Abstract
Background:
Progressive high-load exercise (PHLE) has led to positive clinical results in patients with patellar and Achilles tendinopathy. However, its effects on rotator cuff tendinopathy still need to be investigated.
Purpose:
To assess the clinical effects of PHLE versus low-load exercise (LLE) among patients with rotator cuff tendinopathy.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
Patients with rotator cuff tendinopathy were recruited and randomized to 12 weeks of PHLE or LLE, stratified for concomitant administration of corticosteroid injection. The primary outcome measure was change from baseline to 12 weeks in the Disabilities of the Arm, Shoulder, and Hand (DASH) questionnaire, assessed in the intention-to-treat population.
Results:
A total of 100 patients were randomized to PHLE (n = 49) or LLE (n = 51). Mean changes in the DASH questionnaire were 7.11 points (95% CI, 3.07-11.16) and 8.39 points (95% CI, 4.35-12.44) in the PHLE and LLE groups, respectively; this corresponded to a statistically nonsignificant adjusted mean group difference of −1.37 points (95% CI, −6.72 to 3.99; P = .61). Similar nonsignificant results were seen for pain, range of motion, and strength. However, a significant interaction effect was found between the 2 groups and concomitant corticosteroid use (P = .028), with the largest positive change in DASH in favor of PHLE for the group receiving concomitant corticosteroid.
Conclusion:
The study results showed no superior benefit from PHLE over traditional LLE among patients with rotator cuff tendinopathy. Further investigation of the possible interaction between exercise type and corticosteroid injection is needed to establish optimal and potentially synergistic combinations of these 2 factors.
Registration:
NCT01984203 (ClinicalTrials.gov identifier): Rotator Cuff Tendinopathy Exercise Trial (RoCTEx).
Shoulder pain is a frequent complaint in primary and secondary care. 23,39,43 Subacromial impingement syndrome (SIS) is estimated to account for one-third of all shoulder-related health care contacts. 13,56 The definition of SIS covers an umbrella of different underlying pathological disorders, such as rotator cuff syndrome, tendinitis, and bursitis, rather than a specific diagnosis. 22,36 Consequently, some authors avoid the term SIS. 34,35,47 The diagnosis of rotator cuff tendinopathy is often used synonymously with SIS, but rotator cuff tendinopathy should be confined to tendon-related pain with weakness, especially during elevation and external rotation, largely preserved range of motion, and minimal resting pain. 34,36,37 These clinical signs and symptoms roughly differentiate patients with rotator cuff tendinopathy from those having signs of bursitis, which include more constant pain and, often, more pronounced decreased range of motion. 36 The diagnostic accuracy of existing orthopaedic tests are too low to clinically differentiate rotator cuff tendinopathy from other shoulder disorders. 19,47 However, ultrasound may be used to increase the diagnostic accuracy of patients with shoulder pain. 47 Ultrasound has high specificity in diagnosing changes related to tendinopathy, 41,45,47 and it is reliable when performed in a standardized manner. 25 High specificity is especially important when allocating patients to treatment strategies in a randomized controlled study.
Danish national guidelines recommend a minimum of 3 months of physical therapy before surgery is considered. 53 However, methodologically rigorous studies are necessary to evaluate the effectiveness of specific exercise regimens for rotator cuff tendinopathy. 36 These studies should focus on strengthening exercises of different intensity and frequency, as compared with an active control group. 36
Eccentric/progressive heavy slow-resistance training has shown positive results on tendinopathy in the knee and ankle, with clinically relevant improvement in pain and function, as well as structural changes verified by ultrasound. 2,31,32 For rotator cuff tendinopathy, loaded eccentric exercises have proved superior to unloaded exercises 20 and equal to traditional rotator cuff training 40 in reducing pain and improving function. The superiority of progressive high-load exercise (PHLE) has been demonstrated only when compared with passive controls. 38
Concomitant corticosteroid injection has frequently been administered to enhance the beneficial effects of exercise. 53 Corticosteroids decrease inflammation and edema in the surrounding tissue, thereby decreasing pain in these structures. The precise mechanism of action of corticosteroids in rotator cuff tendinopathy is not entirely clear, 6 and the influence of corticosteroid injection on exercise is unknown.
The primary aim of this study was to evaluate whether PHLE is superior to traditional low-load exercise (LLE) among patients with rotator cuff tendinopathy. We hypothesized that after 12 weeks of exercise, the intervention group would have a significant improvement in the primary outcome measure (Disabilities of the Arm, Shoulder, and Hand [DASH] questionnaire score), 21 with a decrease in shoulder pain and an increase in shoulder strength. The secondary aim was to evaluate the interaction of a concomitant corticosteroid injection in both PHLE and LLE exercise programs.
Methods
Trial Design
The multicenter RoCTEx (Rotator Cuff Tendinopathy Exercise) trial was a randomized controlled superiority trial stratified by corticosteroid injection (yes/no), with observers and patients blinded. The primary endpoint was 12 weeks after baseline. Details on the trial are published in Ingwersen et al. 24
Participants, Settings, and Locations
Between November 2013 and June 2015, participants with rotator cuff tendinopathy were recruited from 3 orthopaedic shoulder clinics at hospitals in western Denmark, supplemented by advertisements in local newspapers and sports/fitness clubs. Patients were seen at the nearest shoulder clinic for a standard clinical examination and assessment. Eligible participants were adults aged 18 to 65 years with shoulder symptoms lasting for a minimum of 3 months, in addition to pain in the proximal lateral aspect of the upper arm, aggravated by abduction. Further eligibility criteria were signs of rotator cuff tendinopathy verified by ultrasound, including at least 1 of the following: tendon swelling, presence of hypoechoic areas, calcification, fibrillar disruption, or neovascularization in the supraspinatus tendon. The primary investigator performed all ultrasound measurements, in a standardized manner, which has shown good to excellent reliability for detecting pathological changes of rotator cuff tendinopathy. 25 Additionally, participants had to have a positive full can, 28 Jobe, 26 or resisted external rotation test 29 as well as a positive Hawkins-Kennedy 18 or Neer test. 46
Patients were excluded if they had resting pain >40 mm on a visual analog scale (VAS; range, 0-100; 0 = no pain), 5 <90° active elevation of the arm, a full-thickness rotator cuff tear (verified by ultrasound), a corticosteroid injection within the previous 6 weeks, or competing diagnoses. 24
Exercise Programs
Patients were randomized to the PHLE (intervention) or LLE (comparator) group. Both groups received the same exercises, attention, and basic information. Patients were seen by a physical therapist for initial exercise instruction (60 minutes) in week 1 and for supervised exercise sessions (30 minutes) in weeks 2-4, 6, and 9 and were instructed to perform home-based exercises 3 times per week.
The exercise program consisted of 2 exercises for the scapula-stabilizing muscles, 2 for the rotator cuff muscles, and 2 mobility exercises for the rotator cuff and scapulo-thoracic complex. The specific rotator cuff exercises were chosen according to studies showing the highest amount of activation in relation to the deltoid and taking the risk of impingement into consideration. 12,49 –51 The only between-group difference was progression of load for the rotator cuff exercises. The PHLE group gradually increased its load from a 15-repetition maximum in week 1 to a 6-repetition maximum in weeks 9 to 12, allowing patients to perform isometric exercises if pain exceeded 50 mm on VAS. The control group performed a 20- to 25-repetition maximum from weeks 1 to 12 (for further description, see Appendices A and B).
Orthopaedic specialists, in accordance with the local standard procedures and in collaboration with the patients, based a potential concomitant administration of corticosteroid injection on their clinical evaluation. In general, a corticosteroid injection was considered applicable in patients with increased pain at night or with active movements of the arm, presence of inflamed bursa (verified on ultrasound), or signs of Doppler activity around calcifications. The corticosteroid injection was administered to patients after baseline assessment but before randomization.
Outcomes
The primary outcome was a change in DASH score (primary/activities of daily living = 0-100, 0 = no problems) from baseline to week 12. 21 Secondary outcomes included DASH scores at weeks 1-4, 6, and 9; perceived pain at rest, during general activities, and at night and maximum pain during the previous 24 hours (on VAS) 5 ; and maximum isometric voluntary contraction (IsoForce Dynometer EVO2; Medical Device Solutions AG) and active/passive range of motion (ROM) (HALO digital goniometer; HALO Medical Devices) for scaption and external/internal rotation (see Appendix C). Pilot studies of strength (intraclass correlations = 0.84-0.92) and ROM measurements (intraclass correlations = 0.75-0.96) have shown excellent 14 interrater reliability. Furthermore, explorative measurements included standardized ultrasound measurements obtained to verify changes consistent with tendinopathy, including tendon thickening versus the contralateral nonaffected tendon, signs of fibrillar disruption, presence of calcification, and neovascularization (dichotomized: yes/no). 25,48 These ultrasound changes are associated with tendinopathy. 54,57
Patients were instructed to complete an exercise diary. Good compliance was defined as attending at least 4 of 6 visits to the physical therapist and completing >80% of the exercises at home (reported in the diary). 24
Data Collection
The primary investigator performed all baseline and follow-up assessments. Fifteen pilot patients were assessed for adjustment and training of inclusion and baseline/follow-up procedures. The treating physical therapists were trained to teach patients how to perform the exercise programs and administer the DASH questionnaire.
Sample Size and Power Calculations
Given studies on patients with SIS, 4,8,20,27,33,42,55 we expected a mean baseline of 40 ± 17 points in the DASH questionnaire, 50% change in the PHLE group, and a 25% change in the LLE group. For a 2-sample pooled t test of a normal mean difference (2-sided significance level of 0.05, P ≤ .05), 100 patients per group were required to obtain a power of at least 98.5% for detecting a mean difference of 10 DASH points between the 2 groups. For the second study objective of examining the interaction between corticosteroid use and exercise group, 260 patients in total were required to achieve 90% power.
A priori, the final deadline for inclusion for this study was March 2015. 24 However, this was postponed to June 2015. From November 2013 to June 2015, 100 participants were included; a substantially lower inclusion rate than expected resulted in a power of 0.84 for the primary aim.
Randomization and Allocation Concealment
Patients were randomly assigned to PHLE or LLE with a 1:1 allocation per center, stratified by administration of concomitant corticosteroid injection. A computer-generated randomization schedule with permuted blocks of random sizes (2-6) was used to prepare opaque, sealed, sequentially numbered envelopes placed in 2 separate ring binders (corticosteroid: yes/no) for each center. A university secretary prepared the envelopes for each center, while local secretaries performed the allocation procedure by taking the next prepared and sealed envelope.
Blinding
Baseline measurements were performed before group allocation. At follow-up, the primary investigator strongly encouraged patients not to disclose their allocated exercise program. Patients were informed that 2 active exercise strategies were compared, but they were blinded regarding group differences and the hypotheses.
Statistical Analysis
For primary efficacy analysis in the intention-to-treat (ITT) population (all randomized patients independent of compliance and withdrawals), the between-group difference in DASH change (baseline to 12 weeks) was calculated, with baseline observation carried forward used for missing data. Analysis of covariance was used to compare mean changes from baseline in DASH scores, as well as the secondary continuous outcomes. The primary model included change from baseline as the dependent variable, with treatment group (PHLE or LLE), corticosteroid status (yes or no), and center site (Nos. 1-3) as the main effects and with baseline score as additional covariate. In the analysis of the secondary aim, the interaction for group and corticosteroid was added to the primary model. Results are expressed as the difference between group means (95% CIs) with associated P values.
To analyze the longitudinal element of time effects on the DASH score (repeated measures at 1-4, 6, 9, and 12 weeks), a mixed linear model approach was used, based on restricted maximum likelihood estimates of the parameters, with treatment group (PHLE or LLE), corticosteroid status (yes or no), and center site (Nos. 1-3) as main effects and with baseline score as an additional covariate.
For the effect on ultrasound-verified structural changes, differences in change between groups were analyzed by a chi-square test.
All ITT analyses were performed blinded to group allocation (groups were named A and B in the data set). Sensitivity analyses for missing values were performed on the ITT population, first with imputation of the mean change value of group A as a positive value and group B as a negative value, and then combined oppositely. All ITT data analyses were carried out according to the pre-established analysis plan. 24 An external statistical consultant performed analyses on the primary outcomes. Before group allocation was unblinded, the primary conclusion was approved by all authors. Analyses were performed with Stata/IC 14 (2015; StataCorp), with P < .05 considered statistically significant.
Ethics and Registration
The Regional Scientific Ethics Committee of Southern Denmark approved the trial on June 27, 2013 (project S-20130071). The trial was registered with the Danish Data Protection Agency and approved on May 30, 2013 (registration 2008-58-0035). The trial is registered at Clinicaltrials.gov (NCT01984203). The study followed the principles of the Declaration of Helsinki, 58 and informed consent was obtained from all participants before participation.
Results
A total of 103 patients from hospitals and 47 patients from external recruitments were assessed for eligibility. Of these, 100 patients who fulfilled the eligibility criteria and signed informed consent to participate constituted the ITT population; 49 patients were randomized to PHLE and 51 to LLE (Figure 1). A total of 93 patients (PHLE, n = 44, 90%; LLE, n = 49, 96%) participated in the follow-up assessment and constituted the “as observed” population. Fifty-nine patients (PHLE, n = 28, 57%; LLE, n = 31, 61%) were compliant with the exercise protocol and constituted the “per protocol” population.

Flowchart of participants with rotator cuff tendinopathy in the RoCTEx trial intention-to-treat (ITT) population. Two patients did not receive allocated intervention: 1 had complications after knee surgery (progressive high-load exercise [PHLE]), and 1 resigned from participation before starting physical therapy (low-load exercise [LLE]). Five patients (4 in the PHLE group and 1 in the LLE group) could not be reached for follow-up appointments. Therefore, the baseline observation carried forward technique was performed on these 7 patients, corresponding to an ITT population of 100 patients. A total of 93 patients (PHLE, n = 44, 90%; LLE, n = 49, 96%) constituted the “as observed” population for the primary outcome. These included 14 patients who discontinued the assigned intervention (PHLE, n = 9, 18%; LLE, n = 5, 10%) because of increased shoulder pain (PHLE, n = 1; LLE, n = 1), referral for a shoulder operation (PHLE, n = 1; LLE, n = 1), other concomitant sickness (PHLE, n = 5; LLE, n = 3), or increased burdens at work (PHLE, n = 2). However, they completed the DASH questionnaire at 12 weeks. From these 14 participants, 10 were not willing or able to participate in the physical follow-up tests assessing ultrasound, visual analog scale, strength, and range of motion, resulting in 83 patients (PHLE, n = 39, 80%; LLE, n = 44, 86%) in the “as observed” population for these measurements. A total of 59 patients (PHLE, n = 28, 57%; LLE, n = 31, 61%) were compliant with the exercise protocol and were included in the per-protocol analysis.
At baseline, demographic and clinical characteristics were not different between groups, except for a statistically significantly higher Hospital Anxiety and Depression score 60 in the PHLE group (Table 1). Furthermore, the DASH baseline score was significantly higher in patients receiving concomitant corticosteroid injection (mean ± SD, 27.64 ± 10.75) than in those not receiving (22.83 ± 11.70, P = .048). Demographic and clinical characteristics of the participants who did not complete the follow-up tests were not different from those with a complete data set (data not shown).
Demographic and Baseline Values for PHLE and LLE (N = 100) a

a Values are presented as mean ± SD and No. (%) unless otherwise noted. ADL, activities of daily living; BMI, body mass index; BW, body weight (kg); DASH, Disabilities of the Arm, Shoulder, and Hand (range, 0-100); HAD, Hospital Anxiety and Depression (range, 0-21); LLE, low-load exercise; N, newton; PHLE, progressive high-load exercise; ROM, range of motion; VAS, visual analog scale (range, 0-100).
b n = 98 (LLE = 51, PHLE = 47).
c n = 93 (LLE = 49, PHLE = 44).
d n = 85 (LLE = 45, PHLE = 40).
e n = 96 (LLE = 51, PHLE = 45).
f n = 99 (LLE = 51, PHLE = 48).
Efficacy Analysis
In the ITT population, there was no significant group difference in the adjusted change in DASH score, with robust results in sensitivity analyses. Furthermore, no significant between-group differences were found in any secondary outcomes (pain, strength, ROM). Both groups had, in general, significant improvements in DASH, pain, and strength, except for pain at rest. ROM significantly changed for only some of the movements (Tables 2 and 3).
Analysis of Changes in Intention-to-Treat Population (N = 100) for Primary and Secondary Outcomes 12 Weeks After Baseline for PHLE and LLE a
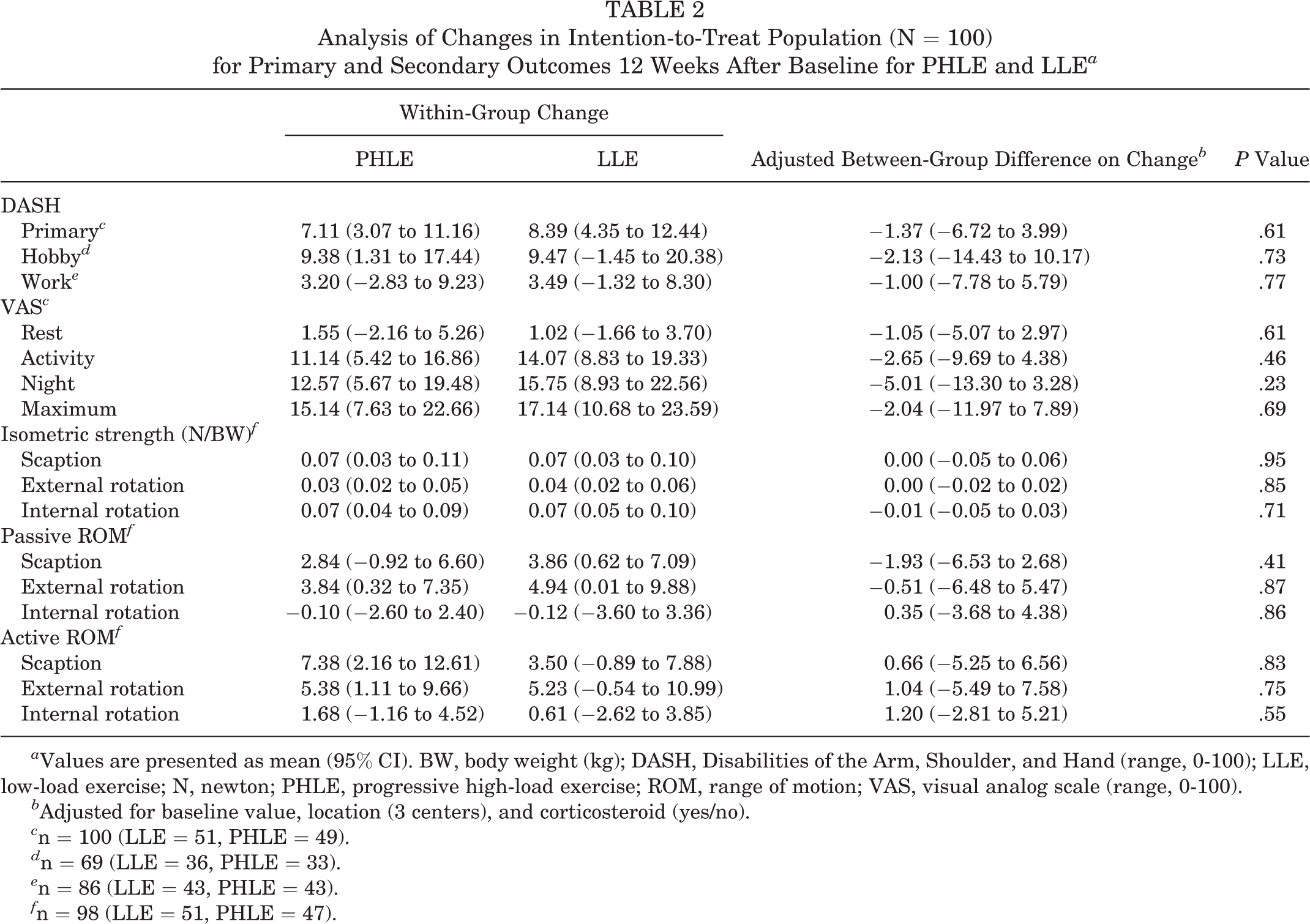
a Values are presented as mean (95% CI). BW, body weight (kg); DASH, Disabilities of the Arm, Shoulder, and Hand (range, 0-100); LLE, low-load exercise; N, newton; PHLE, progressive high-load exercise; ROM, range of motion; VAS, visual analog scale (range, 0-100).
b Adjusted for baseline value, location (3 centers), and corticosteroid (yes/no).
c n = 100 (LLE = 51, PHLE = 49).
d n = 69 (LLE = 36, PHLE = 33).
e n = 86 (LLE = 43, PHLE = 43).
f n = 98 (LLE = 51, PHLE = 47).
Proportion of Participants With Structural Changes Verified by Ultrasound at 12 Weeks in Intention-to-Treat Population (N = 100) a
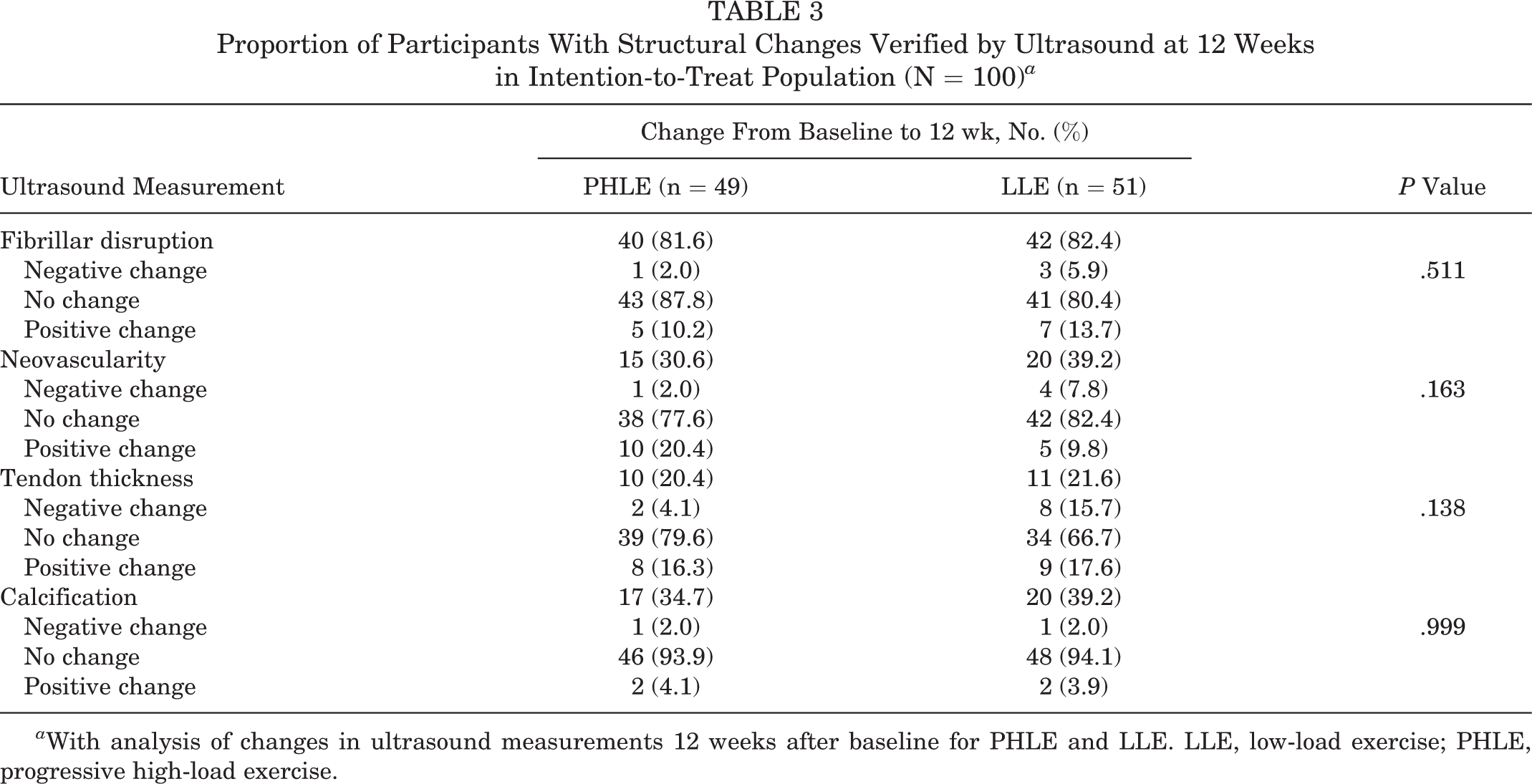
a With analysis of changes in ultrasound measurements 12 weeks after baseline for PHLE and LLE. LLE, low-load exercise; PHLE, progressive high-load exercise.
Analysis of Secondary Aim
A significant interaction was found between the exercise type/groups and concomitant corticosteroid injection (P = .028) on DASH. However, there was no significant main effect of group (P = .82) or concomitant corticosteroid injection (P = .62). The effect of PHLE was higher than that of LLE, although not significantly, among those receiving concomitant corticosteroids (adjusted between-group difference: 6.92 DASH points; 95% CI, −2.11 to 15.96). The effect of PHLE was lower than that of LLE, although not significantly, among those not receiving concomitant corticosteroids (adjusted between-group difference: −5.63 DASH points; 95% CI, −12.10 to 0.83) (Table 4, Figure 2).
Analysis of the Interaction Between the Exercise Groups and Concomitant Administration of Corticosteroid for PHLE and LLE in the Intention-to-Treat Population (N = 100) a

a Values are presented as mean (95% CI). CS−, no concomitant administration of corticosteroid injection; CS+, concomitant administration of corticosteroid injection; DASH, Disabilities of the Arm, Shoulder, and Hand (range, 0-100); LLE, low-load exercise; PHLE, progressive high-load exercise.
b Adjusted for baseline value and location (3 centers).
c n = 100 (LLE = 51, PHLE = 49).
d n = 69 (LLE = 36, PHLE = 33).
e n = 86 (LLE = 43, PHLE = 43).
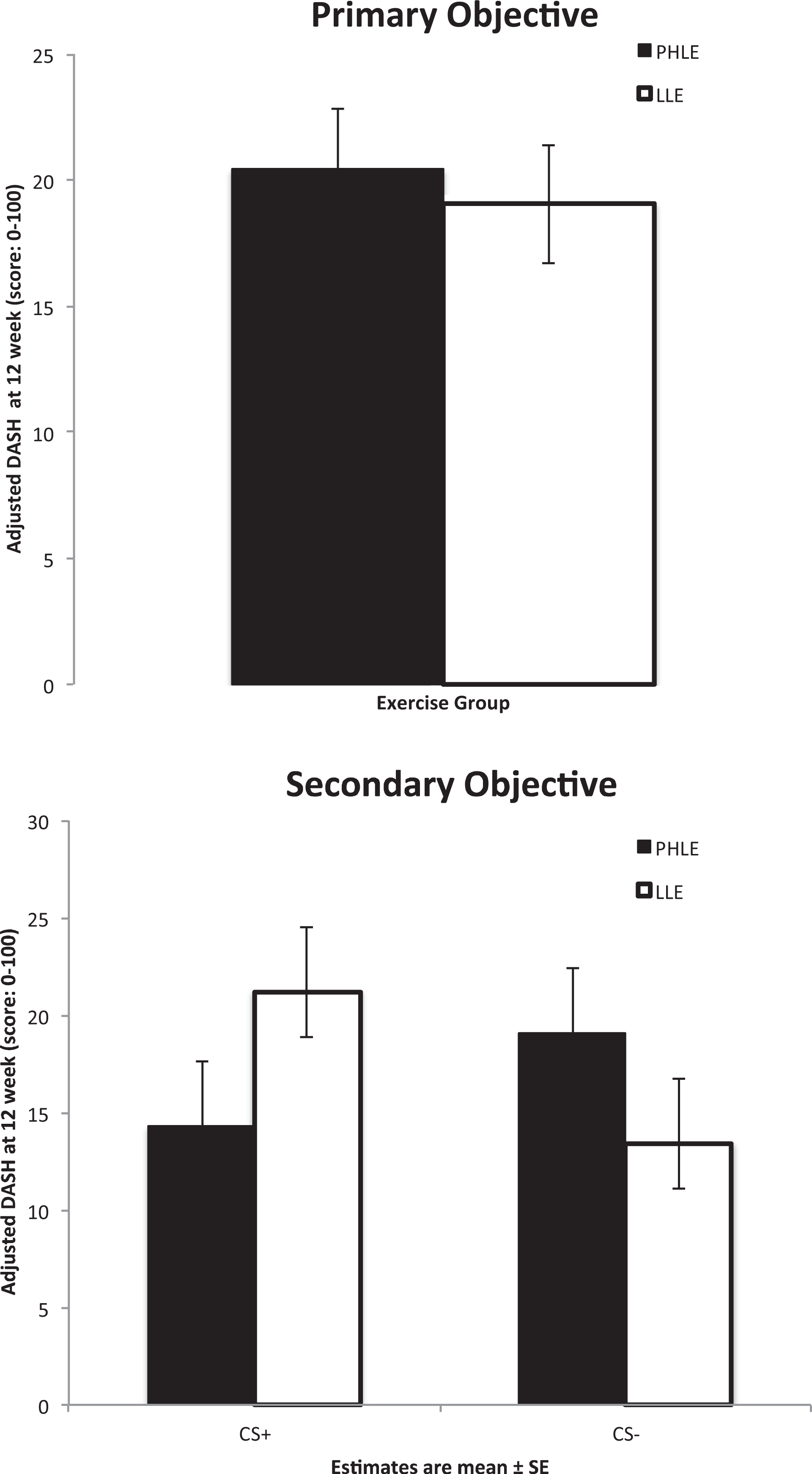
Main outcome: primary and secondary objectives in intention-to-treat analysis (N = 100). Primary objective: illustration of Disabilities of the Arm, Shoulder, and Hand (DASH) score at 12 weeks. Secondary objective: DASH score at 12 weeks stratified for concomitant corticosteroid injection. Estimates are mean ± standard error. CS+, concomitant corticosteroid; CS–, no concomitant corticosteroid; LLE, low-load exercise; PHLE, progressive high-load exercise.
Secondary Analyses
Repeated measures of DASH showed no between-group difference, as was the case with the as-observed (DASH, −1.19; 95% CI, −6.88 to 4.51) and per-protocol (DASH, −0.14 [95% CI: −6.20 to 5.92]) analyses, in the primary outcomes and with the secondary outcomes (pain, strength, and ROM; P > .30) (Appendix D).
A significant effect (P = .039) on ultrasound-verified neovascularity was observed in the per-protocol analysis in favor of PHLE, resulting in a 28.6% decrease in the number of patients presenting with neovascularity in the PHLE group, in contrast to only a 9.7% decrease in the LLE group, at 12-week follow-up.
Adherence to the Exercise Protocol
There was no between-group difference in compliance, corresponding to 28 (57%) being compliant in the PHLE group and 31 (61%) being compliant in the LLE group.
Adverse Events
Three patients were referred for operation within the study period because of worsening of symptoms, and 1 patient received a corticosteroid injection after randomization because of increased pain. There was no significant between-group difference in episodes of increased pain or loss of strength after training (Table 1).
Discussion
No superior benefit of PHLE was seen when compared with LLE in primary or secondary outcomes. In both groups, significant within-group improvements were found in primary and secondary outcomes of pain with activity, pain at night, maximum pain, strength, and passive external ROM. A significant interaction was found between concomitant administration of corticosteroid injection and the type of exercise. This interaction resulted in an increased, although not significant, effect in favor of PHLE for patients receiving corticosteroid injection. The interpretation of the results based on the interaction of exercise and cortisone injection should be considered in relation to the lack of power necessary to prove this secondary aim.
Explanation of Results
This study did not find a significant effect of PHLE in comparison with LLE. In contrast, Lombardi et al 38 and Holmgren et al 20 evaluated the effect of PHLE and eccentric exercises in comparison with a waiting list (passive control) or an unloaded movement exercise program with no progression (active control). Both studies showed a significant difference in the between-group analyses on DASH. However, it is questionable whether Holmgren et al (active control) defined the control exercise program sufficiently to conclude a significant effect of eccentric exercises over general exercises. In comparison, Maenhout et al 40 also evaluated the effect of eccentric exercises, in comparison with an active general exercise program, and they found no between-group differences.
The current lack of effect between exercise groups could arise from several reasons, such as the intervention length, diagnostic criteria, compensation strategies, and equal workload. First, in relation to the length of the intervention, 12 weeks of PHLE exercise may not be long enough to improve the patient’s tendon health, as tendon healing may take a longer time. Therefore, a longer exercise period may have been preferable to evaluate the efficacy of PHLE. Second, as the biomechanics of the shoulder girdle are complex and the differential diagnosis between SIS and rotator cuff tendinopathy is challenging, it remains unknown whether the absence of between-group differences is due to a proportion of patients not having rotator cuff tendinopathy as their main source of pain. The results of this study should be interpreted with this in mind. However, we did follow thorough inclusion procedures to ensure the presence of tendinopathy in the rotator cuff. Based on these methods—consisting of a general assessment performed by experienced orthopaedic specialists to exclude differential diagnoses, a combination of orthopaedic clinical tests to confirm pain arising from the subacromial structures, and a standardized ultrasound procedure 25 —the risk of a heterogeneous study population was considered to be small. Third, the absence of between-group differences may also be explained by the fact that tendon loading is biomechanically different in the complex shoulder joint, as compared with patellar and Achilles tendon loading, where PHLE has shown equal or superior effects versus eccentric exercises in treatment of tendinopathy. 2,7,31,32 Although rotator cuff tendinopathy shows similarities with patellar and Achilles tendinopathy, 3,9,30,36 the tendon loading on painful rotator cuff muscles may evoke inhibition of rotator cuff muscle activity. 11,44 As compensation, the deltoid muscle may increase its activity to perform the required task. 1,44 Consequently, the absorbed load of the rotator cuff tendon may be less than expected in a pain-free shoulder. As loading of the tendon appears important, 7 this potential protection/compensation mechanism may explain why the present study and others of eccentric/PHLE in rotator cuff tendinopathy 40 generally do not result in superior effects when compared with LLE. Fourth, another question may be whether the groups trained with different loads. According to exercise diaries from the per-protocol population, the PHLE group actually exercised with twice the amount of load (normalized to 1-repetition maximum at baseline) of the LLE group in the 2 rotator cuff exercises (data not shown).
This study found a significant interaction effect of corticosteroid injection and exercise type, which resulted in a higher effect of PHLE versus LLE in those patients who had a corticosteroid injection, while the opposite effect was present for those who did not have an injection. This may indicate that concomitant use of corticosteroid injection opens a “window of opportunity,” resulting in decreased pain inhibition in the rotator cuff muscles. This window may ensure that the rotator cuff muscles receive optimal stimulation, resulting in clearer changes, as those previously found in patellar and Achilles tendinopathy. 2,31,32 The role of inflammation within tendinopathy is controversial, 7 but recent evidence supports the occurrence of active inflammation. 10,52 Conversely, the addition of corticosteroids in relation to degenerative tendons is reported to negatively influence regenerative processes in tendons. 16,17,59 The combined risk and benefit must therefore be assessed when administering corticosteroid injection. However, a recent systematic review found that tendon rupture in relation to corticosteroid injections was uncommon, occurring in only 0.1% of tendons in general. 6 To confirm the current results and eventually evaluate the underlying mechanisms, further research of the interaction between exercise type and concomitant corticosteroid injection must be performed.
We found a positive effect of PHLE on structural changes verified by ultrasound among the patients who were compliant with the assigned intervention (per-protocol analyses). The signs of neovascularity disappeared in significantly more patients after the PHLE intervention versus LLE. Furthermore, more patients had decreased tendon thickness in the PHLE group (increased tendon thickness: 33% at baseline and 20% at follow-up in the PHLE group vs 24% and 22%, respectively, in the LLE group), although this difference was not statistically significant. Reduced tendon thickness is anticipated to be a strong indicator of improved function and diminished pain because of the reduced volume occupied by nonedematous tendon in the subacromial space. 34 However, per-protocol analyses are prone to bias, and results should be investigated further before accepting these conclusions.
The current within-group change in DASH score of 7.1 to 8.4 points (29% to 34%) is below the minimal clinically important difference (MCID), which for the DASH has been suggested to be 10.5 points for patients with general shoulder disorders. 15 However, the MCID should generally be considered in relation to the specific population, the specific intervention, and the baseline value. 42
The current baseline DASH was lower than in previous studies of patients with SIS, 4,8,20,27,33,42,55 indicating less discomfort in the current population at inclusion. The addition of recruitment by external advertisements did not appear to cause lower baseline scores, since exploratory analyses (data not shown) showed no differences in baseline scores between recruitment types. Another reason for the low baseline DASH score may be the minimal resting pain and largely preserved ROM often seen in patients with rotator cuff tendinopathy, 36,37 in contrast to higher DASH scores in SIS patients being attributed to a greater variety of symptoms. Camargo et al 4 evaluated the effect of eccentric exercises in a cohort study with similar inclusion criteria, including ultrasound, as in the current trial. They found equally low baseline DASH values. Since none of the previous randomized controlled studies 20,38,40 evaluating eccentric/PHLE used ultrasound measurements to confirm structural abnormalities in the rotator cuff tendon at inclusion, comparisons between the present and previous studies are difficult to perform. In the Holmgren et al 20 study, only clinical signs were used as inclusion criteria. After inclusion, ultrasound measurements of the rotator cuff showed 31% of patients having either partial- or full-thickness rotator cuff tear, which may be the reason for a more heterogenic population and higher baseline score on the DASH. Also, Lombardi et al 38 used only clinical tests to include patients, with equally high levels of DASH baseline results as in the Holmgren et al 20 study. Since no further evaluations of potential differential diagnoses were presented in these studies, the specific differentiation among tendinopathy, bursitis, and rotator cuff tears remains uncertain. Furthermore, the current within-group change of 29% to 34% in PHLE and LLE, respectively, is larger than or similar to the percentage change in the intervention group from Lombardi et al 38 and Camargo et al, 4 indicating that the MCID change in relation to baseline values is similar across studies for rotator cuff tendinopathy, and the results of these studies may therefore seem more consistent.
Strengths and Limitations
A major limitation is the clinical criteria for rotator cuff tendinopathy, which could explain some of the reasons for the lack of between-group differences. However, as described, this problem was approached by strict inclusion procedures, including ultrasound-confirmed structural changes. Another limitation was the inability of blinding the treating physical therapists to the primary hypothesis, which could have influenced administration of exercise. However, because several physical therapists were involved and standardized information for patients was provided through written material and as an exercise DVD describing exercises and progression, this potential bias was minimized. Additionally, the number of compliant participants (60%) was relatively low. However, given that the requirement for being compliant was 80% completion of the exercise sessions, in addition to a high total number of visits to the physical therapists, the current compliance rate is acceptable. Other studies (eg, Holmgren et al 20 ) found approximately 85% compliance on number of days trained, but completion of exercise sessions was not recorded, making comparisons difficult.
A significant interaction occurred between exercise and concomitant administration of corticosteroid injection. However, the current study did not achieve sufficient power to detect a significant between-group difference in the area of concomitant corticosteroid injection. Therefore, further investigation of this interaction is warranted.
The strengths of the current study are the inclusion of an active control group and the blinding of patients. Furthermore, vital methodological strengths of the current study include blinding of the primary examiner, a blinded outcome analysis performed by an external statistician, interpretation of the results prior to unblinding, publication of a detailed study protocol, and a standardized public exercise protocol (Appendices A and B).
Deviations From Protocol
See Appendix E: “Deviations From Protocol.”
Conclusion
Our results showed there was no superior benefit of PHLE over traditional LLE in patients with rotator cuff tendinopathy. Further investigation of the possible interaction between exercise type and corticosteroid injection is needed to establish optimal and potentially synergistic combinations of these 2 factors. This finding suggests that shared decision making between patients and therapists, based on individual preferences, can safely be performed to secure optimal compliance.
Footnotes
Acknowledgment
The authors acknowledge all physical therapists who participated in completion of the interventions and orthopaedic specialists who participated in recruitment of patients.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.C. is a paid consultant for AbbVie, Amgen, Axellus A/S, Bristol-Meyers Squibb, Cambridge Weight Plan, Celgene, Eli Lilly, Hospira, MSD, Norpharma, Novartis, Orkla Health, Pfizer, Roche, Sobi, and Takeda; has received grants from Axellus A/S, AbbVie, Cambridge Weight Plan, Janssen, MSD, Mundipharma, Novartis, and Roche; has received payment for lectures from Abbott, Amgen, Axellus, Bayer HealthCare Pharmaceuticals, Biogen Idec, Bristol-Meyers Squibb, Cambridge Weight Plan, Ipsen, Janssen, Laboratoires Expanscience, MSD, Mundipharma, Norpharma, Novartis, Pfizer, Roche, Rottapharm-Madaus, Sobi, and Wyeth; has received grants for manuscript preparation from Axellus, Bristol-Meyers Squibb, Cambridge Weight Plan, and Aleris-Hamlet (via Norpharma); has received payment for development of educational presentations from Bristol-Meyers Squibb, MSD, and Pfizer; and received reimbursement for travel/accommodations/meeting expenses from Abbott, AbbVie, Axellus, Bristol-Myers Squibb, Cambridge Weight Plan, Celgene, Laboratoires Expanscience, Norpharma, Novartis, Pfizer, Roche, Rottapharm-Madaus, and Wyeth. This work was supported by the Region of Southern Denmark’s Research Fund, the Danish Rheumatism Association, the Ryholts Foundation, and the Danish Physiotherapy Association Foundation for Advancement of Research, Education, and Information. K.G.I. was funded by 1-year fellowships from Hospital Lillebaelt–Vejle Hospital, University of Southern Denmark, and Region of Southern Denmark. The Musculoskeletal Statistics Unit at the Parker Institute (R.C.) is supported by grants from the Oak Foundation.
Ethical approval for this study was obtained by the Regional Committees on Health Research Ethics for Southern Denmark.
Appendix A: LLE Exercise Program
Appendix B: PHLE Exercise Program
Appendix C: Measurement of Strength and Range of Motion
Appendix D
“Per Protocol” Population: Primary and Secondary Outcomes 12 Weeks After Baseline for PHLE and LLE a

| Outcome | Within-Group Change | Adjusted Between-Group Difference on Change b | P Value | |
|---|---|---|---|---|
| PHLE | LLE | |||
| DASH | ||||
| Main c | 12.59 (7.78 to 17.40) | 11.97 (7.53 to 16.41) | –0.14 (–6.20 to 5.92) | .96 |
| Hobby d | 11.84 (1.71 to 21.98) | 12.17 (–4.19 to 28.53) | 1.54 (–17.29 to 20.37) | .68 |
| Work e | 4.5 (–1.39 to 10.39) | 6.94 (0.36 to 13.53 | –2.68 (–9.51 to 4.14) | .75 |
| VAS c | ||||
| Rest | 4.68 (–0.52 to 9.88) | 1.61 (–2.08 to 5.30) | –0.09 (–5.02 to 4.84) | |
| Activity | 17.89 (9.48 to 26.30) | 15.19 (8.80 to 21.58) | 0.74 (–8.20 to 9.67) | .87 |
| Night | 19.82 (8.42 to 31.22) | 15.35 (7.51 to 23.20) | 1.51 (–9.79 to 12.80) | .79 |
| Maximum | 20.43 (8.44 to 32. 42) | 17.84 (10.28 to 25.40) | 0.81 (–13.47 to 15.09) | .91 |
| Isometric strength, N/BW f | ||||
| Scaption | 0.09 (0.03 to 0.16) | 0.08 (0.03 to 0.14) | 0.00 (–0.08 to 0.08) | .99 |
| External rotation | 0.05 (0.02 to 0.07) | 0.05 (0.03 to 0.08) | 0.00 (–0.03 to 0.03) | .92 |
| Internal rotation | 0.09 (0.04 to 0.13) | 0.09 (0.06 to 0.13) | –0.01 (–0.07 to 0.05) | .73 |
| Passive ROM f | ||||
| Scaption | 5.01 (–1.20 to 11.23) | 6.12 (1.48 to 10.76) | –3.47 (–10.96 to 4.03) | .36 |
| External rotation | 3.78 (–1.81 to 9.36) | 4.30 (–2.97 to 11.58) | –3.56 (–12.66 to 5.54) | .44 |
| Internal rotation | 0.43 (–3.45 to 4.32) | 0.33 (–3.01 to 3.68) | 0.41 (–3.96 to 4.78) | .85 |
| Active ROM f | ||||
| Scaption | 11.51 (2.99 to 20.02) | 7.79 (2.45 to 13.12) | –3.00 (–11.20 to 5.20) | .47 |
| External rotation | 8.00 (1.21 to 14.80) | 4.12 (–4.60 to 12.83) | 0.14 (–10.17 to 10.45) | .98 |
| Internal rotation | 2.67 (–2.18 to 7.51) | 0.91 (–2.21 to 4.04) | 1.82 (–3.05 to 6.69) | .46 |
a Values are presented as mean (95% CI). BW, body weight (kg); DASH, Disabilities of the Arm, Shoulder, and Hand (range, 0-100); LLE, low-load exercise; N, newton; PHLE, progressive high-load exercise; ROM, range of motion; VAS, visual analog scale (range, 0-100).
b Adjusted for baseline value, location (categorical: 3 centers), corticosteroid (yes/no).
c n = 59 (LLE = 31, PHLE = 28).
d n = 38 (LLE = 19, PHLE = 19).
e n = 52 (LLE = 27, PHLE = 25).
f n = 58 (LLE = 31, PHLE = 27).
Appendix E: Deviations from Protocol
The following deviations from the protocol to the RoCTEx trial 1 were made.
