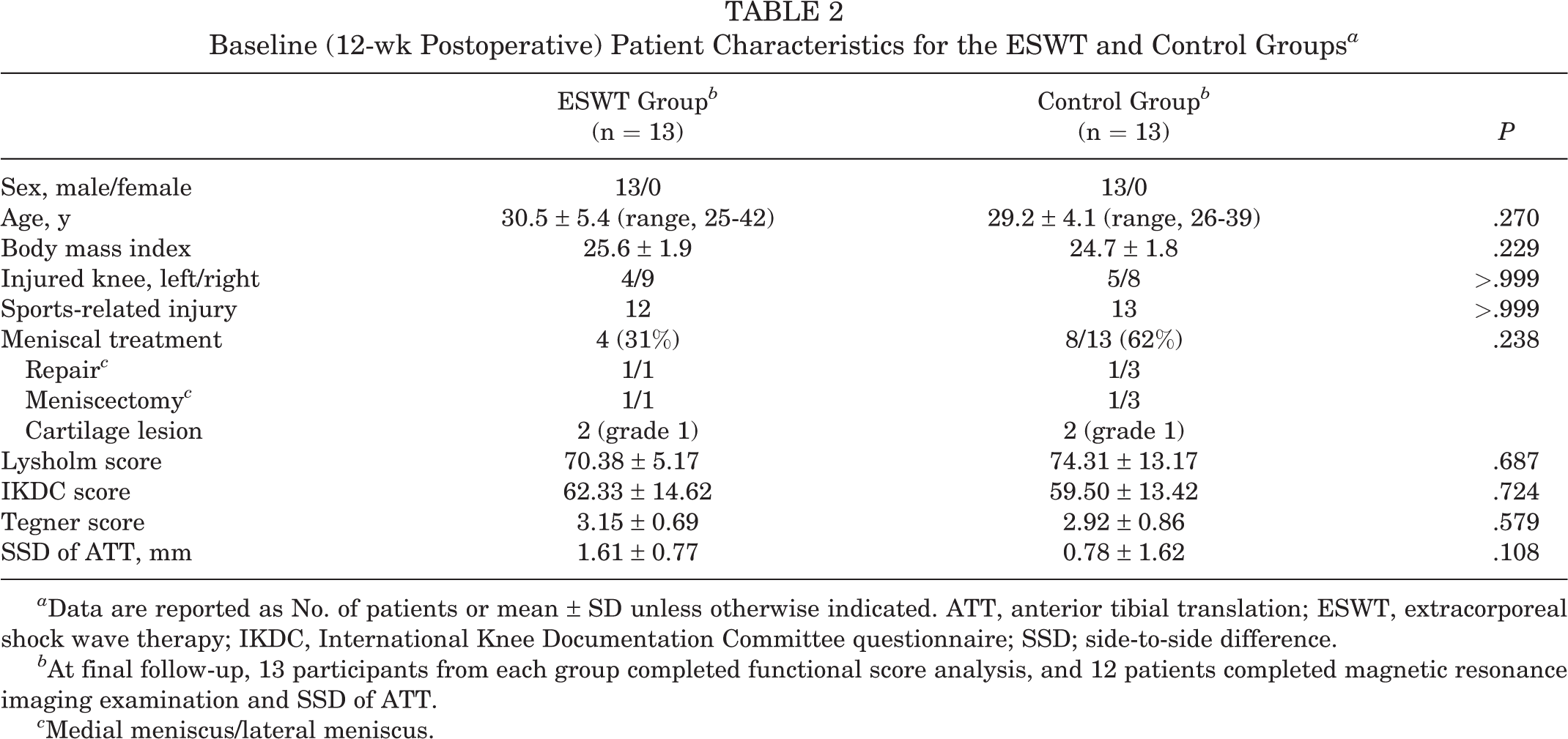
a Data are reported as No. of patients or mean ± SD unless otherwise indicated. ATT, anterior tibial translation; ESWT, extracorporeal shock wave therapy; IKDC, International Knee Documentation Committee questionnaire; SSD; side-to-side difference.
b At final follow-up, 13 participants from each group completed functional score analysis, and 12 patients completed magnetic resonance imaging examination and SSD of ATT.
c Medial meniscus/lateral meniscus.
Primary Outcome Measurement
The Lysholm score was acquired from 13 patients in both groups. In both groups, the scores increased during the 24 months of follow-up, and at 24-month follow-up, they were significantly higher than those of the 3-month baseline assessment or 6-month follow-up (ESWT group, P = .003; control group, P < .001). At 6-month follow-up, no significant difference was detected between the ESWT and control groups (P = .087), but at 24-month follow-up, the Lysholm score of the ESWT group was significantly higher compared with that of the control group (P = .012) (Figure 5A).
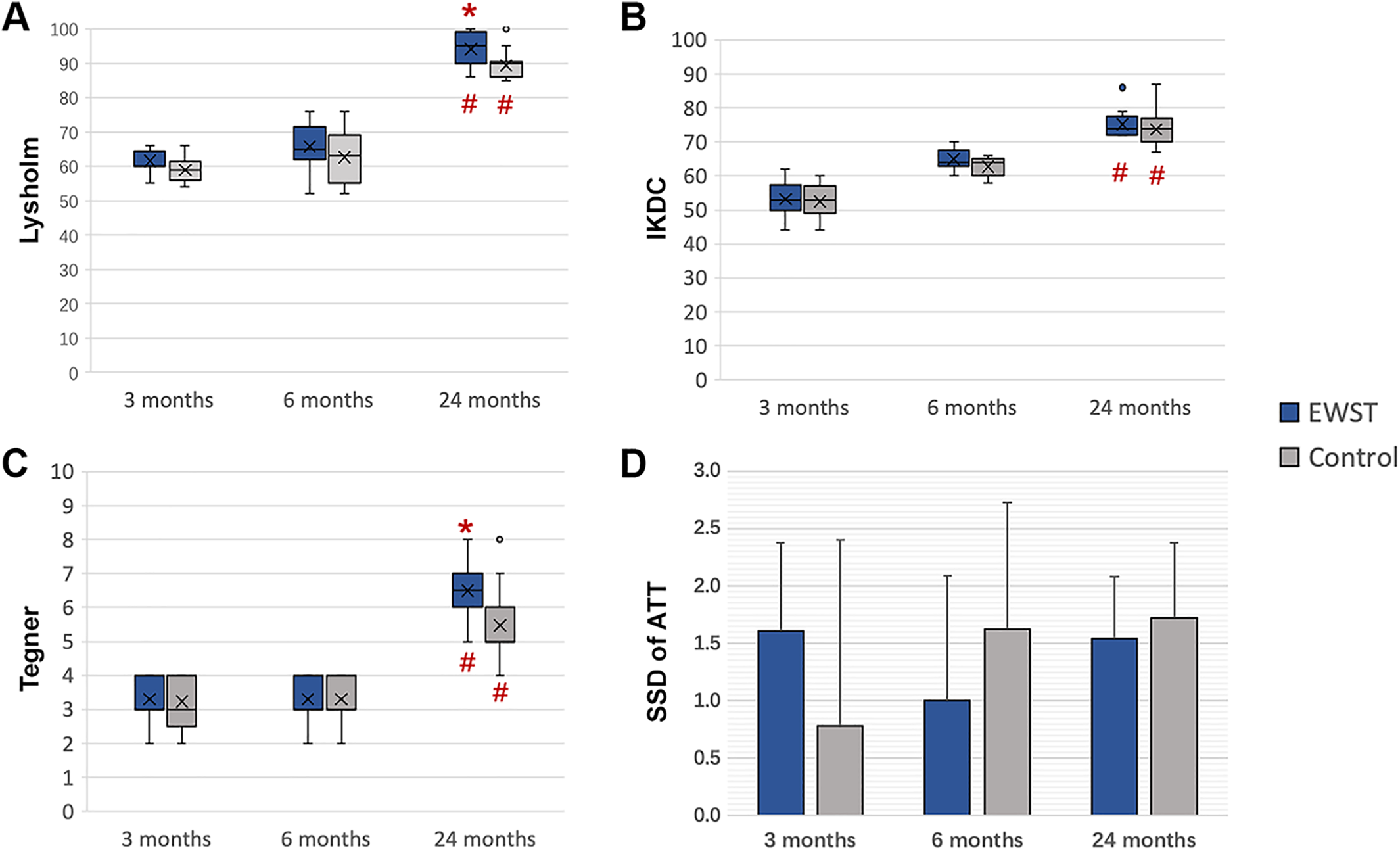
(A-D) Comparison of clinical outcomes between ESWT and control groups. In (A) to (C), the line within the box represents the mean, the “x” represents the median, the top and bottom boundaries of the box represent the interquartile range, and the error bars represent the range. In (D), the box represents the mean and the error bars represent the standard deviation. Significant difference: *between ESWT group and control group and #within the group compared with the other time points (P < .05). ATT, anterior tibial translation; ESWT, extracorporeal shock wave therapy; IKDC, International Knee Documentation Committee questionnaire; SSD, side-to-side difference. The dots in the boxplot represent those who ate a lot more than normal or a lot less than normal (outliers).
Secondary Outcome Measurements
In both groups, the IKDC and Tegner scores increased during the 24 months of follow-up. At the 24-month follow-up, they were significantly higher than those of the 3-month baseline assessment or the 6-month follow-up (P < .01 for all). At the 6-month follow-up, no significant difference was detected between the ESWT group and the control group (IKDC, P = .495; Tegner, P = .314). At 24-month follow-up, the Tegner score of the patients who received ESWTs was significantly higher than that of the patients who did not (P = .017), while no significant difference was found in IKDC scores (P = .415) (Figure 5, B and C).
Regarding clinically relevant values of the MCID, 3 of 13 patients (23.1%) who received ESWT showed improvement in Lysholm score exceeding 8.9 points at 6-month follow-up; all patients (100%), exceeding MCID at 24-month follow-up. In the control group, 2 of 13 patients (15.4%) showed improvement in Lysholm score exceeding 8.9 at 6-month follow-up; all patients (100%), exceeding 16.7 at 24-month follow-up. For the results of MCID of IKDC score, the percentages of patients exceeding 16.7 in the ESWT group were 30.8% (4/13) and 84.6% (11/13) at 6- and 24-month follow-up, respectively, whereas in the control group, the results were 23.1% (3/13) and 53.8% (7/13). Regarding the Tegner score, the percentages of patients exceeding 1 in the ESWT group were 30.8% (4/13) and 100% (13/13) at 6- and 24-month follow-up, respectively, whereas in the control group, the results were 23.1% (3/13) and 38.5% (5/13), respectively.
For the SSD of ATT, there were no significant differences between the ESWT and control groups at the 6-month (P = .157) or 24-month (P = .415) follow-up. Further, no significant differences in SSD of ATT were found within each group at different follow-ups (ESWT, P = .097; control, P = .555) (Figure 5D).
MRI Measurements
In both groups, 15 patients received the MRI examination at 3-month follow-up, and 12 completed the MRI examinations at the 6- and 24-month follow-ups (Figure 6). The oblique sagittal view was used for evaluation. All the patients had intact ACL grafts before they participated in the study, and no graft failure was diagnosed on MRI scan at the 6- or 24-month follow-up in either group.

Sagittal view magnetic resonance imaging scans of knees in the extracorporeal shock wave therapy (ESWT) and control groups at 3-, 6-, and 24-month follow-up. Yellow arrows indicate the graft.
The intra- and interobserver reliability values for SNQ measurement were 0.876 and 0.935, respectively, which was considered good-to-excellent agreement. Overall, the SNQs of TG, IG, and FG decreased over time, which indicated graft maturation during the 24 months of follow-up (P < .01 for all) (Figure 7). At the 3-month assessment, there were no significant differences between the 2 groups for each parameter (TG, P = .157; IG, P = .314; and FG, P = .415). At 6-month follow-up, the SNQ of TG in the ESWT group was significantly smaller than that of the control group (P = .006), but no differences were found for the SNQ of IG (P = .314) or FG (P = .321). At 24-month follow-up, the SNQs of FG and IG were significantly smaller in the ESWT group than in the control group (FG, P = .020; IG, P = .044), whereas the SNQ of TG was similar between the 2 groups (P = .579).
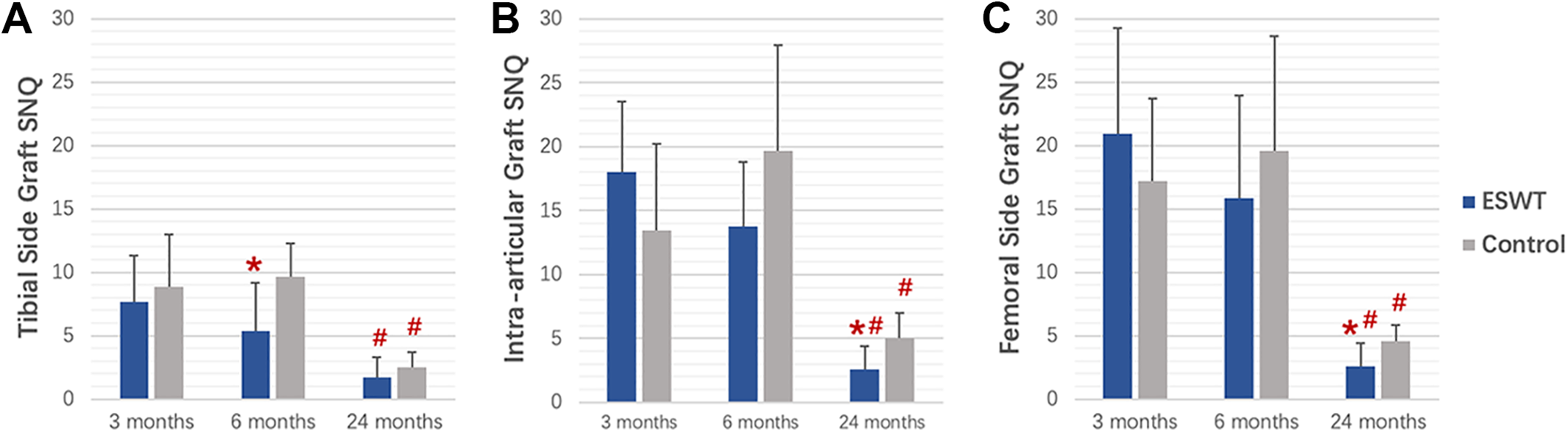
Postoperative mean signal-to-noise quotients (SNQs) of the anterior cruciate ligament grafts at the (A) tibial-side, (B) intra-articular, and (C) femoral-side sites at 3-, 6-, and 24-month follow-up. Error bars represent SDs. Significant difference: *between extracorporeal shock wave therapy (ESWT) group and control group and #within the group compared with the other time points (P < .05).
Correlation Analysis of Functional Scores and Graft SNQs
The results from the control group showed that the Lysholm, IKDC, and Tegner scores were negatively correlated with graft SNQs at the 24-month follow-up (P < .05 for all), which proved the natural healing process of graft maturation after ACLR. The functional scores in the ESWT group were also significantly negatively correlated with graft SNQs in most cases (P < .05), except there was no significant correlation between the Lysholm score and femoral intraosseous graft SNQ or the IKDC score and tibial intraosseous graft SNQ (Table 3).
Correlation Analysis of Functional Scores and Graft SNQs a

a Boldface P values indicate statistical significance (P < .05). ESWT, extracorporeal shock wave therapy; IKDC, International Knee Documentation Committee questionnaire; SNQ, signal-to-noise quotient.
Discussion
ESWT has been used widely in clinical practice for managing musculoskeletal disorders, 17 but evidence of the application of ESWTs in patients after ACLR is lacking. To our knowledge, this is the first randomized controlled trial to investigate the effect of ESWT on the autograft maturation after ACLR. We found that the Lysholm score at 24-month follow-up (the primary outcome) was significantly improved after ESWT treatment in patients who received ACLR. Therefore, the hypothesis of similar clinical outcomes to those of controls was rejected. Further, compared with controls, the ESWT group saw enhanced maturation of intraosseous graft at the tibial side at 6-month follow-up and enhanced maturation of grafts at the intra-articular and femoral sides at 24-month follow-up.
The biological effect of shock wave is closely related to the energy density, timing of treatment, and treatment duration. 7 In a previous study by Wang et al, 37 a single dose of focused ESWT was applied to patients during the operation for ACLR. In their research, a high energy flux density of 0.298 mJ/mm2 was administered to the middle third of the tibial tunnel, and the results showed that ESWT could improve Lysholm scores and decrease tibial tunnel size at 2-year follow-up. Compared with their study, our study applied a radial ESWT weekly with the lower energy density of 0.05 to 0.11 mJ/mm2 at 3 months postoperatively for 5 weeks. Therefore, the major findings in our study were the effects of ESWT on graft maturation after ACLR.
Graft healing after ACLR undergoes 3 staged biological changes: inflammation, revascularization, and remodeling. 3 Some previous animal studies reported that applying focused ESWT with high energy density (≥0.19 mJ/mm2) during surgery could enhance tendon-bone healing in ACLR, 36,38 chronic rotator cuff repair, 6 and delayed patellar–patellar tendon rupture. 39 The possible biological mechanism behind these findings was that the focused high-energy ESWT could promote osteogenesis and neovascularization so as to promote new bone growth at the tendon-bone interface immediately, which might be beneficial to the proliferation stage of tendon-bone healing. However, in a human biopsy study, Rougraff et al 28 reported that the highest remodeling activity occurred from 3 to 10 months after ACLR.
It is postulated 28 that the grafts might experience a shift from revascularization to remodeling between 3 and 6 months, indicating that more turnover occurred in the extracellular matrix during this period. It has been reported that a single application of ESWT could improve blood flow, and repetitive ESWT application could prolong this phase. 15 Other in vitro studies have found that ESWT could stimulate the proliferation and osteogenesis of human osteoblasts, as well as chondrogenesis of human stem cells, 26,32 which are the major cells for extracellular matrix formation. These aforementioned studies might lay the theoretical foundation for radial ESWT promoting ACL healing, suggesting that the low energy density and 5-week treatment course of radial ESWT at 3 months after ACLR might affect graft remodeling.
In this study, we analyzed the graft maturation of 3 segments, which gave more insight into the therapeutic responses of intraosseous and intra-articular grafts separately. Consistent with previous studies, 18,45 we found that the SNQs of the femoral intraosseous graft, intra-articular graft, and tibial intraosseous graft remained stable between 3 and 6 months and decreased at 24-month follow-up, suggesting that the graft underwent revascularization during the early phase after ACLR and devascularization around 2 years after ACLR. Functional outcomes also improved with graft maturation, and correlations were found between graft SNQs and functional scores in both the ESWT and the control groups, which support the findings of previous studies. 18,20
Interestingly, in the ESWT group, graft maturation at the tibial side was seen earlier than at the femoral side or intra-articular side. This difference could be caused by fixation methods and tunnel locations. In the autograft ACLR in our study, a cortical suspension device was used on the femoral side, and interference fixation was used on the tibial side. Therefore, the “bungee effect” and “windshield-wiper effect” caused by suspension fixation can introduce excessive micromotion of the graft in the tunnel, 13,24,40,43 which could compromise graft maturation within the femoral tunnel. 31 In addition, compared with the intra-articular graft and the femoral intraosseous graft, the tibial intraosseous graft was more superficial to the skin. Since the radial ESWT was applied only on the surface projections of the tibial tunnels, the intraosseous grafts from tibial sites were then more responsive to treatment. As the intra-articular graft is a deep structure in the knee joint, radial ESWT might not affect the intra-articular graft directly, or the intra-articular graft might exhibit later response to treatment.
Limitations
There were several limitations to this study. First, all the participants were male patients. There may have been too few female patients receiving ACLR during the enrollment period, and even fewer women were willing to participate in this clinical study. Therefore, caution should be taken when interpreting the results of the present study. Second, although different stages of the ligamentization process have been described by many authors, no agreement exists on their time frame or endpoint. 1,5,28,30 The timing of ESWT application at 3 months postoperatively was selected based on clinical experience. At that time, patients felt safer receiving additional treatment compared with earlier interventions, and they had a strong wish to recover faster. Third, although it was proved that the graft maturation was related to the postoperative outcomes within 2 years of follow-up, we did not establish causality between graft maturation and clinical outcomes because of the study design. Furthermore, we noticed that patients in the control group underwent meniscal repair or meniscectomy with double frequency compared with those in the experimental group. While no statistical difference was observed because of the small number of participants, possible biases may have come from the higher incidence of meniscal injuries in patients following a more restricted immobilization rehabilitation protocol resulting in lower signal intensity of graft on MRI scans. In addition, the achievements of MCID of all clinical measurements at 6-month follow-up were very low in both arms, but they were significantly improved at 24-month follow-up. We were curious to know the results at 12 months; however, we were unable to collect these data because of the COVID-19 outbreak preventing patients from coming to the hospital. Further, our cohorts were too small to calculate MCID. The MCIDs from previous literature to which this study referred may introduce deviation on the external validation. For example, when assessing the Tegner score, we cited the study of Weng et al, 42 who reported that a 1-point improvement in Tegner score reached MCID. However, in their study, the patients were older than 50 years, which is significantly older than our cohort. In addition, meaningful improvement may vary among individuals. Since younger patients have a higher activity level, the actual percentage of patients who reached the MCID for improvement in Tegner score in our cohort may have been less than reported. Fourth, SNQ has been used in various studies to predict the biomechanical and histologic properties of an ACL graft after ACLR, and the SNQ assessed on MRI scans was used as an indirect parameter to evaluate graft maturation in the present study. Nevertheless, the MRI scan acquisition and evaluation methods were very heterogeneous, 33 and quantitative MRI-based biomarkers of ACL graft healing are greatly needed to guide the appropriate time to return to sports after ACLR.
Conclusion
In patients who underwent hamstring autograft ACLR, 5 weeks of radial ESWT not only improved clinical function but also enhanced graft maturation at 24-month follow-up. ESWT may be a promising nonoperative treatment modality for these patients.
Footnotes
Acknowledgment
The authors acknowledge Quanxu Zhao and Zhouyue Yan, who contributed their expertise on the workout and prepared tutorial videos and images for the rehabilitation protocol.
Final revision submitted March 31, 2022; accepted May 16, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by the grants 18Z004 and 18Z002 funded by Shanghai Sports Science and Technology, “Comprehensive Plan” Project funded by Huashan Hospital, Fudan University, as well as by grant 82002367 funded by the National Natural Science Foundation of China. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Huashan Hospital of Fudan University.
