Abstract
Background:
Good-to-excellent midterm results after high tibial osteotomy (HTO) to treat medial compartment cartilage defects or osteoarthritis (OA) have been published, but little is known about long-term survival rates in terms of conversion to total knee arthroplasty (TKA) using angular stable internal plate fixation.
Purpose:
To determine TKA-free survival rates and functional and radiological outcomes at 10 years after HTO. A subgroup analysis of patients who underwent combined HTO and autologous cartilage implantation (ACI) was also performed.
Study Design:
Case series; Level of evidence, 4.
Methods:
Included were 125 patients with a mean follow-up of 9.90 ± 2.25 years; 90 patients underwent HTO for medial OA, and 35 patients underwent ACI and HTO for medial focal cartilage defects. Functional outcome measures included visual analog scale (VAS) for pain, Lysholm, International Knee Documentation Committee (IKDC), and Knee injury and Osteoarthritis Outcome Score (KOOS) subscales and KOOS4 (average of 4 KOOS subscales: Pain, Symptoms, Sport, and Quality of Life). Radiological outcomes included lateral distal femoral angle, medial proximal tibial angle, and joint line convergence angle.
Results:
Overall, 16 patients required conversion to TKA at a mean 86.75 ± 25.73 months (10-year survival rate, 87.2%). Only 2 patients in the HTO+ACI subgroup required a conversion to TKA (10-year survival rate, 94.3%). The complication rate for all patients was 8.8%. In both the HTO and HTO+ACI subgroups, VAS pain levels decreased and Lysholm scores increased significantly from pre- to postoperatively (P < .001). A higher preoperative Tegner score led to a significantly lower risk for conversion to TKA (P = .001), and a preoperative body mass index of ≥35 was associated with a significantly higher risk (P = .019), as was female sex (P = .046). Radiological parameters remained within physiological ranges. The postoperative joint line conversion angle did correlate with postoperative functional outcome but not with TKA conversion.
Conclusion:
Long-term results of HTO for medial compartment OA or cartilage defects with underlying varus deformity were good to excellent. In particular, patients who underwent HTO+ACI presented excellent long-term survival rates. HTO, therefore, delays or prevents TKA implantation, especially in young, active patients with medial compartment damage.
Cartilage defects of the medial compartment are influenced primarily by alignment deformities; this lowers the quality of life of patients in a manner similar to those with severe osteoarthritis (OA). 13 While total knee arthroplasty (TKA) delivers good-to-excellent functional outcomes in the older patients, patients aged <55 years have a 25% to 30% lifetime risk of revision and a high percentage of dissatisfaction. 1,42 As varus- or valgus-alignment pressure peaks are highest at the edge of these cartilage defects, the dimensions of the defect increase over time. Mina et al 21 proved the unloading efficiency of medial open-wedge high tibial osteotomy (HTO) for such cartilage defects. In comparison with lateral closed-wedge osteotomy of the proximal tibia, historically associated with high complication rates, HTO has proven to be a safe and efficient surgical therapy with good midterm results. 2,39,41
There is little evidence on long-term results after HTO using internal plate fixation in TKA-free survival terms. Nowadays, HTO is recommended for patients evaluated with metaphyseal varus deformity, a tibial bone varus angle >5°, and requires meticulous planning of the correction aim according to the specific indication. 6,15,27
Functional midterm results have proven the beneficial effect of optimized preoperative planning and a standardized surgical technique. 2 However, long-term results after HTO using angular stable internal plate fixators are still rare in the literature, and comparable results after HTO with autologous cartilage implantation (ACI) are missing altogether. 14,15,29 In an earlier study, we reported midterm results after open-wedge HTO. 2 The purpose of the present study was to add long-term results (up to 10 years) in a larger cohort, including analyses of postoperative radiological parameters. The aim was to identify risk factors for conversion to TKA in the later course as well as risk factors for lower functional outcome. Secondarily, a subgroup analyses of patients undergoing HTO and ACI with a mean follow-up of 10 years was performed. The working group’s hypothesis was relatively high TKA-free survival rates after a 10-year follow-up as well as still satisfactory functional results. A larger postoperative joint line convergence angle was expected to lead to lower survival and functional results.
Methods
The study was designed as a prospective case series, and the study protocol received ethics committee approval. Data of patients who underwent open-wedge HTO were reviewed if performed for 2 different indications: (1) progression of symptomatic OA of the medial compartment without a concomitant cartilage-repair procedure and (2) focal cartilage defects (HTO in combination with ACI). All patients were treated via an HTO using an angular stable internal plate fixator (Tomofix Synthes) between January 1, 2004, and December 31, 2013, in a clinic for orthopaedic and trauma surgery.
Patients were not eligible for the study if there was significantly restricted flexion or if they had inflammatory arthropathy, extensive loss (more than two-thirds of its surface) or absence of the lateral meniscus, high-grade ligamentous instabilities, or severe general OA including the lateral and patellofemoral compartment. Patients were asked before surgery about their pain in the lateral compartment. If they had pain, they did not qualify for medial open-wedge osteotomy. 8 Smoking was not considered a contraindication except in case of severe nicotine abuse (>20 pack-years). In terms of age, patients up to 70 years of age at the time of surgery were included. Flexion/extension range of motion had to be at least 120° to 0°. Extension deficits were integrated in preoperative planning.
All patients matching the study criteria were included in this study and were divided into 2 subgroups according to the indication for surgery: HTO for medial OA and HTO+ACI for medial focal cartilage defects. Detailed patient characteristics of both subgroups at the time of follow-up in comparison with the initial population of interest are listed in Table 1.
Patient Characteristics of the Initial and Final Samples of the HTO and HTO+ACI Subgroups a

a Data are reported as mean ± SD unless otherwise indicated. There were no statistically significant differences between the initial and final samples in either subgroup, minimizing sample bias. ACI, autologous cartilage implantation; BMI, body mass index; HTO, high tibial osteotomy; JLCA, joint line convergence angle; mLDFA, mechanical lateral distal femoral angle; mMPTA, mechanical medial proximal tibial angle; preop, preoperatively; VAS, visual analog scale for pain.
Inclusion and Exclusion Criteria
Patients with asymptomatic focal cartilage lesions of the lateral or patellofemoral compartment were included in the study if they fulfilled the criteria for enrollment. Patients were excluded if they had postoperative over- or undercorrection of the leg axis (defined as a correction exceeding the 65% intersection or undercutting of the 50% intersection point on the tibial plateau, if the medial border of the tibial plateau represents the 0% intersection point and the lateral border represents the 100% intersection point of the weightbearing line at 6 weeks postoperatively), had incomplete or unavailable postoperative imaging, or did not give their informed consent or were not available for follow-up evaluation in previous studies 13,14 (Figure 1).

Patient flowchart. Data of patients were enrolled if HTO was performed between 2004 and 2013 matching the criteria for inclusion and exclusion. During the follow-up process, patients were lost to follow-up. Revision surgery was necessary in 11 out of 125 patients (revision rate 8.8%). OA, osteoarthritis; HTO, high tibial osteotomy.
Preoperative Management and Surgical Technique
The necessary preoperative diagnostics have been described in prior studies. 2,14,15 Limb alignment was assessed via the Paley technique, 30 including measuring the lateral distal femoral angle (LDFA), medial proximal tibial angle (MPTA), and joint line convergence angle (JLCA) on full-leg radiographs.
Patients received general anesthesia, intravenous antibiotics, and standard thromboembolic prophylaxis. Before their HTO, patients underwent 1-stage arthroscopy to assess the medial, lateral, and patellofemoral compartments and to identify patients with severe degenerative anomalies. Cartilage defects were classified according to the Outerbridge classification. Routine arthroscopy was performed, and ACI was indicated according to the German Society of Orthopaedic Surgery and Traumatology's recommendation. 24 Chondrocytes were harvested using a standardized cartilage biopsy tool (Storz) from the intercondylar notch. 25 ACI was performed as described elsewhere after cell expansion (Cartigro; Stryker). 26 A total of 1 to 2 billion chondrocytes per square centimeter of cartilage defect were applied.
HTO was performed according to the technique recommended by a knee expert group, as previously described. 2,9 The extent of correction planned preoperatively was intraoperatively secured via a navigation system (Orthopilot; Aesculap Co; Software: Orthopilot software for HTO). All osteotomies were performed in a biplanar manner and were stabilized using the Tomofix system (Tomofix; Synthes); correction was aimed at a mild valgus alignment. 15
Postoperative Rehabilitation Protocol
Postoperatively, mobilization started on day 1, and continuous passive motion lasting 4 to 6 hours daily was recommended for the first 6 weeks. Limited weightbearing was allowed 3 weeks postoperatively and extended to 6 weeks in case of ACI. Knee-flexion limitations depended on the individual cartilage defect. Patients who underwent HTO exclusively were not limited in their range of motion at any time. After full weightbearing was achieved, full-leg radiographs were taken to analyze the postoperative weightbearing axis. Digital analyses of pre- and postoperative full-leg radiographs were done before statistical analyses (mediCAD; Hectec).
Clinical Outcomes and Survival
Patient data were collected preoperatively as well as at 1 and 5 years postoperatively, and final patient interviews took place between February and July 2019 after they had reconfirmed, via written consent, participation in the study. Follow-up was defined as the time period from the day of surgery until the day of interview. The need for total knee replacement was defined as a failure and analyzed via Kaplan-Meier curve. Consequently, event-free survival was defined as the primary outcome parameter. Complications were classified as any major or minor complication leading to revision surgery. Major complications included popliteal aneurysm, large overcorrection resulting in immediate revision, delayed union, and deep tissue infections, whereas delayed wound healing was defined as a minor complication. In addition, discomfort due to the implant was recorded.
Secondary outcome parameters included the comparison and acquisition of functional outcome scores as well as radiological parameters. Functional outcome was evaluated by applying the standardized Lysholm score (pre- and postoperatively), International Knee Documentation Committee score (IKDC), Tegner score (pre- and postoperatively), Knee injury and Osteoarthritis Outcome Score (KOOS), and KOOS4 (average score of 4 KOOS subscales: Pain, Symptoms, Sport, and Quality of Life). Pre- and postoperative pain levels were evaluated via a visual analog scale (VAS). 4,34,43 In addition, patients were asked the dichotomous question “Would you undergo HTO surgery again if you had the choice?”
Radiological Assessments
Postoperatively, the extent of valgus correction, the LDFA, MPTA, and JLCA, was compared with preoperative values. Preoperative varus deformity and postoperative valgus correction were defined as medial or lateral deviation from the mechanical weightbearing axis (hip-to-ankle line through the center of the knee).
Statistical Analysis
SPSS for Windows (Version 27; SPSS) was used for statistical analysis of the data ascertained in this study. Quantitative variables at baseline were expressed as mean ± SD. Categorical variables were compared using the chi-square test. Continuous variables were compared using the Student t test; ordinal variables, using the Mann-Whitney U test.
Fixed-effects logistic regression was used to measure associations between the dependent variable of conversion to TKA (yes/no) and the following independent variables: patient characteristics (age, body mass index [BMI] at the time of surgery, sex, duration of symptoms, prior surgery), Outerbridge classification and defect size of medial compartment cartilage lesions in the HTO+ACI group, Kellgren-Lawrence score in the HTO group, and preoperative functional scores (VAS, Lysholm score, Tegner score). Independent variables were chosen based on the assumed effect according to current scientific knowledge, expert opinion, and data availability.
Before conducting the logistic regression analysis, we excluded statistical outliers, as well as multicollinearity via analysis of the variance inflation factor, condition index, and bivariate correlation.
Pearson and Spearman correlations were used to measure associations between 2 variables. Survival rates were shown as a Kaplan-Meier curve and compared using log-rank test. Accordingly, P < .05 was considered statistically significant.
Results
Of the total of 173 patients, 125 were available at the time of data collection (follow-up rate, 72.3%; mean follow-up, 118.84 ± 27.09 months; range, 73-171 months). Mean defect size was 3.89 ± 2.63 cm2 with a mean underlying varus deformity measuring 5.93° ± 2.37°. HTO resulted in a mild overcorrection of 2.64° ± 1.71° of valgus. Detailed patient characteristics, as well as pre- and postoperative radiographic values, are listed in Table 2, and age distributions in both subgroups are displayed in Figure 2. The subgroups differed significantly in terms of age (P < .001) and BMI (P = .015) at the time of surgery and in the extent of preoperative varus deformity (P = .007).
Patient Characteristics in Both Subgroups a
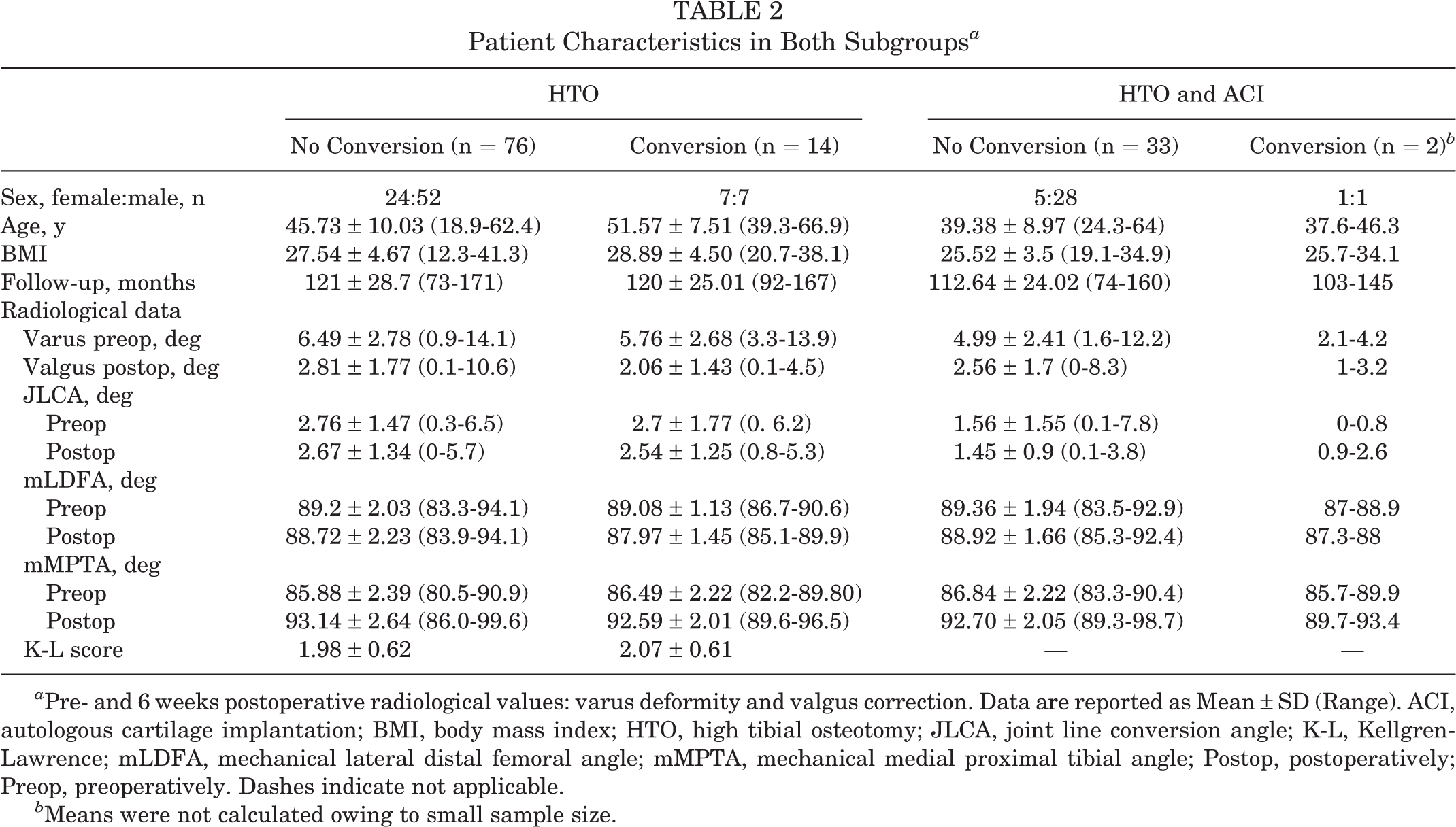
a Pre- and 6 weeks postoperative radiological values: varus deformity and valgus correction. Data are reported as Mean ± SD (Range). ACI, autologous cartilage implantation; BMI, body mass index; HTO, high tibial osteotomy; JLCA, joint line conversion angle; K-L, Kellgren-Lawrence; mLDFA, mechanical lateral distal femoral angle; mMPTA, mechanical medial proximal tibial angle; Postop, postoperatively; Preop, preoperatively. Dashes indicate not applicable.
b Means were not calculated owing to small sample size.

Age distribution and age peaks at the time of surgery in the HTO and HTO+ACI subgroups. The patients with HTO+ACI were significantly younger. ACI, autologous cartilage implantation; HTO, high tibial osteotomy.
JLCA and LDFA did not change significantly, while the MPTA did change significantly. Concerning the influence of JLCA on postoperative functional outcome, JLCA did not correlate significantly with the TKA conversion rate at the 10-year follow-up. Furthermore, postoperative JLCA correlated with several functional scores at the 10-year follow-up (Lysholm: r = −0.182, P = .044; KOOS-Symptoms: r = −0.190, P = .034; KOOS-Activities of Daily Living: r = −0.230, P = .010; KOOS-Sport: r = −0.250, P = .005; and KOOS4: r = −0.200, P = .026).
Complications
Revision surgery (Figure 1) was performed in 11 patients in this cohort (revision rate, 8.8%). Major complications were observed in 5.6% of the patients (nonunion, popliteal aneurysm, and large overcorrection resulting in immediate revision and infection). Minor complications in terms of superficial wound infections occurred in 3.2% of patients. Discomfort due to the implant was described by 48.8% of all patients.
Functional Outcome, Sport and Activity Level
Patients not requiring a TKA in their further follow-up course had a significantly higher preoperative Tegner score than did the conversion-to-TKA group (P = .001); they did not differ in terms of preoperative Lysholm and VAS scores. Both cohorts (conversion to TKA vs no conversion to TKA) experienced a significant increase in their Lysholm scores 10 years after HTO in comparison with preoperative measures; those patients who did not need a TKA even earned higher functional outcome scores in comparison with their 5-year results (Figure 3), except for the KOOS-Sport score, which remained on a constant level. In both subgroups (HTO, HTO+ACI), patients profited significantly from surgery when pre- and postoperative VAS and Lysholm scores were compared (P < .001) (Figure 3).

Lysholm, IKDC, and KOOS4 scores 5 and 10 years after HTO showed significant increase over the long term and significantly higher results in the group without TKA conversion (P < .001). Only 2 of the 16 patients had undergone conversion to TKA before the 5-year follow-up. High postoperative functional scores were seen in the HTO+ACI subgroup. The center bar represents the median, the shaded area represents the interquartile range, and the whiskers show minimum and maximum values except for outliers (circles) and extremes (asterisks). ACI, autologous cartilage implantation; HTO, high tibial osteotomy; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; KOOS4, average of 4 KOOS subscales (Pain, Symptoms, Sport, and Quality of Life); Postop, postoperatively; Preop, preoperatively; TKA, total knee arthroplasty.
In both subgroups, KOOS-Sport and Tegner score levels indicating postoperative levels of physical activity varied broadly among patients, from highly active patients to patients with a low level of activity (Figure 4). KOOS-Sport scores remained at a constant level from 5 to 10 years postoperatively. If no conversion was necessary, the Tegner score decreased to a significantly lower level during the 10-year follow-up but still remained at the level 4 recreational sports level (P = .001).

Sport and physical activity levels in patients in both subgroups without conversion to TKA. KOOS-Sport levels remained on a constant level, with a wide range at the 10-year follow-up. Tegner scores decreased significantly but remained overall at a recreational sports level (eg, recreational sports-cycling, cross-country skiing, jogging on even ground at least twice weekly). The center bar represents the median, the shaded area represents the interquartile range, and the whiskers show minimum and maximum values except for outliers (circles). ACI, autologous cartilage implantation; HTO, high tibial osteotomy; KOOS, Knee injury and Osteoarthritis Outcome Score; PostOP, postoperatively; PreOP, preoperatively; TKA, total knee arthroplasty.
In the HTO subgroup, a higher BMI at the time of surgery was associated with a significantly higher preoperative pain level (VAS: P = .03, R = .228) and a lower preoperative Lysholm score (P = .040, r = −0.217), as well as lower KOOS subscores (KOOS4: P = .019, r = −0.248; KOOS-Pain: P = .022, r = −0.242; KOOS-Activities of Daily Living: P = .001, r = −0.343; KOOS-Sport: P = .004, r = −0.303), IKDC score (P = .026, r = −0.235), and Tegner score (P < .001, r = −0.383) at the 10-year follow-up, whereas there was no correlation between a higher BMI and a lower preoperative Tegner score. There was no significant change in preoperative to 10-year follow-up BMI.
In the HTO+ACI subgroup, age at the time of surgery correlated only with preoperative Lysholm scores (P = .048, r = −0.337), and BMI at the time of surgery correlated only with preoperative VAS scores (P = .031, r = 0.365). Age at the time of surgery correlated significantly with a lower preoperative Lysholm score (P = .048, r = −0.337), and BMI at the time of surgery correlated with higher preoperative VAS scores (P = .031, r = 0.365) (correlations we could no longer establish at the 10-year follow-up). Patients undergoing combined HTO and ACI surgery revealed high functional results (Figure 3).
The question “Would you undergo HTO surgery again if you had the choice?” was answered “no” by 13 out of 125 patients (10.4%) overall: 9 out of 109 of patients who did not undergo conversion to TKA (8.3%) and 4 out of 16 (25%) in the conversion-to-TKA group. In the subgroup of patients who underwent HTO+ACI, only 3 out of 35 patients (8.6%) would not repeat HTO if asked again.
Survival
At the time of data collection, in 16 patients (12.8%), HTO had been converted to TKA. Hence, the 10-year TKA-free survival of HTO was 87.2%. Conversion to TKA was done at a mean 86.75 ± 25.73 months after HTO (range, 45-122 months) (Figure 5); only 2 of the patients underwent conversion to TKA during the first 5 years after HTO.

Survival rates after HTO ± ACI showing good long-term survival rates after isolated HTO and even excellent results for combined HTO and ACI. ACI, autologous cartilage implantation; HTO, high tibial osteotomy; TKA, total knee arthroplasty.
In the HTO+ACI subgroup, only 2 patients had undergone conversion to TKA (5.7%, after 98 and 122 months) resulting in a 10-year TKA-free survival rate of 94.3%. In the HTO subgroup, we documented an 84.4% survival rate after 10 years.
Risk Factors for HTO Failure
After evaluating all the parameters obtained, we identified age at the time of surgery as a significant risk factor for HTO failure in terms of TKA conversion (P = .018; Exp(B) = 1.081) (Figure 6). Furthermore, a higher preoperative Tegner score led to a significantly lower risk for conversion to TKA (P = .001). A BMI of ≥35 was associated with significantly lower survival (P = .019), as was female sex (P = .046) (Figure 7). In the subgroup of HTO for medial OA, the duration of symptoms before surgery (in months) led to a significantly higher risk for conversion to TKA (P = .04; Exp(B) = 1.025). Diverging survival function was observed from age 55 years despite not reaching statistical significance.

Odds ratios of patient characteristics and preoperative functional scores with 95% CIs of the logistic regression for the binary outcome conversion to TKA. The vertical line at 1 represents the neutral effect. BMI, body mass index; Preop, preoperatively; TKA, total knee arthroplasty; VAS, visual analog scale.

Survival rates after HTO ± ACI showing a significantly lower cumulative survival of female patients (P = .046) and of patients with obesity grade 2 to 3 (P = .018).
Discussion
The most important finding of the present study was a long TKA-free survival even after 10 years and very low rates of TKA conversion in case of HTO+ACI. Five-year results of a smaller cohort have already been published. 2 Compared with those, 10-year survival rates decreased, but they remained within probable ranges reported in similar studies. 5,37 The following independent risk factors were identified for conversion to TKA: BMI ≥35, female sex, a low preoperative Tegner score, and age. In HTO for progressing medial OA, we found that the duration of symptoms before surgery was another risk factor. Functional outcome and patient satisfaction remained remarkably high. These findings are in accordance with present studies and disprove earlier descriptions of higher complication rates for TKA after HTO. 36 Patients remained physically active on a recreational level. In HTO for progressing medial OA, a higher BMI at the time of surgery resulted in a lower functional outcome.
Patients in the combined HTO and ACI subgroup presented good-to-excellent functional outcome in line with their very high TKA-free survival rates even after 10 years. Compared with preoperative values, radiological parameters remained within physiological ranges postoperatively as well. JLCA correlated with functional outcome but not with TKA conversion.
Remaining questions regarding the target of correction, changes in patella height, and influence of a lateral compartment cartilage defect were recently answered. 14,17 Several studies have proved the importance of mild correction depending on the initial indication, in contrast to Fujisawa et al’s 10 earlier findings. 6,15 The influence of postoperatively altered radiological parameters remains controversial. 18,38 Feucht et al 7 recently pointed out that less than one-third of patients (28%) with mechanical varus ≥3° have a tibial deformity. If slight overcorrection is accepted (medial MPTA, ≤95°), 57% of patients can undergo correction via isolated HTO, whereas 33% of patients would still require a double-level osteotomy. While these findings result from detailed analyses of long/leg weightbearing radiographs without clinical data, recent clinical studies have tried to determine the influence of the postoperative oblique joint line on postoperative functional outcome. While a postoperative oblique joint line ≥4° seems to correlate with worse functional outcome, its influence on survival rates has not been examined so far. 38
Since the development of the open-wedge HTO technique, there have been few investigations reporting long-term results. While initial reports described survival rates after closed-wedge technique ranging from 51% to 98%, recent studies have consistently reported high survival rates after HTO. 5,8,11,16,23,37 Because of the inhomogeneous use of implants, bone grafts, intraoperative navigation, etc, findings are difficult to compare. However, the present study is the first of its kind to describe long-term survival rates after combined HTO using angular stable internal plate fixators without bone graft, focusing especially on survival rates after HTO and ACI.
In a recent study analyzing HTO survival rates of 1576 patients from the Californian Office of Statewide Health Planning and Development, Pannell et al 31 reported 5-year survival rates of 80% and 10-year rates of only 56%. The relative risk of TKA implantation rose by 8% with each year of increasing age, and they identified female sex and severe OA as risk factors for HTO failure. While their failure rate appears remarkably high compared with that of other studies, their subgroup analysis of patients without severe OA revealed survival rates of 85% after 5 years and 67% after 10 years.
Patient age is often identified as a risk factor. Flecher et al 8 and Hui et al 16 reported hazard ratios of 2 and 3.7, respectively, for patients aged >50 years. A 1% increase in conversion rates per year of age was observed as well. 11 Age at the time of surgery was also registered as a risk factor for TKA conversion in this study, with an 8.1% increase in relative chance for conversion to TKA per year (odds ratio, 1.081). In the subgroup of HTO for progressing medial OA, diverging curves of survival function from age 55 years were noted. The duration of symptoms before surgery was associated with a higher chance of conversion to TKA in the postoperative course. Age limits in the HTO+ACI subgroup could not be established probably because of the rather young age of those patients at the time of surgery.
After age, being female was an additional significant risk factor: the likelihood of TKA conversion rose by 38%; an increased hazard ratio of 1.26 has also been reported. 28,33 In this series, female sex turned out to be a risk factor for later conversion to TKA. BMI of ≥35 (obesity grade 2) was also identified as a risk factor for lower cumulative survival.
Lau et al 19 published 10-year results after conversion to TKA after open-wedge osteotomy, amounting to 87.1% in their 31 patients, whose characteristics resemble those of the present study; however, they employed 3 different implants for osteotomy fixation. Thus, to the best of our knowledge, this is the largest cohort study presenting the results of HTO using internal plate fixators without bone grafts after a 10-year period.
Postoperative radiological parameters all remained within common ranges, even though MPTA changed significantly. While TKA conversion did not correlate with any radiological parameter collected, postoperative JLCA seems to be an especially important risk factor for a poorer postoperative functional outcome.
These findings concur with those of Song et al 38 and Kubota et al, 18 underlining the importance of meticulous preoperative patient selection and of analyzing the preoperative long leg weightbearing axis in order to correct each deformity at its origin, as proposed by Feucht et al. 7
With regard to functional outcome, comparison with earlier studies is again limited because of the inhomogeneous outcome scores used to describe patient outcomes.
Patients in the present study experienced a significant rise in functional scores 10 years after HTO compared with both their pre- and postoperative outcome measures and their 5-year follow-up results. In the subgroup of HTO for progressing medial OA, a high BMI at the time of surgery was associated preoperatively with higher pain levels and lower Lysholm scores but not with a lower Tegner score. Postoperatively, higher BMI levels led to lower functional outcome scores during daily and sports activities. Older age at the time of surgery did not correlate with a lower postoperative functional outcome in either subgroup. In contrast to the Lysholm, KOOS, and IKDC scores, the Tegner score in patients in both subgroups without conversion to TKA failed to rise between the pre- and postoperative or 5-year follow-up and 10-year follow-up levels; instead, they decreased significantly. Nevertheless, it remained on an adequate level for recreational sports, considering the increasing ages of patients over the 10-year follow-up interval. Mean KOOS-Sport scores remained constant on an average level but with a wide range.
In recent studies, functional outcomes after HTO ranged from results resembling preoperative function to a significant improvement in IKDC and KOOS scores. Compared with the postoperative IKDC and KOOS scores listed previously, 12,35 patients in the present study achieved similar results: a low preoperative Tegner score increased the risk of conversion to TKA.
While the feasibility of combined ACI and HTO has been reported earlier, published evidence concerning survival rates is rare. 3 The patient characteristics of this subgroup differ significantly from those of patients who undergo HTO for medial OA. In the present cohort, patients in the HTO+ACI subgroup were significantly younger and had a lower BMI at the time of surgery; moreover, their extent of varus deformity was significantly less.
Schuster et al 37 reported survival rates of 81.7% at a mean follow-up of 12 years for 79 knees treated with combined HTO and arthroscopic abrasion of severe cartilage defects in the medial condyle. While their survival rates appeared rather low, patients’ age and severe degree of anomalies need to be taken into consideration. Nonetheless, it is hard to compare their results with the present ones, as those patients underwent no regenerative cartilage therapy. Pascale et al 32 observed no significant differences in their randomized control trial comparing patients undergoing HTO and microfracturing to those undergoing HTO only. Their results were in line with those of Lee et al, 20 who questioned the effect of adjunctive therapies while performing HTO. Anyway, the different patients’ characteristics have to be considered, as microfracturing is often applied for smaller cartilage defect diameters or as a salvage procedure for degenerative anomalies. ACI, on the other hand, is the standard for cartilage defects measuring 3 cm2 to 4 cm2 or even more. 24 Furthermore, the target group of patients for ACI treatment is younger and used to engaging in higher activity grades than are older patients with degenerative anomalies. 4 Sterett et al 40 observed 5-year survival rates of 97% for HTO with microfracturing and 91% at 7 years. Comparable survival rates were published by Minzlaff et al, 22 with 90.1% after 8.5 years for patients treated with combined HTO and osteochondral autograft transplantation.
A limitation of the present study is the lack of a control group as well as not all outcome measures being collected preoperatively. Radiographic assessment for alignment was performed only 6 weeks postoperatively. Furthermore, the present cohort is comparatively young. That factor might have influenced the indication for TKA revision on the one hand. On the other hand, age seems to be a serious risk factor for early conversion, and younger patients do not tend to have severe degenerative anomalies (a known risk factor for early conversion to TKA). This point is supported by the high rate of concomitant regenerative cartilage procedures in this series. Another limitation is the follow-up rate of 72.3% during the long-term follow-up. Nonetheless, the probability of a sampling bias was minimized by comparing preoperative records of the initial to the follow-up sample without significant differences.
Conclusion
Open-wedge HTO for patients with medial OA or focal cartilage defects in the medial compartment due to underlying varus deformities yielded satisfactory long-term survival rates, satisfactory functional outcome scores, and even excellent results in combination with ACI for medial compartment cartilage defects.
Footnotes
Final revision submitted October 6, 2021; accepted November 8, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: the article processing charge was funded by the German Research Foundation (DFG) and the University of Freiburg in the funding program for open access publishing. H.S. has received speaking fees from Arthrex. G.B. has received speaking fees from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Freiburg University (ref No. 290/18).
