Abstract
Background:
The modified Broström-Gould (MBG) procedure is the gold standard for patients with chronic ankle instability (CAI), but it is relatively contraindicated for patients with higher body weight or generalized ligamentous laxity (GLL). The use of the ligament augmentation reconstruction system (LARS) is an alternative.
Hypothesis:
It was hypothesized that clinical outcomes would be similar in patients with increased body weight (>90 kg) or GLL, relative to controls.
Study Design:
Cohort study; Level of evidence, 2.
Methods:
A total of 66 patients satisfying the inclusion criteria were invited to participate and were divided into 3 groups: controls (no risk factors for inferior clinical outcome), patients with body weight >90 kg, and patients with GLL (Beighton score, ≥5 of 9). All patients underwent imbrication of the lateral collateral ligament complex augmented with the LARS. Primary outcomes of interest were Tegner activity scale (TAS) and Foot and Ankle Outcome Score (FAOS) subscale scores. Secondary outcomes were recurrence of ankle instability, the need for further surgery, and/or complications. Patients were reviewed at 2 and 5 years postoperatively, and outcomes between groups were compared using repeated-measures analysis of variance.
Results:
Complete data were available for 63 patients (21 patients in each group). TAS improved in all groups from preoperatively to 2 years and 5 years postoperatively (P < .001 for all). Relative to the controls, the TAS scores were lower in the >90-kg group at 2 years and 5 years (P < .001 for both periods), while the GLL group had similar scores to controls at both postoperative periods. Both the >90-kg and the GLL groups showed no significant difference in improvement on any FAOS subscale scores relative to the controls, at both 2 and 5 years postoperatively. There were no recurrences, repeat surgeries, or major complications.
Conclusion:
Relative to controls, patients with body weight >90 kg or GLL had similar FAOSs, and TAS scores were lower in the >90-kg group, at 2 and 5 years, after the use of the LARS to augment lateral collateral ligament imbrication for CAI. Use of the LARS in this manner is a viable option in patients for whom the MBG procedure is relatively contraindicated.
Ankle sprains are among the most common musculoskeletal injuries, accounting for up to 40% of injuries among physically active individuals. 10 –12,18,19,37,45,52,58 Most are acute in nature, and the majority involve the lateral ligament complex (LCL). 3 Injuries to the LCL complex most commonly involve the anterior talofibular ligament (ATFL) and the calcaneofibular ligament (CFL), with the incidence being 7 per 1000 persons per year. 21,35
Although most ankle sprains completely heal with appropriate nonoperative management, up to 40% of those who sustain them develop chronic ankle instability (CAI). 7,55 CAI has been associated with many long-term sequelae, such as altered ankle kinematics, 46 chondral injuries, 48,57 and early-onset osteoarthrosis of the ankle joint. 20,48,54 For these reasons, surgical repair of the ruptured LCL complex is indicated when nonoperative management has failed. 1,4
Although contentious, several studies have revealed that individuals with higher body weight and body mass index and those with generalized ligamentous laxity (GLL) are at increased risk of ankle sprains. 2,5,6,43,55 GLL is characterized by abnormally increased range of motion (ROM) at multiple joints compared with that of the general population and is related to conditions with abnormal collagen laxity such as Marfan syndrome. 59 The prevalence is between 5% and 15% of the population, and most cases have no known genetic aberrancy. 23 The increased laxity is a contributing factor to increased risk of CAI after acute injury and the risk of recurrence after ligament reconstruction. 18
The modified Broström-Gould (MBG) procedure has evolved as the surgical treatment of choice for chronic lateral ligament instability of the ankle. 8,28,29,40 There are concerns about the strength of this procedure for some patients, particularly those with GLL, increased body mass, and long-standing instability, as well as those who participate in competition at an elite level. 38,55,60 Alternative procedures have reconstructed the LCL complex using an autologous graft, but this is associated with the risk of graft-site morbidity. 12,25 Another option is to augment an imbrication of the LCL complex using a synthetic ligament. The ligament augmentation reconstruction system (LARS) device made of polyethylene terephthalate (PET; Corin Group) has been used in this manner and has the advantages of a biological repair of native ligaments, protected by a strong synthetic ligament during the reparative phase. 40,41
A recent randomized controlled trial (RCT) with 5-year follow-up found that the use of the LARS resulted in higher patient-scored outcomes and activity levels with lower recurrence rates relative to the MBG procedure, in physically active patients with CAI. 41 That study excluded patients who were regarded to be at a higher risk of failure after the MBG procedure, including body weight >90 kg, GLL, failed previous ankle reconstructions, and long-standing chronic instability. The authors of that paper chose these exclusion criteria to avoid biasing the study in favor of the LARS procedure.
Given that the LARS has been demonstrated to have superior results in patients without these risk factors, the objective of our research was to determine if the LARS has similar outcomes when used in patients with a higher risk of failure (ie, body weight >90 kg or GLL). Our null hypothesis was that the outcomes would be similar even in the presence of these risk factors.
Methods
Study Design and Population
After receiving approval from the hospital ethics committee, we conducted a prospective cohort clinical study. The supervising author (M.D.P.), a subspecialist sports orthopaedic surgeon, performed all procedures. Patients evaluated by the surgeon who satisfied the inclusion and exclusion criteria in Table 1 were invited to participate in the study. All patients had a sports-related traumatic onset of ankle instability. The diagnosis of disruption of the ATFL and CFL was confirmed by magnetic resonance imaging and physical assessment. The physical signs were a grade 3 talar tilt test and grade 3 anterior drawer test.
Inclusion and Exclusion Criteria for Patient Enrollment a

a ATFL, anterior talofibular ligament; CFL, calcaneofibular ligament; MRI, magnetic resonance imaging.
Patients were regarded as physically active if they took part in recreational or competitive physical activity at least 3 times per week. All patients were informed of the rationale behind the study, and all consented to undergo the LARS procedure. All patients followed the same rehabilitation program after surgery (Table 2). Patients were informed that if they opted not to take part in the study, this would not result in any change to the treatment provided.
Rehabilitation Protocol for All Patients After Ankle Stabilization Surgery a

a ROM, range of motion; WBAT, weightbearing as tolerated.
Treatment Group Allocation
Patients were divided into 3 groups. Those who had no known risk factors for recurrence, weighed <90 kg, and did not have GLL (Beighton score, ≤4 of 9) were allocated to the control group; patients with increased body weight were placed in the >90-kg group, and those with a Beighton score ≥5 were placed in the GLL group. None of the patients in the study had both GLL and body weight >90 kg.
Surgical Technique
All procedures were performed with the patient under a general anesthetic, in a supine position, with a sandbag under the ipsilateral pelvis and an inflated tourniquet. Examination under anesthesia and arthroscopy were performed before the stabilization. The LARS procedure was performed as described in the literature and shown in Figure 1. 40,41 An anterolateral approach was used to approach and divide the anterolateral capsuloligamentous structures to expose the anterior surface of the lateral malleolus. A 5-mm fibular tunnel was drilled posterosuperiorly from a point 5 mm proximal to the tip of the malleolus and angled in such a manner that it was at least 20 mm in length before it passed through the posterior cortex of the fibula.
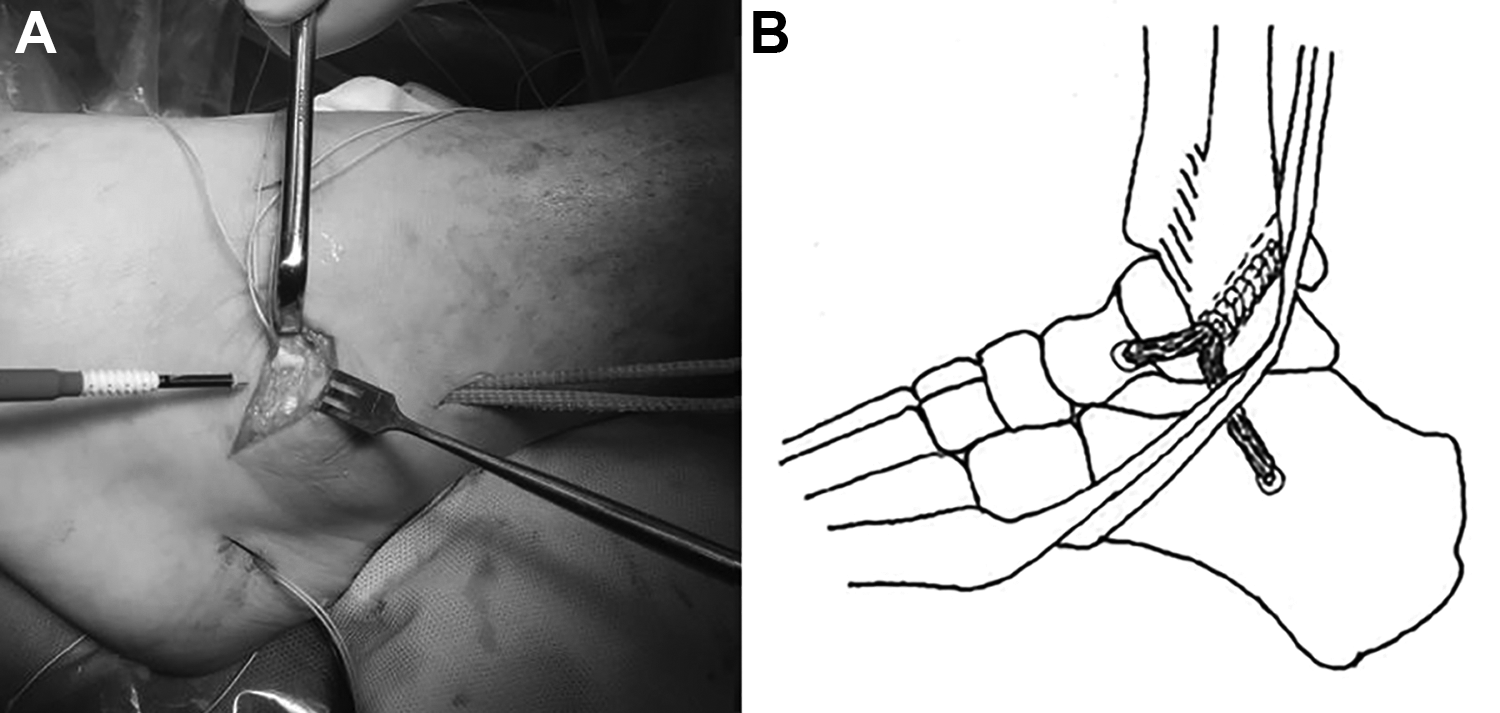
The surgical technique performed using the Ligament Augmentation Reconstruction System (LARS). (A) Intraoperative photograph showing the LARS in situ, before final tensioning and fixation. (B) Final positioning of the LARS, which is cut flush with the posterior surface of the fibula.
We used the LAC 20 LARS ligament (Corin Group), which was a straight, porous, cordlike ligament made of interlocked PET fibers. The diameter was 4 mm and compressible. The looped end of the folded ligament was passed from distal to proximal along the 5-mm fibular tunnel. The looped end was retrieved via the proximal 1-cm skin incision. The CFL limb was passed from the distal end of the fibular tunnel, deep to the peroneal tendons and sheath, and was retrieved via the distal 1-cm skin incision. Using this incision, the CFL limb was secured in a 4.5-mm bone tunnel at its insertion site using a 4.75-mm suture anchor (BioComposite SwiveLock; Arthrex). The ATFL limb was passed from the fibular tunnel, superficial to the capsuloligamentous structures and fixed in a 4.5-mm bone tunnel at its distal attachment using a similar 4.75-mm suture anchor. Traction on either side of the looped end of the LARS ligament allowed the tension of the ATFL and CFL limbs to be adjusted independently, until the anterior drawer and talar tilt were corrected. Before final fixation with a third SwiveLock in the fibular tunnel, the surgeon checked that the LCL reconstruction had not been overtensioned and did not restrict the passive ROM in the ankle joint. The capsuloligamentous structures were then closed, maintaining the LARS in an extracapsular position and double-breasting the attenuated tissues.
At the end of the procedure, all patients were placed in a removable fracture boot. They were asked to remain nonweightbearing but encouraged to perform active postoperative ROM work for the toes, knee, and hip. As prophylaxis against deep venous thrombosis, all patients were placed on subcutaneous enoxaparin 40 mg daily between surgery and the first postoperative review. They were also asked to remove the fracture boot to perform active ROM in dorsiflexion and plantarflexion as tolerable. At the first postoperative review, the wound was checked, and thereafter patients were allowed to begin weightbearing as tolerable in the fracture boot. The rehabilitation program detailed in Table 2 was commenced under the supervision of the treating physical therapist. The rehabilitation protocol aimed to return patients to full activity within 4 months of surgery.
Primary Outcomes of Interest
The primary outcomes of interest were Tegner activity scale (TAS) and Foot and Ankle Outcome Score (FAOS). Both outcome measures were emailed to patients and returned to the supervising author. Patients completed the FAOS before undergoing surgery and then again at 2 years and 5 years after surgery. This is a patient-scored questionnaire validated for use in ankle lateral ligament injury. 44 It consists of 5 domains that are scored out of a possible maximum of 100 points (higher values represent better clinical outcomes), from which an average score, or total, is computed. The FAOS domains are pain, other symptoms, activities of daily living, sports/recreational activities, and overall quality of life.
TAS scores were recorded based on preinjury status, then again immediately before surgery (0 years), and then at 2 and 5 years after surgery. The TAS is a numerical scale with values from 0 to 10 representing specific activities. Values of 10 represent the highest activity level (eg, one of the codes of football at an elite level) and 0 the lowest (no sports activities and unable to do even sedentary work). It has a minimal detectable value of 1.
Secondary Outcomes of Interest
Secondary outcomes were recurrence of ankle instability, need for further surgery, and/or complications. Patients were routinely asked to report any complications to the surgeon. All complications requiring more than normal postoperative care, including readmission to the hospital, infection, further surgery, thromboembolism, nerve injury, or recurrence of instability, were recorded in the patient’s notes over the 5-year period. Patients were asked again to document any complications that had occurred and report these at the same time as completing the FAOS and TAS questionnaires.
Statistical Analysis
A power analysis was performed based on prior research performed on the use of the LARS ligament, with documented recurrence rates and range of values for FAOS and TAS. 26 With an SD of 5 points for the total FAOS, 19 patients were required in each group to achieve a power of 80% with 95% significance. If 1 point is regarded as clinically important, then to detect a significant difference between the mean TAS, 20 patients were required in each group.
Repeated-measures analysis of variance was performed to compare the control group with the high-risk groups (>90-kg and GLL) regarding changes in FAOS subscales at 0, 2, and 5 years after surgery, and changes in the TAS at preinjury and 0, 2, and 5 years after surgery. FAOS for each subscale was then adjusted for age and sex, and TAS was adjusted for preinjury scores, age, and sex. Changes were compared between and within groups, with P <.05 regarded as significant.
Data entry was performed in Microsoft Excel, and statistical analysis was conducted using SPSS Version 26 (IBM).
Results
Patient Characteristics
A total of 66 patients underwent lateral ankle stabilization using LARS between April 2009 and May 2011. No patients refused to take part in the study. Three patients were lost to follow-up, all in the >90-kg group, and their data were not included for analysis. These patients had changed postal address and internet address and/or had moved overseas. To the best of our knowledge, none of these patients had further ankle instability and/or surgery or any significant complications. Thus, 63 patients were enrolled in the study (21 patients in each group).
Table 3 summarizes general patient characteristics overall and by group. Although the male:female ratio was relatively equivalent for the control group, it was 1:1.6 for the GLL group and 3.2:1 for the >90-kg group. There was no significant difference in preinjury TAS scores between either high-risk group relative to the control group (Table 3).
Patient Characteristics a
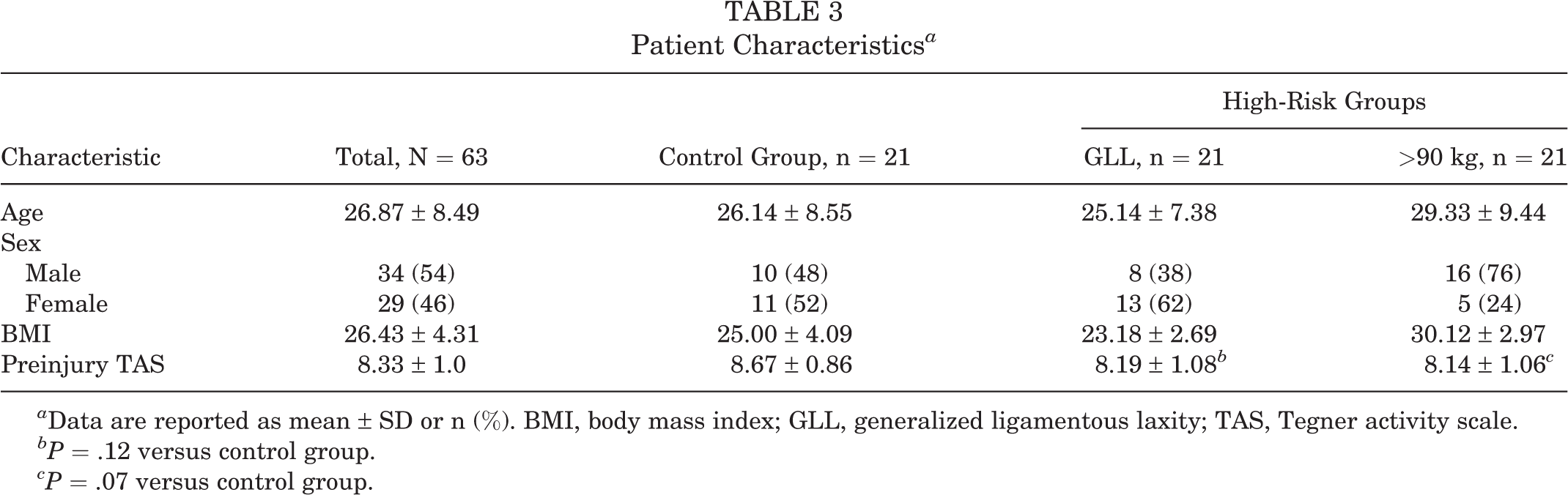
a Data are reported as mean ± SD or n (%). BMI, body mass index; GLL, generalized ligamentous laxity; TAS, Tegner activity scale.
bP = .12 versus control group.
cP = .07 versus control group.
TAS and FAOS Outcomes
Patients in all 3 study groups saw significant improvements in TAS scores from preoperatively to 2 years after surgery, and this improvement was maintained at 5 years after surgery (P < .001 for all). There was no difference between the GLL and control groups at 2 and 5 years postoperatively (P = .18 and .14, respectively). The improvement in TAS scores was significantly lower in the >90-kg group at 2 and 5 years relative to the controls (P = .02 and .03, respectively). The comparison of TAS scores between the control group and the high-risk groups indicated significant differences at both 2 years and 5 years postoperatively (P < .001 for both). These results were unaltered after adjustments made for preinjury scores, age, and sex (P < .001) (Table 4).
Comparison of Postoperative TAS and FAOS Results Between the Control Group and High-Risk Groups a

a Data are reported as mean ± SD. ADL, activities of daily living; ANOVA; analysis of variance; FAOS, Foot and Ankle Outcome Score; GLL, generalized ligamentous laxity; LARS, ligament augmentation reconstruction system; QoL, quality of life; TAS, Tegner activity scale.
b Adjusted for age and sex.
c TAS adjusted for preinjury scores, age, and sex.
There were no significant differences in any FAOS subscale between either high-risk group when compared with the control group at 0, 2, or 5 years after surgery (Table 4). No significant differences were found on any subscale, even with adjustments for age and sex, between either high-risk group when compared with the control group.
Secondary Outcomes of Interest
Over the 5-year period, no patient reported recurrent instability, nor were there any complications requiring additional surgery or admission to the hospital. There were only 5 complications recorded in the study population. These included 3 cases of superficial wound infection that responded to a course of oral antibiotics, 2 in the >90-kg group and 1 in the control group. There was 1 case of deep venous thrombosis in the >90-kg group and 1 case in the control group, and both responded to oral anticoagulants.
Discussion
The findings in this study demonstrated that physically active patients with GLL or body weight >90 kg had similar outcomes in terms of the FAOS when compared with the control group at the 5-year follow-up. However, only the GLL group had similar TAS scores. This outcome persisted when adjustments were made for age and sex among all groups. Therefore, we accept our null hypothesis. There were no clinical failures in any of the patients. The study was not adequately powered to compare the low incidence of complications between the groups, with an 8% risk overall for any complication.
Patients with body mass >90 kg are relatively common in sports such as tackle football. Patients with ligamentous laxity may be disproportionately represented in certain sporting activities, such as gymnastics, swimming, and dance. Such patients have been found to be more susceptible to ankle sprains with recurrent instability and the subsequent development of CAI. 5,6,43 Ankle instability in these 2 groups is predominantly mechanical because of anatomical defects such as arthrokinematic changes, ligamentous tear, and/or degenerative pathological laxity. 26,27,34 However, there may also be an element of functional instability that was not measured in this study. 14
The ultimate goal of surgery for treatment of CAI is to restore normal hindfoot biomechanics, 42 and the anatomic MBG procedure remains the gold standard. 30,36 However, several studies have shown poorer outcomes after the MBG procedure for patients with a high body weight and/or GLL, even if the interior extensor retinaculum is included, 29 rendering these conditions relative contraindications. 24,28,29,38,39,60 Other factors attributable to poorer outcomes include repair strength achieved after imbrication of tissues that have already been damaged 1,17,25,33,56 and the difficulty in repairing the extracapsular CFL. 47 Waldrop et al 56 has shown that the initial mechanical properties of the ligaments in the standard Broström repair were 50% weaker than those of native ATFL immediately after repair.
Alternative surgical procedures for these higher-risk patients include the nonanatomic reconstructive procedures, but these have been gradually replaced with anatomic ligament reconstruction procedures. However, there is morbidity associated with harvesting the autologous graft, while the use of allografts increases costs and there is the risk of disease transmission. The use of the synthetic LARS is an alternative without the associated risks of graft harvesting.
Originally used for the reconstruction of the anterior cruciate ligament, the LARS may be more suited to use as an extracapsular augmentation device. When used to augment an imbrication of the LCL complex of the ankle, the synthetic graft is placed extracapsularly, thereby avoiding the potential occurrence of polyethylene synovitis, as has occurred with reconstruction of the anterior cruciate ligament of the knee. 13,15,31,51 The ankle joint is more constrained by bone anatomy than the knee and is less reliant on extra-articular soft tissues than the knee joint, rendering this synthetic device potentially more suitable for ankle LCL reconstruction. This may be even more important in patients with GLL given the already increased ROM compared with normal, across multiple joints. 5,6
There is a paucity of published level 1 studies investigating the use of synthetic ankle ligaments. A recent meta-analysis performed by Lei et al 32 attempted to compare the use of artificial ligaments with the MBG procedure for ankle instability. Their study concluded that there was no evidence that the LARS technique improved the clinical outcome relative to the MBG procedure for lateral ankle instability, on the basis of the data from what they described as 4 different RCTs. 32 The paper pooled the results of techniques that used synthetic ligament irrespective of the design of the ligament or which components of the LCL complex were reconstructed. Two studies included in the meta-analysis were published by Porter et al. 40,41 Both of these RCTs used the PET LARS device to augment both the ATFL and the CFL and found a superior outcome relative to the MBG procedure at the 2-year and 5-year follow-up visits. The other 2 studies were by Ulku et al 53 and Cho et al, 9 and both used an internal brace device (InternalBrace; Arthrex). This internal brace device consists of a braided ultra—high molecular weight polyethylene/polyester suture tape with knotless bone anchors. The paper by Cho et al 9 was in fact an uncontrolled case series rather than an RCT, and all patients were women and weighed <70 kg. The Ulku et al paper was an RCT, but all patients underwent either an ATFL repair or augmentation of the ATFL only. The results of the meta-analysis are not applicable to patients who undergo a reconstruction of both the ATFL and the CFL using the LARS and who are male and/or weigh >70 kg.
The LARS is a strong synthetic ligament with an ultimate tensile load of 1000 N, 49 and it acts a scaffold for the ingrowth of collagen tissue. 51 The LARS induces biointegration and ultimately incorporates the remnants of the native ligament itself, thereby facilitating healing. With biomechanical behavior of the ankle joint being nonlinear and dependent on the magnitude of the load applied, 50 this is of particular importance in those with high body weight, as any excess weight negatively affects healing ability because of increases in mass moment of inertia on the talocrural joint. 3,16,43 Although coated with collagen, there is no published evidence that the internal brace device stimulates ingrowth of fibrous tissue in vivo, and although stronger than native tissue, its ultimate tensile load is less than that of the LARS. 22 Hence, there are some salient differences between the 2 devices, and pooling the data from the use of the 2 devices may not be appropriate.
Limitations
This study has several limitations. Although the data at 5 years are consistent with an excellent clinical outcome in these patients, longer-term studies are required to fully evaluate the role of the LARS in patients with CAI. The study is not an RCT design, but the goal was to compare the outcome after the same surgical procedure in different patient groups. Patients were sequentially enrolled in the study, and none refused to take part. The study did not investigate the potential role of functional instability as a confounding factor. The small sample size in each group reduces the generalizability of the results, but the power analysis demonstrated that it was large enough to answer the hypothesis. In common with the MBG procedure, the internal brace procedure, and many ligament reconstruction techniques, the surgeon cannot be sure that the final tension in the reconstruction is the same in all patients. However, the LARS technique used allows the tension to be adjusted before final fixation to avoid overconstraint of the ankle joint. Although the patient-scored outcomes used in this study have been validated for use in lateral ankle instability, the scoring remains subjective in nature. However, there is no practical valid objective imaging modality or other technique to measure functional instability of the ankle. Any additional radiation exposure or expense when clinically unnecessary was deemed an unacceptable departure from standard treatment of patients for ethics approval. The outcome after the ankle lateral ligament reconstruction is also influenced by the level of physical activity, and all 3 groups had similar levels of activity. Sex may be a confounding factor. The GLL group had a higher percentage of women, and the >90-kg group had a higher percentage of men relative to the control group. The study was not powerful enough to compare the incidence of complications in the treatment groups.
Conclusion
The findings of the current study demonstrated that patients weighing >90 kg or those with GLL have similar excellent clinical outcomes compared with patients without these risk factors, in terms of the TAS and FAOS when reviewed 5 years after lateral ankle reconstruction using the LARS, with a low risk of recurrence or significant complications. The use of the LARS may be specifically indicated for these patients.
Footnotes
Final revision submitted January 21, 2022; accepted February 8, 2022.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Barton Private Hospital (reference No. BPH EC O2).
