Abstract
Background:
The combination of cells with platelet-rich plasma (PRP) may fulfill tendon deficits and help overcome the limited ability of tendons to heal.
Purpose:
To examine the suitability of 3 human cell types in combination with PRP and the potential impact of the tenocyte-conditioned media (CM) to enhance tendon healing.
Study Design:
Controlled laboratory study.
Methods:
Tenocytes, bone marrow–derived mesenchymal stem cells, and skin fibroblasts were cultured in 3-dimensional PRP hydrogels supplemented or not with CM, and cell proliferation and migration were examined. The effect of tendon-derived CM on matrix-forming phenotype and secretion of inflammatory proteins was determined through their administration to mesenchymal stem cells, tendon, and skin fibroblasts by reverse transcription quantitative polymerase chain reaction and enzyme-linked immunosorbent assay, respectively.
Results:
Differences were found in the matrix-forming phenotype between each of the cell types. The ratio of collagen I:collagen III was greater in bone marrow–derived mesenchymal stem cells than in skin fibroblasts and tenocytes. The bone marrow–derived mesenchymal stem cells expressed increased levels of cartilage-related genes than tenocytes or skin fibroblasts. The presence of the tenocyte-CM stimulated basic healing mechanisms including proliferation and chemotaxis in all cell types. In addition, the tenocyte-CM modified the matrix-forming phenotype of every cell type when cultured in PRP hydrogels. Each cell type secreted interleukin-6, interleukin-8, and monocyte chemotactic protein-1 in PRP hydrogels, but mesenchymal stem cells secreted less interleukin-8 and monocyte chemotactic protein-1 than tenocytes or skin fibroblasts.
Conclusion:
The tenocyte-CM combined with PRP stimulated tenogenesis in mesenchymal stem cells and in skin fibroblasts and reduced the secretion of inflammatory proteins.
Clinical Relevance:
Modifying the target tissue with PRP prior to cell implantation may optimize the effect of cell therapies. Skin fibroblasts and bone marrow–derived mesenchymal stem cells combined with PRP could be used to regenerate tendons.
Chronic musculoskeletal diseases, such as tendinopathy, are largely refractory to therapy. Healing capabilities are restricted because of the acellular, aneural, and avascular nature of tendons. Biological interventions, including the combination of cells with multimolecular preparations, may fulfill tendon deficits and help overcome the limited ability of tendons to self-repair.
Platelet-rich plasma (PRP) is a multimolecular preparation composed of plasma and platelets with a platelet count above that of peripheral blood and optional leukocytes. 8 It has been widely used in orthopaedics and sports medicine to manage tendon conditions. 2 However, after meta-analysis of current clinical studies, several authors agreed that its efficacy is still controversial. 1,3,24,29 This has been attributed not only to the heterogeneity of tendon conditions but also to limitations in identifying biomarkers that describe the progression of the disease and discriminating different subsets of patients for which these biological interventions may be indicated. 24,29 These particulars, together with our incomplete knowledge of PRP biology and its interactions with cells, have hastened the need for the development of experimental foundations that support the potential benefits provided by the combination of PRP with in vitro–expanded adult cells. The final goal is to gain insight from the synergy of biological tools in chronic and acute tendon conditions.
Skin fibroblasts, tenocytes, and mesenchymal stem cells (MSCs) have been explored for their therapeutic applications in tendon conditions and represent potential choices for tendon repair. In a preliminary study that included 12 patients with a 6-month follow-up, skin fibroblasts were injected within the common elbow extensor and a decrease in the number of tears was reported. 7 A subsequent randomized controlled trial was performed in 46 patients with 60 patellar lesions; the application of skin fibroblasts combined with PRP into hypoechogenic sites and intrasubstance tears was superior to PRP injections. 6 As an alternative cell source, the safety and efficacy of autologous tenocytes that were previously expanded in vitro was examined in a case series of 20 patients with chronic resistant lateral epicondylitis; the results showed that pain decrease and functional improvements were maintained at the 4.5-year follow-up without adverse events. 27,28 Moreover, in vitro–expanded bone marrow–derived MSCs (BM-MSCs) have been used as clinical therapy to treat small tears (<1.5 cm) in shoulder tendinopathy with significant clinical improvements. 5
Although adult cell therapies have reached the clinical phase for tendon conditions, both preclinical and clinical investigations are insufficient to develop efficient clinical procedures, as several issues still require careful consideration.
The selection of the cell source is crucial in designing a tendon regeneration strategy. 11 Moreover, conceiving a delivery approach for improved clinical efficacy involves the use of activated PRP that, on fibrin formation and coagulation, will confine cells to the injection site. 12,13 Nevertheless, the success of clinical therapies may also be hindered because injected or transplanted cells encounter a hostile environment that hampers their differentiation and regenerative potential. 10
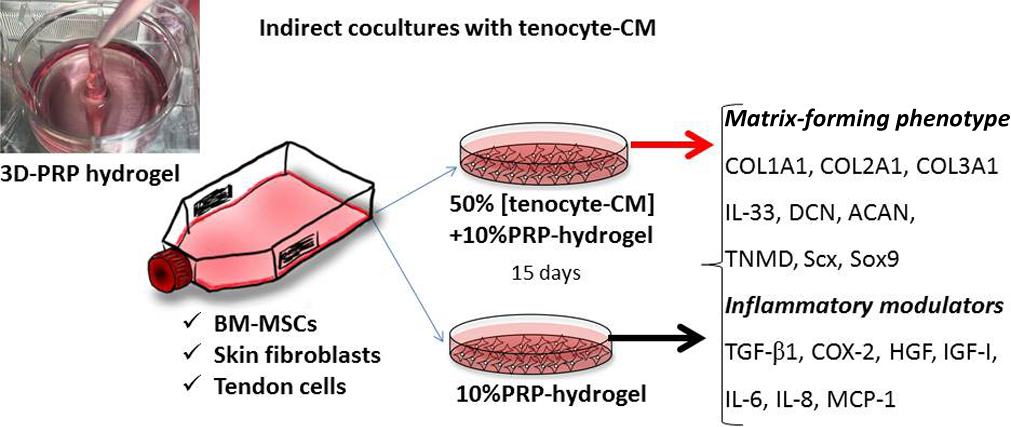
In this study, we addressed 2 main issues related to the current clinical problem. First, using 3-dimensional (3D) hydrogel cultures that closely imitate the in vivo environment when cells are delivered in combination with PRP, 3 we explored the most appropriate cell source to fulfill the requirements of the injured tendon to specifically enhance extracellular matrix formation and to modulate inflammation. Second, we focused on modifying the target tissue prior to cell transplantation to improve its efficacy. 10 We hypothesized that one strategy to turn a hostile microenvironment into a proregenerative milieu might involve preconditioning the host tissue with PRP. Basic processes of tissue regeneration include cell proliferation, migration, and the synthesis of extracellular matrix. To explore this premise, we treated tenocytes with PRP, harvested their conditioned media (CM), and by adding the CM of tenocytes to the culture media, we explored whether biological modifications induced by PRP on the tendon could help optimize the regenerative capacity of transplanted cells (tendon cells, BM-MSCs, and skin fibroblasts). We hypothesized that conditioning host tissue with PRP prior to cell implantation would enhance the regenerative potential of implanted cells.
Methods
Cell Expansion
Human tendon samples were obtained from semitendinosus tendon biopsies from 3 volunteers (3 men; mean age, 27 ± 1.4 years) undergoing anterior cruciate ligament reconstruction. The local ethics committee approved the study (PI2014108), and written informed consent was obtained from all donors. Tenocytes were isolated by explant culture as previously described. 4 Human BM-MSCs (2 men, 1 woman; mean age, 32 ± 6 years) and skin fibroblasts (2 men, 1 woman; mean age, 39 ± 17 years) (not matched) were purchased from Lonza. The cells were cultured at 37°C in 5% CO2, passaged at subconfluence, and used until passage 3; BM-MSCs were expanded in Dulbecco modified Eagle medium (DMEM) GlutaMAX (Gibco, Life Technologies) with 10% fetal bovine serum (FBS) (Hyclone; GE Healthcare), and skin fibroblasts and tenocytes were expanded in DMEM F12 supplemented with 10% FBS. Trypan blue was used to assess cell viability before plating.
Preparation of PRP
Peripheral blood was withdrawn in citrate tubes (Vacuette; Greiner BioOne) from participants within the control group of a randomized PRP clinical trial after they provided informed consent. PRP was prepared using a single-step centrifugation at 570 × g for 7 minutes. The centrifugation procedure enriched the platelet concentration by 1.84 ± 0.42–fold over peripheral blood, and more than 99% of red blood cells and white blood cells were eliminated.
Preparation of Tenocyte-CM
For the preparation of the tenocyte-CM, we set up PRP hydrogel cultures in T75 flasks. Briefly, tenocytes were resuspended in culture media at a density of 3 × 105 cells per flask with 10% PRP and were maintained in 3D PRP hydrogels at 37°C and 5% CO2 for 96 hours. Thereafter, the hydrogels were centrifuged to obtain the aqueous CM that was used as a supplement in the 3D cultures of BM-MSCs, skin fibroblasts, and tenocytes. To examine the effects of this particular tendon CM (representing preconditioning of the host tissue with PRP), indirect cocultures of BM-MSCs, skin, and tendon fibroblasts were performed.
Hydrogel Cultures and Treatments
Prior to the start of the experiment, expanded cells were starved for 24 hours in T75 flasks and harvested by trypsinization (TryPLE select; Gibco, Life Technologies). Samples of each cell phenotype (bone marrow, skin, and tendon) were collected as time 0 samples for constitutive gene expression or were seeded at a concentration of 4 × 104 cells per well in 3D plasma hydrogels in 6-well plates, as previously described. 22 The presence of Ca2+ in the culture medium (1.05 mM) induces thrombin formation and the subsequent cleavage of fibrinogen and polymerization of fibrin dimers. On fibrin formation, cells are captured within the hydrogel.
Wells were filled with 2 mL of DMEM supplemented with 10% PRP and 50% CM. On clotting, PRP hydrogels hold the liquid CM. Other wells were treated with DMEM supplemented with 10% PRP and were used as controls to test the effects of CM. Cells were maintained in 3D hydrogels for 15 days. Every 3 days, extruded supernatant was replaced with media and PRP or with media and PRP plus CM, as appropriate. Experiments were performed in parallel for 3 BM-MSC donors, 3 skin donors, and 3 tendon donors and were repeated using PRP from 3 donors and CM obtained from tenocytes harvested from 3 different donors (Figure 1).
Schematic diagram illustrating the study design.
Proliferation of Human BM-MSCs, Skin Fibroblasts, and Tenocytes With CM
To assess the mitogenic ability of CM, BM-MSCs, skin fibroblasts, and tenocytes were seeded at a density of 4000 cells per well in 96-well plates and cultured with DMEM supplemented with 50% CM. For the PRP hydrogels, cells were loaded at a density of 4000 cells per well in 3D PRP hydrogels. 23 Cell proliferation was measured at 0, 24, 48, 72, and 96 hours using a colorimetric assay XTT (2,3-bis (2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino) carbonyl]-2H-tetrazolium hydroxide), following the manufacturer’s instructions (Cell Proliferation Kit II; Roche). Cell doubling times (hours) were calculated at the exponential phase (http://www.doubling-time.com/compute.php).
Directional Migration of Cells Toward CM
Cell migration toward 50% CM was assessed using a µ-Slide Chemotaxis 3D (Ibidi GmbH) slide filled with 12,000 viable cells. This µ-slide allows for the creation of a gradient of a chemotactic compound or solution (eg, CM) along the slide and thus for the study of cell movement toward it. One side of the slide was filled with CM and the other was filled with DMEM (CM/DMEM). Positive (CM/CM) and negative controls (DMEM/DMEM) with no gradients were implemented for all the cell phenotypes. Video microscopy was performed over a period of 24 hours for each cell phenotype and for the controls. Time-lapse microscopy was performed using a Nikon Eclipse TE2000-E microscope (Nikon) at 10× magnification and an ORCA ER camera (Hamamatsu) with a 10-minute interval between images. Data were imported into the “Manual Tracking” plugin (Fabrice P. Cordelières; Institut Curie) for ImageJ software (National Institutes of Health). A minimum of 30 cells were tracked in each experiment. Tracking was further analyzed with the ImageJ plugin “Chemotaxis and Migration Tool” (Ibidi GmbH). The center of mass (COM), forward migration indices (FMI) in directions parallel and perpendicular to the gradient, speed (µm/min), directness (µm), and significance (P) for the Rayleigh test were obtained. In addition to the trajectory and velocity, these parameters describe the cell’s tendency to travel toward the chemotactic gradient.
Gene Expression Analysis
The RNA from 3D hydrogels was extracted using TRIzol Reagent (Invitrogen, Life Technologies) according to the manufacturer’s instructions, and its quality and amount were assessed using a Nanodrop ND-1000 (Thermo Scientific) spectrophotometer. One microgram of RNA was reverse transcribed to cDNA using the SuperScript III First-Strand Synthesis System (Invitrogen, Life Technologies). Reverse transcription quantitative polymerase chain reaction (RT-qPCR) was performed on an ABI-7900 Real Time System (Applied Biosystems, Life Technologies) at a final volume of 20 μL using Power SYBR Green PCR Master Mix (Applied Biosystems, Life Technologies), 600 nM of primers, and 20 ng of cDNA. Reactions were performed in triplicate for each sample.
Gene expression was assessed for the matrix components collagen type I alpha 1 (COL1A1), collagen type II alpha 1 (COL2A1), collagen type III alpha 1 (COL3A1), aggrecan (ACAN), decorin (DCN), and tenomodulin (TNMD). Additionally, gene expression was assessed for the transcription factors scleraxis (Scx) and SRY (Sex Determining Region Y)-Box 9 (Sox9) as well as for the cytokines, growth factors, and inflammatory mediators transforming growth factor–beta 1 (TGF-β1), interleukin-33 (IL-33), hepatocyte growth factor (HGF), insulin-like growth factor–1 (IGF-1), and cyclooxygenase 2 (COX-2).
Relative expression was calculated using the mean expression of 2 stably expressed housekeeping genes: glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and TATA box binding protein (TBP). Gene expression data were calculated using 2−ΔCt. ΔCt was calculated by the difference between Ct (cycle threshold) values of the target genes and the mean Ct of GAPDH and TBP. The 2−ΔΔCt method was used to evaluate the effect of CM in PRP hydrogel 3D cultures.
Assessment of Protein Secretion
Cytokine production in the extruded supernatant of 3D PRP hydrogels was determined by enzyme-linked immunosorbent assay (ELISA). The levels of interleukin-6 (IL-6), interleukin-8 (IL-8), and monocyte chemotactic protein–1 (MCP-1) were measured using Peprotech mini ELISA Development kits (Peprotech). SureBlue TMB Microwell peroxidase was purchased from KPL.
Abbreviated terms used in this article are shown in Table 1.
Abbreviated Terms
Statistical Analysis
Data are expressed as the mean ± standard error or as 95% confidence intervals of the averaged technical triplicates of 6 to 9 independent cultures: 6 independent cultures (2 cell donors × 3 PRP donors) for cell proliferation and migration and 9 independent cultures (3 cell donors × 3 PRP donors) for gene expression and protein secretion assessments. Comparisons between gene expressions, protein secretion, velocity of migration, and accumulated and Euclidean distances were performed using the Kruskal-Wallis test, followed by the Mann-Whitney U test. Correlations were calculated using the Spearman coefficient (ρ). A P value ≤ .05 was considered to be significant. Data were analyzed using SPSS for Windows, version 18.0 (IBM Corp).
Results
CM Induces Cell Proliferation
PRP stimulated proliferation of all of the cell phenotypes. CM also supported proliferation, especially in BM-MSCs and skin fibroblasts, but in a less powerful manner than PRP. The doubling times of tenocytes, BM-MSCs, and skin fibroblasts cultured in 3D PRP hydrogels and in the presence of CM are shown in Table 2.
Doubling Times of Tenocytes, BM-MSCs, and Skin Fibroblasts Under Different Experimental Conditions a

a Results are shown as the mean (SEM) of the experimental duplicates. DMEM, Dubelcco modified Eagle medium.
CM Induces Directed Cell Migration
The 3 cell phenotypes migrated toward CM gradients, as shown in Figure 2 (Rayleigh test significance). Directness (directionality), defined as the displacement divided by the total path length of the cell, was greater in BM-MSCs than in tenocytes (P = .017) in the CM gradient. Migration was similar between the different cell types, with no differences in velocity or distance traveled by the COM. Intrinsic cell movement, observed in the negative control experiment (DMEM/DMEM), was greater in skin fibroblasts than in BM-MSCs (P < .001) and tenocytes (P = .004) (see Appendix Table A1).
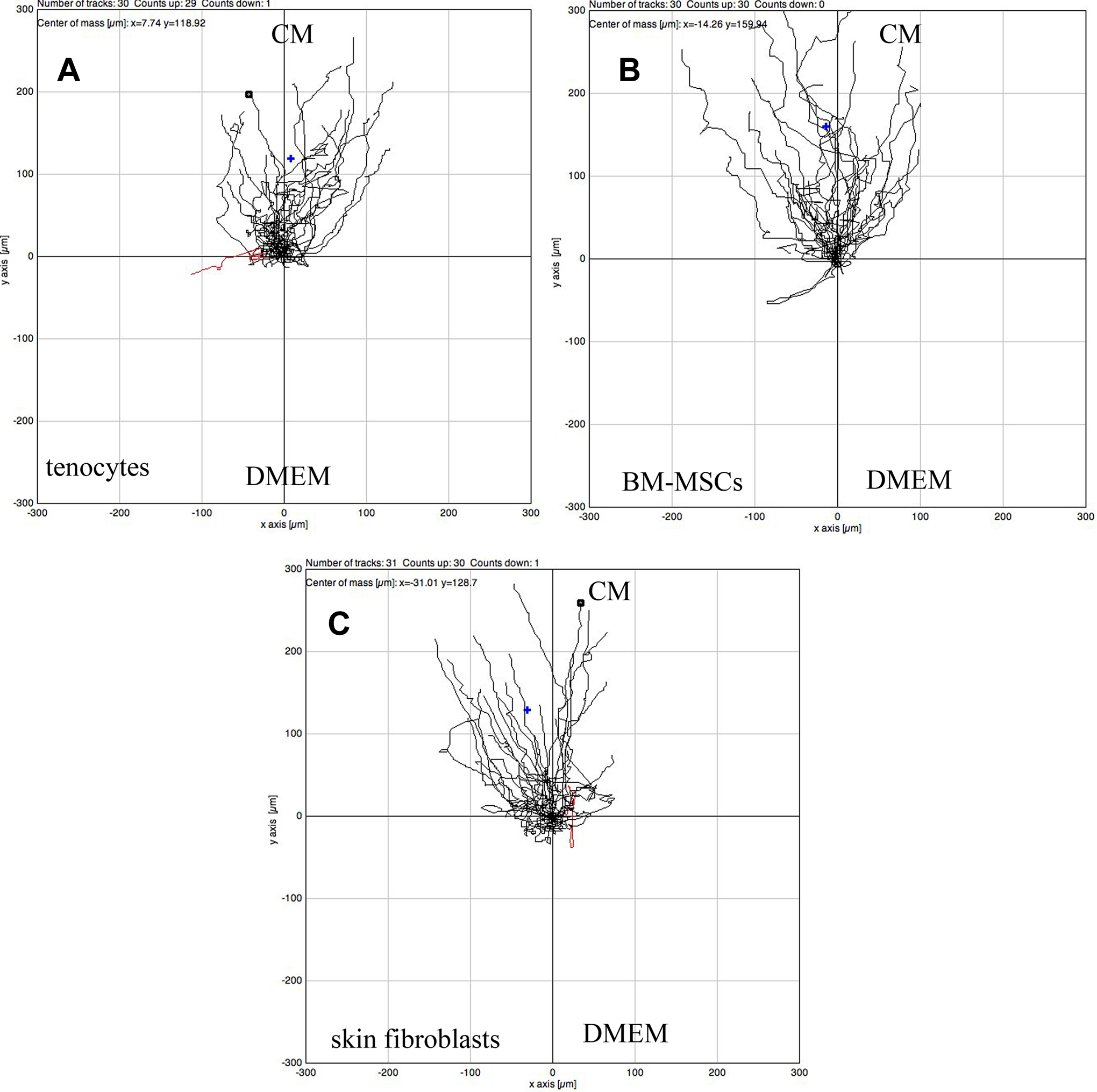
Representative plots of (A) tenocyte, (B) BM-MSC, and (C) skin fibroblast migration over 24 hours toward a gradient created by CM (n ≥ 30). CM is initially injected in the upper part of the μ-slide and a gradient is progressively created by diffusion. DMEM, Dubelcco modified Eagle medium.
Gene Matrix-Forming Phenotypes
Constitutive Expression
The constitutive gene expressions of matrix-forming molecules and growth factors of the cells prior to loading in 3D PRP hydrogels are shown in Appendix Table A2.
No significant differences were found in the constitutive gene expressions between BM-MSCs and skin fibroblasts, but tenocytes showed greater expression levels of Scx, TGF-β1, and COX-2 but lower expression levels of COL1A1, COL3A1, and ACAN compared with both BM-MSCs and skin fibroblasts (P < .05).
Differential Gene Expression
We found differences in the matrix-forming phenotype between tenocytes, BM-MSCs, and skin fibroblasts cultured in 3D PRP hydrogels for 15 days (Figure 3). The relative expression of COL1A1 was 4-fold greater in BM-MSCs than in skin fibroblasts (P = .004) and 7-fold greater than in tenocytes (P = .004). Skin fibroblasts also expressed greater levels of COL1A1 than tenocytes (P = .002) (Figure 3A). Although there were no differences in COL3A1 expression among cell phenotypes, the ratio between COL1A1 and COL3A1 was to a great extent lower in tenocytes (10.9; 95% CI, 6.6-15.2) than in BM-MSCs (139.3; 95% CI, 46.3-232.4) and skin fibroblasts (79.7; 95% CI, 17.9-141.9) (P = .002 for both). Remarkably, the relative expression of IL-33 was undetectable in BM-MSCs but appeared moderately low in tendon and skin cells.

Gene expression, as assessed by reverse transcription quantitative polymerase chain reaction (RT-qPCR), of (A-C) the matrix-forming phenotype and (D) inflammatory modulators for tenocytes, bone marrow–derived mesenchymal stem cells (BM-MSCs), and skin fibroblasts cultured for 15 days in 3-dimensional (3D) platelet-rich plasma (PRP) hydrogels. Box plots show the 2−ΔCt relative gene expressions with the medians and 25th to 75th percentiles. Representative phase-contrast photomicrographs of (E) tenocytes, (F) BM-MSCs, and (G) skin fibroblasts in 3D PRP hydrogels. Magnification 10×.
Regarding cartilage-related molecules, BM-MSCs expressed higher levels of COL2A1, ACAN, and Sox9 in PRP hydrogels than tenocytes or skin fibroblasts, and there were no differences between skin fibroblasts and tenocytes (Figure 3B). TNMD expression was greater in BM-MSCs than in the other cell types (P = .004) (Figure 3C). COL1A and COL2A1 showed positive correlations with TNMD (ρ = 0.727, P = .001, and ρ = 0.955, P < .001, respectively). In agreement with the anatomic cell source, the levels of expression of Scx were significantly greater in tenocytes than in BM-MSCs or skin fibroblasts (P = .002 for both) (Figure 3C).
We also investigated the levels of expression of inflammatory modulators and growth factors. The relative expression of TGF-β1 was higher in BM-MSCs than in tenocytes (P = .010) or skin fibroblasts (P = .015). TGF-β1 also showed a positive correlation with COL1A1 and COL2A1 (ρ = 0.808, P < .001, and ρ = 0.534, P = .041, respectively). BM-MSCs expressed greater levels of COX-2 than tendon and skin fibroblasts (P = .026 and P = .015, respectively) (Figure 3D).
Skin fibroblasts, tenocytes, and BM-MSCs expressed moderately low levels of HGF, without differences between them. Interestingly, tenocytes expressed greater levels of IGF-1 than skin fibroblasts (P = .026), and we found a positive correlation between the expression levels of IGF-1 and Scx (ρ = 0.565, P = .004).
CM-Induced Changes in Matrix-Forming Phenotype and Growth Factor Expression
As depicted in Figures 3 and 4, tenocytes showed spindle-shape morphology and were aligned only in PRP hydrogels (Figure 3E) in the absence of CM (Figure 4C). In contrast, BM-MSCs and skin fibroblasts exhibited a spindle-shape morphology and were aligned when exposed to CM for 15 days (Figure 4, F and I), but not in PRP hydrogels without CM (Figure 3, F and G).

Gene expression of the matrix-forming phenotype and inflammatory modulators in tenocytes, bone marrow–derived mesenchymal stem cells (BM-MSCs), and skin fibroblasts after 15 days in culture in 3-dimensional (3D) platelet-rich plasma (PRP) hydrogels supplemented with tenocyte-conditioned media. Data obtained from reverse transcription quantitative polymerase chain reaction (RT-qPCR) are displayed as 2−ΔΔCt relative to the 3D PRP hydrogels. Box plots show the medians and 25th to 75th percentiles.
In skin fibroblast cells, CM induced more changes. Interestingly, CM enhanced the expression of Scx by 3-fold in BM-MSCs (P = .002) and by 5-fold in skin fibroblasts (P = .001). CM also influenced the expression of fibrillar proteins, including COL1A1 and COL3A1 but not COL2A1. In particular, COL1A1 was increased by 2.3-fold in skin fibroblasts (P = .005) but did not change in BM-MSCs. COL3A1 expression was upregulated by 3.6-fold in MSCs (P = .002) and by 2.7-fold in skin fibroblasts (P = .002). Consequently, after CM exposure, the ratio of COL1A1:COL3A1 became higher in skin fibroblasts than in BM-MSCs.
Skin fibroblasts also showed concomitant upregulation of TGF-β1 by 2-fold (P = .008) and of ACAN expression by 6-fold (P = .018).
CM strongly downregulated the expression of several molecules in tenocytes, including COL1A1 and COL3A1 (P = .018 and P = .005, respectively). In addition, Scx and TGF-β1 were reduced by half (P = .050 and P = .001, respectively) (Figure 4, A and B).
Cell Secretion of Inflammatory Proteins in 3D PRP Hydrogels Exposed to CM
Dissimilarities in Inflammatory Protein Secretion Between Cell Types in Nonsupplemented PRP Hydrogels
There were no differences in IL-6 secretion between the 3 cell types, but BM-MSCs secreted less IL-8 than tenocytes (P = .009) and skin fibroblasts (P = .002). Furthermore, BM-MSCs secreted a smaller amount of MCP-1 than skin fibroblasts (P = .015) (Figure 5A).

Secreted inflammatory proteins as assessed by enzyme-linked immunosorbent assay (ELISA). Interleukin (IL)-6, IL-8, and monocyte chemotactic protein–1 (MCP-1) were measured after 15 days in culture in (A) 3-dimensional (3D) platelet-rich plasma (PRP) hydrogels and (B) 3D PRP hydrogels supplemented with tenocyte-conditioned media (CM).
CM Modified the Secretion of Inflammatory Proteins
In BM-MSCs, CM induced a rise in IL-6 secretion (P = .002). The levels of secretion of MCP-1 and IL-8 remained unchanged. In skin fibroblasts, CM induced a reduction in IL-8 secretion (P = .036) and a robust decrease in MCP-1 secretion (P < .001) (Figure 5).
Comparing the different cell types, data showed that BM-MSCs secreted less IL-8 and less MCP-1 than tenocytes (P < .001 for both) or skin fibroblasts (P < .001) for IL-8 (P = .001) and MCP-1 (Figure 5B).
Discussion
Biological interventions based on implantation of in vitro–expanded cells, such as skin fibroblasts, tenocytes, or BM-MSCs, combined with PRP are a potential strategy for tendon repair. 7,15,24,29 For improved outcomes, the optimal cell source, mode of delivery, and conditioning of the host tissue prior to cell implantation must be investigated.
Preconditioning the target tendon with PRP injections stimulates cell proliferation, chemotaxis, and secretory activities of endogenous tenocytes (ie, CM). As shown in this study, tenogenesis of skin fibroblasts and BM-MSCs is enhanced by PRP combined with CM. Thus, implantation of skin fibroblasts or BM-MSCs combined with PRP subsequent to preconditioning the target tendon with PRP injections would have more opportunities to regenerate the tendon. BM-MSCs could have advantages because of their increased synthetic activities and lower inflammatory profile.
Depending on the anatomic source, cells can respond differently to diverse molecular environments through a change in the matrix-forming phenotype and secretion of inflammatory proteins. In vitro plasticity provides insights into how these cell populations may behave when delivered with PRP to a tendon. A common concern when implanting cells locally is the large proportion of cells that do not remain in the target tissue and are lost. 15 To cope with this crucial problem, cells could be delivered with PRP to confine them within the chosen site. In fact, cells injected in a liquid carrier can diffuse into surrounding tissues. Instead, on activation, PRP is gelatinized, comprising cells within the PRP scaffold.
Local cell migration and proliferation are important issues to be considered when designing biological therapies. PRP has been shown to have chemotactic and mitogenic properties for tenocytes. 22 The CM of PRP-treated tenocytes is also chemotactic and mitogenic, with the ability to induce endogenous cell migration. However, it may be necessary to engraft additional exogenous cells when treating severe conditions.
The optimal cell source for tendon conditions may depend on whether the lesion is in the midpoint of the tendon or in fibrocartilaginous enthesis. In this regard, BM-MSCs expressed greater levels of cartilage matrix–forming molecules (collagen 2, ACAN, and the transcription factor Sox-9) as well as TNDM than tenocytes or skin fibroblasts. TNDM is an angiogenesis inhibitor that is predominantly expressed in avascular regions of tendon and cartilage. 30 Although this issue cannot be based on gene expression data and requires mandatory in vivo confirmation, in vitro results might suggest that the combined therapy of BM-MSCs with PRP could be an appropriate approach for regenerating the fibrocartilaginous enthesis by which the tendon is attached to the bone. It has been observed in vivo that, after surgical transection of the Achilles tendon enthesis, a group of rats injected with BM-MSCs showed better enthesis organization and improved biomechanical properties than controls or rats with destroyed enthesis that were treated with chondrocytes. 20 Although this surgical transection model does not reflect overuse or intrinsic tendon degeneration, it informs about the healing potential of BM-MSCs in tendons.
Skin fibroblasts and BM-MSCs in PRP hydrogels do not express Scx at the same level as tenocytes, and consequently, they are not considered to be differentiated into tenocytes. Scx is a key transcription factor that is expressed in tendon progenitors and in tenocytes. 17,19 The molecular microenvironment provided by CM, but not by the PRP environment, stimulated Scx expression in both BM-MSCs and skin fibroblasts, in agreement with other coculture models involving MSCs and tenocytes. 16,18,25
BM-MSCs showed higher expression of collagen I, the major fibrillar component of tendon extracellular matrix, and had the greatest collagen I:collagen III ratio. Tendons are mechanically functional tissues, and a low ratio of collagen I:collagen III is associated with weak mechanical properties. 9,21 Supporting these data, BM-MSC implantation in naturally occurring tendinopathy in equine superficial digital flexor tendons has been shown to enhance normalization of biochemical and compositional parameters without fibrosis at a 6-month follow-up. 26
A shortcoming of BM-MSCs is that when they were exposed to CM, they showed a decreased ratio of collagen I:collagen III. Under these conditions, skin fibroblasts can be more strongly involved in the activation of midsubstance tendon healing because, in addition to tendon markers, they showed a convenient collagen I:collagen III ratio. Thus, skin fibroblasts could be more suitable than BM-MSCs, provided that the tendon is conditioned with PRP prior to cell implantation. Further confirmation of these results in animal models with naturally occurring tendinopathy is imperative.
Typically, studies have not focused on modifying the target tissue to optimize the effect of cell therapies. One possibility is that the host tissue is not receptive to transplanted cells and does not supply normal trophic signals because of inflammation. 10 A link between inflammation and matrix synthesis in tendons has been proposed, as represented by IL-33, because, in addition to being involved in the collagen III synthesis, it operates in tenocytes via the IL-1R signaling pathway and elevated the production of IL-6, IL-8, and MCP-1 in vivo and in vitro. 19 The latter has been recently involved in early tendinopathic changes, more specifically in matrix deregulation toward collagen III expression. 19 Accordingly, we found that cells that express IL-33 (tenocytes and skin fibroblasts) secreted a greater amount of IL-8 and MCP-1.
CM induced a rise in the secretion of pleiotropic IL-6 by BM-MSCs, but after 15 days of exposure to CM, the BM-MSCs secreted less IL-8 and less MCP-1 than tenocytes and skin fibroblasts. This may indicate a weaker inflammatory response that could be linked to an improved matrix-forming phenotype.
One downside of BM-MSCs is that they are harvested via invasive painful punctures, and the reproducibility of the harvesting procedures is controversial. Instead, skin and tendon biopsies can be easily obtained, avoiding the problems of inconsistent tissue sampling found in bone marrow. Tendon biopsies can be harvested from the patellar tendon while skin fibroblasts used in the clinical studies referenced here were obtained with a 4-mm-diameter punch biopsy needle from the lateral aspect of the thigh. Moreover, clinical-grade PRP can be used to accelerate ex vivo cell expansion to decrease the delay between harvesting and implantation.
Several limitations should be noted in this study. We have not included in our study adipose-derived stromal cells, which present sourcing advantages over BM-MSCs. Moreover, the different cell types have been obtained from various healthy donors not matched for age and sex. Another weakness of the study is that no mechanism is explored. Although observations are made that the 3 cell types behave differently, we still do not know why they behave differently. To improve our basic science understanding of the process-signaling pathways such as IL-1R and nuclear factor–κB, activation should be explored. 14 In gene profiling, we explored a limited amount of genes and additional fibrillar collagens, including COL5A1 and COL11A1, and fibril/fiber assembly regulatory genes, such as COL12A1 and COL14A1, were not assessed. To reach robust conclusions, the 3 cell types must be tested in animal models of tendinopathy; the resulting composition and biomechanical properties of the regenerated tendon can substantiate the choice of the most efficient cell type for proper tendon or enthesis regeneration. Finally, only a single formulation of PRP was tested, whereas other options are available clinically.
Conclusion
We have provided information about how the choice of the cell source can modify the biological parameters involved in tendon repair. CM combined with PRP stimulated tenogenesis in BM-MSCs and skin fibroblasts. BM-MSCs synthesize less IL-8 and MCP-1 inflammatory proteins. However, because of the accessibility of dermal fibroblasts, their high collagen I:collagen III ratio, and the reduced secretion of MCP-1 compared with tenocytes, they appear to be a better choice than BM-MSCs to treat tendon lesions. These results are relevant to the design of clinical trials with autologous cells and PRP.
Footnotes
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by the Provincial Council of Alava, Grant “Alava Innova.”
Ethical approval for this study was obtained from the Ethics Committee of the Basque Country.
Appendix
Constitutive Gene Expression of Tendon Cells, BM-MSC, and Skin Fibroblasts Assessed by RT-qPCR a
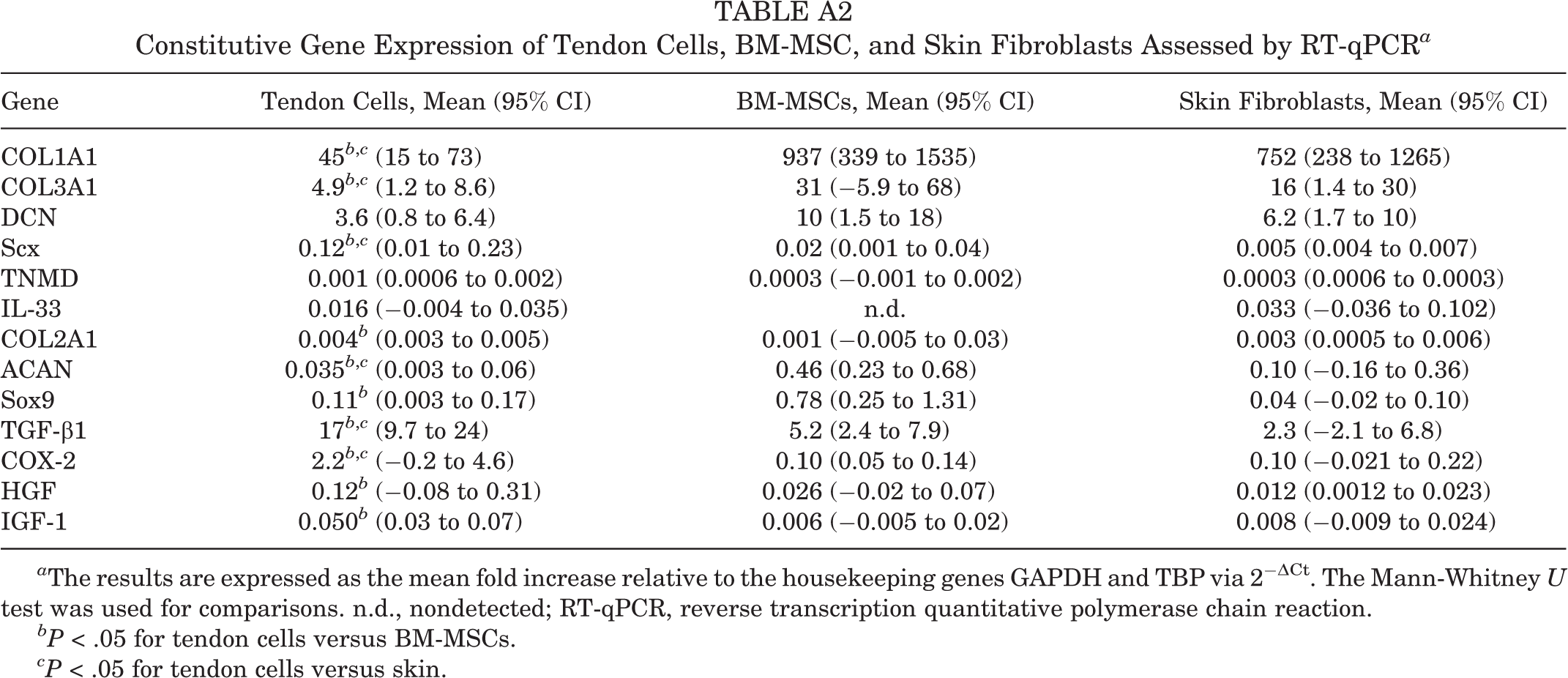
| Gene | Tendon Cells, Mean (95% CI) | BM-MSCs, Mean (95% CI) | Skin Fibroblasts, Mean (95% CI) |
|---|---|---|---|
| COL1A1 | 45 b ,c (15 to 73) | 937 (339 to 1535) | 752 (238 to 1265) |
| COL3A1 | 4.9 b ,c (1.2 to 8.6) | 31 (−5.9 to 68) | 16 (1.4 to 30) |
| DCN | 3.6 (0.8 to 6.4) | 10 (1.5 to 18) | 6.2 (1.7 to 10) |
| Scx | 0.12 b ,c (0.01 to 0.23) | 0.02 (0.001 to 0.04) | 0.005 (0.004 to 0.007) |
| TNMD | 0.001 (0.0006 to 0.002) | 0.0003 (−0.001 to 0.002) | 0.0003 (0.0006 to 0.0003) |
| IL-33 | 0.016 (−0.004 to 0.035) | n.d. | 0.033 (−0.036 to 0.102) |
| COL2A1 | 0.004 b (0.003 to 0.005) | 0.001 (−0.005 to 0.03) | 0.003 (0.0005 to 0.006) |
| ACAN | 0.035 b ,c (0.003 to 0.06) | 0.46 (0.23 to 0.68) | 0.10 (−0.16 to 0.36) |
| Sox9 | 0.11 b (0.003 to 0.17) | 0.78 (0.25 to 1.31) | 0.04 (−0.02 to 0.10) |
| TGF-β1 | 17 b ,c (9.7 to 24) | 5.2 (2.4 to 7.9) | 2.3 (−2.1 to 6.8) |
| COX-2 | 2.2 b ,c (−0.2 to 4.6) | 0.10 (0.05 to 0.14) | 0.10 (−0.021 to 0.22) |
| HGF | 0.12 b (−0.08 to 0.31) | 0.026 (−0.02 to 0.07) | 0.012 (0.0012 to 0.023) |
| IGF-1 | 0.050 b (0.03 to 0.07) | 0.006 (−0.005 to 0.02) | 0.008 (−0.009 to 0.024) |
a The results are expressed as the mean fold increase relative to the housekeeping genes GAPDH and TBP via 2−ΔCt. The Mann-Whitney U test was used for comparisons. n.d., nondetected; RT-qPCR, reverse transcription quantitative polymerase chain reaction.
bP < .05 for tendon cells versus BM-MSCs.
cP < .05 for tendon cells versus skin.
