Abstract
Background:
Intra-articular infiltration of platelet-rich plasma (PRP) is an alternative therapeutic option to classic hyaluronic acid for the treatment of symptomatic knee osteoarthritis (KOA). However, variation in preparation methods and quality assessment of PRP makes the study of its real clinical efficacy difficult.
Purpose:
To (1) evaluate the clinical efficacy of a characterized PRP product prepared in a standardized manner and in a closed-system for the treatment of KOA and to (2) evaluate the association of the clinical response to PRP-related variables.
Study Design:
Case series; Level of evidence, 4.
Methods:
We recruited 130 patients with nonoperative KOA and evaluated them for 1 year. PRP was prepared from a donation of autologous blood, obtaining 3 aliquots of approximately 10mL of product, which were frozen, allowing platelet disruption, platelet factor release, and long-term storage, until administration. Patients were treated 3 consecutive times every 4 weeks with an intra-articular PRP knee injection under sterile conditions. Complete blood count was performed on the whole-blood sample and the processed PRP before freezing it, for product quality assessment. Patients were assessed using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) and basic satisfaction scale at 3 months, 6 months, and 1 year after intervention.
Results:
Quality assessment confirmed a leukocyte-poor PRP product (white blood cell count, 0.09 ± 0.09 × 109/L) with a high platelet purity (platelet count, 630.86 ± 191.75 × 109/L). WOMAC scores improved, and basic satisfaction was achieved in 70% of patients. No adverse events were reported. No correlations were observed between PRP quality parameters and clinical results. PRP complete treatment production costs were €108/US$125 (€36/US$41.6 per injection).
Conclusion:
This standardized PRP production method resulted in improved WOMAC scores at 1 year postoperatively in 70% of patients with KOA. This technique was safe and affordable and ensured consecutive infiltrations with the same product to each patient.
The lifetime risk of developing knee osteoarthritis (KOA) is estimated to be around 14% in the United States, with prevalence of at least some degree of KOA in almost 40% of patients older than 45 years. 32 In Europe, the KOA prevalence in the general population ranges depending on the study, 11 and in Spain it is around 14% (range of 95% CI, 12.66%-15.11%). 5 Platelet-rich plasma (PRP) injection is a well-known nonsurgical therapeutic option indicated for the management of symptomatic KOA. 1,22 PRP contains a cocktail of platelet-derived growth factors, which are crucial in tissue regeneration. 30 They contribute to the regulation of cartilage anabolism and articular homeostasis, including menisci and synovia. 19 Furthermore, according to in vitro studies, PRP appears to stimulate endogenous hyaluronic acid (HA) production, 54 inducing chondrocyte and mesenchymal stem cell proliferation, and proteoglycan and type 2 collagen deposition. 52
There is an important effort worldwide to establish therapeutic strategies to modify the natural history of KOA, increasing the interest in orthobiologic products. PRP has demonstrated its supremacy in comparison with placebo and tends to be considered superior to HA in several clinical trials, 4,21,30 being considered by Dhillon et al 13 as “the best option available that could modify the disease process in early KOA.”
However, most studies addressing the efficacy of PRP in orthobiologic medicine highlight the same conclusion: evidence is limited by the substantial heterogeneity among PRP preparation methods, processing, administration form, storage, or reporting of quality parameters. 8 In fact, a 2020 evidence-based-guideline published by the American College of Rheumatology and the Arthritis Foundation 29 strongly discourages PRP treatment in patients with KOA, based on “the heterogeneity and lack of standardization in available preparations of PRP, as well as techniques used, making it difficult to identify exactly what is being injected.” It is calculated that only 5% to 6% of reports specify the type and cellular composition of the PRP used. 6,17 In addition, the high fee burden to patients is recognized, as there is no consensus or regulation on production costs, and many private clinics use expensive kits for its production. 10
There have been several attempts to categorize PRP according to various variables (the preparation method; fibrin content; the presence of white blood cells [WBCs] or red blood cells [RBCs]; final volume; platelet concentration or enrichment; activation method, including freezing; and delivery method), with more or less complexity. 12,14,35,36 Gradually, this field is improving thanks to the crosstalk of various disciplines (traumatologists and hematologists, among others), and the need for standardization has been recognized by the scientific community. 24 We have recently reviewed this issue and have proposed a tentative nomenclature system to make possible comparison among groups. 1 In addition, an expert consensus with specifications for the Minimum Information for Studies Evaluating Biologics in Orthopedics (MIBO) for PRP has been published that should be reported by clinical studies involved in this field. 42
In Spain, PRP has been considered “medicine for human use” by The Spanish Agency of Medicines and Medical Devices since 2013 (V1/23052013). 3 Since 2018, our certified local blood bank has implemented a closed-system PRP standardized preparation method, which allows the production of autologous PRP at low cost (€36/US$41.6 per injection). 43 This product ensures safety (closed system and leuko reduced) and an optimal platelet enrichment (approximately 2- to 3-fold). Furthermore, it is stored frozen in aliquots, which allows the release of platelet factors in the plasma fraction (through freezing-induced platelet lysis) and the autologous use of the same product in consecutive infiltrations per patient (reproducibility). In the present study, we evaluated the clinical efficacy of a characterized PRP product prepared in a standardized manner and in a closed system as KOA treatment, and the association of the clinical response to PRP-related variables in 130 patients, during a 1-year follow-up period.
Methods
Patients
Patients with symptomatic KOA without surgery indication were recruited during their orthopaedic clinic visit at our hospital from August 2018 to January 2020. The study was approved by our local hospital ethical committee, and all patients participated upon informed consent. The protocol is registered in a specific website to assess (https://www.precis-2.org), with its tool, the real grade of pragmatism of this study (Figure 1). 33

Pragmatic Explanatory Continuum Indicator Summary (PRECIS-2) wheel showing the grade of pragmatism for this study design.
Study inclusion criteria were symptomatic KOA (femorotibial and/or femoropatellar), regardless of the Kellgren-Lawrence (KL) radiological grade, without response to conservative management (nonsteroidal anti-inflammatory drugs [NSAIDs], physical rehabilitation, weight loss, or other infiltrations such as corticoids or HA) during at least 6 months and with no indication of knee surgery (based on medical criteria or patient rejection), and signed informed consent. Exclusion criteria were corticosteroid infiltrations 6 weeks before or HA less than 1 year before PRP infiltration, loss of follow-up during the study, severe effusion of the knee, knee surgery candidates, serious heart disease, hemostasis disorders, severe autoimmune disease, history of neoplasia at the site of PRP application, hematological alteration in recent blood analysis, positivity to serological tests, or genomic detection of hepatitis B and C viruses, human immunodeficiency virus, or syphilis.
Study Design and Intervention
This was a single-center and prospective interventional case series with pretest-posttest design, conducted in accordance with the CONSORT (Consolidated Standards of Reporting Trials) guidelines (extension for pragmatic trials). It was performed at our hospital’s orthopaedics department outpatient clinic, and all patients were recruited during the daily clinical practice of 2 orthopaedic consultants (J.F.F., M.A.R.) , after clinical and radiological evaluation. Full weightbearing knee radiographs were obtained the day of recruitment (around 3 months before the first injection). Patients were considered for the study after assessment at the hematology department outpatient clinic, where the autologous blood harvesting (150mL) took place.
Blood was processed at the local blood bank to obtain the 3 PRP aliquots of around 10mL for autologous use as described previously. 43 In brief, donated blood underwent 2 rounds of differential centrifugation in order to separate the PRP fraction, using the infrastructure of a certified blood bank, which allows the processing to be completed within a closed system. Aliquots were stored frozen at −40°C until use. Complete blood counts (CBCs) were performed on the donated blood and the PRP prior to freezing. All quality-control parameters were displayed (including labeled information about patient identification, as it is for autologous use, blood group, platelet count, serological test results, and blood screening by nucleic acid testing) under local guidelines, which accomplished Good Manufacturing Practices. Patients were treated in the same orthopaedic outpatient clinic, proceeding knee intra-articular injection under sterile conditions, through superolateral approach, of a thawed 10mL aliquot each. The infiltration scheme consisted of 3 consecutive infiltrations, every 4 weeks. The study design and workflow are shown in Figure 2. A specific rehabilitation protocol was not applied to patients. Patients were allowed to continue with mild analgesics such as acetaminophen (not NSAIDs).
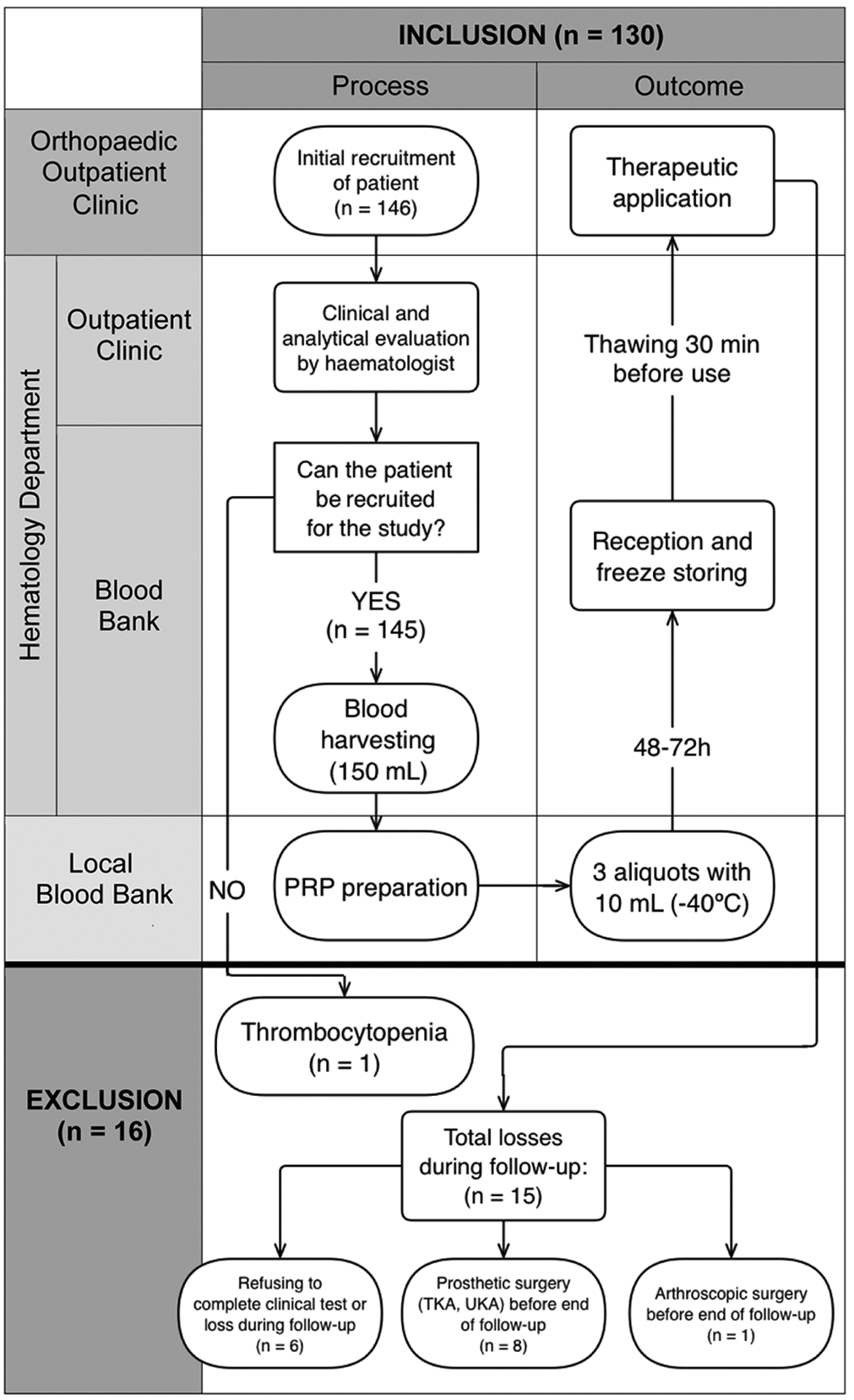
Study design and workflow, with inclusion and exclusion final data. PRP, platelet-rich plasma; TKA, total knee arthroplasty; UKA, unicompartmental knee arthroplasty.
Data and Outcomes Assessed
The following information was registered from each patient: sex, age, body mass index (BMI), knee side, labor disputes, and comorbidities such as diabetes mellitus, dyslipidemia, thyroid disease, rheumatologic conditions, and mental disease.
CBCs from each blood harvesting and processed PRP were obtained using a Sysmex XS-1000i hematocounter. From the harvested whole-blood CBC we used the platelet and WBC counts, and from the PRP CBC we used the platelet, WBC, and RBC counts.
The following PRP quality assessment parameters were calculated as previously described: platelet enrichment, dose of injected platelets, platelet and leukocyte efficiency, purity (percentage of total cells) in platelets, WBC, and RBC. 34
Patients were prospectively assessed for clinical response at baseline, and at 3-month, 6-month, and 1-year follow-up after the first injection. The primary outcome measure was the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score (through its validated Spanish version), 16 and cellular parameters were measured for PRP characterization. We also stratified patients according to baseline WOMAC scores (low, <35.3; medium, 35.4-51.4; and high, >51.5), as suggested by Tubach et al. 55
As a secondary outcome measure, patient satisfaction was surveyed by a simple categorical scale (none/low/enough/high). During the coronavirus disease 2019 (COVID-19) alarm state, these data were collected by telephone interview. The KL classification system was used to assess radiographic KOA. 28 In addition, we noted any adverse events, such as knee septic arthritis, knee pain not controlled with usual analgesic, and deep venous thrombosis.
Statistical Analysis
The sample size for this study was calculated based on the mean prevalence of KOA in Spain (around 10%) and our city population (300,000), using a 95% CI, a 6% precision, and an expected proportion of loss to follow-up of 10%. We calculated the final sample size needed to avoid sampling errors (adjusted for losses) was 109 patients. The Kolmogorov-Smirnov or chi-square test were used to assess normality. Paired-samples or independent Student t test, Mann-Whitney U test, paired-samples Wilcoxon test, analysis of variance, and Kruskal Wallis H test were employed to determine statistical significance. Correlation for quantitative data was calculated by Pearson or Spearman coefficient. We stratified in subgroups related with age and sex. All data were analyzed using SPSS (Version 16.0). Statistical significance was considered at P < .05.
Results
Demographic Data
From the 130 patients included in the study, 60 were men (46.2%) and 70 were women (53.8%), with a mean age of 63.04 ± 10.7 years (range, 40-90 years) and median BMI of 28.37 ± 3.98 (range, 21.2-41.6). The right knee was treated in 73 cases (56.2%) and the left knee in 57 cases (43.8%). Regarding comorbidities, 13 (10%) had diabetes mellitus, 30 (23.1%) had dyslipidemia, 26 (20%) had thyroid pathologies, 58 (44.6%) had history of mental disease (depression or anxiety), and 25 (19.2%) had other rheumatologic conditions (rheumatoid arthritis, gout, polymyalgia rheumatica); 28 patients (21.5%) were involved in labor disputes.
The most frequent radiographic osteoarthritis grade at study enrollment was KL grade 2 (53 cases; 40.8%), followed by grade 3 (39 cases; 30%), grade 1 (22 cases; 17%), and grade 4 (16 cases; 12.2%).
Product Characterization
Our PRP production method is summarized in Table 1. 43 CBC analysis of whole-blood samples showed a mean platelet count of 232.97 ± 59.51 × 109/L (range, 101-434 × 109/L) and a mean WBC count of 6.05 ± 1.50 × 109/L (range, 3.72-12.19 × 109/L).
PRP Production Characteristics a

a PRP, platelet-rich plasma; RBC, red blood cell; WBC, white blood cell.
PRP cytological and quality characteristics are shown in Table 2 and Supplemental Table S1.
Based on these results, we can identify our PRP according to the different classifications: pure-PRP, leukocyte poor (as defined by Dohan Ehrenfest et al 14 ), platelet count greater than baseline levels to 750 × 109/L and below/equal baseline WBC (based on the PAW [Platelets, Activation, White Cells] classification 12 ), ACA (very high dose of platelets, >90% efficiency of platelet recovery rate, very pure PRP, based on the DEPA (Dose Efficiency Purity Activation) classification 36 , frozen-thawed preparation, platelet count less than 900 × 109/L obtained by gravitational centrifugation techniques based on the International Society on Thrombosis and Haemostasis classification 24 , and PRP obtained from whole-blood donation that has been frozen-thawed once before application based on the classification by Acebes-Huerta et al 1 .
PRP Complete Blood Count and Calculated Quality Variables a

a PRP, platelet-rich plasma; RBC, red blood cell; WBC, white blood cell.
Clinical Results
The baseline and follow-up scores for the WOMAC global, WOMAC subscales, and baseline WOMAC categories are shown in Table 3 and Supplemental Table S1.
WOMAC Scores at Baseline and 3-Month, 6-Month, and 1-Year Follow-up a

a Data are presented as mean ± SD. WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
b Low, <35.3; medium, 35.4-51.4; high, >51.5. 55
Differences in WOMAC scores between baseline and 3-month, 6-month, and 1-year follow-ups are shown in Table 4. Statistical analysis revealed significant improvement at all time points studied versus baseline. Improvement was observed already at 3 months postoperatively and was maintained throughout the complete follow-up period (Table 4 and Figure 3). Similar results were seen across all WOMAC subscales, with only 1 particularity: WOMAC Stiffness improved further between 3- and 6-month follow-up (P = .03).
Difference in WOMAC Scores Between Follow-up Time Points a

a Data are presented as mean ± SD unless otherwise indicated. Bold P values indicate statistically significant difference between time points (P < .05). WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
b Wilcoxon test (Z statistic) was used for WOMAC Stiffness (nonparametric value).

Mean Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores with subscales and categories at baseline, and at 3-month, 6-month, and 1-year follow-up. Error bars indicate 95% CIs. *P < .05, ***P < .0001.
When considering the clinical significance of the results, the difference in WOMAC scores between baseline and 1-year follow-up represented a reduction of 25.72% ± 46.04%, which was considered clinically relevant according to Hmamouchi et al 25 (minimal clinically importance difference [MCID] >16%). The mean percentage reduction on the WOMAC subscales was 28.42% ± 47.24% for WOMAC Pain and 24.66% ± 48.36% for WOMAC Function, with a median (±SD) percentage reduction of 33.33% ± 87.4% for WOMAC Stiffness.
When stratified by WOMAC category, improvement from baseline was clinically significant according to Tubach et al 55 at 1-year follow-up in all 3 categories (mean difference, 5.81 ± 14.29, P = .01 for low WOMAC; 14.25 ± 19.04, P = .0001 for medium WOMAC; and 17.11 ± 18.61, P = .0001 for high WOMAC). Furthermore, the MCID was achieved in the low WOMAC and medium WOMAC categories, according to data and ranges reported by Tubach et al. Patients in the high WOMAC group did not reach the proposed MCID (20.4). Nevertheless, MCID was achieved on the global WOMAC (12.32 ± 17.95), according to the same authors (MCID, 9.1) (Table 4 and Figure 3). 55
Patient satisfaction at 1-year follow-up was “none” in 3.8% of patients, “low” in 25.4%, “enough” in 27.7%, and “high” in 43.1%. No adverse events were reported except, occasionally, local pain due to the infiltration procedure, which was treated with usual analgesics (eg, acetaminophen).
Correlation of Age, Sex, and PRP Quality With Clinical Response
We did not find any correlation between age and clinical results (WOMAC pre- vs 1-year posttreatment, P = .16; satisfaction at 1 year, P = .26) or PRP quality parameters (dose, P = .13; enrichment, P = .36; efficiency, P = .26, purity, P = .53), even when age was stratified into <65 and ≥65 years (clinical results: WOMAC pre- vs 1-year posttreatment, P = .64; satisfaction at 1-year, P = .06; PRP quality:dose, P = .39; enrichment, P = .39; efficiency, P = .41; purity, P = .58). Sex was also not correlated with clinical results (WOMAC score pre-vs 1-year posttreatment, P = .62; satisfaction at 1 year, P = .31), or PRP quality parameters (dose, P = .07; enrichment, P = .53; efficiency, P = .40; purity, P = .07). Finally, no significant correlations were found between PRP quality parameters (dose, efficiency, enrichment, or purity) and initial WOMAC score (P = .38, P = .78, P = .20, and P = .36, respectively), WOMAC at 1-year follow-up (P = .66, P = .90, P = .62, and P = .76, respectively), WOMAC pre- versus 1-year posttreatment (P = .72, P = .69, P = .52, and P = .60, respectively) or satisfaction at 1-year follow-up (P = .96, P = .21, P = .21, and P = .19, respectively) (Figure 4).

Correlation of (A) platelet enrichment and (B) platelet dose with clinical results as extrapolated from Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) and satisfaction scores. PLT, platelet; PRP, platelet-rich plasma.
Discussion
This pragmatic and prospective interventional pretest-posttest study evaluates the clinical efficacy in the treatment of KOA of a PRP product prepared using a standardized method, which is safe (closed system), affordable, and reproducible. Of the total patients recruited, 130 completed 1-year follow-up. We considered a treatment regime of 3 infiltrations every 4 weeks, as previous studies support that only multiple injections sustain anti-inflammatory effects in the long term compared with single injections in KOA. 9,21,56
Clinical Response
We found statistically significant improvement in WOMAC scores at all time points of observation (3-month, 6-month, and 1-year follow-up) compared with baseline (Figure 2 and Table 3). In addition, our data revealed that the improvement is achieved already at 3 months and is maintained, at least, through the 1 year follow-up, coinciding with published outcomes in recent level 1 evidence randomized controlled trials. 31,44,47 Clinical response is not influenced by age or sex in our study; however, there are discrepancies regarding this issue. 7,18,31,45,48 When analyzing the results of WOMAC subscales, we noted the same results, besides an improvement in stiffness from the 3-month to 6-month follow-up time point observation. Globally, at 1-year follow-up, the pain score improved 28%, the stiffness score around 33%, and the function score near 25% (24.8%). Furthermore, although patient satisfaction is not reported in many published studies, in our study, satisfaction at 1-year follow-up was rated as “enough” or “high” by 70% of the patients.
MCID is also an important aspect to consider regarding the assessment of clinical response and significance. MCID for global WOMAC score data was achieved, according to both Hmamouchi et al 25 (21% vs 16%) and Tubach et al 55 (12.32 vs 9.1). Besides, and according to the latter author, WOMAC categories may associate with the clinical response to treatment because, as hypothesized, the higher the baseline score, the better the improvement. 55 Supporting this notion, WOMAC score differences showed significant improvement in all categories (high, medium, low). Interestingly, MCID was achieved in low and medium WOMAC score group (5.81 vs 5.3 and 12.83 vs 11.8), but not in the high score group (17.11 vs 20.4). Of note, these latter 2 studies calculated these indicators based on treatment of KOA with NSAIDs, not PRP, and the follow-up period was 4 to 6 weeks, rather than 1 year follow-up. Further studies of this indicator involving PRP treatment and longer follow-up should be done to validate the results.
PRP Quality and Characterization
Many studies aim to define the appropriate and most efficient “therapeutic platelet dose” when using PRP. While a substantial enrichment of the platelet physiological count is desired, some in vitro studies have pointed out that too much of it could have an inhibitory and detrimental effect. 23 In a recent systematic review, the platelet count enrichment ranged between 1.3- and 8-fold, although the majority ranged between 2- and 4-fold. 30 In our study, the average PRP enrichment was 2.75 ± 0.65 times, with the reported and expected donor-dependent differences. 37 Interestingly, despite the mean platelet capture efficiency’s being 54.9%, classified as “C” (low), 36 the mean platelet purity was 96.64%, which ensures a very safe product (with absence of, or only residual, WBC and RBC, the presence of which may cause undesired side effects of an inflammatory nature). 38,39,45,49,53,54 Furthermore, in our previous study, 43 the concentration of 6 growth factors (epidermal growth factor, hepatocyte growth factor, platelet-derived growth factor—BB, vascular endothelial growth factor A and D, and fibroblast growth factor 23) was measured by multiplex technology in processed PRP samples during the validation period (from 14 different donors), and in the 3 aliquots obtained from each one. While donor-dependent variation was evident, the concentration of analyzed factors remained constant in the 3 frozen aliquots from each single donor, which assures the homogeneity of the PRP composition during the therapeutic regime (3 aliquots from the same donation). Further studies will aim at studying the association of PRP molecular composition with clinical responses in PRP KOA-treated patients.
The amount of growth factors in the PRP product is highly dependent on platelet-enrichment, but also on the postprocessing or activation method (ie, mechanical [through a freeze-thaw process] vs chemical [calcium chloride]) and is also a matter of debate. Several studies show that these methods may be equivalent, 2,27,50 highlighting that the freeze-thaw method is a simple and clinically compliant approach to assure the availability of platelet-derived growth factors and bioactive molecules in the final product. 15 Furthermore, a recent study suggests that frozen-thawed PRP should be preferred for chronic pathologies such as KOA, where injections have to be carried out more regularly. 27
The PRP volume used in the treatment of KOA is frequently reported in the literature as less than 6mL, 30 but other authors suggest that the ideal volume is at least 8mL, to ensure that platelets and plasma can diffuse throughout the joint, reaching all areas. 45,51 We used aliquots of approximately 10mL, with no adverse events observed. This volume enables us to achieve a “very high dose” of injected platelets, classifying the PRP used in this study as “A,” according to the DEPA classification, with a mean dose of injected platelets of 6.31 ± 1.92 × 10 9 per infiltration. The dose of injected platelets has been described as the most relevant parameter with which to assess clinical efficacy. 36
Regarding economic issues, and according to Scientific American, each PRP injection price can range from US$500 to US$2500, usually as out-of-pocket fees. 46 In Spain, a median cost of €194/US$224.10 per PRP injection (range, €90-€389/US$104-$450) is estimated. 41 However, final quality assessment of these PRP products, or serological tests or blood screening (nucleic acid testing), is often not performed. These factors were included in the price of the PRP used in this study, and the final production costs per PRP infiltration were €36/US$41.6 (complete treatment €108/US$125). This huge difference is possibly due to the reduction in production costs, as we used a blood bank infrastructure already available and in use. This price is considerably lower than the regular cost of HA in Spain (€200-€400/US$230-$462 per injection), but higher than that of corticosteroid injection (€2-€3 for each triamcinolone ampoule/US$2.3-$3.4). Nevertheless, this product is completely different from PRP (and, therefore, so are its results and effectiveness).
Relationship Between PRP Quality and Clinical Results
Of note, no association was found between PRP quality parameter values and clinical response. This fact concurs with the general belief that the larger number of platelets, the better the clinical results, with some caveats: it is possible that, above a certain quality threshold, a satisfactory clinical response is obtained. If producing PRP in this standardized manner ensures a PRP product with sufficient quality to produce positive clinical responses, 43 the monitoring of the PRP preparation methodology and its quality control should be given first priority in treatments involving PRP products.
The PRP used in this study comprises the characteristics required to classify it as “superdose PRP” and, against the idea that apheresis-based methods are currently the only way to have a reproducible PRP product with highly enriched platelet counts, our study shows that positive clinical responses can be also achieved when standardizing PRP production by differential centrifugation of whole-blood harvesting, using the infrastructure of a blood bank, assuring a leuko-depleted PRP product, with freezing-induced rupture of platelets and release of platelet growth factors. 13,26,40
Strengths and Weakness of the Study
Our study is nonrandomized and uncontrolled. In pragmatic trials, the question of placebo (as it has shown some improvements by itself as intra-articular injection in patients with KOA) and masking should be raised 20 ; in this case, at the present moment we do not have the possibility of an equivalent control group, as there is no therapeutic alternative used for symptomatic KOA in the daily practice, as most of our patients have tried them before (usually NSAIDs, corticoids, or HA infiltration) without good results. Future studies may address this issue by comparing this PRP product with platelet-lysates, growth factors such as sprifermin (rhFGF18), or other platelet-derived bioproducts on study (such as secretomes). Another limitation of the study is the minimum exclusion criteria in terms of prior nonoperative measures, surgical history, comorbidities, or KOA grade, which results in variability within the patient cohort. We did not assess outcomes as a function of KL severity or BMI. This study did not include assessment of the growth factors in the aliquots after freezing/thawing. Further studies with a more restrictive exclusion criteria and a larger cohort will be necessary to overcome this limitation.
The strengths of this study are mainly its pragmatic point of view, which allowed the recruitment of a high number of patients (N = 130). Another strength is that the posttest results were assessed at 3 different time points (at 3-month, 6-month, and 1-year follow-up), minimizing potential placebo effects. Finally, in this study we report almost all of the MIBO recommended for PRP. 42
Conclusion
In this study, we present a standardized PRP production method for symptomatic KOA with very good clinical results, with statistical (WOMAC) and clinical (MCID) significance and 70% patient satisfaction at 1-year follow-up. This product is safe, highly reproducible, affordable, and very convenient for the patient. Despite donor-dependent differences, the production method ensured PRP products above a minimum quality threshold, required for positive clinical responses in the treatment of KOA.
Supplemental material for this article is available at http://journals.sagepub.com/doi/suppl/ 10.1177/23259671221076496.
Supplemental material
Supplemental Material, sj-xlsx-1-ojs-10.1177_23259671221076496 - Clinical Response After Treatment of Knee Osteoarthritis With a Standardized, Closed-System, Low-Cost Platelet-Rich Plasma Product: 1-Year Outcomes
Supplemental Material, sj-xlsx-1-ojs-10.1177_23259671221076496 for Clinical Response After Treatment of Knee Osteoarthritis With a Standardized, Closed-System, Low-Cost Platelet-Rich Plasma Product: 1-Year Outcomes by Judit Fernández-Fuertes, Tamara Arias-Fernández, Andrea Acebes-Huerta, Marlene Álvarez-Rico and Laura Gutiérrez in Orthopaedic Journal of Sports Medicine
Footnotes
Acknowledgment
The authors thank all the institutions and personnel involved in this study: Hospital Universitario de Cabueñes (especially Carmen Fernández, chief of the hematology department, and all the workers involved at the Banco de Sangre-Blood Bank, as well as all the orthopaedic clinicians who contributed to this work); Centro Comunitario de Sangre y Tejidos de Asturias (especially María Carmen Muñoz and all the workers involved in the PRP production, and to Ana María Ojea for her support); Instituto de Investigación Universitaria del Principado de Asturias and University of Oviedo-Asturias (Spain). Finally, the authors thank Álvaro Camblor and Lorién García for their assistance with data collection.
Final revision submitted December 9, 2021; accepted December 23, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was partially supported by an intramural 2018 postdoctoral grant and an intramural 2019 fomento proyectos grant (Fundación para la Investigación y la Innovación Biosanitaria de Asturias [FINBA]) to A.A.-H. and I+D 2017 and 2020 grants (SAF2017-85489-P and PID2020-117265GB-I00, respectively; Ministerio de Ciencia e Innovación, Spain and Fondos FEDER) to L.G. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Hospital Universitario Central de Asturias (study 33/19).
Supplemental material
Supplemental material for this article is available at http://journals.sagepub.com/doi/suppl/
![]()
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
