Abstract
Background:
Injured anterior cruciate ligament (ACL) tissue retains proprioceptive nerve fibers, vascularity, and biomechanical properties. For these reasons, remnant ACL tissue is often preserved during the treatment of ACL injuries.
Purpose:
To assess through a cadaveric model whether reorienting and retensioning the residual ACL via an osteotomy improves knee stability after partial ACL tear, with substantial remnant tissue and intact femoral and tibial attachments.
Study Design:
Controlled laboratory study.
Methods:
In 8 adult cadaveric knees, we measured anterior tibial translation and rotational laxity at 30° and 90° of flexion with the ACL in its native state and in 3 conditions: partial tear, retensioned, and ACL-deficient. The partial-tear state consisted of a sectioned anteromedial ACL bundle.
Results:
In the native state, the translation was 10 ± 2.7 mm (mean ± SD) at 30° of flexion and 8.4 ± 3.6 mm at 90° of flexion. Anterior translation of the knees in the partial-tear state (14 ± 2.7 mm at 30° and 12 ± 2.7 mm at 90°) was significantly greater than baseline (P < .001 for both). Translation in the ACL-retensioned state (9.2 ± 1.7 mm at 30° and 7.2 ± 2.1 mm at 90°) was significantly less than in the ACL-deficient state (P < .001 for both), and translation was not significantly different from that of the intact state. For ACL-deficient knees, translation (20 ± 4.3 mm at 30° and 16 ± 4.4 mm at 90°) was significantly greater than all other states (P < .001 for all). Although rotational testing demonstrated the least laxity at 30° and 90° of flexion in the retensioned and intact states and the most laxity in the ACL-deficient state, rotation was not significantly different among any of the experimental states.
Conclusion:
In a cadaveric model of an incomplete ACL tear, a reorienting and retensioning core osteotomy at the tibial insertion of the remnant ACL improved anteroposterior translation of the knee without compromising its rotational laxity.
Clinical Relevance:
The findings of this study support the concept of ACL tissue reorienting and retensioning in the treatment of ACL laxity as an area for future investigation.
Anterior cruciate ligament (ACL) reconstruction (ACLR) is the gold standard for restoring knee stability after an ACL tear. In this technique, the torn ACL is removed and replaced with a new tissue graft. However, even after an injury, ACL tissue retains its proprioceptive nerve fibers, vascularity, and biomechanical properties. 16 Thus, preserving native, remnant ACL tissue may have benefits over complete resection when treating ACL laxity.
The potential benefits of preserving native ACL tissue after ACL injury have been recognized. 13,16 For ACLR, techniques on preserving femoral and tibial-sided ACL stumps, as well as intact ACL bundles, have been described. 1,8,9 While the former techniques help graft healing and incorporation, the latter improves postoperative proprioception function. In the spectrum of ACL tear patterns, there are instances in which most of the macroscopic ligament is in continuity, though redundant. These patterns include tibial spine fractures, in which both ACL bundles are often in continuity and lax, and partial ACL tears, in which a single-bundle ACL tear can be completely torn, with an incomplete tear of the other ACL bundle. When these partial tears cause instability that would benefit from surgical intervention, an alternative tissue-preserving treatment may be possible. 7 With a substantial ACL remnant that is continuous but lax, reorienting and retensioning the remaining ACL tissue may be an option.
Thus, we propose an alternative approach to complete resection and reconstruction for a subset of incomplete ACL tears by reorienting and retensioning the remaining native fibers. We hypothesized that by using an osteotomy, the intact posterolateral bundle in an incomplete ACL tear can be centralized in the tibial insertion and advanced distally to improve joint stability. Our goal was to assess whether a core osteotomy improved joint stability in a cadaveric model of an incomplete ACL tear.
Methods
Specimen Preparation
After obtaining cadaveric lower extremities from our state anatomy board, we examined and excluded specimens that had undergone prior knee surgery and lacked intact cruciate ligaments. In 8 specimens, we dissected and removed the soft tissue structures around the knee, leaving the collateral ligaments, joint capsule, and intra-articular structures intact. We osteotomized the femur and tibia at set distances, as determined by the mounting apparatus, from the joint line. An anterior parapatellar approach to the knee was utilized, with the quadriceps tendon transected proximally to reflect the patella distally, to provide access to the ACL. The cruciate ligaments were visually inspected to confirm their integrity.
Biomechanical Testing
Each specimen was positioned in a custom-made mount that allowed adjustment of the knee flexion angle. 15 The mount was attached to a servohydraulic testing machine (MTS 858 Material Testing System; MTS Systems) that pemitted measurement of tibial translation and rotation (Figure 1). During mounting, the axis of knee flexion was aligned with the actuator axis, and the degrees of knee flexion were confirmed with a goniometer. Translational and rotational testing loads were based on prior testing experience. 15

The test apparatus allows measurement of anterior tibial translation as well as internal and external tibial rotation. (A) Actuator from materials testing machine and direction of translational force (double arrow). (B) Transducer to measure degrees of tibial rotation. (C) Torque is applied here on the tibial mounting jig and direction of rotational force (curved arrow). (D) The femoral mounting jig allowed for variable knee flexion angles (dashed line).
In the native state, we established the neutral point for each knee in relation to anterior and posterior translation according to methods utilized in a prior study. 15 The knee was then cycled between –30 N and 30 N at 0.01 Hz. The resulting load-versus-displacement curve displayed a central section with no slope. The midpoint of this no-slope section (inflection point) was defined as the neutral point. Translational testing at this anteroposterior neutral point ensured that all measured tibial translation would be anterior subluxation of the tibia. Additionally, this served as the neutral point for both internal and external rotation testing.
Testing of anteroposterior translation was performed at 30° and 90° of knee flexion to simulate standard clinical tests of ACL laxity (ie, a Lachman and anterior drawer test). Translation testing was performed by applying a monotonically increasing load to 134 N over 60 seconds. The translation tests were repeated for each specimen in each experimental state.
For rotational laxity testing at 30° and 90°, a ±6.5-N·m torque was applied by a hanging 15-lb. weight to the rotational axis of the tibial mount. The resulting maximum internal and external rotation were recorded. Two cycles were completed for each specimen in each experimental state.
Experimental States
First, baseline testing was performed with the ACL in its native state. Second, we identified and sectioned at least 50% of the ACL, including all of the anteromedial bundle of the ACL, and testing was repeated (herein, “partial-tear state”). When there appeared to be an intermediate ACL bundle, or if the border between the anteromedial and posterolateral bundles was unclear, care was taken to section more of the ACL to ensure that only the posterolateral bundle remained (Figure 2A). Third, we performed a coring osteotomy of the intact posterolateral ACL bundle using a coring reamer (Coring Reamer and Collared Pin Set, 10 mm, AR-1224 S; Arthrex) (Figure 2B). The osteotomy started distally on the anteromedial face of the tibia, just proximal to the pes anserinus. The proximal portion of the osteotomy centered on the ACL, including the intact posterolateral bundle. The osteotomy trajectory was first established by a guide pin then cut with the cannulated coring reamer. This osteotomy was then rotated, centralizing the posterolateral bundle by bringing it more anterior and medial, placing the remaining bundle in the anterior half of the circular osteotomy. Rotation was performed with a clamp on the bone core distally. A No. 2 nonabsorbable FiberWire suture (Arthrex) was passed through the base of the ACL at its insertion in osteotomy core. The osteotomy was then distalized within the coring tunnel, with the knee at 30° of flexion, with manual tension on the Fiberwire sutures, tensioning the posterolateral bundle. The FiberWire suture was tied over a tibial post and an interference screw (Figure 2C). Testing was repeated for the repaired knees (herein, “ACL-retensioned state”). Fourth, the remaining ACL was sectioned, and testing was repeated (herein, “ACL-deficient state”).

Illustrations and corresponding arthroscopic images of experimental steps. (A) Sectioning of at least 50% of the ACL. (B) A coring osteotomy of the intact posterolateral ACL bundle (anteromedial bundle removed for clarity). (C) The centralized and distalized osteotomy within the coring tunnel. Illustrations courtesy of Ransel Palatino. ACL, anterior cruciate ligament.
Statistical Analysis
Descriptive analyses consisted of means, medians, and standard deviations. We performed t tests, with significance set at P < .05. Analyses were performed using Excel (Microsoft) and SPSS Version 23 (IBM).
Results
Stability Testing at 30º
At 30º of knee flexion, mean anterior tibial translation was 10 ± 2.6 mm in the intact state, increasing significantly to 14 ± 2.7 mm in the partial-tear state (P < .001) (Figure 3). In the ACL-retensioned state, mean anterior tibial translation decreased to 9.2 ± 1.7 mm, which was significantly different from the ACL-deficient state (P < .001). Mean anterior tibial translation in the ACL-retensioned state was not different from the intact state (P = .38). Five of the ACL-retensioned knees had similar translation to the intact state, and 3 had less translation than in the intact state. The ACL-deficient knees had greater translation at 30° of knee flexion compared with all other experimental states (P < .001), including the partial tear state (Table 1).
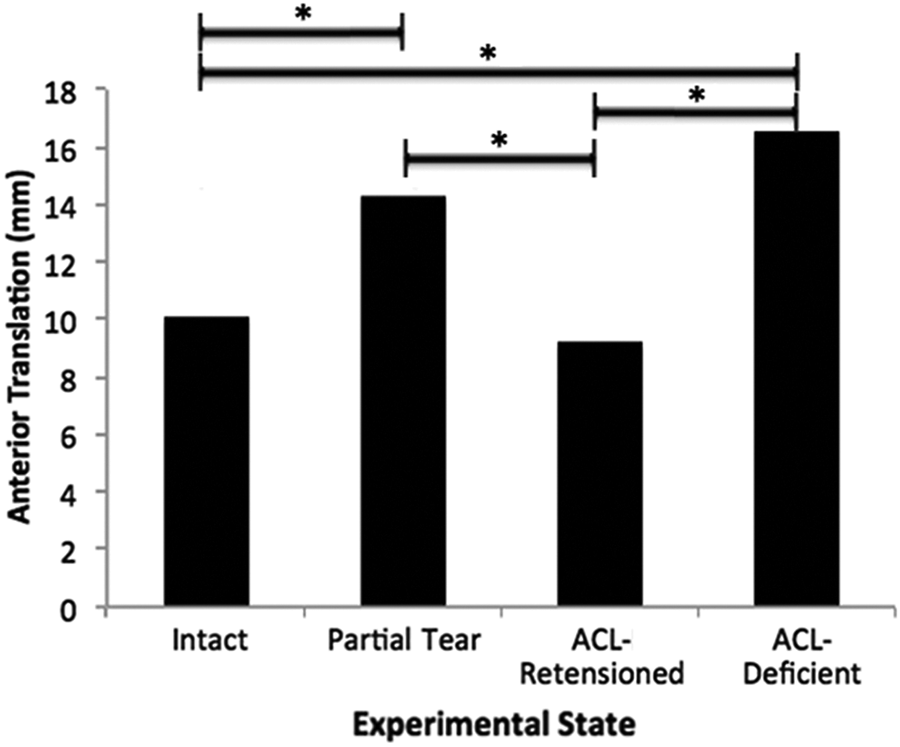
Mean anterior tibial translation in each experimental state in the 8 cadaveric knees at 30° of knee flexion (simulated Lachman test). *P < .001. ACL, anterior cruciate ligament.
Anterior Tibial Translation of the 8 Cadaveric Knees in the 4 Experimental States a
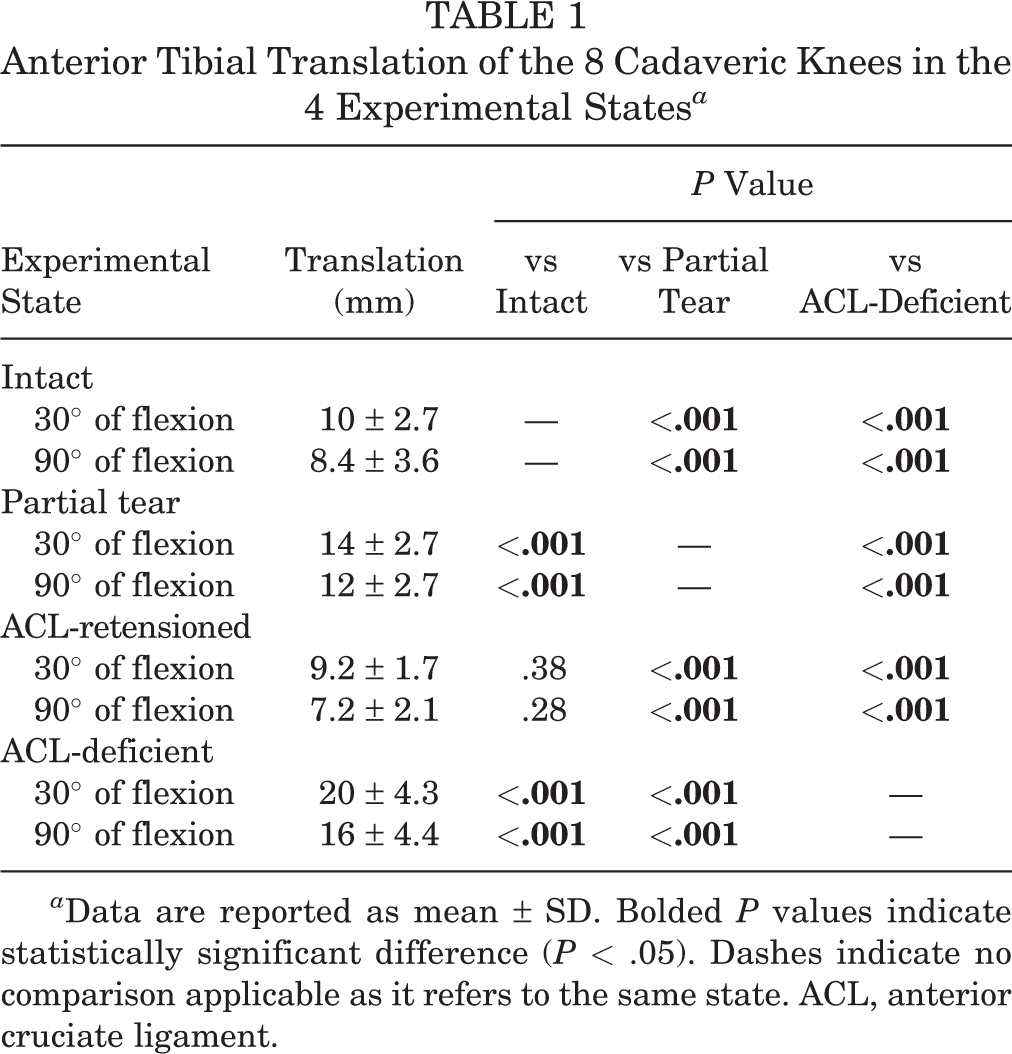
a Data are reported as mean ± SD. Bolded P values indicate statistically significant difference (P < .05). Dashes indicate no comparison applicable as it refers to the same state. ACL, anterior cruciate ligament.
At 30° of knee flexion, mean rotational laxity was least in the ACL-retensioned state, followed by the intact state, the partial-tear state, and the ACL-deficient state (Table 2). However, no experimental state was significantly different from any other in terms of rotational laxity.
Rotational Laxity of the 8 Cadaveric Knees in the 4 Experimental States a

a Data are reported as mean ± SD. Dashes indicate no comparison applicable as it refers to the same state. ACL, anterior cruciate ligament.
Stability Testing at 90º
At 90° of knee flexion, the mean anterior tibial translation was 8.4 ± 3.3 mm in the intact state, increasing significantly to 12 ± 2.7 mm in the partial-tear state (P < .001) (Figure 4). In the ACL-retensioned state, the mean anterior tibial translation decreased to 7.2 ± 2.1 mm, which was significantly different from the partial-tear state (P < .001). The ACL-retensioned state was not significantly different from the intact state in terms of mean anterior tibial translation (P = .28). Four knees in the ACL-retensioned state had similar translation to the intact state, and 4 had less translation than in the intact state. The ACL-deficient knees had greater translation at 90º of knee flexion compared with all other experimental states (P < .001), including the partial-tear state.
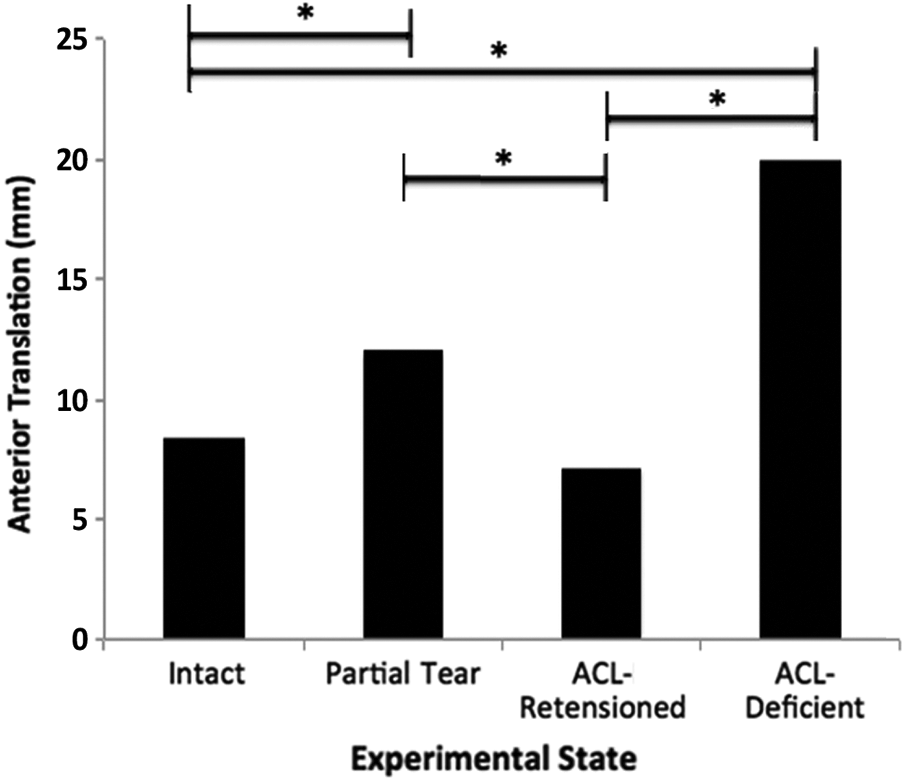
Mean anterior tibial translation in each experimental state in 8 cadaveric knees at 90° of knee flexion (simulated anterior drawer test). *P < .001. ACL, anterior cruciate ligament.
At 90° of knee flexion, the mean rotational laxity was least in the intact state, followed by the partial-tear state, the ACL-retensioned state, and the ACL-deficient state. However, no experimental state was significantly different from any other in terms of rotational laxity (Table 2).
Discussion
Using a cadaveric model, we found that a reorienting and retensioning osteotomy of the residual posterolateral ACL fibers can improve knee anterior translational stability after an incomplete ACL tear. Currently, the standard operative treatment for ACL tears is ACLR with debridement of the torn ligament and placement of a new graft substitute. Because remnant ACLs retain some of their neurovascular and biomechanical properties, preserving the native ACL while restoring stability may have benefits over ACLR.
The ACL anatomy has been well-described, with early studies reporting the existence of 2 or 3 bundles. 3,14 Subsequent studies described the femoral and tibial attachment points with degrees of variability reflecting natural variations in human anatomy. 2,4,17 For ACLR, reconstructing the ACL as a single bundle versus 2 bundles has been explored. Single- and double-bundle reconstructions have produced similar clinical outcomes and failure rates, 6,10 suggesting that a single-bundle ACLR may provide sufficient translational and rotational stability of the knee. We thus hypothesized that a remnant ACL, appropriately positioned and tensioned, could improve translational and rotational stability in a cadaveric model.
Our cadaveric models, progressing from a partial ACL tear to ACL retensioning and then finally to complete ACL disruption, confirmed our hypothesis regarding the ability of a coring osteotomy to improve translational stability. The anteromedial ACL bundle primarily provides anterior translational stability. 3,14 Accordingly, sectioning of the anteromedial bundle produced a significant increase in anterior translation. In contrast, the posterolateral bundle primarily provides rotational stability. 3,14 Thus, in the final step of our model (the only state in which the posterolateral bundle was completely disrupted, with complete removal of the ACL), rotational laxity was greatest, though the difference was not significant. In terms of our primary aim of using the remaining ACL to improve stability, we found a significant decrease in translational laxity after ACL retensioning compared with the partial-tear stage. Though some specimens had more laxity than the intact state, while others were overconstrained, all had translation similar to baseline levels. Therefore, we have shown that the posterolateral bundle, when repositioned and tensioned anteromedially, can restore anterior translational stability. In the final state, the ACL-deficient knee had a combined increase in anterior translation and rotation. Thus, the osteotomized, centralized, and distalized tibial insertion of the posterolateral bundle can improve translational stability and maintain rotational stability in a cadaveric model.
In terms of the value of native tissue after an ACL tear, remnant ACL fibers have beneficial properties that can make preservation worthwhile. After injury, the proprioceptive nerves and vasculature of the ACL remain. Sonnery-Cottet et al 16 showed that in partial ACL tears, a well-vascularized synovial sheet and proprioceptive neuroreceptors remain. Though these properties support reinnervation and revascularization of soft-tissue ACLR grafts, these preserved histologic characteristics point to the potential benefits of salvaging the remnant ACL fibers. Furthermore, the proprioceptive function of the remnant ACL may improve knee biomechanics and help reduce cartilage wear. Murray and Fleming 13 evaluated ACL repair with a bioenhanced scaffolding in a porcine model. They found that knees that underwent biologically enhanced ACL repair had less macroscopic cartilage damage than those that underwent ACLR. This occurred despite the fact that the ACLR group had better anteroposterior stability than an ACL-transection control knee and that the repair group did not. Preserving the ACL remnant via repair still resulted in significantly less posttraumatic osteoarthritis than did reconstruction. This concept supports the potential benefits of preserving native ACL fibers. Salvage of the native ACL, such as with our proposed osteotomy, may preserve the proprioceptive nerves and vasculature and protect the cartilage.
While the incomplete ACL tear covers injuries ranging from microscopic collagen fiber disruption to near complete tears with few remaining strands of collagen, there is likely a specific subset of ACL tears for which retensioning could be considered. In our experimental state, if the demarcation of bundles was unclear, more ACL was sectioned, creating a small posterolateral bundle. Though the retensioned state demonstrated that a small remnant ACL can improve anterior translational laxity, in practical application, a more substantial ACL remnant should be present. This technique could be considered on the end of the spectrum in which collagen fiber disruption occurs on a microscopic level but where, grossly, a nearly intact ACL remnant remains. In this situation, the substantial remaining tissue would make reconstruction with bundle preservation difficult to achieve without disrupting intact ACL fibers. However, the substantial remaining tissue also makes the addition of graft tissue via reconstruction unnecessary, which would otherwise be difficult with the creation and advancement of a bone core. Application of this retensioning concept would require careful selection of the appropriate ACL remnant. Selection could include confirmation preoperatively of ACL fiber continuity on magnetic resonance imaging and intraoperatively of the preservation of the majority of both ACL bundles. Additionally, the ACL remnant should have an intact femoral origin and tibial insertion to be considered an appropriate ACL remnant.
In addition, there are analogous circumstances outside of incomplete tears in which intact femoral and tibial ACL attachments are intact and a continuous ligament exists between them. These scenarios may be amenable to tensioning of a lax ACL. A similar procedural concept already exists for tibial spine fracture, which often involves a primary bony avulsion with a secondary partial ACL tear. 5,11,12 Simple reduction and fixation of the bony avulsion often results in increased laxity caused by intrasubstance ACL injury. Thus, it is common to attempt to compensate for this laxity by advancing the bone fragment to retension the ACL. An osteotomy would be conceptually similar, addressing laxity through a bony procedure. In addition, ACL repairs can have a healed ACL ligament with residual laxity. 13 There may be redundant ACL tissue in the setting of connective tissue disease, and secondary laxity is often present after a primary collateral ligament injury or lower extremity malalignment. Patients with such conditions could benefit from an osteotomy that achieves distalization, without rotation and reorientation of the fibers. The concept of ACL retensioning may be applicable to other scenarios beyond the specific scenario tested in this study.
Our study is limited by our usage of a cadaveric model and the inherent heterogeneity in knee anatomy. Sectioning a portion of the ACL in isolation may not replicate an in vivo injury, because the remaining bundle typically sustains an injury as well. However, for the purposes of our study, sectioning the anteromedial bundle consistently created measurable changes in laxity through each testing stage. In addition, our goal was to demonstrate that the remnant could be reoriented for stability, and, in theory, any in vivo laxity of the remaining bundle could be addressed through the distalization component of the osteotomy. Future in vitro and in vivo knee testing is required. We were also limited to measurements with the knee flexed in fixed positions rather than dynamically. For translational stability, we simulated the Lachman test at 30° and the anterior drawer test at 90° of knee flexion, which are the most clinically relevant positions for testing translational stability. For rotational stability, we assessed rotation in isolation. Our testing does not simulate a true pivot-shift test, which includes a simultaneously valgus and axial force in a knee with an intact iliotibial band. From our results, we were unable to distinguish the expected rotational differences between states. However, the variability in rotation recorded suggests that the translational stability gained by altering the posterolateral bundle may need to be balanced against maintaining, rather than overconstraining, the natural rotational laxity of the knee. Also, in vivo application of this concept has to balance distalization of the remnant ACL against overconstraining the knee, which could lead to a flexion contracture. The possibility of overconstraint is reflected in the lower absolute values of anterior translation in the retensioned state. For practical purposes, tensioning the remnant ACL closer to full extension, rather than 30°, may be a consideration for in vivo application. Finally, technical considerations for further in vitro and future in vivo study of this technique include the femoral origin of the retensioned ACL, coring of a broad ACL tibial insertion, the absence of open physes, and the careful selection of incomplete ACL injuries with a substantial remnant.
In conclusion, in a cadaveric model of an incomplete ACL tear, a coring osteotomy at the tibial insertion of the remnant posterolateral ACL bundle improved anteroposterior translation of the knee without compromising rotational laxity. Retensioning remnant ACL tissue can be explored as a tool for addressing ACL-related instability.
Footnotes
Final revision submitted October 23, 2020; accepted December 14, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research supplies for this study were provided by Arthrex. R.J.L. has received education payments from Supreme Orthopedic Systems and research support from OrthoPediatrics and Vericel. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
