Abstract
Background:
Massive rotator cuff tears associated with greater tuberosity bone loss are challenging to treat. Repairing the rotator cuff without addressing the greater tuberosity deficiency may result in poorer clinical outcomes.
Hypothesis:
Utilizing an Achilles tendon–bone block allograft to address both the massive rotator cuff tear and greater tuberosity bone loss concurrently can result in improved clinical outcomes.
Study Design:
Case series; Level of evidence, 4.
Methods:
The authors performed a retrospective study of patients treated between January 2011 and December 2018 with Achilles tendon–bone block allograft for massive rotator cuff tears associated with greater tuberosity bone loss. The inclusion criteria were massive rotator cuff tear and bony deficiency of the greater tuberosity; patients with a history of bony metabolism disease, connective tissue disease, and previous surgery to the wrist or elbow of the affected limb were excluded. Range of motion, visual analog scale for pain, Constant score, and Single Numeric Assessment Evaluation score were assessed preoperatively and at a minimum of 2 years postoperatively. Radiographs and ultrasound images were evaluated to assess allograft union and rotator cuff integrity.
Results:
Five patients (3 male and 2 female; mean age, 54.0 ± 12.2 years) were included in the study. The mean follow-up was 80.6 ± 33.7 months. Preoperative to postoperative values improved significantly on the visual analog scale (from 45.8 ± 25.5 to 14.5 ± 14.1; P = .04), Constant score (from 36.8 ± 7.9 to 73.5 ± 3.1; P < .001), and Single Numeric Assessment Evaluation score (from 42.5 ± 26.3 to 82.5 ± 10.4; P = .04). Forward flexion improved significantly from 53° ± 47° to 149° ± 17° (P = .03). Four of the 5 patients achieved bony union. One patient required removal of symptomatic hardware at 6 months postoperatively, and 1 patient required revision surgery at 1 year postoperatively because of progressive osteonecrosis of the humeral head.
Conclusion:
In patients with massive rotator cuff tears and a greater tuberosity bony deficiency, utilizing an Achilles tendon–bone block allograft to restore the bony defect and reinforce the rotator cuff repair was safe and effective. At a minimum of 2 years postoperatively, most patients demonstrated improved clinical outcomes, tendon healing, and graft incorporation.
Keywords
Massive rotator cuff tears with concomitant greater tuberosity bone loss are challenging to treat. 18 Proximal humeral bone loss can occur as a result of trauma with consequent nonunion or bony resorption of the greater tuberosity bone. 27 Other causes of greater tuberosity bone loss include large degenerative humeral cysts 2,7,32 and osteolytic reaction from implants. 30 As the greater tuberosity forms the insertion point of the supraspinatus, infraspinatus, and teres minor tendons, the loss of bone in this region makes repair of the tendons challenging. In addition, the absence of the greater tuberosity bone may limit deltoid wrapping and the associated deltoid tension, affecting clinical outcomes. 33 In cases of a combined massive rotator cuff tear with greater tuberosity bony defect, surgeons need to achieve both a robust rotator cuff repair and restoration of the greater tuberosity bone stock.
In patients aged >60 years, reverse shoulder arthroplasty (RSA) is an acceptable solution. However, because of longevity concerns, RSA is not an ideal option for young patients. 5,12,25 Achilles tendon–bone block allograft (ATBA) has been described as an alternative to RSA in a young patient with greater tuberosity and rotator cuff deficiency. 20 There has also been growing interest in utilizing the Achilles tendon allograft for superior capsular reconstruction (SCR). 15,16,24 However, there remains a paucity of clinical literature analyzing outcomes of an ATBA for concomitant massive rotator cuff tears and greater tuberosity bone loss.
In this study, we aimed to report the minimum 2-year clinical and radiological outcomes of patients who underwent an ATBA for massive rotator cuff tears with greater tuberosity bone deficiency. The hypothesis was that utilization of such reconstruction would result in significant improvement in range of motion (ROM) and patient-reported outcomes.
Methods
Patient Selection
This study obtained ethical approval from the institutional review board of our institution, and written informed consent was obtained from all patients. A retrospective review was conducted of ATBAs performed by a single surgeon (A.L.) between January 2011 and December 2018. The inclusion criteria included massive rotator cuff tear and bony deficiency of the greater tuberosity with a minimum follow-up of 2 years (Figure 1). Massive rotator cuff tears were defined as a complete tear of ≥2 tendons, with at least 1 tendon retracted beyond the top of the humeral head. 21 The exclusion criteria included history of bony metabolism disease, history of connective tissue disease, and previous surgery to the wrist or elbow of the affected limb. All patients who fit the inclusion criteria were recruited.

(A) Anteroposterior plain radiograph of a left shoulder (patient 5) showing significant bone loss in the greater tuberosity after proximal humeral fracture, previous osteosynthesis followed by hardware removal, and open rotator cuff repair. (B) Magnetic resonance imaging (MRI) scan showing a retracted (Patte 2) supraspinatus tear in the same patient.
Surgical Technique
Surgery was performed with the patient in the beach-chair position under general anesthesia with an interscalene nerve block. An anterosuperior incision was used for a deltoid split approach to expose the greater tuberosity. The remnant rotator cuff tendon was identified, debrided, and preserved (Figure 2). The proximal humeral head defect was then exposed and debrided to a viable bony bed. A fresh-frozen, gamma-irradiated ATBA (Maxxeus; Community Tissue Services) was thawed in warm saline for 15 minutes (Figure 3A). The calcaneum bone was then shaped using a saw such that the allograft bone approximated the proximal humeral head defect. A shelf of bone adjacent to the Achilles tendon was preserved to allow for tendon ingrowth after native rotator cuff repair (Figure 3B and Video Supplement).

Lateral view from a right shoulder. The remnant rotator cuff tendon is identified, and sutures are passed through it. The proximal humeral head defect is debrided to a viable bony bed.

(A) fresh-frozen, gamma-irradiated Achilles tendon–bone block allograft is thawed and prepared. (B) The Achilles tendon is partially lifted off the bone block to create a shelf of bone adjacent to the tendon. This is to allow for improved rotator cuff tendon ingrowth after the native rotator cuff repair.
Depending on the size of the bone defect, the bony portion of the allograft was secured to the proximal humerus via one or two 4-mm malleolar screws under fluoroscopic control (Figure 4). The preserved native rotator cuff was repaired onto the medial remnant native bone using 4.5-mm double-loaded suture anchors in a horizontal mattress configuration (Healix; DePuy Mitek, Inc) (Figure 5 and Video Supplement). The Achilles tendon was then trimmed to an appropriate length to reinforce the native rotator cuff. The Achilles tendon was sewn onto the remnant cuff using high-strength sutures (Ethibond 6; Ethicon) so as to reinforce the repair (Figure 6 and Video Supplement).

Intraoperative fluoroscopic image of a left shoulder. Restoration of bone stock and good fixation of the calcaneum graft is confirmed.

The Achilles tendon (AT) is flipped laterally to allow for repair of the preserved native rotator cuff (white arrow) via suture anchors placed into the medial remnant bone. The bone block is secured into the greater tuberosity defect via a 4-mm malleolar screw. The AT has been trimmed to an appropriate length for reinforcing the rotator cuff tendon repair.

The Achilles tendon (AT) is sewn onto the remnant cuff to reinforce the repair.
Postoperatively, all patients underwent the same rehabilitation protocol. Patients wore an abduction pillow sling for 6 weeks. The sling was removed, and passive mobilization was initiated at 6 weeks postoperatively. Strengthening exercises were initiated at 12 weeks postoperatively.
Study Variables
The primary outcome measure was improvement in clinical outcomes such as ROM, visual analog scale (VAS) score for pain (maximum score of 100), the Constant score, 10 and the Single Numeric Assessment Evaluation (SANE) score. 35 These clinical instruments were selected for their ease of administration and clinical validity. 6 Furthermore, the prevalence of complications such as graft nonintegration, infection, or reoperation was evaluated. The following baseline characteristics were assessed: age, sex, diagnosis, previous surgery, shoulder side, and limb dominance. Postoperative histology of the graft was obtained in 1 patient at the 6-month postoperative period.
Clinical and Radiological Outcomes
A combination of the Jobe test, Patte test, and external rotation lag sign was used to diagnose posterosuperior rotator cuff tears. 9,22 Clinical outcomes measured included ROM and the VAS, Constant, and SANE scores. These scores were obtained preoperatively and at a minimum of 2 years postsurgery. For the clinical assessment of ROM, we used a goniometer to measure active anterior forward flexion and external rotation. External rotation was measured with the patient's arm by the side of the body, and internal rotation was assessed to be the highest vertebral spinous process reached by the extended thumb of the patient. The clinical ROM measurements were performed by the senior author (A.L.).
The radiological assessment included preoperative plain radiology and magnetic resonance imaging (MRI) and computed tomography (CT). The rotator cuff tear was assessed on MRI scans and classified as per the classifications of Collin et al 8 and Lädermann et al 17 by 2 fellowship-trained orthopaedic surgeons (S.W.L.H., X.C.). The Collin classification divides the rotator cuff into 5 components: supraspinatus, superior subscapularis, inferior subscapularis, infraspinatus, and teres minor. The tear pattern is then classified into 5 types depending on the location of the tear: A (supraspinatus and superior subscapularis), B (supraspinatus and entire subscapularis), C (supraspinatus, superior subscapularis, and infraspinatus), D (supraspinatus and infraspinatus), and E (supraspinatus, infraspinatus, and teres minor). The Lädermann classification divides rotator cuff lesions into 4 major types: A (involvement of bone), B (involvement of tendon), C (musculotendinous junction), and D (involvement of muscle). Tendon retraction was determined using the Patte classification, 29 muscle atrophy using Tangeant sign 36 and fatty infiltration of the affected muscles was graded via the Goutallier classification. 14 Bone loss was measured by the same 2 reviewers (S.W.L.H., X.C.) on the axial section of the MRI scan on the cut, where the bone loss was deemed maximal. The bone loss was assessed from the anterior-most margin of the bone defect to the posterior-most margin. Plain radiography and ultrasonography 31 were repeated at a minimum of 2 years postsurgery. These images were reviewed by the same 2 reviewers (S.W.L.H., X.C.). Radiological factors such as graft union, implant failure, rotator cuff integrity, and thickness of the rotator cuff were assessed.
Statistical Analysis
The descriptive analysis consisted of frequencies and percentages for discrete data. Means and SDs were used for continuous data. Differences between the pre- and postoperative VAS, Constant, and SANE scores were statistically analyzed using the paired Student t tests for patients with complete data. A 2-tailed significance level of .05 was used for all the tests. All statistical analysis was conducted using Stata 13 (StataCorp).
Results
Clinical Outcomes
A total of 5 patients (3 male and 2 female) underwent the ATBA procedure during the study period, and all 5 were available for follow-up at a mean of 80.6 ± 33.7 months (range, 48-135 months) postoperatively. The mean age of the patients was 54.0 ± 12.2 years (range, 44-71 years). The dominant arm was affected in 3 of the 5 patients (Table 1).
Patient Characteristics a

a ATBA, Achilles tendon–bone block allograft.
b Bone loss occurred as a result of a chronic greater tuberosity fracture.
Four of the 5 patients had previous surgery to the affected shoulder. Four of the 5 patients were evaluated with either pseudoparalysis 21 or pseudoparesis of the affected shoulder. All the patients had the same configuration of massive rotator cuff tears, with both supraspinatus and infraspinatus tears (type D) 16 and concomitant tuberosity insufficiency (type A3). 17 All patients had minimum grade 2 retracted tears and 2 complete tendons, being thus considered as massive rotator cuff tears. 21 All the patients had low-grade atrophy (grade 1 or 2) per the Goutallier classification. The preoperative radiological characteristics are summarized in Table 2.
Clinical outcomes are summarized in Table 3. Significant improvements were seen in all the clinical scores and anterior forward flexion.
Preoperative Rotator Cuff Radiological Characteristics
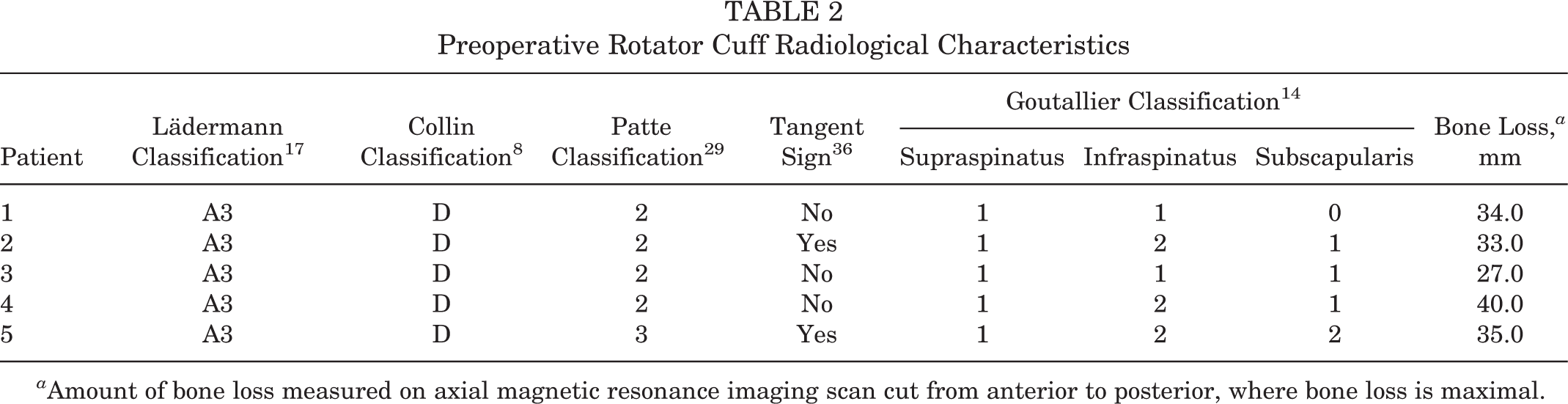
a Amount of bone loss measured on axial magnetic resonance imaging scan cut from anterior to posterior, where bone loss is maximal.
Comparison of Preoperative and Postoperative Clinical Outcomes a

a Bold values indicate significant P-values (P < .05). AFF, anterior forward flexion; ATBA, Achilles tendon-bone block allograft; ER, external rotation; IR, internal rotation, hand in the back; SANE, single numeric assessment evaluation; VAS, visual analog scale.
a AFF, anterior forward flexion; ATBA, Achilles tendon-bone block allograft; ER, external rotation; IR, internal rotation, hand in the back; SANE, single numeric assessment evaluation; VAS, visual analog scale.
b Patient underwent revision to reverse total shoulder arthroplasty at 1 year after the ATBA procedure because of progressive osteonecrosis of the humeral head and was excluded from the data analysis of the clinical outcomes.
There was 1 patient with clinical failure who required revision surgery for progressive osteonecrosis of the whole humeral head. The patient was aged 48 years and did not improve after initial ATBA surgery. An MRI scan revealed progressive osteonecrosis of the humeral head (Figure 7). At 1 year post–ATBA surgery, revision RSA was performed on the patient (patient 5). Relook arthroscopy was performed in 1 patient (patient 1) for removal of symptomatic hardware. At 6 months postoperatively, the patient had discomfort arising from an impingement of the malleolar screw on abduction, and removal of hardware was performed. A biopsy specimen of the Achilles tendon graft was taken at the same setting. Histology showed angiogenesis and incorporation of the Achilles tendon allograft tissue (Figure 8).

(A) Anteroposterior radiograph of the left shoulder showing nonunion and resorption of the Achilles tendon–bone block allograft (ATBA). (B) Coronal T1-weighted magnetic resonance imaging scan of the left shoulder showing osteonecrosis of the humeral head with a retracted tear of the supraspinatus tendon. White arrow, retracted supraspinatus tear. *Malleolar screw in situ.

Histopathology of the right Achilles tendon graft (patient 1). The slide revealed a mostly viable tendon-like fibrous tissue with recognizable nuclei. It is rearranged with areas of neovascularization and edematous areas with myxoid degeneration. A minimal inflammatory infiltrate with rare lymphocytes and occasional plasma cells are also observed on one of the fragments in the most vascularized zone. On less than one-third of the surface of the sample, there are areas where the tendon tissue is eosinophilic, devitalized, and without a nucleus.
Radiological Outcomes
Postoperative radiographs showed graft incorporation in 4 of the 5 (80%) patients (Figure 9, A-C). In 1 patient (patient 5), there was nonunion of the allograft associated with progression of humeral head osteonecrosis, likely as a result of the original trauma. Ultrasound examination showed an intact rotator cuff tendon in all patients (Figure 9D), except the 1 who underwent revision via an RSA (80%) at the final follow-up. In 1 patient (patient 2), glenohumeral arthritis (asymptomatic) was noticed at the 7-year follow-up.

Anteroposterior radiograph of a right shoulder (A) preoperatively, (B) at 6 months postoperation, and (C) at 7 years postoperation. Graft incorporation of the bone block allograft is seen. Observe the remodeling of the inferior acromion and the mild glenohumeral arthritis at 7 years postoperation. (D) Ultrasound image of the same patient (patient 2) shows supraspinatus healing with a tendon width of 6 mm (purple line). White arrow, humeral head; red arrow, artifacts related to the metallic screw.
Discussion
The primary finding of the current study is that an ATBA can result in functional improvement in patients with a massive rotator cuff tear and greater tuberosity insufficiency. The combination of these 2 pathologies is an extremely difficult situation to manage, and after the ATBA procedure, only 1 of 5 patients went on to require RSA. The ATBA may thus be a joint preservation option in young patients with bone loss and reparable massive rotator cuff tears without concurrent glenohumeral arthritis.
As observed in the current study, massive rotator cuff tears with greater tuberosity insufficiency often present with pseudoparalysis. Despite an adequate rotator cuff repair, there is concern that patients will still have suboptimal clinical outcomes, as the greater tuberosity bone loss results in a loss of the “deltoid-wrapping” effect. 33 This effect is a growing concept in shoulder arthroplasty, which states that a loss of the deltoid wrapping angle subsequently reduces the tension within the deltoid. 33 While there is no current literature on the deltoid-wrapping effect in cases of native humeral head bone loss, the same principle may also apply. Without a greater tuberosity contour, there is a loss of the deltoid-wrapping effect over the native humeral head. This results in a poorer deltoid tension and subsequently poorer function. Surgeons should thus correct the greater tuberosity bone loss in cases of rotator cuff repair. Notably, the ATBA effectively reversed pseudoparalysis/pseudoparesis in 4 of our 5 patients, which provides support for restoring the bone in addition to the tendon alone. 11
Some surgeons advocate primary bone grafting of the greater tuberosity bony defect where possible. 2,23,27 Impaction bone grafting is an option that has been described, which potentially allows for secure placement of suture anchors within bony defects of the greater tuberosity. 19,27 However, impaction bone grafting is best performed with contained defects. In massive or uncontained defects, this option may not be viable. Furthermore, despite the initial security of the suture anchors within the grafted material, there still remains some risk of suture anchor pullout, particularly if the graft material does not incorporate into the native bone. In addition, the bone loss itself may represent a significant factor in poor healing of the tendon. 13 Another potential option would be to perform a staged procedure. There are several potential disadvantages. First, patients may not be keen on staged procedures, and while awaiting the second-stage procedure, patients will continue to have symptoms. Second, there is a risk of failure of graft incorporation. If the graft does not incorporate, an alternative surgical plan will have to be considered. Finally, in cases of delayed incorporation, the consequent delay in the second-stage surgery may result in irreversible fatty infiltration into the tendon, which in turn increases the failure risk of rotator cuff retear. Given these reasons, the preference for both the surgeon and patient should be to address the greater tuberosity bone loss in a single-stage setting. Using a calcaneal bone allograft shaped to fit the proximal humeral bone loss is effective and requires minimal implants for fixation. Our series showed that 4 of 5 patients had allograft bony union, with the 1 case of nonunion attributed to progressive humeral head osteonecrosis from the primary injury.
In terms of rotator cuff repair, the authors utilized the soft tissue portion of the ATBS as an augmentation to the rotator cuff repair. Our positive findings are consistent with previous reports of rotator cuff repair reinforced with allograft. 1,3,4,34 Agrawal 1 performed revision rotator cuff repair with acellular dermal allograft augmentation. He showed that after 1 year of follow-up, MRI evaluations revealed that 85% of the rotator cuffs were intact, with significant improvement in preoperative Constant scores. These findings were replicated in a randomized controlled trial by Barber et al, 4 who compared acellular dermal allograft augmentation versus none in single-row repairs of large, 2-tendon tears. There were significantly improved American Shoulder and Elbow Surgeons (ASES) and Constant scores in the augmented group at a mean follow-up of 2 years.
Previous studies of Achilles tendon allograft for irreparable rotator cuff tears have used it primarily for the soft tissue tendon, with the added advantage of bone-to-bone healing. Moore et al 28 described the usage of various allografts for massive irreparable rotator cuff tears in 32 patients, 5 of whom received an ATBA. In these patients, there was no concomitant bone loss, but a bone shelf in the intact greater tuberosity was created for the ATBA to achieve bone-to-bone healing. The allograft tendon was sewn medially to the native rotator cuff tissue when it was deemed irreparable. Overall, the authors did not find this method of treatment significantly better than those reported for subacromial decompression, acromioplasty, and rotator cuff debridement alone. This led them to conclude that allograft reconstruction for massive, irreparable rotator cuff tears was not recommended. There are also studies that have utilized an ATBA for SCR in irreparable rotator cuff tears. 15,16,24 Kholinne et al 15 published a case series of 6 patients with massive irreparable rotator cuff tears in which an ATBA was used for SCR. There was a high graft failure rate at 1 year (83%), and only 50% of the patients had improvements in the ASES scores that met the minimal clinically important difference of 21.0 points. The failures in the graft occurred in the midsubstance, and the side-to-side repair of the graft to the remnant native tissue had a high failure rate. Despite using similar ATBAs in the current study, we observed more positive clinical outcomes. This difference is likely due to the repair of the native rotator cuff. In our series, all patients had repairable massive rotator cuff tears without significant atrophy or fatty infiltration. As such, we were able to employ our technique, in which we utilized the graft as an augmentation to a rotator cuff repair, with a large surface of contact between the native tendon and the allograft. This technique differs substantially from those of Kholinne et al, 15 who utilized the allograft tendon as an SCR, and Moore et al, 28 who sewed the allograft to the ends of the native rotator cuff tissue. The rotator cuff repair may explain the improved clinical outcomes, as well as the graft integration noted on histology. In fact, Mihata et al 26 published a series of patients in whom a fascia lata SCR was used to reinforce rotator cuff repair. Their results showed that a graft SCR reinforcement prevented retear 1 year after arthroscopic rotator cuff repair and improved the quality of the tendon on MRI scans. Therefore, when utilizing an ATBA for massive rotator cuff tears, surgeons should still attempt to perform an adequate rotator cuff repair.
Limitations
The main limitation of this study is the small number of patients. However, to the best of our knowledge, this is the first study that provides at least 2 years of postoperative clinical and radiological outcomes for this procedure. More biomechanical studies should be performed to further detail the exact biomechanical benefits of restoring bony contour of the greater tuberosity.
Conclusion
In patients with massive rotator cuff tears and a greater tuberosity bony deficiency, utilizing an ATBA to restore the bony defect and reinforce the rotator cuff repair was safe and effective. At a minimum of 2 years postprocedure, most patients demonstrated improved clinical outcomes, tendon healing, and graft incorporation.
A Video Supplement for this article is available at http://journals.sagepub.com/doi/suppl/10.1177/2325967121990629.
Footnotes
Final revision submitted November 2, 2021; accepted November 8, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding was received from FORE (Foundation for Research and Teaching in Orthopaedics, Sports Medicine, Trauma and Imaging in the Musculoskeletal System; grant No. FORE 2021-18). P.J.D. has received education payments from Steelhead Surgical; consulting fees from Arthrex, Integer Holdings, and Pacira Pharmaceuticals; royalties from Arthrex; and compensation for other services from Arthrex and Steelhead Surgical. P.C. has received personal fees from Advanced Medical Applications, ConMed, Smith & Nephew, Stortz, and Stryker. A.L. has received grants from Medacta; has received personal fees from Arthrex, Medacta, and Stryker; and is the founder of BeeMed and the president of FORE. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Ethics Commission of the Canton of Geneva (AMG 12-26).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
