Abstract
Background:
Although nonoperative management of acute Achilles tendon rupture (ATR) is a reasonable option, surgical repair has attracted attention for young and active patients. More reliable Achilles tendon repair techniques are needed to enhance recovery after ATR in this population.
Purpose/Hypothesis:
To biomechanically analyze the panda rope bridge technique (PRBT) and compare it with other minimally invasive repair techniques over a simulated, progressive rehabilitation program. It was hypothesized that PRBT would result in better biomechanical properties and enhanced recovery after ATR.
Study Design:
Controlled laboratory study.
Methods:
An Achilles tendon rupture was created 4 cm from the distal tendon insertion site in 40 bovine lower extremities, and specimens were then randomly allocated to 5 Achilles tendon repair techniques: (1) Achillon, (2) modified Achillon, (3) Percutaneous Achilles Repair System (PARS), (4) modified PARS, and (5) PRBT. Each group was subjected to a cyclic loading protocol that was representative of progressive postoperative rehabilitation for ATR (250 cycles at 1 Hz for each loading stage: 20-100 N, 20-200 N, 20-300 N, and 20-400 N).
Results:
The PRBT technique demonstrated significantly less elongation (1.62 ± 0.25 mm) than the 4 other repair techniques after the first loading stage of 20 to 100 N (P < .05). All specimens in the 4 other groups developed a large gap (elongation ≥5 mm) at the 20- to 200-N loading stage. When overall biomechanical performance was examined, the PRBT group exhibited higher strength (20-400 N) and more mean loading cycles (984 ± 10) compared with the 4 other groups (P < .05).
Conclusion:
In this bovine model, PRBT biomechanically outperformed the other minimally invasive Achilles tendon repair techniques that were tested and could therefore meet the requirements of accelerated rehabilitation.
Clinical Relevance:
The reduced tendency for premature rerupture and the overall improved biomechanical properties of PRBT suggest that ATR patients treated with PRBT may more readily complete early and aggressive postoperative rehabilitation protocols. In addition, they may have a lower risk of early irreversible suture failure.
Keywords
Acute Achilles tendon rupture (ATR) is frequent in athletes as well as the general public. 11,18 The primary objective for ATR repair is to restore the normal biomechanical properties of the Achilles tendon and to avoid rerupture during the early stages of biological healing. Although nonoperative management of ATR is a viable option, surgical repair is preferred in healthy and active populations. 3,12,23
Because of the long duration of recovery and delayed return to function after ATR repair, an enhanced recovery period would greatly benefit patients. Another benefit of a shorter recovery time is that appropriately applied stress stimulation can assist and accelerate tendon healing. 4,5 However, the stress applied to Achilles tendon stumps may exceed the capability of some commercially available suture techniques, which eventually results in elongation of the healing tendon, a large gap between the previously repaired tendon, and even irrevocable suture failure. Therefore, it is imperative to explore a novel repair technique that provides enough initial biomechanical support to meet the immediate enhanced recovery after surgery (ERAS) protocol.
A previous study showed that excessive tension at the fixation point as well as insufficient contact area between the healing tendon stumps were the main causes of repair failure in the early postoperative stage. 28 Another study examined whether the panda rope bridge technique (PRBT), a novel repair technique for ATR, could solve these problems. 30 It was found that proximal bridge fixation at the gastrocnemius myotendinous junction, combined with the distal calcaneal anchor, ensured stability of the fixed points, maintaining moderate tension between tendon stumps throughout the rehabilitation protocol. Patients who received PRBT were allowed to actively move the ankle immediately after surgery and initiated full-weightbearing walking without crutches or a cast within 3 days, in full compliance with the ERAS protocol. The mean time for patients to return to work and sports was 1.7 months and 4.6 months, respectively. 30 Although no cases of rerupture or repair failure were observed clinically, the authors set out to clarify the original research questions: Can PRBT provide better biomechanical properties than other established, minimally invasive repair techniques? Can PRBT completely meet the requirements of ERAS protocol for ATR?
Methods
Specimens
A total of 40 fresh-frozen bovine Achilles tendons (2-4 years old; obtained from the experimental animal center of Chongqing Medical University) were used to conduct the biomechanical study. The fresh bovine lower extremities were frozen to –20° C within 24 hours after being severed. The length and cross-sectional area of each Achilles tendon sample were measured, and the differences were <10%, which presented no statistical significance. The mean ± SD length of the 40 bovine Achilles tendons was 16.2 ± 0.6 cm, and the cross-sectional area of the thinnest anatomic position of the Achilles tendons was 1.2 ± 0.3 cm2.
A typical Achilles tendon rupture was created 4 cm from the distal tendon insertion site in all specimens, and the specimens were randomly allocated to 5 different repair technique groups (n = 8 specimens each): (1) Achillon group, (2) modified Achillon group, (3) Percutaneous Achilles Repair System (PARS) group, (4) modified PARS group, and (5) PRBT group. The senior sports medicine physician who was most skilled at each respective (T.W., L.J.Y, or another physician) performed all repairs for that technique. The same suture material (No. 2 Ultrabraid; Smith & Nephew) was used for all groups.
Surgical Technique
Illustrations of the 5 repair techniques are shown in Figure 1. For all techniques, repair sutures were tied with 6 square knots and 1 surgical knot while the foot was held in gentle plantarflexion to reapproximate the tendon ends.

Diagrams illustrating 5 different suture configurations: (1) Achillon repair, (2) modified Achillon repair, (3) Percutaneous Achilles Repair System (PARS) repair, (4) modified PARS repair, and (5) panda rope bridge technique (PRBT) repair.
The Achillon repairs were performed in accordance with the manufacturer’s instructions for the Achillon Achilles Tendon Suture System (Integra LifeSciences) and consisted of 3 pairs of sutures passed transversely through the 2-cm proximal and distal stumps of the ruptured tendon. The sutures were brought together and tightened until the stumps of the tendon were approximated; the sutures were then tied securely using standard square and surgical knots. 3 The modified Achillon repairs were performed in the same manner as Achillon repairs at proximal stumps of the ruptured tendon, and 3 pairs of sutures were threaded through a coronal calcaneal tunnel about 2 cm below the Achilles tendon insertion.
The PARS repairs were performed in accordance with the manufacturer’s instructions for the PARS Jig System (Arthrex) and consisted of 3 pairs of sutures passed crosswise through the 2-cm proximal and distal stumps of the ruptured tendon. The sutures were then brought together and tightened until the stumps of the tendon were approximated; afterward, the sutures were tied securely using standard square and surgical knots. 22 The modified PARS repairs were performed in the same manner as PARS repairs at the proximal stumps of the ruptured tendon, and 3 pairs of sutures were threaded through a coronal calcaneal tunnel about 2 cm below the Achilles tendon insertion.
The PRBT repairs were performed as described by Yin et al. 30 In brief, 4 pairs of sutures were passed through a coronal calcaneal tunnel about 2 cm below the distal Achilles tendon insertion, and 4 paired Krackow-type locking loops were sutured at the medial and lateral sides of the proximal tendon.
Biomechanical Testing
The repaired Achilles tendons were placed in an adjustable fixture attached to the base of a dynamic tensile testing machine (Instron). A loading protocol that represented conditions seen in an enhanced rehabilitation program was applied to all specimens. 6 No preload was applied. The protocol consisted of 4 cyclic loading stages at 1 Hz with 250 cycles in each stage: stage 1, 20-100 N; stage 2, 20-200 N; stage 3, 20-300 N; and stage 4, 20-400 N. Therefore, each sample was designed to go through 1000 loading cycles unless the specimen exhibited repair failure during the loading process.
The loading stages and target parameters were designed to simulate the ERAS protocol for patients with ATR. The loading parameters (100, 200, 300, and 400 N, respectively) for each cyclic stage were determined based on the biological tensile stress that the Achilles tendon sustains in different positions, and the load was gradually increased to reflect the continuous recovery process. A frequency of 1 Hz was chosen to reflect a normal walking pace.
Repair failure was defined as tendon rerupture, bone tunnel collapse, or tendon elongation >30 mm (the displacement exceeded the limit of the instrument measurement). Any large gaps, defined as ≥5 mm of separation between the Achilles tendon ends, were also recorded. Such gaps might seriously impede tendon healing.
Statistical Analysis
Based on the data from preliminary tests, at least 6 specimens in each group were necessary to meet the design requirements of the experiment. Therefore, 8 specimens were included in each group to ensure implementation of the experiment. One-way analysis of variance was applied for continuous variables and chi-square or Fisher exact test for categorical variables. P < .05 was considered statistically significant. Analyses were performed using Stata for Mac; Version 15.0 (StataCorp).
Results
Tendon Elongation
Although the specimens in all groups survived the first 250 cycles of 20-100 N loading, significant differences were seen during this early loading phase (Figure 2). The mean elongation for the PRBT group was 1.62 ± 0.25 mm, which was the lowest among all 5 groups; the highest elongation was found in the PARS group, 3.81 ± 0.18 mm. The remaining groups ranked by the length of elongation were the modified PARS group, the Achillon group, and the modified Achillon group, with a mean elongation of 3.41 ± 0.20, 2.95 ± 0.23, and 2.55 ± 0.26 mm, respectively. The differences between each group were significant (P < .05).
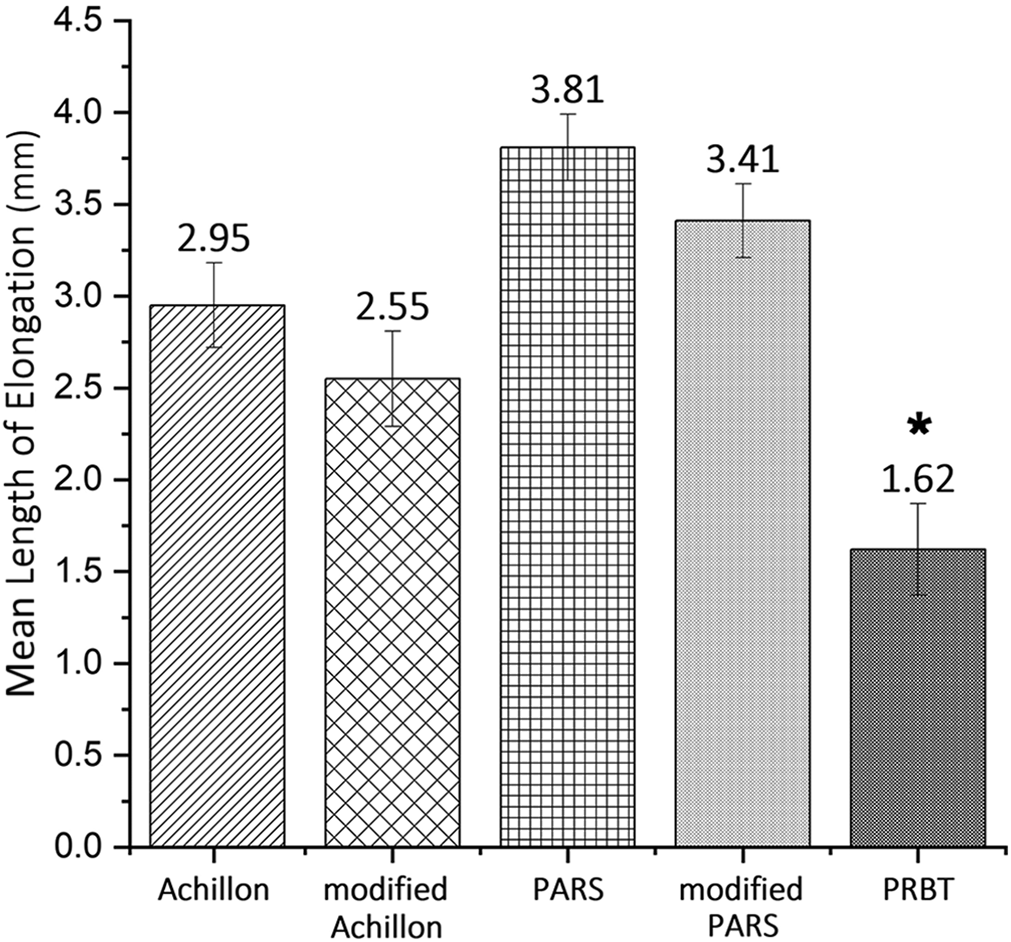
Mean elongation of each repair technique over the first cyclic loading stage (20-100 N for 250 cycles at 1 Hz). *The panda rope bridge technique (PRBT) repair presented significantly less elongation than the other 4 repair techniques in the first cyclic loading stage (P < .05). Error bars represent SDs. PARS, Percutaneous Achilles Repair System.
Significant differences were also observed in formation of a large gap (elongation ≥5 mm). Except for the PRBT group, all specimens in the other 4 groups showed formation of a large gap at the 20-200 N stage. In the PRBT group, 3 of the 8 specimens developed a large gap at 20-300 N, and all specimens showed a large gap at 20-400 N. Figure 3 shows examples of formation of a large gap.
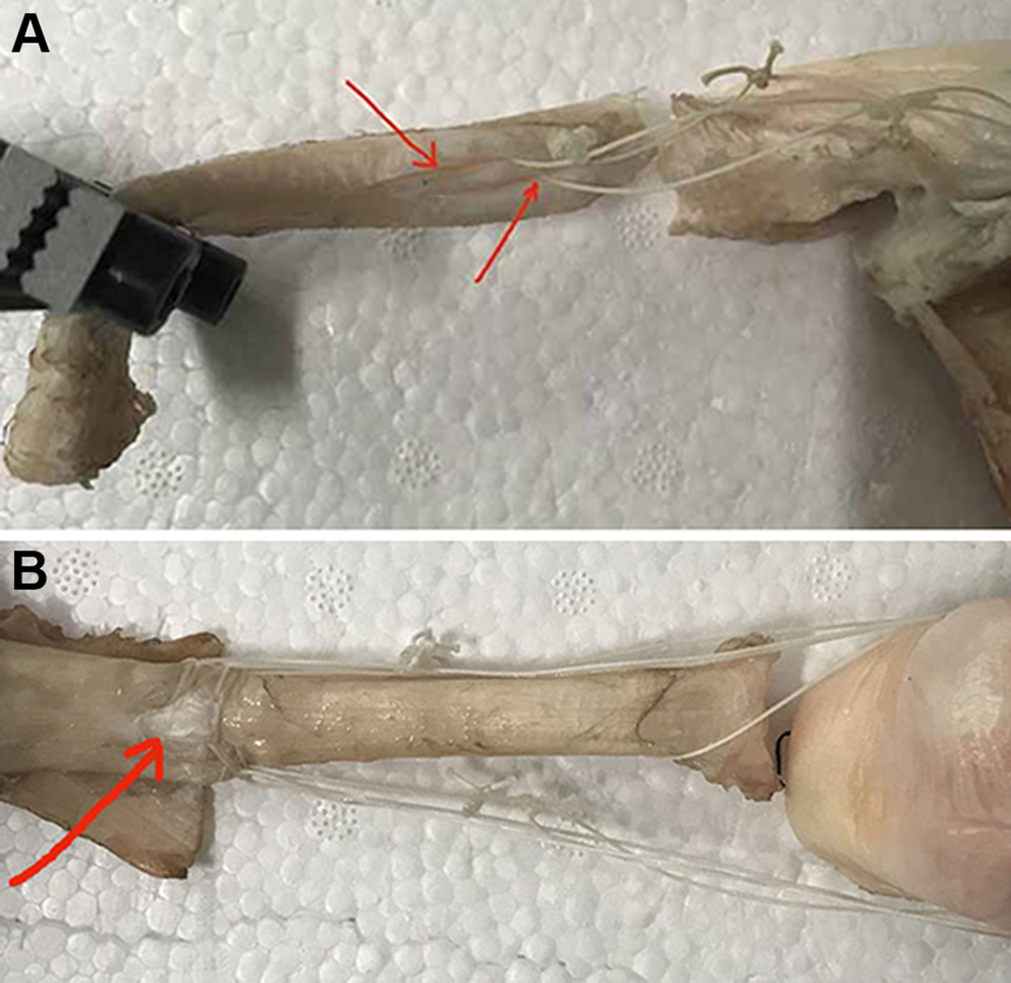
(A) The modified Percutaneous Achilles Repair System (PARS) suture cut the tendon tissues (red arrows) at the point of fixation and formed a large gap during the 20-200 N cycle. (B) The normal tendon tissues of the panda rope bridge technique (PRBT) group were torn (red arrow), and a large gap appeared during the 20-400 N cycle.
Loading Stages
No group survived the entire 4 stages of 1000 loading cycles, yet the maximal loading stage varied among the 5 groups (P < .05) (Table 1). The mean number of cycles also presented a corresponding statistical difference among the 5 groups according to Kruskal-Wallis rank test, with 384 ± 9 cycles in the Achillon group, 503 ± 1 cycles in the modified Achillon group, 540 ± 6 cycles in the PARS group, 622 ± 17 cycles in the modified PARS group, and 984 ± 10 cycles in the PRBT group (P < .05) (Figure 4). Ruptures in the PRBT group appeared at the normal tendon tissues proximal to the Krackow-type locking sutures; in the other 4 groups, all ruptures occurred at the suture-tendon interface.
Maximal Loading Stage (Ultimate Survival Load) for Each Repair Technique (n = 8 Specimens Each) a

a Each of the 4 cyclic loading stages was conducted at 1 Hz and consisted of 250 cycles. PARS, Percutaneous Achilles Repair System; PRBT, panda rope bridge technique.
b PRBT repair resulted in a significantly higher loading stage (P < .05) compared with the other 4 repair techniques throughout the 4 progressive loading stages.

The mean number of cycles to failure for each repair group. *The panda rope bridge technique (PRBT) group exhibited significantly more loading cycles than the other 4 repair techniques throughout the 4 progressive loading stages (P < .05). PARS, Percutaneous Achilles Repair System.
Discussion
The study results demonstrated that among the 5 tested repair techniques, PRBT presented the minimum tendon elongation (1.62 ± 0.25 mm) in the first 250 cycles of 20-100 N loads and also the lowest rate of a large gap throughout the loading cycles. Several related studies reported that tendon elongation led to gait disorders and impaired the biomechanical properties of the Achilles tendon. 1,24,27 Excessive elongation may interfere with tendon healing and thus reduce the muscle strength and endurance of the triceps surae. Silbernagel et al 26 found that greater Achilles tendon elongation corresponded to greater deficits in single-leg heel-raise height. Similarly, in a randomized controlled clinical trial involving 50 patients, Kangas et al 16 found that tendon elongation was negatively correlated with clinical outcome scores when using the Leppilahti scoring method.
In the current study, PRBT provided a more reliable initial strength, which is essential for enhanced postoperative recovery and Achilles tendon healing. In addition to higher initial strength, PRBT exhibited greater ultimate strength and more stable fatigue-resistant properties compared with the other tested techniques. The ultimate strength of PRBT was reached at the 20-300 N stage, whereas all of the other 4 repair methods failed at 20-200 N; further, PRBT demonstrated a mean of 750 loading cycles compared with 500 cycles in the other 4 techniques.
Repair with PRBT does not guarantee that an aggressive early postoperative rehabilitation protocol can be undertaken. However, the research data presented here can guide the development and implementation of a postoperative rehabilitation protocol for patients who have ATR. Previous studies have estimated that the Achilles tendon sustains 100 N in passive motion, 191 N when walking on a 1-inch heel lift, 282 N when walking on a 0.5-inch heel lift, and 369 N when walking without a heel lift. 2,9,19 Therefore, an ATR patient treated with PRBT can start ankle movement exercises (20-100 N) immediately after surgery without the risk of irreversible tendon elongation or a large gap at the repair site. Present research also proved that the seemingly radical plan of walking on a 30-mm (1.18-inch) heel on day 5 postoperatively was safe for patients who underwent PRBT, because the ultimate strength of the PRBT repair (300 N) was far more than 191 N, and no large gap would form at up to 200 N. According to the experimental data in the current research, these results could not be achieved by other 4 repair techniques.
Surgical restoration of the ATR is commonly performed, mostly in young and active patients, but soft tissue complications are common. 13,17 Minimally invasive and percutaneous techniques are increasingly preferred given the lower rate of sural nerve damage and skin complications as well as the achievement of a satisfactory cosmetic appearance. However, minimally invasive repair results in insufficient tensile strength, with some of the percutaneous repair configurations having only 50% of the strength compared with traditional open sutures. 15,25
The core of PRBT is the rope bridge technique, which ensures moderate tensile stress throughout the ERAS process of ATR. As shown in Figure 1, the major differences between PRBT and the other techniques were the proximal Krackow-type locking loop sutures and the distal calcaneal tunnel fixation. In a cadaveric experiment, Watson et al 29 demonstrated that a Krackow-type locking loop suture was substantially stronger than either of the other 2 standard configurations in Achilles tendon repair. In another study, the number of suture strands crossing the repair site was correlated with the mechanical strength of Achillon tendon repair techniques. 21 Moreover, the greater load dispersion at the proximal and distal fixation of the tear site enhanced the stability of the sutures. 25 In the current study, the biomechanical superiority of bone tunnel fixation was reflected in the results of modified Achillon and PARS techniques. Therefore, PRBT provided increased elongation and gap resistance with a greater portion of the proximal tendon held by the sutures and a rigid distal fixation in the calcaneus, which dispersed the load within a larger volume of tissues. Further, the tension was distributed more evenly on the suture and the tissue itself.
The weakness of either distal or proximal fixation might be the inherent defect responsible for the unsatisfactory biomechanical performance of the traditional repair techniques studied here. Simple sutures on tendon tissues in Achillon and PARS groups were the primary strength-limiting components, which might lead to cutting of the tendon tissues at the fixed points and further unexpected extension stress predominately located between the tendon stumps. It was difficult to evenly transmit the biological strength to the entire ankle flexion-extension system, so that excessive elongation or the large gap appeared during the early cyclic loading, and rerupture occurred when the ultimate load was notably lower in the traditional repair groups compared with the PRBT group. 7,14,20
To our knowledge, there has been no previous report of a rope bridge technique. In theory, PRBT better maintains the contact of the stumps and neutralizes the tensile stress at the repair site by virtue of the two solid fixed points, thereby promoting tendon healing. The results of this study also distinctly demonstrated that PRBT presented more satisfactory in vitro biomechanical properties compared with Achillon and PARS.
In the current study, to better examine the advantages of PRBT, we included two modified repair methods based on the classic Achillon and PARS techniques; these modified techniques moved the distal fixed point from tendon tissues to calcaneus, thereby strengthening the distal fixation of the sutures. Interestingly, the two modified techniques presented an intermediate level of ultimate strength and fatigue resistance, with biomechanical results that fell between the results of the classic techniques and PRBT. The modified techniques improved the distal fixation compared with PRBT, but the proximal fixation was still fragile, so that the biomechanical properties of Achilles tendon repaired with the modified techniques were still inferior to results achieved with PRBT.
Authors have observed that patients with ATR who received PRBT could start a standard enhanced rehabilitation protocol immediately after surgery without excessive extension or rerupture of the Achilles tendon. This observation is consistent with the theoretical reasoning and experimental data of the current study, and previous biomechanical studies have reached similar conclusions. Clanton et al 6 reported that displacement of Achillon and PARS repairs after the first 250 cycles of 20-100 N was 9.9 and 12.2 mm, respectively. The mean number of cycles to failure for Achillon and PARS in that study was 362 and 424, which was similar to our findings. Clanton et al noted that most of the elongation occurred in the first 10 cycles. Preloading prior to fixation would remove some of the repair elongation that occurs during the initial loading; however, we chose not to apply a preload in order to keep our model in line with current clinical practice. In another study, both Achillon and PARS repair methods resulted in a large gap ≥5 mm during the 20-200 N cyclic loading. 8 Thus, technical theory, in vitro biomechanical tests, and clinical outcomes indicate that PRBT could yield better biomechanical effectiveness for repaired Achilles tendon; further, compared with other percutaneous repair techniques, PRBT might have advantages in terms of postoperative enhanced recovery.
This study has several limitations. Because this biomechanical experiment included destructive mechanical tests, it was not suitable for in vivo evaluation; due to ethical restrictions, the number of fresh human Achilles tendons available was limited and could not fully meet the grouping requirements of the research. However, several biomechanical studies demonstrated that the viscoelastic, structural, and material properties of bovine tendons were similar to those of humans, having satisfactory stiffness and resistance to high failure loads that were comparable with human cadaveric tendons 9,10 ; thus, we used fresh bovine Achilles tendon as a substitute to obtain the best biomechanical results. However, this experiment did present some different results from Clanton et al, 6 who used human tendons. Another limitation was that we performed only cyclic testing and did not look at initial load to failure, affecting the comprehensiveness of the study results.
An additional limitation was that the frozen bovine tendon could represent only time-zero Achilles tendon rupture, whereas in the clinic, the Achilles tendon progressively heals during the recovery protocol, thus making it possible to withstand greater mechanical stress. Indeed, in our clinical observation, none of the >100 patients we have treated with PRBT have yet experienced failure. Furthermore, in this experiment, we did not compare the percutaneous techniques with traditional open techniques. Therefore, the results of this study should not be overly extrapolated. Despite these limitations, we believe that the results of the present study are worth consideration.
Conclusion
PRBT had better ability to resist cyclic loading than other percutaneous repair techniques. None of the techniques were strong enough to withstand cyclic loading representative of a progressive postoperative rehabilitation. We urge caution when designing early rehabilitation using any of these techniques.
Footnotes
Final revision submitted November 20, 2020; accepted January 3, 2021
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was funded by the Chongqing Science and Technology Commission’s Technology Innovation and Application Demonstration Fund (cstc2017shmsA130049 and cstc2018jscx-msybX0088) and the Kuanren Talents Program of The Second Affiliated Hospital of Chongqing Medical University (KY2019L013). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
