Abstract
Background:
Glenoid cartilage defects may contribute to anterior shoulder instability recurrence and progression to osteoarthritis, but their morphology remains unknown.
Purpose/Hypothesis:
The purpose was to determine the shape, size, and location of glenoid cartilage defects and the prevalence and risk factors for cartilage defects in the setting of anterior glenohumeral instability. It was hypothesized that glenoid cartilage defects would be common, would be associated with recurrence of dislocation, and would share similar morphology with glenoid osseous defects.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
In this retrospective study, all patients who underwent arthroscopic surgical treatment for anterior glenohumeral instability between January 2012 and May 2019 were included; excluded were patients with documented posterior or multidirectional instability or previous glenohumeral surgery. For each patient, the operative report, arthroscopic images, and preoperative magnetic resonance imaging (MRI) scans were reviewed to determine the prevalence of cartilage injury. For those patients with an Outerbridge grade 3 or 4 defect, the cartilage surfaces on the MRI scans were segmented to make 3-dimensional (3-D) segmentations. From these 3-D segmentations, we measured length, width, and surface area of the glenoid and defect, and the orientation of the defect relative to the superior and inferior poles of the glenoid. A multivariable analysis was conducted to determine correlates with cartilage damage.
Results:
In 322 patients treated operatively for anterior glenohumeral instability, 38% had a concomitant cartilage defect. The mean cartilage defect was located directly anteriorly at the 3:07 clockface position (range, 2:10-4:05) and encompassed 6.5% ± 3.5% of the glenoid surface area. However, defects ranged up to >56% of glenoid length and up to 27% of glenoid width, and the largest defect encompassed 19.5% of the glenoid cartilage surface area. Patients with a cartilage defect were more likely to be male (
Conclusion:
Cartilage defects were common in patients with operatively treated anterior glenohumeral instability, occurring in 38% of patients. These defects were located directly anteriorly at 3:07, similar to osseous glenoid defects. Future prospective studies with cartilage-specific MRI sequences should be conducted.
Numerous studies have been performed detailing the prevalence of, morphology of, and risk factors for glenoid osseous defects after anterior shoulder instability. Biomechanical studies regarding glenoid osseous defects have reported a relationship between the width of the defect and recurrent instability. 13,35 In these studies, glenoid osseous defects were located anteriorly, 2,15,30 and 25% of the glenoid width is generally regarded as the critical width, although one study has suggested a lower width. 35
Anterior shoulder dislocations are associated with anterior glenoid cartilage defects in up to 64% of cases. 7,17,24,34 Normally, the concavity in cartilage deepens the glenoid socket, 32,36 broadening the curved articular surface and lengthening the arc length of the glenoid (Figure 1). The presence of osseous defects alters contact surface, arc length, and socket depth, so isolated cartilage defects could play a similar biomechanical role in instability. Glenoid labrum articular disruption injuries have been shown to be associated with an increased risk of recurrence. 28 Without surgical treatment, instability recurs in 38% to 80% of individuals. 1,3,11,16,29 Even with surgical treatment, instability recurs in up to 26% of individuals. 21 Glenohumeral instability results in osteoarthritis (OA) in 20% to 60% of cases with long-term follow-up. 4,8,9,12,23,25,27,33 Current surgical treatment does not restore preinjury stability, and abnormal postoperative translation of the humerus relative to the glenoid may contribute to OA. 26 Cartilage defects may partially explain both recurrent instability and the progression to OA despite surgical stabilization. 5,9,28
No studies have examined isolated glenoid cartilage defects in the setting of anterior shoulder instability. Isolated cartilage loss might contribute to recurrent instability. 18,19,28 To understand the contribution of cartilage defects to instability recurrence and OA progression, we must first understand their morphology, prevalence, and risk factors. Cartilage-specific sequences add time and cost to magnetic resonance imaging (MRI) scans, and thus there must be sufficient rationale to justify a prospective study incorporating these sequences. Therefore, the purpose of this study was to determine the morphology, prevalence, and risk factors for glenoid cartilage defects in the setting of anterior glenohumeral instability to understand whether a future prospective study is indicated. It was hypothesized that cartilage defects would be common and associated with recurrence of dislocation and would share similar morphology to osseous glenoid defects.
Methods
Study Cohort
Institutional review board approval was received for this retrospective study and the requirement for informed consent was waived. Included were all patients who underwent arthroscopic surgical treatment for anteroinferior glenohumeral instability at a single institution (University of Utah) between January 1, 2012, and May 8, 2019. The case logs of our institution were reviewed, and all patients were included if the Common Procedural Terminology codes 29806, 23455, 23466, 23462, 23460, and 23465 were used (507 patients). We then excluded revision procedures (79 patients), as surgical stabilization may contribute to cartilage injury. We also excluded patients who had been documented to have posterior instability (63 patients) or multidirectional instability (43 patients). This left 322 patients who underwent primary surgery for anterior glenohumeral instability.
Data Collection
For each patient, the following data were collected: age, sex, body mass index (BMI), tobacco use, whether the patient was documented to have been a contact or collision athlete, the number of dislocations before surgical treatment, the length of time from the first dislocation to surgical treatment, patient positioning during surgery, and the number of anchors used in the repair. We also noted concomitant procedures, including superior labral repair, posterior labral repair, remplissage, rotator interval closure, biceps tenodesis, distal clavicle excision, subacromial decompression, rotator cuff repair, and repair of humeral avulsion of the glenohumeral ligaments (HAGLs). The operative report, arthroscopic images, MRI scans, and, if available, computed tomography (CT) images were reviewed to determine whether a cartilage defect was present at the time of surgery. If any of these sources described a defect, then the patient was considered to have a defect; however, only those patients in whom the defect was visualized preoperatively on an MRI scan were included in the defect modeling portion of the study.
Defect Modeling From MRI Scans
MRI scans were obtained with patients in the supine position in a Magnetom Avanto Fit scanner (Siemens) using shoulder coils. These images were intra-articular gadolinium-enhanced arthrograms, and all defect measurements were made on MRI scans, with no defect size measurements made intraoperatively. T1 fat suppression axial acquisition was performed using 1.2 mm × 1.2 mm pixels at a 3.3 mm slice thickness. Imaging parameters included a repetition time of 650 ms, echo time of 13 ms, and a 320 × 320-voxel field of view. All images were saved in DICOM (Digital Imaging and Communications in Medicine) format and were reviewed by a fellowship-trained orthopaedic shoulder and elbow surgeon (P.N.C.). This surgeon ensured there was no OA, fracture (other than Hill-Sachs), or implants from previous surgery.
The DICOM image stacks were imported into Mimics (Version 21.0 Innovation Suite; Materialise). All MRI scans were obtained in the supine position, so axial images were not orthogonal to the long axis of the glenoid, defined as the line connecting the superior pole and inferior pole of the glenoid. Therefore, the axial images were resliced so as to be orthogonal to the long axis of the glenoid. After reorientation, segmentation was performed to generate a 3-dimensional (3-D) representation of the remaining cartilage (Figure 1). First, the glenoid cortical bone was semiautomatically segmented using built-in thresholding functions (Figure 1, blue area). The area of lower intensity than contrast and higher intensity than cortical bone area adjacent to the subchondral bone area was defined as the cartilage. As the chondrolabral junction is difficult to determine from MRI scans, we defined the perimeter of the cartilage using the perimeter of the subchondral bone (Figure 1, green area). 14 If the cartilage area ended within the perimeter of the subchondral bone, this was defined as a cartilage defect and separately segmented (Figure 1, red area). In cases of cartilage thinning, if the cartilage was >50% thinner than the expected thickness (as judged via the posterior cartilage), then it was considered as a defect. After segmentation, the resulting 3-D surfaces were smoothed (iterations, 10; smooth factor, 1; with shrinkage compensation) and wrapped (smallest detail, 0.3 mm; gap closing distance, 0.5 mm) to fill voids and reduce stair-step artifact.

Segmentation of cartilage volume on MRI scans for shoulders with (A) no cartilage defects and (B) cartilage defects. The glenoid cortical bone was first semiautomatically segmented using built-in thresholding functions (blue area). The lower intensity than contrast and higher intensity than cortical bone area adjacent to the subchondral bone area was defined as the cartilage (green area). As the chondrolabral junction is difficult to determine on MRI scans, we defined the perimeter of the cartilage using the perimeter of the subchondral bone (yellow line). If the cartilage area ended within the perimeter of the subchondral bone, this was defined as a cartilage defect and separately segmented (red area). MRI, magnetic resonance imaging.
Cartilage Defect Measurement
Once 3-D models of both the cartilage and defect were made (Figure 2), measurements were performed using 3-Matic (Version 13.0; Materialise). From these 3-D reconstructions, the following points were identified (Figure 2): glenoid superior pole (point
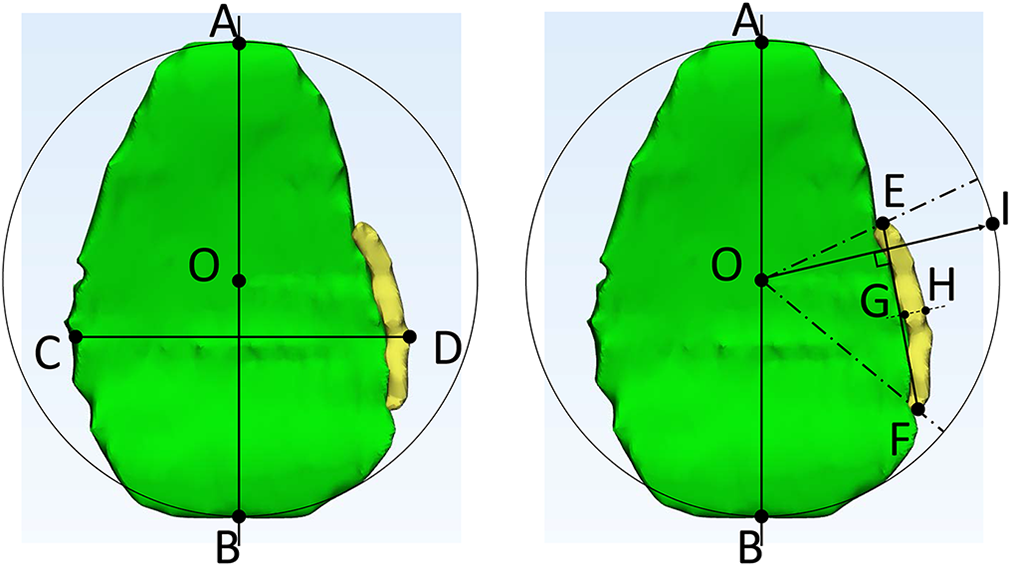
Measuring glenoid cartilage (left) and glenoid cartilage defect (right). Cartilage is shown in green; defect, in yellow. The following points were identified: glenoid superior pole (point
These same points were then used to determine the location of superior and inferior extent of the defect as previously described (angle
All measurements were performed by 2 orthopaedic surgeons (P.N.C. and J.K.) blinded to each other’s measurements, as well as 1 orthopaedic surgeon (J.K.) separated by 2 weeks for interrater reliability. The intraclass correlation coefficient (ICC; 2-way mixed effects, single rater, absolute agreement) was calculated for each outcome for interrater and intrarater reliability, and 95% confidence intervals (CIs) were reported. For these ICCs, the a priori level of acceptability was 0.75. 6 No standard data were available, and thus we did not calculate accuracy.
Statistical Analysis
Study variables were summarized descriptively. Continuous variables were summarized as mean, standard deviation, and range, and categorical variables were summarized as frequency and percentage. Preoperative and operative variables, including age, sex, BMI, tobacco use, whether the patient was documented to have been a contact or collision athlete, the number of dislocations before surgical treatment, the length of time from the first dislocation to surgical treatment, and whether concomitant procedures were performed, were compared between the subgroups of patients who had a glenoid cartilage defect and those who did not have a glenoid cartilage defect to determine risk factors for cartilage defects. Concomitant procedures for which <5 were performed were excluded. No analysis was conducted to compare number of dislocations or mechanism of injury with defect size or location as it was believed that the study would be underpowered for such an analysis. Continuous variables were compared using the Student
Results
Study Cohort
Of the 322 included patients, 121 (38%) had a cartilage defect. Of the 262 who had an MRI scan available in the system, 96 (37%) had a cartilage defect. Of the 60 patients who did not have MRI scan data, 25 were found to have a cartilage defect on the basis of review of operative reports and intraoperative imaging. The included patients were mostly male, nonsmokers, and not contact or collision athletes and had recurrent dislocations (Table 1). Intraoperatively, most patients did not undergo concomitant superior labral repairs or posterior labral repairs, underwent an anterior labral repair with 3 to 4 anchors, and did not undergo concomitant remplissage (Table 2).
Characteristics of Included Patients

Surgical Characteristics of Included Patients

Defect Characteristics
Reliability for all of the glenoid and defect measurements was excellent (Table 3). Of the 96 patients with cartilage defects, 27 had osseous glenoid fractures, and these were excluded. A total of 69 patients (21%) had an isolated glenoid cartilage defect (Figure 3); all were aged <50 years, and thus these defects were deemed unlikely to be degenerative. Of the 69 patients, 26 had Outerbridge grade 3 lesions, and 25 had Outerbridge grade 4 lesions; only these were included in cartilage segmentation. Of these 51 patients, 6 had delamination defects or island defects without continuity with the rim, and these were excluded from cartilage segmentation, as it was believed that they could not be segmented accurately. The remaining 45 defects were segmented and measured (Table 3).
Defect Characteristics and Measurement Reliability


Flowchart of selection of defects for segmentation. 3D, 3-dimensional; Shlds, shoulders.
The glenoid cartilage defects ranged between 0:59 and 5:07 on the clockface of the glenoid. The mean location was between 2:10 ± 0:31 and 4:05 ± 0:39 (Figure 4). The mean defect midpoint and orientation (3:07 ± 0:28 and 2:43 ± 0:16, respectively) were both nearly directly anterior. One distinct subset of cartilage defects was anterosuperior at the glenoid attachment of the upper border of the anteroinferior glenohumeral ligaments (n = 6/45; 13%) (Figure 5). Defects ranged from >56% of glenoid length and up to 27% of glenoid width. The largest defect encompassed 19.5% of the glenoid cartilage surface area.

Schematic of the mean cartilage defect extent on the clockface. Grey area shows SDs.

A subset of patients displayed the cartilage defect subtype of an anterosuperior defect shown. This was thought to be an avulsion of the anteroinferior glenohumeral ligament complex from the glenoid.
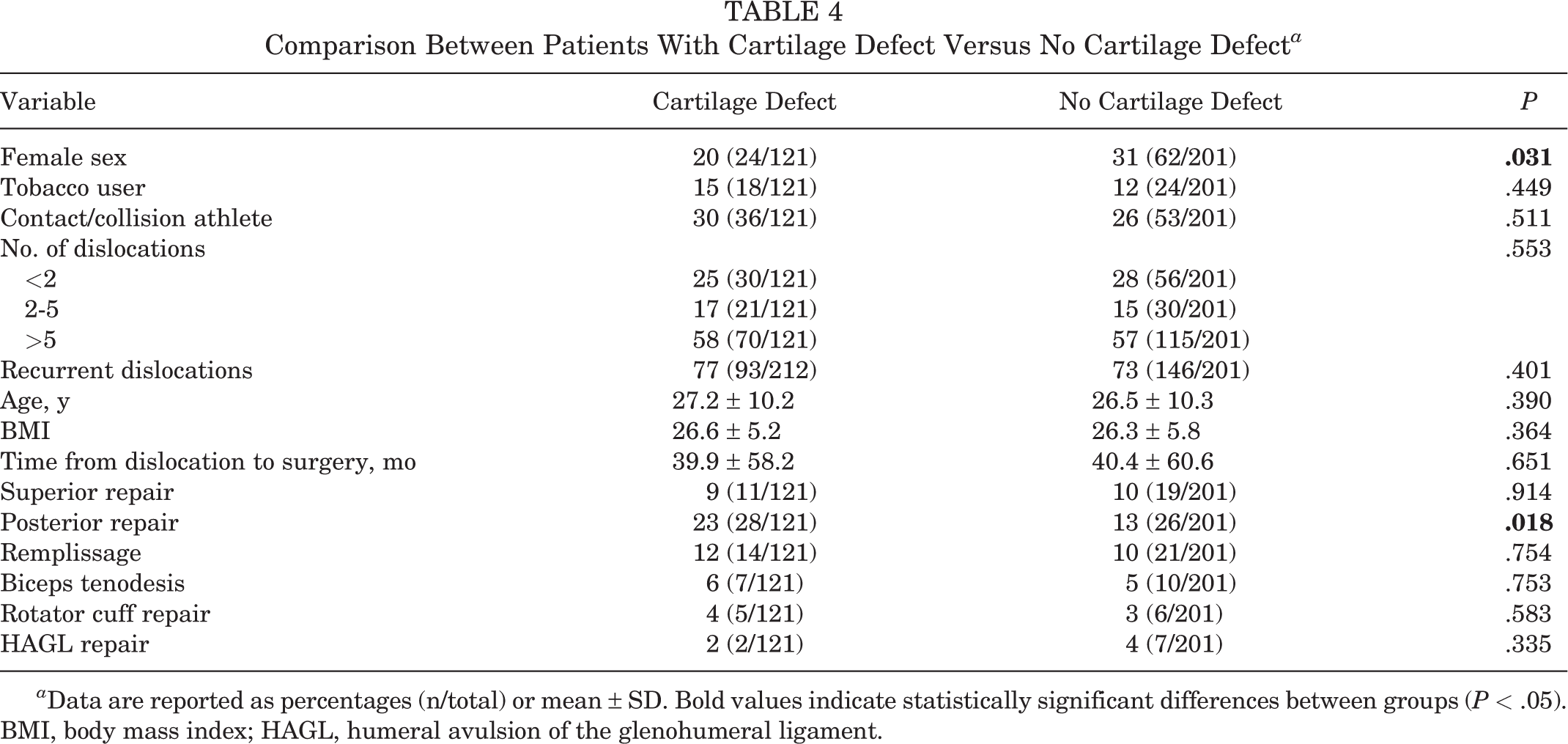
Risk Factors
When comparing the cartilage defect and noncartilage defect groups, we observed significant sex-based differences, with females having a significantly lower risk for cartilage defects (Table 4,
Comparison Between Patients With Cartilage Defect Versus No Cartilage Defect
Discussion
Defects were common in the current study, with 38% of patients having a concomitant cartilage defect. The defects were oriented directly anteriorly, similar to the orientation of osseous defects. While the mean cartilage defect size was small, encompassing 6.5% ± 3.5% of the glenoid surface area, the largest defect encompassed 19.5% of the glenoid cartilage surface area.
Despite several previous studies having examined the prevalence of isolated cartilage defects in the setting of glenohumeral instability, there is no current agreement as to how common cartilage defects are in this setting. Duchman et al 7 conducted a retrospective review of glenoid bone and cartilage lesions encountered in the Multicenter Orthopaedic Outcomes Network (MOON) and found these lesions in 10.9% of primary cases. Krych et al 17 conducted a retrospective review of 87 shoulders and found 63% to have glenoid isolated cartilage injuries. O’Brien et al 24 conducted a retrospective review of magnetic resonance arthrograms in 101 patients with instability and found 36% had glenoid isolated cartilage lesions. There is no agreement as to the prevalence of glenoid isolated cartilage loss, which has been described as 10.9%, 7 63%, 17 36%, 24 and 57%. 34 The prevalence of glenoid bone loss has been reported to be 70% to 97% of recurrent instability cases. 10,20,22,31 So, the prevalence of cartilage defects when bone loss is included is likely much higher. Our own results suggested that glenoid cartilage defects are common.
As the mean cartilage defect was oriented directly anteriorly in our study, future biomechanical studies should study direct anterior defects, not anteroinferior defects, to best replicate the pathology. 18 No previous studies have been conducted to describe the size and orientation of glenoid cartilage defects. However, numerous previous studies have been conducted to describe the morphology of glenoid bone loss. Saito et al 30 conducted a retrospective review of 123 shoulders and found these lesions to range most commonly between 2:30 and 4:20 on the clockface, with a mean extent of 107° and a mean orientation of 3:01, which is nearly perfectly anteriorly directed. Bockmann et al 2 confirmed a nearly straight anterior osseous defect orientation in 44 shoulders. Ji et al 15 revealed the common location of the glenoid osseous defect was the 3:20 clockface position. Our own results suggested that glenoid cartilage defects are oriented directly anterior relative to the glenoid, similar to osseous lesions.
Although the measurement techniques described in this study are too onerous for routine clinical application, better understanding the morphology of the pathology provides insights into pathogenesis. For instance, in our study, a distinct subset of cartilage defects were on the anterosuperior aspect of the glenoid (Figure 2). It has been reported that the superior band of the inferior glenohumeral ligaments attach at the 2- to 3-o’clock positions. 14 Therefore, it is possible that this cartilage defect occurred via an avulsion of the superior band of the inferior glenohumeral ligament pulling the junction between the labrum and the cartilage. Thus, bone and cartilage defects may occur both via humeral head impaction and inferior glenohumeral ligament avulsion.
In our study, patients with a cartilage defect were more likely to be male and to have undergone a concomitant posterior labral repair. Duchman et al 7 conducted a retrospective review of bone and cartilage lesions encountered in the MOON and found bone and cartilage lesions to be associated with male sex, increased BMI, and increased patient age. The Duchman study agreed with our own results, while the MOON study did not. Krych et al 17 conducted a retrospective review of articular cartilage injuries and found that glenoid cartilage injuries were associated with the number of dislocations and the need for a closed reduction. Further studies are necessary to understand the relationship between recurrence and cartilage defects. In addition, further studies are necessary to understand whether cartilage defects contribute to future bone loss.
Limitations
There were several important limitations to the current study. First, this was a retrospective study using available data in our medical record. Second, this retrospective study used existing MRI scans. It is possible that more subtle or smaller cartilage defects may not be evaluated fully using these scan types, as we did not use a cartilage-specific sequence as this was a retrospective study. Future prospective studies using cartilage-specific sequences are planned, but, given the sample size necessary to establish prevalence, this will take years. Because these sequences have not been standard of care in the past, it may take several years for sufficient data to be available to conduct a study using these sequences. To mitigate this limitation, we have used MRI scans, MR arthroscopic images, and the surgeon’s description of the cartilage surfaces to determine cartilage defect presence and thus to increase sensitivity, although this does not mitigate defect measurements. Third, we could not include the erosion-type bone loss as a type of cartilage loss as mentioned in the Discussion section. CT images of the affected shoulder, ideally with the contralateral shoulder for comparison, would be necessary to accurately measure erosion-type bone loss. Fourth, this was a single institution study, and thus our findings may not be generalizable to other centers. Fifth, as this was a retrospective study, there were varying indications for concomitant posterior labral repair, limiting our ability to extrapolate that risk factor. Sixth, no follow-up data on recurrence or analysis of bipolar bone loss were included.
Conclusion
Study findings indicated that cartilage defects are common in patients with operatively treated anterior glenohumeral instability, occurring in 38% of patients. These defects were located directly anteriorly at the 3:07 clockface position, similar to osseous glenoid defects in this study. Future prospective studies with cartilage-specific MRI scan sequences should be conducted.
Footnotes
Final revision submitted December 7, 2021; accepted January 10, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.T. has received consulting fees from Cayenne Medical, Mitek, Stryker, and Wright Medical; speaking fees from Wright Medical; and royalties from Shoulder Innovations, Wright Medical, and Zimmer and has stock/stock options in Conextions, IntraFuse, and KATOR. P.N.C. has received research support from Major League Baseball, education payments from Active Medical, consulting fees from DJO and DePuy, nonconsulting fees from Arthrex and DePuy, and royalties from DePuy. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Utah (ref No. 00046622).
