Abstract
Background:
Arthroscopic capsular release is an effective treatment for refractory shoulder stiffness, yet there are no basic studies that can explain the extent of the release.
Purpose:
This study aimed to compare the genetic expression of inflammation- and fibrosis-related factors between the anterior and posterior capsules in patients with shoulder stiffness and rotator cuff tear.
Study Design:
Descriptive laboratory study.
Methods:
Enrolled in this study were 35 patients who underwent arthroscopic capsular release for shoulder stiffness along with the rotator cuff repair. Anterior and posterior glenohumeral joint capsular tissues were obtained during the capsular release. For the control tissue, anterior capsule was obtained from 40 patients without stiffness who underwent arthroscopic rotator cuff repair. The gene expression of collagen types I and III, fibronectin, extracellular matrix, basic fibroblast growth factor, transforming growth factor–β, connective tissue growth factor, matrix metalloproteinase (MMP)-1, MMP-2, MMP-9, tissue inhibitor of metalloproteinase (TIMP)-1, TIMP-2, intercellular adhesion molecule 1, interleukin 1, and tumor necrotizing factor–α were analyzed using real-time reverse transcription polymerase chain reaction. Differences in gene expression between the anterior capsule, the posterior capsule, and the control tissue were compared with the Kruskal-Wallis test.
Results:
The expression levels of collagen types I and III were significantly higher in the anterior capsule with stiffness com (pared with both the posterior capsule with stiffness (P = .010 for both) and the control (P = .038 and .010, respectively). The levels of fibronectin, MMP-2, and MMP-9 in the anterior capsule were significantly higher than in both the posterior capsule (P = .013, .003, and .006, respectively) and the control (P = .014, .003, and .005, respectively).
Conclusion:
Genetic analysis of the shoulder capsule revealed that more fibrogenic processes occur in the anterior capsule compared with the posterior capsule in patients with shoulder stiffness.
Clinical Relevance:
Capsular release for shoulder stiffness should be more focused on the anterior capsule than on the posterior capsule.
Shoulder stiffness is a common pathological condition that manifests as a restriction of both passive and active shoulder motions. Even though nonoperative treatment is recommended as the first-line modality, surgical treatment is recommended for those who do not respond to conservative treatment. Among many surgical procedures, arthroscopic release of the joint capsule has shown promising results, especially in patients with refractory shoulder stiffness. 13,34,42 –45 In patients diagnosed with rotator cuff tear with concomitant stiffness, simultaneous arthroscopic capsular release can be conducted in tandem with the rotator cuff repair.
Many studies have reported that surgical release of the rotator interval tissue, including the anterior capsule, increases flexion and external rotation ranges. 14,28,33 However, there have been many controversies regarding the usage of extended posterior capsular release. Several authors have recommended posterior capsular release for the improvement of internal rotation. 19,30 On the other hand, according to Snow et al 39 and Chen et al, 5 additional posterior capsular release was not associated with any significant difference in outcomes compared with anterior capsular release.
Several studies have revealed that shoulder stiffness involves both inflammatory and fibrotic processes. 2,37 The pathophysiology includes synovial inflammation, subsynovial fibrosis, and subsequent capsular fibrosis and thickening, collectively leading to glenohumeral joint contracture. 37 Extensive analysis of cytogenetic factors involved in the pathology of shoulder stiffness has led to the publication of many notable results. The levels of inflammatory cytokines, such as interleukin (IL)-6 and IL-1β, as well as fibrogenic cytokines, such as the matrix metalloproteinases (MMPs) and their natural inhibitor, tissue inhibitor of metalloproteinase (TIMP), are increased in the synovia of these patients. 3,22 Platelet-derived growth factor and basic fibroblast growth factor (bFGF) are also known to be involved in shoulder stiffness inflammatory and fibrotic processes. 3,37
Rotator cuff tears are one of the most common conditions related to inflammation of the shoulder leading to joint stiffness. The rate of concomitant stiffness is high owing to the persistent pain or capsular contracture that occurs gradually over time. 7,41 In fact, the prevalence of shoulder stiffness was reported to be from 12.3% to 41.7% of all rotator cuff tears. 6,15,16,31,41 Many factors can lead to this shoulder stiffness in patients with rotator cuff tears. 7 The pain from the torn tendon may result in joint disuse, whereas contracture of the joint capsule or secondary muscular weakness may facilitate joint stiffness. In addition, secondary adhesive capsulitis, which is precipitated by inflammation from the rotator cuff tear, can contribute to shoulder stiffness. 41 For the treatment of rotator cuff tear with shoulder stiffness, successful results on simultaneous treatment of rotator cuff tear and capsular release for shoulder stiffness has been reported in several studies. 7,8,25 In fact, simultaneous rotator cuff repair and capsular release is commonly practiced in clinical settings, whereas isolated capsular release is performed restrictively, with relatively narrow indications.
This study aimed to compare the genetic expression of inflammation- and fibrosis-related factors between the anterior and posterior capsules in patients with shoulder stiffness and rotator cuff tear. We hypothesized that there will be overall differences in the expression of inflammation- and fibrosis-related cytokines between the anterior and posterior capsules, with a significant increase in expression when compared with control tissue.
Methods
Inclusion and Exclusion Criteria
Between January 2015 and March 2017, a total of 35 consecutive patients with rotator cuff tears and concomitant shoulder stiffness were prospectively enrolled. Patients were required to have had more than 3 months of shoulder stiffness and pain, without a history of intra-articular injection. We used the following criteria for stiffness: forward flexion less than 100° (maximum 150°; forward flexion was defined as glenohumeral motion without scapulohumeral rhythm 20 ), external rotation less than 30°, and internal rotation behind the back at a level equal to or lower than the fifth lumbar spine junction. Passive range of motion (ROM) was checked twice, before and after the general anesthesia prior to the surgical procedure, to rule out subjective limitation of joint motion owing to pain. 24 –26 We did not evaluate the contralateral shoulder for ROM as a comparison.
The size of the rotator cuff tear was limited to small to medium; only full-thickness tears were included. Tear size was classified based on greatest dimension as either small (<1 cm) or medium (1-3 cm). 10 Plain radiographs of the shoulder (true anteroposterior, supraspinatus outlet, and axial view) and shoulder magnetic resonance images were preoperatively evaluated in all patients. We excluded patients with concomitant glenohumeral lesions (eg, Bankart lesions or superior labrum anterior to posterior lesions), arthritic changes, and large or massive rotator cuff tears (tear size larger than 3 cm, or more than 2 concomitant tendon tears). Large or massive tears were excluded because they tend to have more functional deficit that can be misinterpreted as joint stiffness. Patients with a history of operation or fracture and bilateral stiffness were excluded from the study, as were patients older than 70 years to avoid restriction of joint motion as a natural aging process together with capsular changes. To minimize the intervention of confounding factors that are known to be related to pathogenesis of shoulder stiffness, patients with diabetes mellitus, thyroid problems, and a history of treatment for shoulder stiffness prior to surgery were excluded. For the control group, 40 rotator cuff tear patients without shoulder stiffness were enrolled. The same inclusion criteria were applied to the control group, except for the ROM. Patient assessment was conducted by one of the authors (H.S.P.)of this study. This study was approved by the institutional review board of our hospital.
Capsular Tissue Harvesting Technique
All surgical procedures were performed by 1 senior shoulder surgeon (Y.S.K.) with the patients under general anesthesia. The patients underwent standard glenohumeral arthroscopy in the lateral decubitus position. Anterior and posterior capsular tissues were obtained during the procedure of capsulectomy from the patients with stiffness. Anterior capsular release began below the bicep origin, preserving the glenoid labrum. Without releasing the subscapularis tendon, capsular release was carried out down to the 7 o’clock position and involved both the anterior and the posterior bands of the inferior glenohumeral ligament. From the posterosuperior recess, posterior capsular release was carried out down to the 7 o’clock position, where it met with the previous anteroinferior capsular release. Glenohumeral joint capsular tissue was obtained intraoperatively from the anterior (4 to 5 o’clock position) and posterior capsules (8 to 10 o’clock position) during the process of release. For the control group, tissue from the anterior capsule just underneath the middle glenohumeral ligament was obtained without actual release of the capsule. Tissue from the posterior capsule was not obtained for the control group.
Gene Expression Evaluation of Human Extracellular MMP, Collagens, and Growth Factors in the Capsular Tissues
The obtained capsular tissues were stored in RNAlater solution (Applied Biosystems/Ambion) at –20°C. Total RNA was extracted using a RNeasy Fibrous Tissue Mini Kit (QIAGEN); the RNA purification was performed with the 260-nm/280-nm light absorption ratio set at more than 1.7. Collagens I and III; MMP-1, MMP-2 and MMP-9; TIMP-1 and TIMP-2; extracellular matrix (ECM); fibronectin; and intercellular adhesion molecule (ICAM-1) were analyzed to assess ECM gene expression. In addition, bFGF, transforming growth factor–beta (TGF-β), connective tissue growth factor (CTGF), IL-1, and tumor necrosis factor (TNF)-α were evaluated.
Quantitative real-time reverse transcriptase polymerase chain reaction (PCR) was performed using the iTaq Universal SYBR Green Supermix (Bio-Rad Laboratories) and the cDNA Synthesis Kit-iScript cDNA Synthesis Kit (Bio-Rad Laboratories) on a CFX96 Real-Time Detection System (Bio-Rad Laboratories). The final solution was used directly for PCR amplification. Each cDNA reaction was diluted 5 times, along with a calibrator sample containing the transcript of interest and run in 25 µL of Bio-Rad SYBR Green using 10 pmol of primers designed with Primer 3 software (http://frodo.wi.mit.edu/primer3/bioinfo.ut.ee/primer3-0.4.0/). The PCR program consisted of 1 initial denaturation step (3 minutes at 94ºC) and 40 cycles of denaturation (10 seconds at 95ºC), annealing (20 seconds at a temperature optimal for each gene), and amplification (30 seconds at 72ºC), followed by melt curve determination consisting of 1 denaturation step (10 seconds at 95ºC) and annealing (1 step of 0.5 seconds at 65ºC). A calibrator reverse transcription (RT) sample containing the cDNA of interest was run to obtain a standard curve for each primer set; individual primer reaction efficiencies were calculated from this curve using CFXmanager software. Calculation of relative mRNA levels was performed with CFXmanager software according to the 2–▵▵CT method. 36 Values obtained for each gene were normalized to the β2 microglobulin housekeeping gene. Gene-specific oligonucleotide sequences are listed in Table 1.
Gene-Specific Primer Sequences a

a bFGF, basic fibroblast growth factor; CTGF, connective tissue growth factor; ECM, extracellular matrix; ICAM-1, intracellular adhesion molecule 1; IL-1, interleukin 1; MMP, matrix metalloproteinase; TGF-β, tumor growth factor beta; TIMP, TIMP metallopeptidase inhibitor; TNF-α, tumor necrosis factor alpha.
Immunohistochemistry
Hematoxylin-eosin staining and immunohistochemical staining were performed for the histological assessment of capsular tissue. After overnight fixing with 10% buffered formalin, the sample was washed and dehydrated through a graded series of alcohol. The specimen was embedded in paraffin and cut into 4-µm thickness for the attachment to the slide. It was treated with Dako REAL Peroxidase-Blocking Solution (DAKO) for 15 minutes, 0.1% Triton x-100 solution for 10 minutes, and 10% normal goat serum (Vector Laboratories) for 20 minutes. The primary antibody mouse monoclonal antihuman type I and III collagen, fibronectin, and MMP-2 and MMP-9 were incubated overnight at 4°C. After washing with phosphate-buffered saline, biotinylated secondary antibody (Vectastain Elite ABC Kit; Vector Laboratories) was applied for 20 minutes. To catalyze chromogen development in 3,3′-diaminobenzidine tetrachloride, streptavidin conjugated peroxidase was applied. The stained sections were examined, and the distribution of type I and III collagen, fibronectin, MMP-2, and MMP-9 was determined under optical microscopy after counterstaining with hematoxylin-eosin.
Statistical Methods
Differences between gene expressions in the anterior and posterior capsules and the control tissue were compared with the Kruskal-Wallis test. Statistical analysis was performed using SPSS, Version 12.0 (IBM). P values less than .05 were considered statistically significant.
Results
The demographic data of patients are listed in Table 2. The gene expression levels of collagen types I and III in the anterior capsule of patients with stiffness were significantly higher than those in both the posterior capsule in the patients with stiffness (P = .010 for both) and the control patients (P = .038 and .010, respectively) (Figure 1). We found that the expressions of fibronectin, ECM, TGF-β, MMP-2, MMP-9, ICAM-1, and IL-1 were higher in the anterior capsule compared with both the posterior capsule and the control tissue. In particular, the levels of fibronectin (P = .013), ECM (P = .036), MMP-2 (P = .003), and MMP-9 (P = .006) were significantly higher in the anterior capsule compared with the posterior capsule, and the levels of fibronectin, MMP-2 and MMP-9 were significantly higher in the anterior capsule compared with the control tissue (P = .014, P = .003271 and P = .005, respectively). The level of TNF-α was significantly lower in the anterior and posterior capsule compared with the control group (Figure 2). All the values for mRNA expression level are listed in Table 3.
Hematoxylin-eosin staining showed prominent inflammatory change with vascular proliferation in the anterior capsule when compared with the posterior capsule. Immunohistochemistry revealed increased type I and III collagen, fibronectin, and MMP-2 and MMP-9 expressions in the anterior capsule (Figure 3).
Demographic Data of Patients a

a ROM, range of motion.
b Internal rotation was based on the level of vertebrae and was numbered as 0 for any level below the sacral region, with 1 additional point for each level higher.
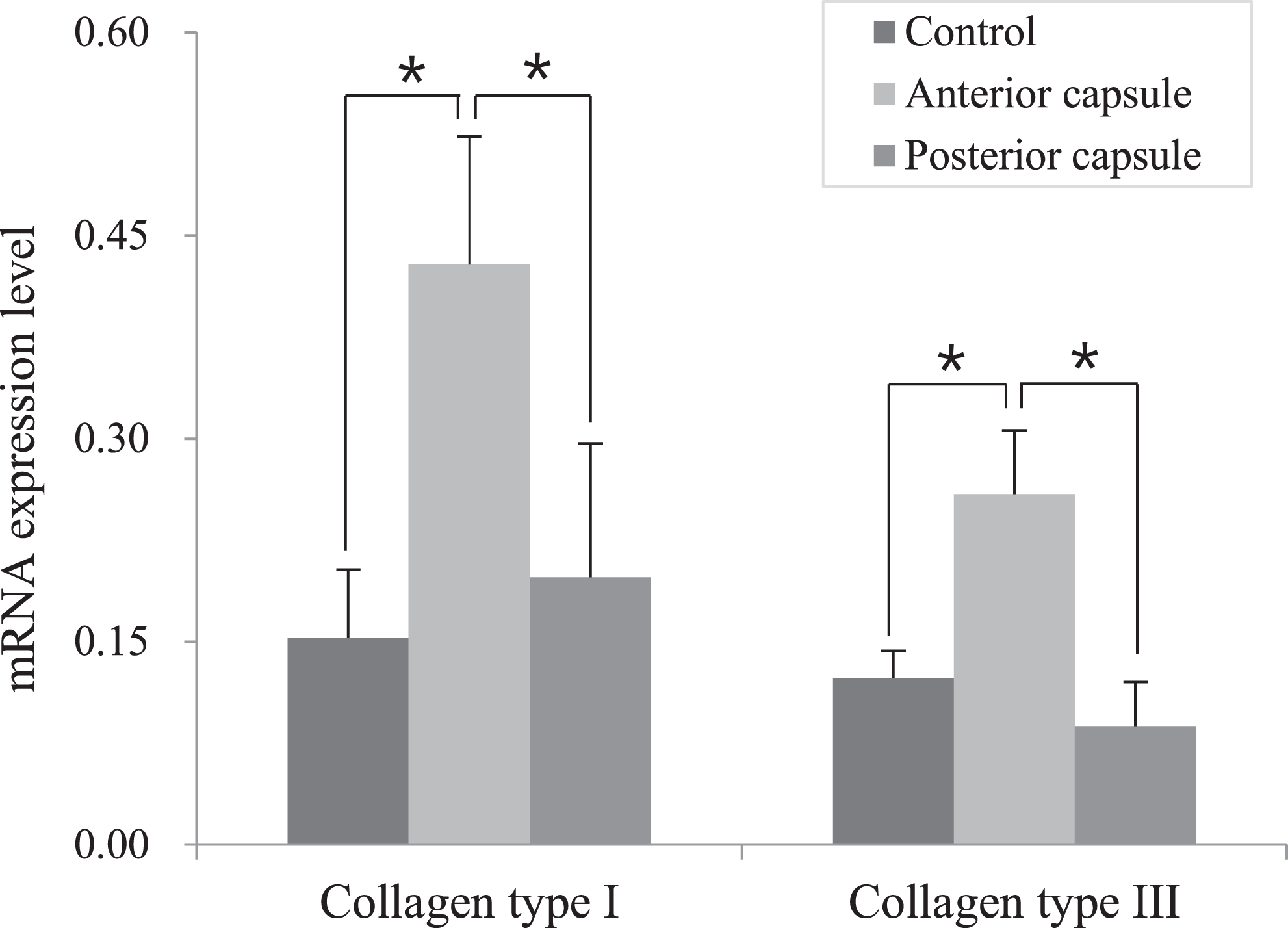
Gene expression levels of collagen types I and III. *Statistically significant difference (P < .05)
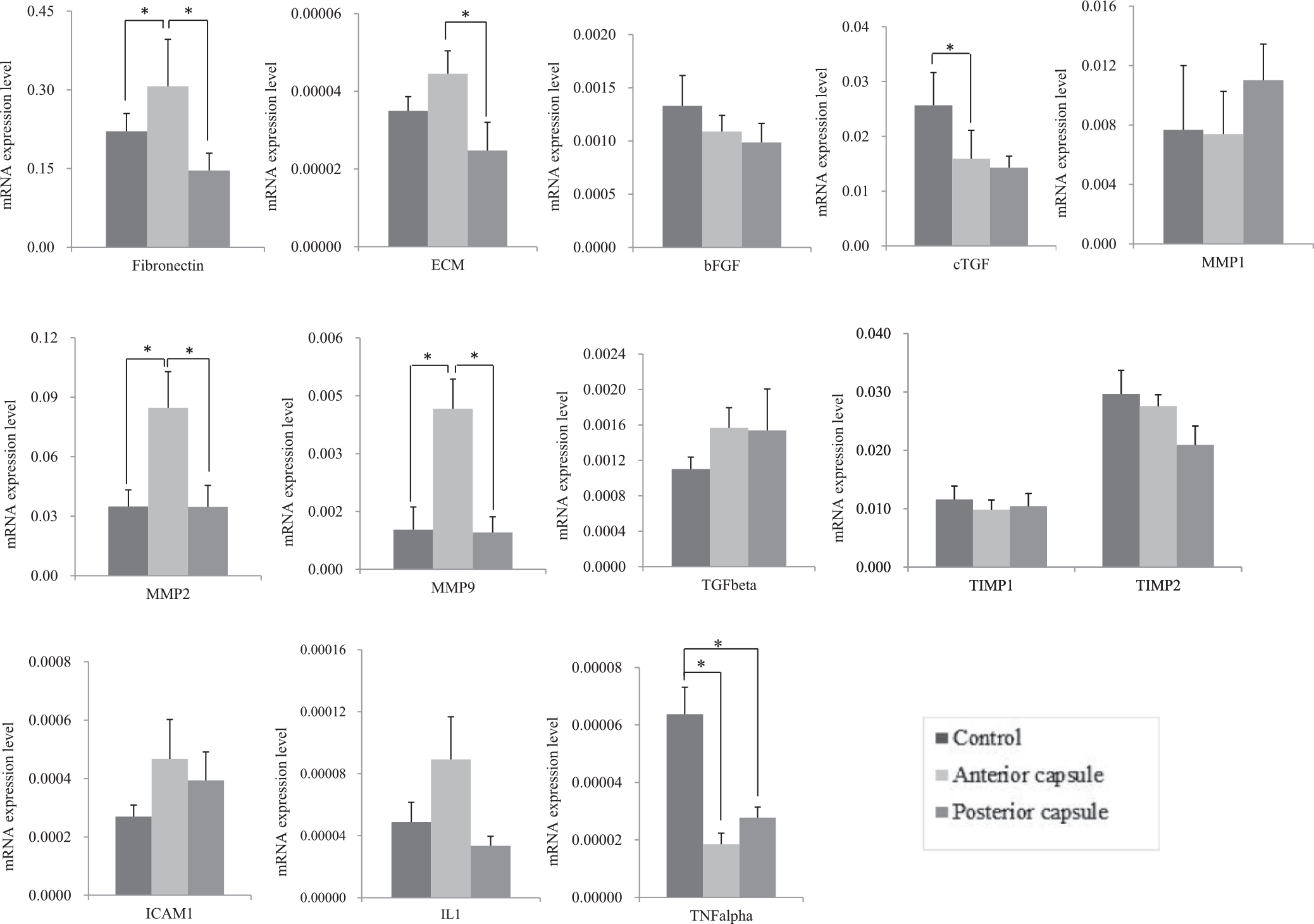
Gene expression levels of inflammatory, fibrogenic, and growth factors in the anterior and posterior capsules as well as control tissue. bFGF, basic fibroblast growth factor; CTGF, connective tissue growth factor; ECM, extracellular matrix; ICAM, intercellular adhesion molecule; IL, interleukin; MMP, matrix metalloproteinase; TGF, transforming growth factor; TIMP, tissue inhibitor of metalloproteinase; TNF, tumor necrosis factor. *Statistically significant difference (P < .05).
mRNA Levels of Evaluated Factors a

a Data are reported as mean ± SD. bFGF, basic fibroblast growth factor; CTGF, connective tissue growth factor; ECM, extracellular matrix; ICAM-1, intercellular adhesion molecule 1; IL-1, interleukin 1; MMP, matrix metalloproteinase; TGF- β, transforming growth factor beta; TIMP, tissue inhibitor of metalloproteinase; TNF-α, tumor necrosis factor alpha.
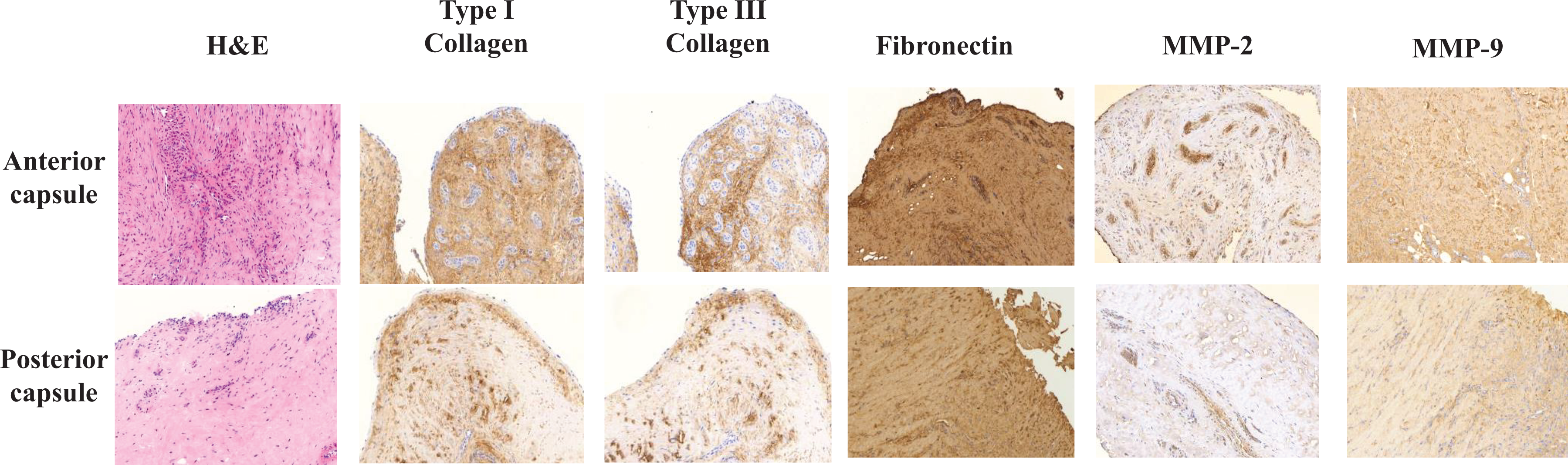
Collagen types I and III, fibronectin, and matrix metalloproteinase (MMP)-2 and MMP-9 expressions in the anterior and posterior capsule of stiff shoulder assessed by an immunohistochemistry. The far-left panels show hematoxylin-eosin (H&E) staining of the capsule (×100 magnification).
Discussion
This study revealed that the expression levels of collagens and genes encoding inflammation- and fibrosis-related factors were higher in the anterior capsules of patients with rotator cuff tear and shoulder stiffness compared with the posterior capsules and with the anterior capsules of patients without shoulder stiffness.
It has been clinically shown that the glenohumeral capsule is the primary pathological site for shoulder stiffness. 12,13,34,38,43,45 However, as different areas of the shoulder joint have been implicated in stiffness, the extent of capsular release remains controversial, especially regarding the significance of posterior capsular release. Some studies have advocated for additional posterior capsular release or circumferential release around the joint capsule for the recovery of internal rotation. 21,44 On the other hand, recent studies have reported that including additional posterior capsular release does not provide actual clinical benefit in terms of gaining ROM. 26,30,39
The sustained production of cytokines involved in tissue fibrosis is known to represent a persistent fibrogenic stimulus, resulting in capsular hypertrophy. 1,3 Modulation by the abnormal production of cytokines and growth factors may lead to the formation of a matrix of collagen populated by fibroblasts and myofibroblasts. 3 In this study, we attempted to verify the extent of capsular release in shoulder stiffness patients by elucidating and comparing the expression of inflammation- and fibrosis-related factors in the anterior and posterior capsules. To objectify the results, anterior capsular tissues from shoulders without stiffness were included for comparison. Only after including a nonpathologic group can we definitively conclude that shoulder stiffness is associated with differential pathologic gene expression.
Among the many cytokines and factors related to fibrosis and inflammation, we selected a few well-validated factors associated with shoulder stiffness in previous studies. In the case of collagen, our results were consistent with previous studies 27,40 showing higher expression in the anterior capsule of patients with shoulder stiffness compared with the posterior capsule or with control tissue. Fibronectin is a high–molecular weight glycoprotein of the ECM that binds to integrins. 32 An elevated level of fibronectin was found in the joint synovium/capsule of patients with idiopathic adhesive capsulitis. 9 High levels of MMPs and MMP inhibitors (TIMPs) have been found in the capsular tissue of frozen shoulders. 17,22 MMPs are protease enzymes that are known to have a critical role in regulating the remodeling of fibroblast-produced ECM. Consistent with previous studies, the levels of fibronectin, MMP-2, and MMP-9 were significantly higher in the anterior capsule when compared with the control tissue. Interestingly, the expressions of these factors were not high in the posterior capsules of the patients with shoulder stiffness, and the levels in the posterior capsules were not significantly different from those of the control tissue. Similar results were shown with ECM, which is also known to be involved in shoulder stiffness. 35 Even though the difference was not statistically significant, similar results showing higher expression in the anterior capsule were found for TGF-β, ICAM-1, and IL-1, which are known to be related to synovial hyperplasia and fibrosis. ICAM-1 is known for its role in the inflammatory response mediated by the immune system and the production of stimulatory signals for leukocyte activation and lymphocyte proliferation. 4 In a 2013 study, the expression of ICAM-1 was higher in the synovial fluid, capsule of the glenohumeral joint, and peripheral blood of patients with shoulder stiffness compared to control patients. 23
Among the analyzed factors, TNF-α and CTGF were the factors with unexpected results, showing higher expression in the control tissue compared with either the anterior or the posterior capsule. However, regarding the lower positive results obtained for TNF-α in a study by Bunker et al, 3 the involvement of TNF-α in the etiology of shoulder stiffness can be relatively weak compared with other factors. Similar to TNF-α, the expression of growth factors in shoulder stiffness, especially that of CTGF, has not shown consistent results in previous studies. 3,11 The expression level of growth factors can be high in capsules with synovial hypertrophy, which are often found in patients without shoulder stiffness. In these cases, the capsules can be in a stage of pre- or active inflammation that does not necessarily lead to joint limitation. Regarding the relationship between growth factors and active inflammation, the expression of growth factors would not be prominent in patients with refractory stiffness involved with rotator cuff tear, which belongs to the phase of fibrosis.
To the best of our knowledge, biological- or molecular-level evidence that could justify the extent of capsular release has not yet been provided in any published studies. The fact that collagens and fibrosis-related factors are more abundant in the anterior capsule suggests that more extensive fibrosis may occur in the anterior capsule than in the posterior capsule, even with patients having definite restriction in internal rotation. This could explain why limited anterior and inferior capsular release, without posterior release, has shown clinically meaningful results for the treatment of refractory shoulder stiffness. 26
The patients enrolled in the current study were treated for both shoulder stiffness and rotator cuff tear. The choice of study population was a practical consideration because surgical repair was required for the treatment of the rotator cuff tears, and simultaneous capsular release is considered an effective treatment for rotator cuff tears with shoulder stiffness. 8,41 On the other hand, candidates for the surgical treatment of primary adhesive capsulitis of the shoulder are limited to patients with refractory stiffness that shows no response to nonoperative treatment for a certain amount of time. A finding of this study was that the inflammatory and fibrogenic process of the capsule, which is known to take place in primary adhesive capsulitis, also contributed to the pathogenesis of secondary stiffness with rotator cuff tear. Because the expression of certain factors related to fibrosis were significantly higher in the anterior capsule, whereas no difference was shown between the control tissue and the posterior capsule, we can conclude that anterior capsular release is important for the surgical treatment of shoulder stiffness with rotator cuff tear.
There are some limitations to this study. First, the number of enrolled patients was relatively small, and power analysis was not performed. Perhaps for this reason, the results among some of the related factors, including MMP-1 and the TIMPs, were not as consistent as expected. Further studies with larger sample sizes based on the results of power analysis could provide a more accurate representation of the population being tested. Second, treating rotator cuff tears with concomitant stiffness can be paradoxical. The actual repair of the rotator cuff tear may lead to the progression of stiffness, because the repair is a joint tightening procedure and, again, postoperative immobilization is important for healing the repaired tendon. 18,29,46 In addition, we could not guarantee that all the enrolled patients were at the same stage of shoulder stiffness. According to Hannafin and Chiaia, 12 there are 5 stages of adhesive capsulitis, and patients in different stages may also have various genetic features within the capsule. However, capsular release conducted with rotator cuff repair not only treats isolated stiffness but also allows a rapid recovery time and improvements in ROM. 8 The results of this study may contribute to clinical practice by providing basic information. Third, even though the control group was selected from among patients with no limitation in shoulder motion, the accompanying rotator cuff tear might have affected the inflammation and expression of cytokines in the glenohumeral joint capsule. From a practical standpoint however, recruitment of tissue from a control group without any existence of pathology seems impossible. Fourth, only tissue from the anterior capsule was obtained from the control sample, and not any from the posterior capsule. It would be of interest to see how control tissue from the posterior capsule contrasts with the tissues affected by stiffness. However, the existence and gene expression of the posterior capsule would not affect the results of this study, as significant differences were shown in both the anterior and the posterior capsule in patients with shoulder stiffness. Last, gene expression only describes what genes are being expressed at the time of harvest, and the progression toward pathology cannot be determined. Additional protein-level analyses or biochemistry would give more objective support to the results of the study.
Conclusion
In patients with shoulder stiffness with rotator cuff tear, a more intense fibrogenic process occurs in the anterior capsule compared with posterior capsule tissues.
Footnotes
Final revision submitted March 7, 2021; accepted March 24, 2021.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Seoul St. Mary’s Hospital, The Catholic University of Korea (study No. KC12OISI0532).
