Abstract
Background:
Platelet-rich plasma (PRP) has gained attention as a therapeutic option for knee osteoarthritis; however, its efficacy varies widely. Leukocytes in PRP raise the concern of aggravating proinflammatory activity. To date, PRP has rarely been investigated with regard to leukocyte concentration.
Purpose:
To provide clinical evidence of the intra-articular injection of PRPs containing different leukocyte concentrations.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
We systematically searched the MEDLINE, Embase, Cochrane Library, CINAHL, and Scopus databases. PRP was classified into leukocyte-poor (LP-PRP) and leukocyte-rich (LR-PRP). Clinical outcomes including Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), visual analog scale (VAS) for pain score, International Knee Documentation Committee (IKDC) subjective score, and adverse reactions were evaluated. The Methodological Index for Non-Randomized Studies criteria were used for quality assessment.
Results:
Included were 32 studies with an evidence level between 1 and 4. Both LP-PRP and LR-PRP showed improvements above the minimal clinically important difference (MCID) in VAS pain score. No significant intergroup difference was seen at 3, 6, or 12 months of follow-up. Regarding function, both LP-PRP and LR-PRP showed improvements above the MCID in the WOMAC and IKDC scores, with no significant difference between the groups. Adverse reactions for pain were significantly higher in LR-PRP than in LP-PRP (odds ratio, 1.64; 95% confidence interval, 1.29-2.10; P = .01). After intra-articular PRP injection, LR-PRP showed a significantly higher rate of swelling than LP-PRP (odds ratio, 1.56; 95% confidence interval, 1.22-1.99; P = .02). The mean Methodological Index for Non-Randomized Studies score of the included studies was 18.6 (range, 10-24).
Conclusion:
Intra-articular PRP injection resulted in improvements above the MCID in terms of pain and function in patients with knee osteoarthritis up to 12 months. The risk of local adverse reactions appeared to be increased after LR-PRP compared with LP-PRP injection. The findings of this review can support the potential use of intra-articular PRP injection for the treatment of knee osteoarthritis. In clinical application, clinicians need to consider selecting a specific type of PRP for knee osteoarthritis.
Platelet-rich plasma (PRP) has gained attention as an alternative biological treatment option because it contains several anabolic and related factors. 55 Several in vitro and in vivo studies have shown beneficial effects of PRP, including an improved healing process, 18,34,43,46,52 anti-inflammatory and analgesic effects, 1,3,73 and chondropromoting and chondroprotective effects. 4,20,40,42 Despite increasing information regarding PRP and its use in the clinical setting for the treatment of knee osteoarthritis (OA), its efficacy remains controversial. 8,15,77
The presence of leukocytes in PRP raises a concern due to their well-known proinflammatory activity. 45 Some in vitro studies have reported that a high leukocyte concentration within PRP could increase the expression of catabolic cascades and inflammatory markers such as interleukin 1 (IL-1) and tumor necrosis factor α. 5,47,48 Similar results have been shown in in vivo animal studies. 18,49 In this regard, some studies have suggested that leukocyte-poor PRP (LP-PRP) would be more suitable for the treatment of knee OA. In contrast, despite the increase of proinflammatory markers in leukocyte-rich PRP (LR-PRP), some in vitro studies have shown that LR-PRP can provide beneficial effects to knee OA via the interaction between platelets and neutrophils. Platelets in association with neutrophils can interfere with the conversion of leukotrienes into lipoxin, thereby promoting the resolution phase of the healing cascade. 37,56 In addition, 1 study reported the production of large amounts of vascular endothelial growth factor from platelets by neutrophils. 17 Moreover, a 5-fold increase in the anti-inflammatory markers IL-4 and IL-10 has been reported. 2 Despite concomitant anabolic and catabolic effects of leukocytes, the effects of their concentrations in PRP on clinical outcomes have not been well investigated.
Therefore, we sought to provide evidence about the clinical efficacy and adverse reactions of intra-articular injection of PRP in patients with knee OA based on leukocyte concentration, which was assessed meticulously for categorization into LP-PRP and LR-PRP.
Methods
This systematic review and meta-analysis was designed based on the Cochrane Review Methods and performed according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 50 The study protocol was registered with the International Prospective Register of Systematic Reviews (registration No. CRD42020158791).
Literature Search
A systematic literature search was performed of the PubMed (MEDLINE), Embase, Cochrane Library, CINAHL, and Scopus databases up to September 1, 2019. No restrictions were placed on language or year of publication. A combination of the following keywords was used in the title, abstract, Medical Subject Headings, and keyword fields: “knee,” “osteoarthritis,” “platelet-rich plasma,” and “leukocyte.” The research question and inclusion criteria were established a priori. Manual searches were performed for articles that could have been missed in the electronic search. The bibliographies of the initially retrieved studies were cross-checked to identify additional relevant articles. Two investigators (Y-B.P. and J-H.K.) independently screened the abstracts and titles of the retrieved studies; those that met the inclusion criteria were subjected to full-text review. Any disagreements between the 2 reviewers were resolved via discussion.
Study Selection
The following inclusion criteria were used: (1) all levels of evidence; (2) cohort of patients diagnosed with knee OA; (3) intervention consisting of intra-articular injection of PRP; (3) comparison of LP-PRP versus LR-PRP; (4) outcomes including patient-reported outcome measures (PROMs) and adverse reactions; and (5) a full report of parameters, including means, standard deviations (SDs), and sample numbers. We excluded studies not clearly reporting parameters; those not clearly reporting the follow-up period for clinical outcomes; animal, biomechanical, and cadaveric studies; technical notes; letters to the editor; expert opinions; review articles; meta-analyses; and scientific conference abstracts. In addition, if studies with similar data of different follow-up periods at the same institutions were found, previous studies with shorter follow-up were excluded to avoid duplicates.
Definitions of LP-PRP and LR-PRP
According to previous studies, 15,39,64 the PAW classification system (absolute number of Platelets, manner in which platelet Activation occurs, and presence or absence of White cells) was used to define LP-PRP and LR-PRP. LP-PRP was defined as PRP with a leukocyte concentration equal to or less than baseline of whole blood, whereas LR-PRP was defined as PRP with a leukocyte concentration greater than baseline of whole blood. 16 After thorough review of the methods section of each article, the leukocyte concentration in the final PRP product was identified. When insufficient information regarding leukocyte concentration was provided, the study authors were contacted, or the manufacturer documentation for the PRP system was reviewed to obtain detailed information about leukocyte concentration. Accordingly, all PRP preparations in the included studies could be categorized into LP-PRP or LR-PRP.
Data Extraction and Synthesis
Two investigators (Y-B.P. and J-H.K.) independently extracted data from each article using a predefined data extraction form. Any disagreements between the 2 reviewers were resolved via discussion. The following data were extracted: first author, year of publication, country, number of patients and knees, patient age, patient sex, patient body mass index, OA severity, follow-up period, main findings of each study, information about PRP preparation (preparation system, spinning frequency, and activation status), details of the interventions (dose, injection frequency, interval, and fresh or frozen), mean platelet and leukocyte concentration of the PRP injection, PROMs including means and SDs, and adverse reactions. For PROMs, the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), visual analog scale (VAS) for pain, International Knee Documentation Committee (IKDC) subjective score, Lysholm score, Knee injury and Osteoarthritis Outcome Score (KOOS), 12-Item Short From Healthy Survey (SF-12), 36-Item Short Form Health Survey (SF-36), Outcome Measurement for Rheumatology Committee and Osteoarthritis–Research Society International Standing Committee for Clinical Trials Response Criteria Initiative (OMERACT-OARSI), and Knee Society Scale (KSS) were aggregated from pooled studies; however, Lysholm score, KOOS, SF-36, OMERACT-OARSI, and KSS were excluded because of a lack of sufficient studies for meaningful analysis (<2 studies for each group). For adverse reactions, local and related reactions after the intra-articular injection such as pain and swelling were aggregated from pooled studies; however, systemic reactions and unrelated reactions were excluded because of heterogeneity.
Assessment of Methodologic Quality
Two investigators (Y-B.P. and J-H.K.) independently assessed the methodologic quality of each study using the Methodological Index for Non-Randomized Studies (MINORS) criteria. 69 The maximum score is 24 for comparative studies and 16 for noncomparative studies according to MINORS checklists. 69 Any discrepancies in scores between the 2 reviewers were resolved via discussion.
Statistical Analysis
The main outcomes of this meta-analysis were the mean differences (MDs) in improvement of PROMs and odds ratios (ORs) of adverse reactions between LP-PRP and LR-PRP intra-articular injection in patients with knee OA. In each study and for continuous outcome variables including WOMAC, VAS score, and IKDC subjective score, we calculated the treatment effect from the difference between the pre- and postintervention changes in mean and SD in the LP-PRP and LR-PRP groups for the reported follow-up periods. For continuous variables, we calculated MDs and 95% confidence intervals (CIs). For dichotomous outcome variables as adverse reactions, we calculated ORs with 95% CIs. Heterogeneity was determined by estimating the proportion of interstudy inconsistencies because of actual differences between studies rather than differences due to random error or chance using the I 2 statistic, where 25% was considered low heterogeneity, 50% was moderate heterogeneity, and 75% was high heterogeneity. 32
Random-effects meta-analysis was performed to pool the outcomes across the included studies. Forest plots were used to show outcomes, the pooled estimate of effect, and the overall summary effect of each study and were constructed using Open Meta-Analyst (Brown University; http://www.cebm.brown.edu/openmeta). Additional analyses were performed using Comprehensive Meta-Analysis software (Biostat) and R statistical software Version 3.4.0 (the metaphor Package: a Meta-Analysis Package for R; R Foundation for Statistical Computing). The standardized MD and standardized variance were calculated from the weighted estimates, standard errors, and sample size of each cohort using the logit method. 75,76 Summary ORs and 95% CIs were calculated based on the standardized MD and standardized variance (George Wilson University). Publication bias was not assessed in this study because it was not generally necessary if there were <10 studies in a comparison. Statistical significance was set at P < .05.
Results
Identification of Studies
vAn initial electronic search yielded 212 studies, and an additional 18 studies were identified through manual searching. After the removal of 116 duplicate studies, 114 remained. After screening of the titles and abstracts and reading of the full texts, 32 studies were finally included in this systematic review and meta-analysis. Details about study identification, inclusion, and exclusion criteria are shown in Figure 1.

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram for the identification and selection of studies included in this meta-analysis.
Study Characteristics and Methodologic Quality Assessment
Of the 32 identified studies, only 1 study 23 performed a direct comparison between LP-PRP and LR-PRP. A total of 22 studies # involving 1070 patients with 1162 knees evaluated LP-PRP results, whereas 11 studies ** involving 593 patients with 628 knees evaluated LR-PRP results. Details of the studies, including sample size, patient characteristics, OA severity, PROMs, follow-up period, results, and MINORS score, are presented in Appendix Table A1. This systematic review included the studies from level of evidence I to IV. Details about PRP preparation and injection protocol as well as platelet and leukocyte concentrations in the PRP injection are summarized in Appendix Table A2. The methodologic quality assessment using MINORS revealed that the pooled mean MINORS score of the LP-PRP group was 18.6 ± 4.6 (range, 11-24) including 16 comparative studies †† (median MINORS, 21.5; range, 17-24) and 6 noncomparative studies 7,11,24,28,30,61 (median MINORS, 11.5; range, 11-14), whereas the pooled mean MINORS score of the LR-PRP group was 18.6 ± 3.8 (range, 10-23) including 9 comparative studies ‡‡ (median MINORS, 20; range, 17-23) and 2 noncomparative studies 44,68 (median MINORS, 12; range, 10-14).
VAS Score
A total of 2 studies 35,67 of LP-PRP and 4 studies 21,38,58,71 of LR-PRP provided relevant data on VAS at 3 months. At this assessment point, the pooled MD of improvement was not significantly different between LP-PRP (MD, 40.82; 95% CI, 36.90-44.74) and LR-PRP (MD, 37.69; 95% CI, 24.98-50.40; P = .64). A total of 8 studies 6,7,12,23,24,30,35,57 of LP-PRP and 8 studies 21 –23,26,29,36,38,71 of LR-PRP provided data on VAS at 6 months, at which point the pooled MD of improvement at 6 months was not significantly different between LP-PRP (MD, 22.33; 95% CI, 12.79-31.87) and LR-PRP (MD, 23.85; 95% CI, 15.83-31.86; P = .81). A total of 5 studies 6,12,23,25,30 on LP-PRP and 4 studies 21 –23,38 on LR-PRP provided VAS results at 12 months. At this point, the pooled MD of improvement was not significantly different between LP-PRP (MD, 16.77; 95% CI, 10.82-22.71) and LR-PRP (MD, 28.92; 95% CI, 21.79-36.04; P = .06) (Figure 2).

Forest plots of studies showing improvements of visual analog scale (VAS) scores in patients with knee osteoarthritis after intra-articular injection of leukocyte-poor (LP) platelet-rich plasma (PRP) and leukocyte-rich (LR) PRP at (A) 3 months, (B) 6 months, and (C) 12 months. Squares represent the mean improvement in the VAS, with the size of the square being proportional to the sample size.
Total WOMAC Score
We found that 5 studies 10,11,41,67,70 on LP-PRP and 5 studies 21,38,44,68,71 on LR-PRP provided relevant data on total WOMAC score at 3 months. At this assessment point, the pooled MD of improvement was not significantly different between LP-PRP (MD, 24.05; 95% CI, 11.95-36.14) and LR-PRP (MD, 25.03; 95% CI, 12.44-37.63; P = .91). A total of 11 studies §§ on LP-PRP and 5 studies 21,38,44,68,71 on LR-PRP provided data at 6 months, showing that the pooled MD of improvement was not significantly different between LP-PRP (MD, 20.93; 95% CI, 16.46-25.40) and LR-PRP (MD, 20.73; 95% CI, 12.78-28.68; P = .97). A total of 6 studies 6,30,41,62,70,74 on LP-PRP and 3 studies 21,38,44 on LR-PRP provided WOMAC data at 12 months, at which point the pooled MD of improvement was not significantly different between LP-PRP (MD, 18.04; 95% CI, 10.61-25.47) and LR-PRP (MD, 18.18; 95% CI, 13.86-22.50; P = .97) (Figure 3).

Forest plots of studies showing the improvement of total Western Ontario and McMaster Universities Arthritis Index (WOMAC) score in patients with knee osteoarthritis after intra-articular injection of leukocyte-poor (LP) platelet-rich plasma (PRP) and leukocyte-rich (LR) PRP at (A) 3 months, (B) 6 months, and (C) 12 months. Squares represent the mean improvement in the total WOMAC score, with the size of the square being proportional to the sample size.
IKDC Subjective Score
A total of 4 studies 12,23,24,41 on LP-PRP and 4 studies 22,23,26,36 on LR-PRP provided relevant data on IKDC subjective score at 6 months. At this follow-up point, the pooled MD of improvement was not significantly different between LP-PRP (MD, 17.19; 95% CI, 14.04-20.33) and LR-PRP (MD, 16.93; 95% CI, 11.25-22.62; P = .94). A total 5 studies 12,23,24,31,41 on LP-PRP and 2 studies 22,23 on LR-PRP provided IKDC data at 12 months, at which point the pooled MD of improvement was not significantly different between LP-PRP (MD, 17.74; 95% CI, 10.61-24.88) and LR-PRP (MD, 15.70; 95% CI, 11.79-19.62; P = .62) (Figure 4).

Forest plots of studies showing the improvement of International Knee Documentation Committee (IKDC) subjective score in patients with knee osteoarthritis after intra-articular injection of leukocyte-poor (LP) platelet-rich plasma (PRP) and leukocyte-rich (LR) PRP at (A) 6 months and (B) 12 months. Squares represent the mean improvement in the IKDC subjective score, with the size of the square being proportional to the sample size.
Adverse Reaction
For pain after intra-articular PRP injection, a total of 14 studies ∥∥ on LP-PRP and 6 studies 23,29,36,58,68,71 on LR-PRP provided relevant data. The mean adverse reaction rates for pain were significantly higher for LR-PRP (0.152; 95% CI, 0.050-0.255) than LP-PRP (0.018; 95% CI, 0.007-0.029) (OR, 1.64; 95% CI, 1.29-2.10; P = .01). For swelling after intra-articular PRP injection, a total of 14 studies ∥∥ on LP-PRP and 6 studies 23,29,36,58,68,71 on LR-PRP were analyzed. The mean adverse reaction rates for swelling were significantly higher for LR-PRP (0.098; 95% CI, 0.027-0.169) than for LP-PRP (0.014; 95% CI, 0.003-0.024) (OR, 1.56; 95% CI, 1.22-1.99; P = .02) (Figure 5).

Forest plots of studies showing the adverse reaction rate after injection of leukocyte-poor (LR) platelet-rich plasma (PRP) and leukocyte-rich (LP) PRP in patients with knee osteoarthritis. Squares represent the mean adverse reaction rate in (A) pain and (B) swelling, with the size of the square being proportional to the sample size. Ev/Trt, event/total sample size.
Discussion
The most important findings of this study were that the intra-articular injection of LP-PRP showed lower adverse reactions than did LR-PRP in knee OA. Among the adverse reactions, pain and swelling after the intra-articular injection of PRP were significantly more common in the LR-PRP group. Although no significant difference was noted between LP-PRP and LR-PRP, pain was significantly improved after the intra-articular PRP injection. Furthermore, our meta-analysis found significant improvement in functional outcomes after intra-articular PRP injection regardless of leukocyte concentration. These results support the potential use of intra-articular PRP injections for the treatment of knee OA. These findings may also help clinicians select a specific PRP type for knee OA.
This meta-analysis revealed that intra-articular PRP injections induced significant pain improvement regardless of leukocyte concentration. The LP-PRP and LR-PRP groups of included studies showed improvement above the minimal clinically important difference (MCID) in pain, with a VAS score of 19.9 at 3 months without a study in LR-PRP group, 58 which showed below the MCID. 72 At the 6-month follow-up, 66.7% (6/9) of studies for LP-PRP 7,12,30,35,57 and 77.8% (7/9) of studies for LR-PRP 21 –23,26,36,38,71 showed improvement above the MCID in pain, with a VAS score of 19.9. 72 At the 12-month follow-up, however, only 42.9% (3/7) of studies for LP-PRP 23,25,30 showed an improvement above the MCID, whereas all (4/4) studies for LR-PRP 21 –23,38 showed an improvement above the MCID in pain with a VAS of 19.9. The MCID is known to be influenced by the initial pain severity. 72 The low baseline showed a low MCID for pain. In this review, the baseline VAS of LP-PRP (41 points) was lower than that of LR-PRP (56 points). Therefore, an improvement of <19.9 in the LP-PRP group at 12-month follow-up could indicate minimal clinical improvement. Three recent meta-analyses reported that PRP injections showed significant pain improvement in knee OA versus hyaluronic acid (HA) or placebo for 12 months of follow-up. 8,15,66 On the contrary, a recent meta-analysis reported a limited efficacy of PRP for pain reduction in knee OA. 77 Although VAS pain scores showed no significant differences at 3- and 6-month follow-up, WOMAC pain scores differed significantly at 12 months of follow-up. A previous meta-analysis that included only 4 studies evaluating VAS pain did not consider the MCID of the VAS pain score. 77 As mentioned, VAS pain scores in knee OA showed improvements above the MCID in this meta-analysis. With these points in mind, intra-articular PRP injections would be an option for pain relief in knee OA for 12 months.
The results of this review revealed that intra-articular PRP injections showed significant functional improvement in WOMAC and IKDC scores regardless of leukocyte concentration. Most included studies (WOMAC score 81.8% and 66.7% in LP-PRP, 100% and 66.7% in LR-PRP at 6 and 12 months, respectively) showed that the improvements in WOMAC at 6 and 12 months were greater than the MCID of 11.5. 72 WOMAC is a validated evaluation system for assessing pain and function in knee OA. 13,72 Although WOMAC pain, stiffness, and function scores were not evaluated separately, improvements in WOMAC total scores were relatively large (95% CI, 16.46-25.40 and 10.61-25.47 at 6 and 12 months, respectively, for LP-PRP; 12.78-28.68 and 11.79-19.62 at 6 and 12 months, respectively, for LR-PRP). Consistent with this result, a recent meta-analysis of comparisons between PRP and HA reported significant efficacy of PRP in functional improvement compared with HA at 12-month follow-up. 15 IKDC was not a specific evaluation tool for knee OA, but IKDC subjective score has long been used to evaluate mixed knee pathologies. 13,14,27,53 All included studies of LP-PRP and LR-PRP showed improvement above the MCID of 6.7 33 in mixed knee pathologies at 6 and 12 months (95% CI, 14.04-20.33 and 10.61-24.88, respectively, for LP-PRP; 11.25-22.62 and 11.79-19.62, respectively, for LR-PRP). The improvement in IKDC scores after PRP injection in knee OA was greater than the MCID of IKDC scores in knee injuries of anterior cruciate ligament, meniscus, and cartilage, which indicated that PRP injection had the ability to improve knee status. Based on these findings, this review suggests that intra-articular PRP injections could be a viable therapeutic option for functional improvement in patients with knee OA.
This review revealed that LP-PRP and LR-PRP showed similar improvements in pain and function in patients with knee OA over 12 months of follow-up. In a previous meta-analysis of 6 randomized controlled trials (RCTs) and 3 prospective comparative studies, LP-PRP showed significantly better WOMAC scores than did HA, but LR-PRP did not. 64 However, there was no significant difference in clinical outcomes between LP-PRP and LR-PRP. These findings indicated that LP-PRP resulted in greater improvements in clinical outcomes than did LR-PRP. Apart from different improvements reported in previous meta-analyses, improvements in pain and functional scores in this review did not differ between LP-PRP and LR-PRP. These differences in results may come from different included studies (9 in the previous review, 32 in this review). To the best of our knowledge, no RCT has compared LP-PRP and LR-PRP in knee OA, and only 1 prospective comparative study has compared LP-PRP and LR-PRP. Filardo et al 23 reported similar improvements in pain and function between LP-PRP and LR-PRP over 12 months. The clinical superiority of LP-PRP versus LR-PRP remains controversial, as the role of leukocytes has been a subject of debate because of their positive as well as negative properties. 37 Leukocytes not only play a role in proinflammatory activity but also interact with platelets and other cell types to drive the resolution phase of the healing cascade. 37,54 Neutrophils secrete cytokines for the chemotaxis of monocytes, which are crucial to induce the inflammatory process required to initiate the healing process, called “regenerative inflammation.” 37,59 In addition, IL-1 receptor antagonist protein, thought to be more abundant in LR-PRP, may be responsible for the beneficial effects of the healing process because it blocks IL-1, decreases the activity of matrix metalloproteinase, and reduces the risk of cartilage degradation. 9,51 These potential benefits of leukocytes in LR-PRP, including IL-1 receptor antagonist protein and monocytes, might compensate for the catabolic effect of leukocytes, and even showed similar clinical efficacy such as pain and functional improvement compared with LP-PRP.
Interestingly, the incidence of adverse reactions after PRP injections was associated with leukocyte concentration. The incidence of adverse reactions after PRP injections compared with HA or placebo remains controversial. Previous reviews have suggested that intra-articular PRP did not increase adverse reactions compared with HA or saline injections. 8,15,77 In contrast, 1 review reported that PRP injections resulted in a higher incidence of adverse reactions than did HA. 64 To the best of our knowledge, few studies have evaluated adverse reactions after PRP injections according to leukocyte concentration. Riboh et al 64 concluded that adverse reactions appeared to be a class reaction of PRP that was not dependent on leukocyte concentration. Inconsistent with the previous meta-analysis, our study showed a significant difference in the incidence of adverse reaction after PRP injections according to leukocyte concentration. Adverse reactions included pain and swelling after injection. Theoretically, the presence of leukocytes in PRP increases proinflammatory activity by the expression of catabolic cascades and release of inflammatory markers. 5,45,47,48 In addition, some in vivo studies have reported that PRP containing leukocytes also contained metalloproteinase 2, 3, and 9 and showed greater inflammatory reactions after injections. 18,60 Moreover, only 1 clinical study comparing LP-PRP and LR-PRP reported that knees receiving LR-PRP showed more swelling and pain reaction immediately after the injections. 18 Therefore, we believe that the proinflammatory activity of leukocytes could exacerbate pain and swelling immediately after LR-PRP injections.
Limitations
Some limitations of this study need to be addressed. First, this study included not only RCTs but also prospective comparative studies and case series, resulting in some inherent heterogeneity attributed to uncontrolled bias. In addition, only 1 study directly compared LP-PRP and LR-PRP in knee OA, which is a major limitation of this review. 64 Because we were unable to perform meta-analysis of studies with direct comparison, we tried to include several studies evaluating LP-PRP or LR-PRP in knee OA without methodologic flaws. Second, heterogeneity in injection frequency, injection volume, and blood draw time was not considered in this review. The main purpose of this review was to compare clinical outcomes between LP-PRP and LR-PRP in patients with knee OA. The optimal injection volume and frequency should be investigated in further studies. Third, PAW classification was applied to define LP-PRP and LR-PRP in this study. The Minimum Information for Studies Evaluating Biologics in Orthopaedics guideline has recently been introduced and has shown strength considering all kinds of leukocytes including lymphocytes, monocytes, and neutrophils. However, it was difficult to perform this review using this guideline because of lack of data in the included studies. Fourth, we noted heterogeneity in the evaluation of degeneration level of the knee joint. The Kellgren-Lawrence classification and the Ahlbäck classification criteria are commonly used to evaluate joint degeneration level, but the 2 criteria have different descriptions of grades. Given this variance, the association between the OA radiographic severity and PRP injection efficacy is difficult to conclude.
The findings of this review can support a potential use of intra-articular PRP injection for the treatment of knee OA. In clinical application, clinicians need to consider selecting the leukocyte concentration of PRP for knee OA. However, these issues should be investigated in further studies to increase the efficacy of the clinical application of PRP.
Conclusion
Regardless of leukocyte concentration, intra-articular PRP injection resulted in improvements above the MCID in terms of pain and function in patients with knee OA up to 12 months. LR-PRP appears to pose an increased risk of local adverse reactions compared with LP-PRP injection.
Footnotes
Final revision submitted December 22, 2020; accepted January 25, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by the National Research Foundation of Korea grant funded by the Korean government (No. NRF-2019R1G1A1009620). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Notes
Appendix
Details of PRP Treatment and Mean Platelet and Leukocyte Concentrations in Included Studies a
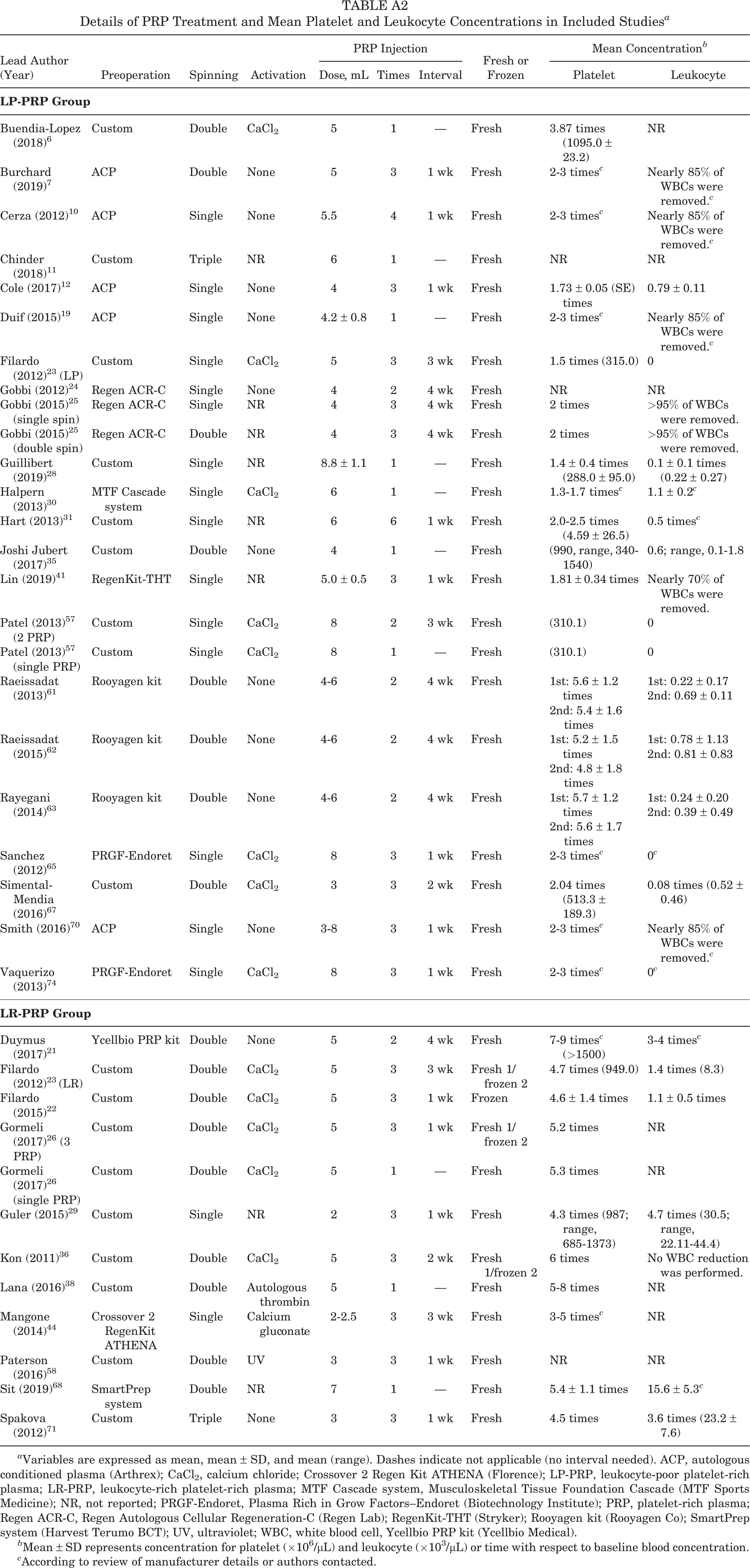
| Lead Author (Year) | Preoperation | Spinning | Activation | PRP Injection | Fresh or Frozen | Mean Concentration b | |||
|---|---|---|---|---|---|---|---|---|---|
| Dose, mL | Times | Interval | Platelet | Leukocyte | |||||
|
|
|||||||||
| Buendia-Lopez (2018) 6 | Custom | Double | CaCl2 | 5 | 1 | — | Fresh | 3.87 times (1095.0 ± 23.2) | NR |
| Burchard (2019) 7 | ACP | Double | None | 5 | 3 | 1 wk | Fresh | 2-3 times c | Nearly 85% of WBCs were removed. c |
| Cerza (2012) 10 | ACP | Single | None | 5.5 | 4 | 1 wk | Fresh | 2-3 times c | Nearly 85% of WBCs were removed. c |
| Chinder (2018) 11 | Custom | Triple | NR | 6 | 1 | — | Fresh | NR | NR |
| Cole (2017) 12 | ACP | Single | None | 4 | 3 | 1 wk | Fresh | 1.73 ± 0.05 (SE) times | 0.79 ± 0.11 |
| Duif (2015) 19 | ACP | Single | None | 4.2 ± 0.8 | 1 | — | Fresh | 2-3 times c | Nearly 85% of WBCs were removed. c |
| Filardo (2012) 23 (LP) | Custom | Single | CaCl2 | 5 | 3 | 3 wk | Fresh | 1.5 times (315.0) | 0 |
| Gobbi (2012) 24 | Regen ACR-C | Single | None | 4 | 2 | 4 wk | Fresh | NR | NR |
| Gobbi (2015) 25 (single spin) | Regen ACR-C | Single | NR | 4 | 3 | 4 wk | Fresh | 2 times | >95% of WBCs were removed. |
| Gobbi (2015) 25 (double spin) | Regen ACR-C | Double | NR | 4 | 3 | 4 wk | Fresh | 2 times | >95% of WBCs were removed. |
| Guillibert (2019) 28 | Custom | Single | NR | 8.8 ± 1.1 | 1 | — | Fresh | 1.4 ± 0.4 times (288.0 ± 95.0) | 0.1 ± 0.1 times (0.22 ± 0.27) |
| Halpern (2013) 30 | MTF Cascade system | Single | CaCl2 | 6 | 1 | — | Fresh | 1.3-1.7 times c | 1.1 ± 0.2 c |
| Hart (2013) 31 | Custom | Single | NR | 6 | 6 | 1 wk | Fresh | 2.0-2.5 times (4.59 ± 26.5) | 0.5 times c |
| Joshi Jubert (2017) 35 | Custom | Double | None | 4 | 1 | — | Fresh | (990, range, 340-1540) | 0.6; range, 0.1-1.8 |
| Lin (2019) 41 | RegenKit-THT | Single | NR | 5.0 ± 0.5 | 3 | 1 wk | Fresh | 1.81 ± 0.34 times | Nearly 70% of WBCs were removed. |
| Patel (2013) 57 (2 PRP) | Custom | Single | CaCl2 | 8 | 2 | 3 wk | Fresh | (310.1) | 0 |
| Patel (2013) 57 (single PRP) | Custom | Single | CaCl2 | 8 | 1 | — | Fresh | (310.1) | 0 |
| Raeissadat (2013) 61 | Rooyagen kit | Double | None | 4-6 | 2 | 4 wk | Fresh | 1st: 5.6 ± 1.2 times 2nd: 5.4 ± 1.6 times |
1st: 0.22 ± 0.17 2nd: 0.69 ± 0.11 |
| Raeissadat (2015) 62 | Rooyagen kit | Double | None | 4-6 | 2 | 4 wk | Fresh | 1st: 5.2 ± 1.5 times 2nd: 4.8 ± 1.8 times |
1st: 0.78 ± 1.13 2nd: 0.81 ± 0.83 |
| Rayegani (2014) 63 | Rooyagen kit | Double | None | 4-6 | 2 | 4 wk | Fresh | 1st: 5.7 ± 1.2 times 2nd: 5.6 ± 1.7 times |
1st: 0.24 ± 0.20 2nd: 0.39 ± 0.49 |
| Sanchez (2012) 65 | PRGF-Endoret | Single | CaCl2 | 8 | 3 | 1 wk | Fresh | 2-3 times c | 0 c |
| Simental-Mendia (2016) 67 | Custom | Double | CaCl2 | 3 | 3 | 2 wk | Fresh | 2.04 times (513.3 ± 189.3) | 0.08 times (0.52 ± 0.46) |
| Smith (2016) 70 | ACP | Single | None | 3-8 | 3 | 1 wk | Fresh | 2-3 times c | Nearly 85% of WBCs were removed. c |
| Vaquerizo (2013) 74 | PRGF-Endoret | Single | CaCl2 | 8 | 3 | 1 wk | Fresh | 2-3 times c | 0 c |
|
|
|||||||||
| Duymus (2017) 21 | Ycellbio PRP kit | Double | None | 5 | 2 | 4 wk | Fresh | 7-9 times c (>1500) | 3-4 times c |
| Filardo (2012) 23 (LR) | Custom | Double | CaCl2 | 5 | 3 | 3 wk | Fresh 1/frozen 2 | 4.7 times (949.0) | 1.4 times (8.3) |
| Filardo (2015) 22 | Custom | Double | CaCl2 | 5 | 3 | 1 wk | Frozen | 4.6 ± 1.4 times | 1.1 ± 0.5 times |
| Gormeli (2017) 26 (3 PRP) | Custom | Double | CaCl2 | 5 | 3 | 1 wk | Fresh 1/frozen 2 | 5.2 times | NR |
| Gormeli (2017) 26 (single PRP) | Custom | Double | CaCl2 | 5 | 1 | — | Fresh | 5.3 times | NR |
| Guler (2015) 29 | Custom | Single | NR | 2 | 3 | 1 wk | Fresh | 4.3 times (987; range, 685-1373) | 4.7 times (30.5; range, 22.11-44.4) |
| Kon (2011) 36 | Custom | Double | CaCl2 | 5 | 3 | 2 wk | Fresh 1/frozen 2 | 6 times | No WBC reduction was performed. |
| Lana (2016) 38 | Custom | Double | Autologous thrombin | 5 | 1 | — | Fresh | 5-8 times | NR |
| Mangone (2014) 44 | Crossover 2 RegenKit ATHENA | Single | Calcium gluconate | 2-2.5 | 3 | 3 wk | Fresh | 3-5 times c | NR |
| Paterson (2016) 58 | Custom | Double | UV | 3 | 3 | 1 wk | Fresh | NR | NR |
| Sit (2019) 68 | SmartPrep system | Double | NR | 7 | 1 | — | Fresh | 5.4 ± 1.1 times | 15.6 ± 5.3 c |
| Spakova (2012) 71 | Custom | Triple | None | 3 | 3 | 1 wk | Fresh | 4.5 times | 3.6 times (23.2 ± 7.6) |
a Variables are expressed as mean, mean ± SD, and mean (range). Dashes indicate not applicable (no interval needed). ACP, autologous conditioned plasma (Arthrex); CaCl2, calcium chloride; Crossover 2 Regen Kit ATHENA (Florence); LP-PRP, leukocyte-poor platelet-rich plasma; LR-PRP, leukocyte-rich platelet-rich plasma; MTF Cascade system, Musculoskeletal Tissue Foundation Cascade (MTF Sports Medicine); NR, not reported; PRGF-Endoret, Plasma Rich in Grow Factors–Endoret (Biotechnology Institute); PRP, platelet-rich plasma; Regen ACR-C, Regen Autologous Cellular Regeneration-C (Regen Lab); RegenKit-THT (Stryker); Rooyagen kit (Rooyagen Co); SmartPrep system (Harvest Terumo BCT); UV, ultraviolet; WBC, white blood cell, Ycellbio PRP kit (Ycellbio Medical).
b Mean ± SD represents concentration for platelet (×106/μL) and leukocyte (×103/μL) or time with respect to baseline blood concentration.
c According to review of manufacturer details or authors contacted.
