Abstract
Background:
Knee abduction moment during landing has been associated with anterior cruciate ligament (ACL) injury. However, accurately capturing this measurement is expensive and technically rigorous. Less complex variables that lend themselves to easier clinical integration are desirable.
Purpose:
To corroborate in vitro cadaveric simulation and in vivo knee abduction angles from landing tasks to allow for estimation of ACL strain in live participants during a landing task.
Study Design:
Descriptive laboratory study.
Methods:
A total of 205 female high school athletes previously underwent prospective 3-dimensional motion analysis and subsequent injury tracking. Differences in knee abduction angle between those who went on to develop ACL injury and healthy controls were assessed using Student t tests and receiver operating characteristic analysis. A total of 11 cadaveric specimens underwent mechanical impact simulation while instrumented to record ACL strain and knee abduction angle. Pearson correlation coefficients were calculated between these variables. The resultant linear regression model was used to estimate ACL strain in the 205 high school athletes based on their knee abduction angles.
Results:
Knee abduction angle was greater for athletes who went on to develop injury than for healthy controls (P < .01). Knee abduction angle at initial contact predicted ACL injury status with 78% sensitivity and 83% specificity, with a threshold of 4.6° of knee abduction. ACL strain was significantly correlated with knee abduction angle during cadaveric simulation (P < .01). Subsequent estimates of peak ACL strain in the high school athletes were greater for those who went on to injury (7.7-8.1% ± 1.5%) than for healthy controls (4.1-4.5% ± 3.6%) (P < .01).
Conclusion:
Knee abduction angle exhibited comparable reliability with knee abduction moment for ACL injury risk identification. Cadaveric simulation data can be extrapolated to estimate in vivo ACL strain. Athletes who went on to ACL injury exhibited greater knee abduction and greater ACL strain than did healthy controls during landing.
Clinical Relevance:
These important associations between the in vivo and cadaveric environments allow clinicians to estimate peak ACL strain from observed knee abduction angles. Neuromuscular control of knee abduction angle during dynamic tasks is imperative for knee joint health. The present associations are an important step toward the establishment of a minimal clinically important difference value for ACL strain during landing.
Keywords
Frontal plane loading during landing has been demonstrably associated with anterior cruciate ligament (ACL) injury and injury risk in athletes. 19,39,41,46,51,71 The influence of these loads is likely magnitude dependent, given that 0 to 15 N·m of isolated knee abduction moment (KAM) applied to the knee at 20° of flexion has exhibited limited effect on ACL strain in live participants. 27 However, 0 to 15 N·m of valgus underrepresents physiologic loading during dynamic athletic tasks, given that during a laboratory-controlled drop from a 31-cm box, mean KAM exceeds 20 N·m, 28,30 classification for high injury risk exceeds KAM of 25.3 N·m, 51 and individual peak KAM can exceed 57 N·m. 16 The higher loads are likely to have greater influence on ligament strain, a concept that is supported by cadaveric impact simulations, because only the highest risk and rupture loading profiles have exhibited statistically significant increases in ACL strain compared with baseline trials. 13,15 These large KAMs play a critical role in ACL strain and subsequent injury onset, given that ACL ruptures can be reliably generated on lower extremity specimens during landing simulations that incorporate frontal plane loading. 15 –17,44,47 Specifically, KAM applied to the knee during multiple simulations of athletic tasks has been shown to induce greater increases in peak ACL strain than has either anterior tibial translation or internal tibial rotation torque. 7,9 –12,14,15,48,63
Along with KAM, knee abduction angle (KAA) is also associated with dynamic athletic tasks and poor neuromuscular control, especially in female athletes. 29,31 Indeed, female athletes who have gone on to develop ACL injury have exhibited greater KAA at initial contact (IC) during landing than have healthy controls. 41 Video analysis of actual ACL ruptures further confirms the presence of KAA deviation and frontal plane collapse of the knee during injury events. 42,45 In addition, controlled laboratory simulation of KAA rotations on cadaveric lower extremities oriented to represent the mean IC position at landing has induced greater increases in ACL strain than have equal magnitude rotations applied in the transverse plane. 9 However, despite the identification and modeling of these frontal plane contributions to ACL strain, a gap in the literature remains. Specifically, the quantified relationship between KAA and intra-articular ACL mechanics during landing tasks and the subsequent potential to use KAA as a predictor for injury risk in vivo remain underexplored.
To evaluate factors associated with increased ACL injury in both male and female populations, biomechanical parameters have been widely examined in the literature. 1,2,40 In particular, peak KAM has been used to identify ACL injury risk status within an in vivo cohort with a high sensitivity and specificity. 41 Unfortunately, kinetic measures, such as KAM, require rigorous laboratory protocols and expensive 3-dimensional (3D) motion analysis equipment to capture and calculate data with precision and reliability. 24,28,49 Due to this limitation, researchers and clinicians have extrapolated surrogate clinical measures, often based on kinematic joint position and angles, such as KAA, to identify patients at high risk for injury in the clinic with similar accuracy to a 3D inverse dynamics analysis. 52 –55 As such, there remains significant interest in the association between kinematic measurements, such as KAA, and ACL injury risk identification. Subsequent correlation between KAA and intra-articular ligament mechanics would amplify the importance of such an association because it would then allow clinician researchers to make quantifiable estimates of mechanical function through visually observable kinematics during landing tasks.
This investigation had 2 primary objectives: to (1) determine whether KAA exhibits similar capacity for ACL injury risk classification to KAM across an in vivo cohort and (2) corroborate data from in vitro cadaveric simulations with in vivo kinematics to estimate ACL strain in live participants during a landing task. Specifically, we aimed to associate KAA from cadaveric and in vivo cohorts. In vivo kinematics were then used in combination with cadaveric measured ACL strain to derive in vivo ACL strain. The hypothesis tested was that KAA would be a significant identifier of ACL injury incidence in the in vivo participants and of ACL strain in the cadaveric specimens. In addition, we tested whether the ACL strain estimates derived for in vivo participants would be higher in those who went on to develop injury than in healthy controls.
Methods
In Vivo Kinematic Analysis
As reported in the literature, 3D motion analysis data were previously collected on a cohort of 205 female team sport athletes. 41 This cohort from high schools local to Cincinnati, Ohio, (mean ± SD age, 16.1 ± 1.7 years; height, 164.4 ± 6.1 cm; mass, 59.2 ± 8.1 kg) was previously used to prospectively examine the role of KAM relative to ACL injury risk and consisted of soccer, volleyball, and basketball players. In brief, 25 retroreflective markers were placed on each participant in a modified Helen Hayes format. These markers were then tracked for position at 240 Hz using an 8-camera motion analysis system (Eagle Cameras; Motion Analysis Corp) while ground-reaction forces were simultaneously collected at 1200 Hz using dual in-ground force platforms (AMTI). Ground-reaction force and marker position data were synched through an electronic trigger, and data were collected continuously throughout the performance of a drop vertical jump task (DVJ) from a 31-cm box. Data were filtered through a low-pass Butterworth digital filter with a cutoff frequency of 9 Hz for positional coordinates and 50 Hz for ground-reaction forces. Data were then submitted to custom software that calculated kinematic and kinetic values for the knee joint. After motion capture analysis, each participant was prospectively tracked for ACL injury for 2 years. At the end of the 2-year tracking period, all participants were separated into 2 groups: an ACL injury group (n = 9) and a healthy control group (n = 196). All 9 participants in the ACL injury group had sustained a noncontact ACL injury during participation in high school sporting activity (8 soccer, 1 basketball). Of the 9 injured participants, 4 exhibited family history of knee injury, 6 exhibited knee laxity, and 8 were postpubertal. Previous injury history was not controlled for in this in vivo cohort. Further, 12 participants had a previous history of knee injury, 2 of whom had ACL injuries. Both participants with a previous history of ACL injury had sustained additional ACL injury to the contralateral limb. Before data collection, informed consent/assent was obtained for each participant. All methods were reviewed and approved by the Cincinnati Children’s Hospital Institutional Review Board.
In Vitro Cadaveric Impact Simulation
In total, 46 cadaveric lower extremity specimens were set up in the custom-designed mechanical impact simulator and examined through simulated drop landings (Figure 1). Of these 46 specimens, 19 specimens completed the full range of prescribed impact simulations before any structural failure. Of these, 11 specimens (8 male, 3 female; mean ± SD age, 38.2 ± 9.5 years; mass, 94.1 ± 25.6 kg; height, 175 ± 10 cm; body mass index, 31 ± 9) were assessed using continuous 3D kinematic data and used for analysis in the present investigation. Details of the method have been previously reported in the literature. 16,17 In brief, specimens were resected of all soft tissue superior to the knee joint with the exception of the quadriceps and hamstrings tendons. The femur was transected 20 cm superior to the patella, and the specimen was then inverted and potted in a custom fixture such that the long axis of the femur aligned with the vertical axis of a 6-axis load cell (Omega160 IP65/IP68; ATI Industrial Automation). A custom fixture was mounted to the tibia and attached to pneumatic cylinders that applied external loads of KAM, anterior tibial shear (ATS), and internal tibial rotation (ITR) to the knee joint. These external loads were derived from a separate in vivo cohort of 44 healthy participants (mean ± SD age, 23.3 ± 4.1 years; mass, 72.6 ± 13.9 kg; height, 172 ± 10 cm) who were active in recreational or organized sports. These athletes performed DVJ tasks under 3D motion analysis as described above. Consent/assent was obtained before testing of these individuals, and all methods were approved by The Ohio State University Institutional Review Board. The subsequent KAM, ATS, and ITR kinetic data from these DVJ tasks were subdivided into relative risk levels as indicated by existing literature. 8,13 –15 These in vivo–based external loads were individually randomized by risk level and applied to the joint immediately before the delivery of an impulse force to the sole of the foot. After this paradigm, each specimen went through repeated-measures testing that consisted of 26 simulations, each with a unique profile of external knee kinetics. 15 These external forces and impulse deliveries emulated the dynamic knee environment experienced when athletes land from a jump and produced clinical representations of ACL injuries in 88% of specimens tested. 17 Testing was ceased once a soft or hard tissue injury was identified. Specimen integrity was evaluated before and after simulation by a board-certified orthopaedic surgeon. All 11 specimens included for analysis in this investigation experienced ACL failure.
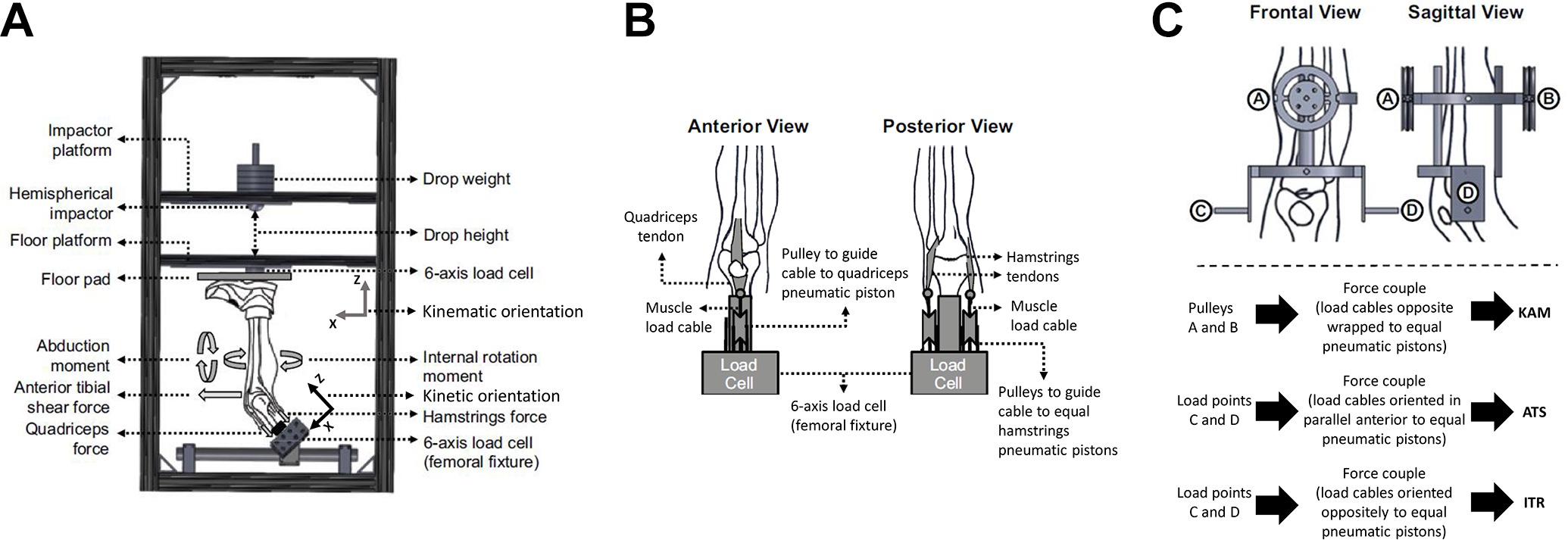
(A) Meta-view of custom-designed mechanical impact simulator for creation of anterior cruciate ligament ruptures. (B) Cable pulley system used to deliver pneumatically actuated loads to the quadriceps and hamstrings tendons. (C) External fixation frame attached to the tibia and used to deliver pneumatically actuated knee abduction moment, anterior tibial shear, and internal tibial rotation loads to each specimen. KAM, knee abduction moment; ATS, anterior tibial shear; ITR, internal tibialrotation. Reprinted with permission from Bates NA, Schilaty ND, Nagelli CV, Krych AJ, Hewett TE. Validation of non-contact anterior cruciate ligament tears produced by a mechanical impact simulator against the clinical presentation of injury. Am J Sports Med. 2018;46(9):2113-2121.
Throughout impact simulations, ACL strain was recorded using a differential variance reluctance transducer strain gauge (Lord MicroStrain) implanted in the distal third of the anteromedial bundle of the ligament, 16 –18,26,47,58 knee joint forces and torques were recorded using the 6-axis load cell and extrapolated to the knee joint center point, 16,17,47,58,61 and 3D kinematic data between the tibia and femur were determined based on Optotrak (Northern Digital Inc) triads that were mounted directly to the femur and tibia. Kinematic data were determined as tibial motion with respect to the femur. Data were sampled at 10,000 Hz, with the exception of the Optotrak system, which sampled at 100 Hz. All data were filtered through a low-pass Butterworth digital filter with cutoff frequency of 50 Hz 62 and then interpolated to 901 points across a 3.0-second trial (100 points per second). Cadaveric kinematic data were calculated as the tibia with respect to the femur coordinate system using Visual 3D (C-Motion). KAA was represented as a negative value. Positive values reported along the same axis were indicative of knee adduction angles (varus). All methods were reviewed and approved as not human subjects research by the Mayo Clinic Institutional Review Board.
Statistical Analysis
Statistical means and standard deviations were calculated for KAA for in vivo injured and uninjured participant groups. Relative to the orientation of our coordinate axes and to maintain consistency with previous literature, 41,51 Student t tests were used to compare values between groups, with significance at α < .05. Logistic regression models were used to create receiver operating characteristic (ROC) curves and obtain sensitivity and specificity between the injured and uninjured groups. Maximum sensitivity and specificity for status prediction were captured from the ROC curves. These analyses were individually performed for peak KAA and KAA at IC. Student t tests were used to compare values between peak KAA and KAA at IC.
Pearson correlation coefficients were calculated between ACL strain (peak magnitude and change from initial value) and frontal plane knee measures (change in KAA from initial value and magnitude of external KAM applied). Significance was set at α < .05, and the predictive threshold was set at r 2 ≥ 0.2. 41,43 Although r2 > 0.2 is considered a low to moderate statistical correlation, no single biomechanical factor is known to exist that can precisely predict future ACL injury events. Instead, multifactorial models are used to predict relative injury risk as a surrogate for likelihood of sustaining ACL injury. Literature has previously presented a 5-factor paradigm for injury prediction models. 29 Following this 5-factor paradigm, an r 2 threshold of ≥0.2 would be a minimal inclusion criterion for any given individual factor. A net benefit analysis 3,67 was used to assess the clinical applicability of this predictive model.
Peak KAA and KAA at IC from the in vivo kinematic data were applied to the resultant cadaveric simulation linear regression equations that were significant and predictive in order estimate in vivo ACL strain. Means and standard deviations were calculated for both in vivo participant groups, and Student t tests were again used to test for significance (α < .05).
Results
In Vivo Kinematic Analysis
Magnitude of peak KAA during landing was significantly different between the ACL-injured group and the uninjured referent group (P < .01) (Figure 2). The mean peak KAA was significantly more deviated from a vertical tibiofemoral alignment in athletes who went on to develop ACL injury (–9.0° ± 3.2°) than their uninjured counterparts (–1.4° ± 7.7°; P < .01) (Figure 3). Timing to peak KAA from IC was not a significantly different factor between groups (P = .30). Logistic regression analysis demonstrated that peak KAA during landing was a significant predictor of ACL injury status (P < .01) in the ACL injured group compared with healthy controls, with a sensitivity of 100% and specificity of 65% (threshold, –6.3°; area under the curve, 0.804). Net benefit analysis using a probability of 4.3% (9 injuries from 205 athletes) yielded a score of 0.020, which indicated that for 100 individuals with a diagnosis of high ACL injury risk by the KAA threshold, 2 individuals would be likely to benefit from interventional treatment.
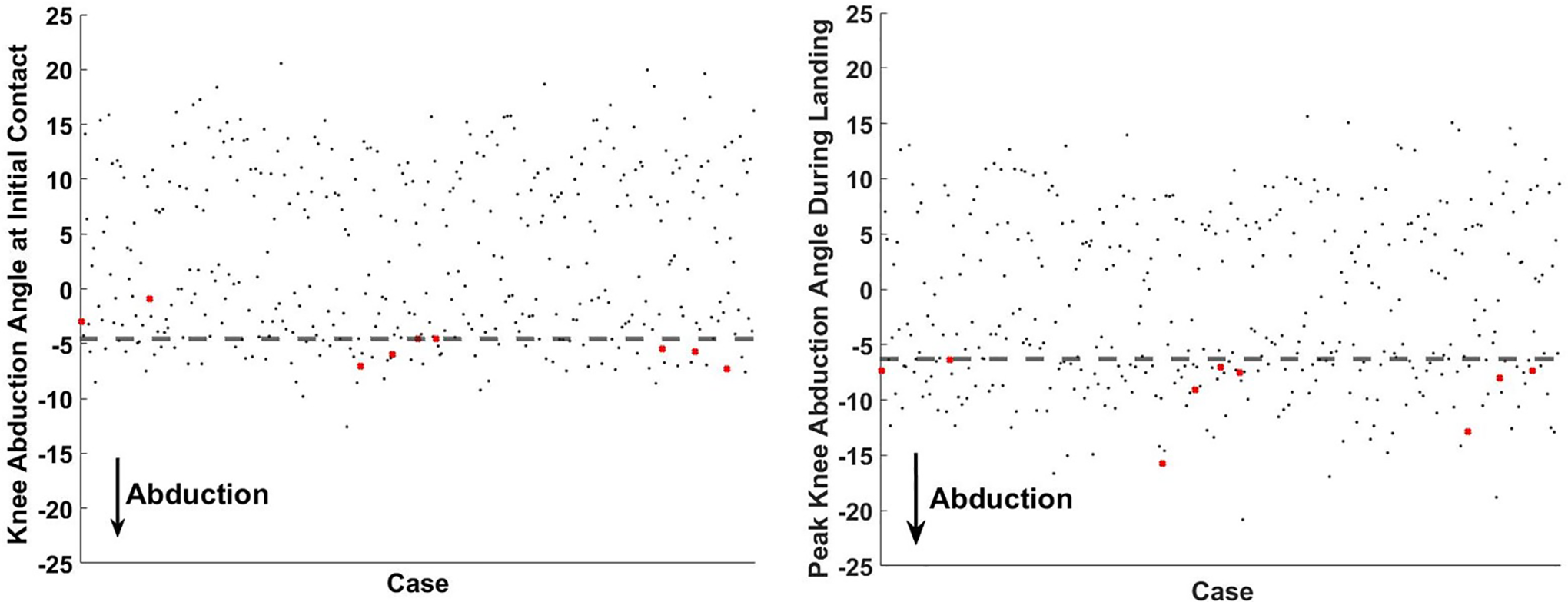
Scatterplot of knee abduction angle (KAA) at initial contact and peak KAA during landing from a drop vertical jump. Participants who had anterior cruciate ligament injury are marked with a red X. Cutoff thresholds for each respective condition are represented by the horizontal dashed lines.

Boxplot of median peak knee abduction angle (KAA) (blue), KAA at initial contact (IC) (red), and ΔKAA (green) during landing from a drop vertical jump. *Significant difference in KAA magnitudes between injury groups. KAA is represented as a negative value in our coordinate axis. The horizontal dashed line indicates no frontal plane rotation between the femoral and tibial axes. **Significant difference in peak KAA and KAA at IC within injury group. ACL, anterior cruciate ligament.
The magnitude of KAA at IC was also significant between those who went on to experience ACL injury and the referent uninjured cohort (P < .01) (Figure 3). KAA at IC was significantly more prevalent in athletes who went on to sustain ACL injury (–5.0° ± 2.0°) than their uninjured counterparts (3.4° ± 7.6°; P < .01). For both groups, KAA expressed reduced magnitude at IC compared with at its peak value (P ≤ .01). Mean knee orientation at IC was in adduction for uninjured athletes and abduction for athletes who had ACL injury. Logistic regression analysis demonstrated that KAA at IC significantly identified ACL injury status (P < .01) in the ACL-injured group compared with healthy controls, with a sensitivity of 78% and specificity of 83% (threshold, –4.6°; area under the curve, 0.843).
In Vitro Cadaveric Simulation
During simulated landings on the cadaveric specimens, KAA was directly correlated with peak absolute ACL strain (P < .01; r 2 = 0.24) (Figure 4) and change in ACL strain from the initial limb position (P < .01; r 2 = 0.25). Absolute ACL strain increased 0.47% ± 0.05% per degree of KAA. Similarly, the change in ACL strain from initial position increased by 0.44% ± 0.04% per degree of KAA. Peak absolute ACL strain (P < .01; r 2 = 0.27) and change in ACL strain from initial position (P < .01; r 2 = 0.22) were likewise directly correlated with applied KAM. Peak absolute ACL strain increased by 0.030% ± 0.003% per N·m of KAM applied. Similarly, the change in ACL strain from initial position increased by 0.025% ± 0.002% per Newton-meter of KAM applied.
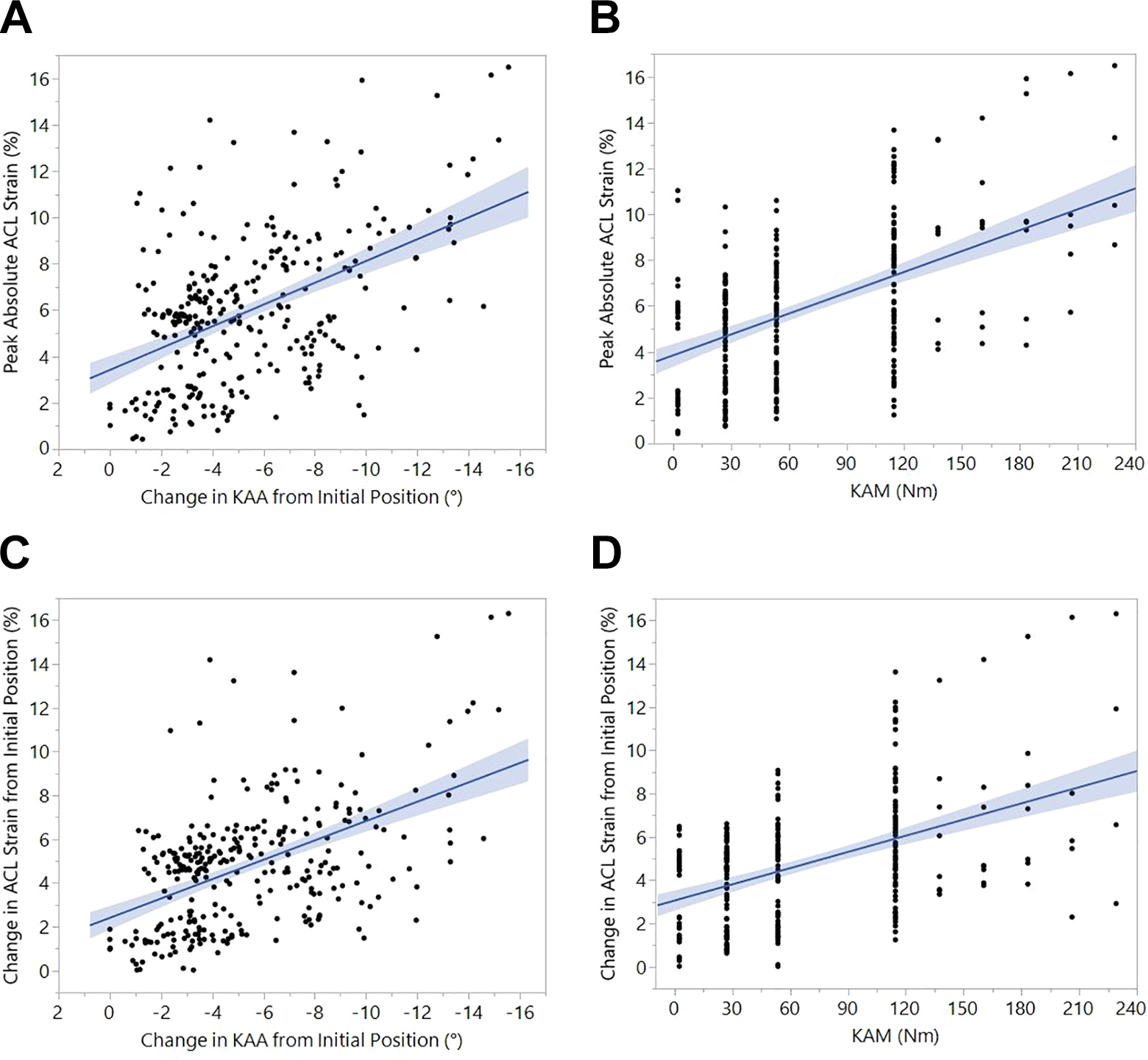
Scatterplots showing the linear correlations from the impact simulations between (A) peak absolute anterior cruciate ligament (ACL) strain and change in knee abduction angle (KAA), (B) peak absolute ACL strain and applied knee abduction moment (KAM), (C) change in ACL strain and change in KAA, and (D) change in ACL strain and applied KAM. Increased change in KAA during landing was correlated with increased peak ACL strain (A, C). Likewise, increased KAM applied to the joint also produced increased peak ACL strain during landing (B, D).
Derivation of In Vivo ACL Strain
When the specimen did not have external loads applied to the knee, the intercept values from the linear regression models indicated that baseline (y-intercept) ACL strain ranged between 3.4% and 3.8%. When used as the intercept value in the linear strain model derived above, these baseline strains along with in vivo KAA measurements estimated that athletes who went on to develop ACL injury achieved mean peak ACL strains of 7.7%-8.1% ± 1.5% while landing from a DVJ (Figure 5). The uninjured cohort achieved mean peak ACL strains of 4.1%-4.5% ± 3.6%. At IC, the estimated mean ACL strain was 5.7%-6.2% ± 0.9% for participants with ACL injury and 1.8%-2.2% ± 3.6% for uninjured participants. ACL strain estimates from our model were greater for participants who went on to ACL injury than for uninjured participants. This was observed at both peak strain and IC (P < .01).

Bar plot depicting the lower and upper ranges of estimated peak anterior cruciate ligament (ACL) strain achieved in both participant groups during a laboratory drop vertical jump task that was performed from a 31-cm drop height. IC, initial contact.
Discussion
The present investigation was designed to synthesize in vivo kinematics and cadaveric simulation data from landing tasks in order to quantify the role of KAA as a predictor of both ACL strain and future ACL injury. Our results supported the primary hypothesis that KAA would be a significant predictor of ACL injury risk via retrospective calculation from in vivo motion analysis data. Peak KAA predicted ACL injury status with 100% sensitivity and 65% specificity, whereas KAA at IC predicted ACL injury status with a sensitivity of 78% and specificity of 83%. These data further corroborate the findings of multiple earlier studies that frontal plane knee dynamics are essential to the screening and identification of athletes at high risk for ACL injuries, as KAA presented greater sensitivity and only slightly lower specificity than did KAM, which has long been used to project relative ACL injury risk. 34,41,51,53 –55 Specifically, previous literature has established that athletes who exceeded a threshold of 25.3 N·m KAM during landing had a 6.8% risk for subsequent ACL injury compared with a 0.4% risk in athletes who were below this threshold. 51 The present results established that KAA thresholds of 4.6° at IC or 6.3° at peak magnitude similarly demonstrate greater risk for subsequent ACL injury. This KAA model demonstrated that athletes who went on to sustain subsequent ACL injury were once again clustered at the cohort’s outermost quartile of frontal plane deviation when landing from a drop (Figure 2), which emphasizes that this subset of the athletic population is at elevated risk for noncontact knee injuries. Because the athletes who did not experience subsequent ACL injury presented mean frontal plane angles with slight knee varus, athletes who exhibit knee valgus angles during controlled athletic tasks should be considered at higher risk for injury than should those who do not exhibit valgus angles.
The evidence of prospective KAM measures as a predictor of an underlying injury risk factor has led to extensive research into the reduction of KAM through targeted neuromuscular intervention, which has subsequently been shown to reduce risk of all ACL injuries by 50% and noncontact ACL injuries in female participants by 67%. 6,38,41,49,65,66,70 The present in vivo motion analysis data indicate that KAA is as strong a predictor as KAM for ACL injury risk. Because KAA is easier to observe clinically than is KAM, this measure offers the potential for more simplified clinical assessment of successful injury risk reduction through interventional training. Athletes at the highest risk for injury also have demonstrated the greatest biomechanical changes after neuromuscular training. 36 The optimal ROC sensitivity and specificity for ACL injured versus uninjured groups in the present study provided a threshold cutoff for high-risk athletes as discerned using KAA.
The additional primary hypothesis that KAA would be a significant predictor of ACL strain during cadaveric simulations of landing was also supported. As the change in KAA from baseline increased, so did both peak ACL strain and change in ACL strain during simulated landings. Because each of the cadaveric KAA versus ACL strain correlations expressed r 2 ≥ 0.24, inclusion of KAA in a predictive model related to ACL loading and injury risk would subscribe to the 5-factor maximum systematic selection of logistic regression. 43 This theory indicates that screening models should limit the collection burden on clinicians through the inclusion of only those independent factors that account for ≥20% of the variance in the dependent factor. 6,41,43 As with the in vivo kinematic data, cadaveric simulation of KAA and KAM presented similarly robust models relative to ACL dynamics, as r 2 values were between 0.22 and 0.27. Similarly, when cadaveric simulation KAA and KAM values were applied to their respective regression models, they produced comparable changes in ACL strain. Peak cadaveric change in KAA ranged from 0.0° to 15.6°, which, based on the linear equation derived in this investigation, estimated an increase of 8.3% in peak ACL strain. Meanwhile, peak cadaveric change in KAM ranged from 2.4 to 229.2 N·m for an estimated increase of 7.8% in peak ACL strain. Previous experiments have demonstrated that KAM magnitude directly alters ACL strain during cadaveric simulations of landing and has a greater influence than do internal tibial rotation moments. 9,15 The present data corroborate that frontal plane knee kinematics are similarly predictive of ACL loading and strain.
ACL injuries remain multifactorial events, the combination of multiplanar biomechanical loading, anatomic geometry, and physiological factors. 19,20,40 Consequently, it may be presumptuous to expect a single factor to account for the majority of variance in predictive modeling of injury risk or strain. Indeed, the present KAA factor accounted for ∼30% of the variance in ACL strain. However, accounting for only a portion of the variance in a model does not preclude single factors from clinical relevance. In the present study, KAA accounted for sufficient variance to be designated for inclusion in predictive models. 43 In the clinical setting, reduction of factors is essential for speed and ease of analysis. Indeed, clinical surrogates are able to predict KAM and ACL injury risk with 84% sensitivity and 67% specificity through the use of several less rigorous factors. 54,55 Similarly, a robust single factor could be used to power a clinically relevant predictive model. Net benefit analysis of the current KAA data indicated that of 100 participants diagnosed as having high risk for ACL injury, 2 participants would benefit from interventions. This relates to literature reporting that the number needed to treat to prevent an ACL injury is 108; however, the previous literature study did not isolate high-risk participants before intervention. 66 Therefore, although use of a single factor increases the variance in a predictive model, the findings may still be generalizable to the clinical setting.
Finally, our results supported the experimental hypothesis that linearly estimated change in ACL strain derived in vivo would be higher in those participants who went on to develop injury than in healthy controls. The ACL strain estimates for the injured group that were derived from our model were greater than those for the uninjured group. During robotically controlled simulations of DVJ landings and sidestep cutting tasks, peak ACL strain in both a male and a female model ranged from 6.1% to 7.0%. 11 These robotic models were constrained by 3D kinematics recorded in vivo from a pair of high-risk athletes (as determined using KAM) who performed the prescribed athletic tasks. 10 The congruency of the present data with these previous cadaveric simulation data is a validation of consistency across 3 unique models (in vivo 3D motion capture, cadaveric robotic articulation, and cadaveric impact simulation) that have been developed within our laboratory. 10,16,41 These current findings demonstrate potential for cadaveric simulation results to be generalized relative to dynamic in vivo environments. The subsequent application of an in sim approach provides the potential to further enhance injury prevention, surgical intervention, and rehabilitation techniques. 59
Techniques for the rapid identification of injury risk in athletes from clinical measures are important because the literature has shown that interventional techniques can effectively alter high-risk biomechanics. 35,36,65 Specifically, athletes who are classified as having the highest risk for ACL injury based on latent profile analysis are subsequently the most susceptible to risk reduction through a targeted neuromuscular intervention focused on the proximal leg and trunk. 36,37 Such intervention has been shown to increase hip external rotation moments and impulses, increase peak trunk flexion, and decrease peak trunk extension during landing. In addition, preventive interventional training that is inclusive of strengthening exercises and proximal control exercises has been found to reduce ACL injury incidence, whereas training that includes balance exercises has not. 65 Biomechanical alterations from training interventions are such that 1 ACL injury is estimated to be prevented for every 108 to 120 athletes who are trained. 66 In addition, athletes who demonstrate increased frontal plane mechanics are more responsive to targeted neuromuscular training. 50,56,64 More simple clinical identification of high-risk athletes through a KAA measurement may further increase this ratio of responsiveness by parsing out individuals who are most apt to have poor biomechanics altered from a neuromuscular intervention.
Ultimate strain to failure in the ACL is between 15% and 18%. 17,21,47,72 Based on this range, the mean predicted strain values for the ACL injury group in this report accounted for between 43% and 54% of this ultimate failure range. Accordingly, even those athletes who eventually went on to injury did not approach the threshold of injury during a controlled, unobstructed, unperturbed landing in a laboratory. The safety factor of a laboratory landing task, as estimated using the current linear equation, was approximately double the value of strain that was calculated. This is empirically supported, as no adverse events have been reported during thousands of DVJ screening tasks performed in our laboratories. The peak ACL strain presented contradicts some existing literature that indicates that peak ACL strain during gait approaches 12.5%. 25 Because gait generates significantly less ground-reaction force than do landing tasks, 4,23 it remains controversial that gait would produce greater ligament strain than has been measured in more dynamic athletic tasks. 11,13,15 Furthermore, in vivo implantation of strain gauges on the ACL has shown baseline strain of 2% to 3% for weightbearing individuals at 20° of knee flexion. 27 Because high-risk external knee loads generate ∼3% ACL strain and impulse delivery likewise contributes ∼3% strain, 14 the ∼8% peak ACL strain during landing that was estimated from the present linear model is better aligned with data collected from sensors directly implanted in tissue in vivo and in vitro. However, the post-IC change in ACL strain estimated for live athletes in the current study was approximately 1% less than what has previously been observed in cadaveric simulation. 15
A first limitation of the current investigation was the inability to directly instrument the ACL of in vivo participants with miniature strain gauges. Such a procedure would provide for the easiest direct assessment of correlation between KAA and ACL strain; however, it would also be highly invasive and introduce unnecessary risk to the participant. Further, in a clinical or athletic setting, direct evaluation of ACL strain will likewise not be available, so the surrogate cadaveric simulation measurements used presently remain appropriate. A second limitation was that the participants in the in vivo cohort and cadaveric limb cohort were different people. Care was taken to minimize the age difference between participants in the in vivo kinematic and cadaveric simulation by exclusion of cadaveric specimens from individuals who were outside the fifth decade of life, had undergone chemotherapy treatments, and were either sedentary or restricted to bedrest. 16,17 A third limitation to this investigation was that the in vivo kinematic cohort consisted of female participants, whereas the cadaveric simulation cohort included both male and female specimens. The female ACL has decreased mechanical failure properties compared with those of the male ACL and on average expressed 3.2% more strain during impact simulations than that of the male ACL, although this value was not statistically significant. 22,32,33,60 The present in vivo kinematic cohort was entirely composed of female participants. Thus, based on the 8:3 male-to-female ratio of cadavers, it is possible that the cadaveric-simulation-derived linear model underestimated the peak in vivo ACL strain by as much as 2.3%. Application of this increase would raise the mean ACL strain in the ACL injury group to 9.5% to 10.0% and account for 53% to 67% of the ultimate failure strain. These values would leave a safety factor for injury avoidance during the performance of DVJ tasks but would indicate that high-risk athletes are significantly closer to injury during controlled tasks than anticipated. A fourth limitation exists in that all in vitro simulations were restricted to 25° of knee flexion. Although this represents the mean knee flexion at IC during in vivo landings for an athletic population, 5 future investigations should explore the role of varied IC flexion angles and passive knee flexion during landing relative to intra-articular ligament mechanics. A fifth limitation as pertains to the in vivo cohort is that neither playing time nor previous injury history was controlled. Playing time affects athletic exposures, which relates to injury incidence. We noted that 2 athletes had previous ACL reconstruction and went on to further ACL injury during the study. It is known that athletes with previous history of ACL injury are significantly more likely to experience subsequent injury than is the general population. 57,68,69 Sixth, the possibility for sampling bias exists because the current cadaveric population expressed greater age, mass, and body mass index than did either in vivo cohort. Unfortunately, this reality is unavoidable when working with cadaveric specimens because young and healthy specimens are painstakingly difficult to acquire. Larger specimens may be indicative of larger ligaments with greater cross-sectional area that can sustain high ultimate failure forces. However, our primary cadaveric outcome measure was ligament strain, which is, by definition, normalized to specimen size. Given that the literature had not associated ultimate ligament strain with specimen characteristics, the expectation of bias influence on the current data is not warranted at present.
Conclusion
KAA identified relative ACL injury risk in vivo and ACL strain during cadaveric simulation. The association between these environments allows clinicians to make an in vivo estimate of peak ACL strain of 0.5% per degree of KAA observed during landing. KAA proved to be similarly robust in prediction of ACL injury risk as a KAM-driven model. Because KAA is simpler to observe clinically than is KAM, the implementation of this factor in neuromuscular interventions could lead to less arduous assessment of training effectiveness in at-risk populations.
Footnotes
Acknowledgment
The authors acknowledge the nonauthorship contributions of Kevin R. Ford, Alexander W. Hooke, and Aaron J. Krych to this investigation.
Final revision submitted April 16, 2020; accepted April 29, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding for this study was provided by an NBA/GE Collaboration grant. Fellowship funding (ALM) was provided by the Mayo Clinic Graduate School of Biomedical Sciences. National Institutes of Health funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases included grants K12HD065987 and L30AR070273 (to N.D.S.), T32AR056950 (to R.F.H.), R01AR055563 (to N.A.B.), and R01AR056259 (to T.E.H.). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from The Ohio State University (ID No. 2012H0281), the Cincinnati Children’s Hospital Medical Center (ID No. 2008-0023), and the Mayo Clinic (ID No. 15-005819).
