Abstract
Background:
Posttraumatic osteoarthritis (PTOA) is secondary to an array of joint injuries. Animal models are useful tools for addressing the uniqueness of PTOA progression in each type of joint injury and developing strategies for PTOA prevention and treatment.
Hypothesis:
Intra-articular fracture induces PTOA pathology.
Study Design:
Descriptive laboratory study.
Methods:
Through a parapatellar incision, the medial tibial plateau was exposed in the left knees of 8 Sprague-Dawley rats. Osteotomy at the midpoint between the tibial crest and the outermost portion of the medial tibial plateau, including the covering articular cartilage, was performed using a surgical blade. The fractured medial tibial plateau was fixed with 2 needles transversely. The fractured knees were not immobilized. Before and after surgery, rat gait was recorded. Rats were sacrificed at week 8, and their knees were harvested for histology.
Results:
After intra-articular fracture, the affected limbs altered gait from baseline (week 0). In the first 2 weeks, the gait of the operated limbs featured a reduced paw print intensity and stride length but increased maximal contact and stance time. Reduction of maximal and mean print area and duty cycle (the percentage of stance phase in a step) was present from week 1 to week 5. Only print length was reduced in weeks 7 and 8. At week 8, histology of the operated knees demonstrated osteoarthritic pathology. The severity of the PTOA pathology did not correlate with the changes of print length at week 8.
Conclusion:
Intra-articular fracture of the medial tibial plateau effectively induced PTOA in rat knees. During PTOA development, the injured limbs demonstrated characteristic gait.
Clinical Relevance:
Intra-articular fracture represents severe joint injury and associates with a high rate of PTOA. This animal model, with histologic and gait validations, can be useful for future studies of PTOA prevention and early diagnosis.
Posttraumatic osteoarthritis (PTOA) is secondary to joint injury. PTOA accounts for 12% of symptomatic osteoarthritis (OA) and affects relatively younger patients than primary OA. 5 The development of PTOA can be divided into acute and chronic phases. Immediately after trauma, the structurally damaged joint has signs of hemarthrosis and inflammation. The chronic phase of the joint injury encompasses degeneration of articular cartilage and chronic inflammation in the joint. Unlike primary OA, which is closely associated with aging and has a subtle onset of the disease, PTOA has a definitive onset at the joint injury. From joint injury to the establishment of PTOA, there is a window of opportunity of prevention. 1 The clinical outcome of PTOA, however, has not significantly improved in the past 50 years. 11
Animal models have been used for investigating the pathogenesis of PTOA and evaluating treatment modalities that can prevent, halt, and cure PTOA. While it is still challenging to simulate primary OA in animals, 15 animal models of joint injury and PTOA are relatively straightforward. The most commonly used animal models of PTOA are meniscectomy with or without transaction of the anterior cruciate ligament or medial collateral ligament. 8,9 The ligamentous damage in these models significantly destabilizes the joints and makes the joints bear loads unequally. 2,3 Shift of loading intra-articularly in those destabilized joints primarily causes PTOA. 14,17 Joint instability has consistently induced PTOA in the knees of rodents and other species. 3,8,9 Joint injury, however, occurs in varied severity and different joints, which are distinct anatomically and functionally.
Intra-articular fracture is a severe joint injury with fracture extended into the joint and damaged articular cartilage. The incidence of PTOA dramatically increases when a joint injury is associated with intra-articular fracture. 13 Simulated in mouse knees, intra-articular fracture that was produced by striking the distal femur induced severe PTOA. 6 This is an excellent model for the natural history of PTOA. The high-energy trauma created is common in intra-articular fracture. In a closed fashion, however, the patterns of intra-articular fracture are uncertain. The consistency of fracture patterns among animals, especially injury to the articular cartilage, is critical for studying PTOA in a controlled manner.
In this study, intra-articular fracture of the medial tibial plateau was produced surgically in rat knees to ensure a consistent fracture pattern. The fracture was treated with internal fixation to mimic clinical management. The development of PTOA in knees with intra-articular fracture was followed with gait analysis and evaluated by histology.
Methods
For this study, 8 male Sprague-Dawley rats (Charles River Laboratory) 12 weeks of age were used (approved by an institutional animal care and usage committee).
Surgical Procedure of Intra-articular Fracture
Rats were placed in the supine position and anesthetized with inhalation of 3% isoflurane. The left hind limbs of the rats were shaved and the skin was sterilized using iodine-povidone swabs. The prepared limb was draped in a sterile fashion. While the left hind limb was maintained in flexion (about 90°) with a customized supporter placed underneath the knee, an incision was made slightly medial to the patellar tendon and around the patella to open the joint capsule. The patella was flipped laterally to expose the medial tibial plateau. The leg was held in complete flexion as an osteotomy was made using the tip of a surgical scalpel (No. 11), which was positioned at the midpoint between the tibial crest and the outermost aspect of the medial tibial plateau. The blade was vertical to the surface of the medial tibial plateau. Driven by a hammer, the blade cut through the articular cartilage and medial tibial plateau posteriorly. Free movement of the medial portion of the tibial plateau ensured the completion of the intra-articular fracture. The fractured tibial plateau was reduced. Two needles (gauge 21) were inserted transversely from the medial side of the fractured medial tibial plateau, crossing the fracture line, and anchored in the cortical bone of the tibia (Figure 1). The ends of the needles were buried subcutaneously. The patella was positioned back into place. The wound was approximated in layers with 3.0 nylon suture.

Radiogram of intra-articular fracture of the medial tibial plateau in a rat knee. Two needles were inserted across the fracture line for internal fixation. Arrows indicate the fracture line.
The procedure took approximately 10 minutes to complete. Carprofen (5 mg/kg) and cefazolin (25 mg/kg) were administered subcutaneously for pain relief and infection prevention, respectively, at the beginning of the procedure and then twice in the first 24 hours postsurgery. The operated limbs were not immobilized. The animals were housed individually in a light-, temperature-, and humidity-controlled environment.
Gait Analysis
CatWalk 7.1 gait analysis system (Noldus Co) was used to evaluate the gait pattern of 8 rats with intra-articular fracture of the medial tibial plateau. The CatWalk gait analysis system comprises a glass walkway that is confined as a corridor to direct and limit the movement of the rats and a digital camera mounted underneath the see-through glass walkway to capture the paw prints as rats walk across the walkway.
One week prior to intra-articular fracture creation, gait was recorded on 8 rats to establish a baseline. After the surgical procedure, gait was recorded every week until sacrifice in week 8. Each rat had to walk across the walkway 3 times, without interruption. Gait recordings of the 3 runs of each rat were averaged to minimize unintended gait alterations.
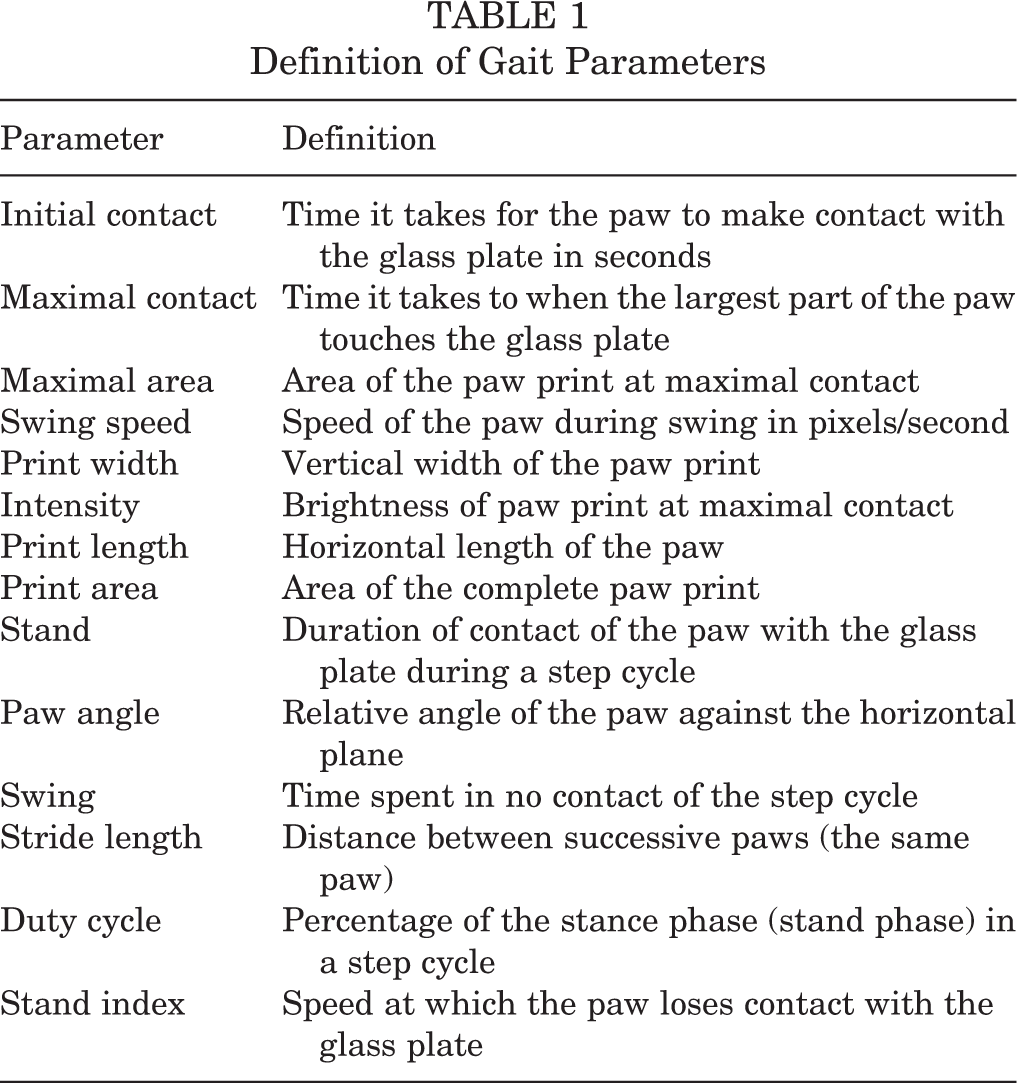
The images of paw prints were analyzed using the CatWalk program for a set of parameters, such as initial contact (time in seconds), maximal contact (time in seconds), maximal area (in pixels), intensity (brightness of pixels; range, 0-255), print length (in pixels), print width (in pixels), mean print area (in pixels), stride length (in pixels), duty cycle (% stand/[stand + swing]; that is, the percentage of the stance phase in a step cycle), stand (time in seconds), swing (time in seconds), paw angle (degree of paw axis with glass plate), swing speed (pixels/second), and stand index (time in seconds). Definitions of these 14 parameters (Reference Manual; Noldus Co) are listed in Table 1.
Definition of Gait Parameters
Histology
Rats were sacrificed 8 weeks after surgery. Both left and right knee joints were harvested for histological evaluation. The joints were fixed in 4% paraformaldehyde and then decalcified in 10% ethylenediaminetetraacetic acid (EDTA). After overnight incubation in 25% sucrose for cryoprotection, the samples were embedded in Tissue-Tek O.C.T. compound (Sakura Finetek USA) and stored at –80°C. Rat knees were sectioned along the frontal plane at a thickness of 10 µm using a cryostat. Tissue sections, including both distal femur and proximal tibia, were stained with safranin-O/fast green. The slides were scored by 2 investigators independently according to modified Mankin score, which quantifies the severity of OA pathology in the following 3 categories: structure (0-6), cells (0-3), and safranin-O stain (0-4). 10 The most severe OA has the highest Mankin score.
Statistical Analysis
The Mankin scores of the rat knees were compared between the hindlimbs with intra-articular fracture of the medial tibial plateau and nonoperated limbs using paired t tests. The gait parameters of the operated knees collected at multiple points, including baseline (week 0), were analyzed with 1-way repeated-measures analysis of variance (ANOVA) followed by a post hoc Tukey test. Gait parameters at week 8 and Mankin scores of the operated limbs were also correlated using Pearson correlation coefficients. Significance was set as P < .05.
Results
All animals survived the surgery and were able to stand and walk on the operated limbs 2 to 3 days postsurgery. No wound infection was observed.
Gait Alterations
In the first 2 weeks after surgery, the stance phase of the operated limbs was drastically increased (>60 times) from the baseline gait recorded before surgery (week 0; P = .0001). The time of stance gradually reduced in the subsequent weeks and returned to baseline in weeks 7 and 8 (P > .999) (Figure 2A). The swing time of the operated limbs increased after surgery from weeks 1 to 5 (except for week 4; P = .0001-.003) and then reduced to the level of baseline in week 6 (P = .06) (Figure 2B). The mean stride length of the operated limbs reduced approximately 10 to 30 times in the first 2 weeks after surgery (P = .0004 and .0001, respectively) and was virtually equal to baseline after 4 weeks (P > .999) (Figure 2C). The duty cycle of the operated limbs was markedly reduced approximately 25 times in weeks 1 and 2 (P = .0001). It was only partially recovered in weeks 4 and 5 (P = .003 for both) and completely recovered by week 6 (P > .999) (Figure 2D).

Gait alterations in the limbs with intra-articular fracture of the medial tibial plateau compared with baseline data (week 0). (A) The stance time of the operated limbs increased in weeks 1 and 2. (B) From weeks 1 to 5, swing time of the operated limbs increased over baseline (it did not reach statistical significance at week 4). (C) Stride length was reduced in the first 2 weeks. (D) Of the operated limbs, the duty cycle was reduced from baseline for weeks 1 to 5. (E) Maximal contact time was increased in the first 2 weeks after intra-articular fracture. (F and G) Both maximal and mean print area were reduced from weeks 1 to 5. (H) Print length of the operated limbs was generally reduced from baseline, especially at weeks 4, 7, and 8. (I) Print intensity was significantly reduced in the first 2 weeks. Statistically significant difference as compared with week 0: *P < .05; **P < .001.
Compared with baseline, the maximal contact time increased approximately 2-fold immediately after intra-articular fracture, and this increase lasted for 2 weeks (P = .04) (Figure 2E). The maximal and mean print area of the operated limbs was altered in the same fashion: compared with baseline, they were reduced from week 1 to 5 after surgery (P = .005-.04) but the reduction became statistically insignificant in weeks 7 and 8 (P = .2 for maximal print area; P = .1 and .09 for mean print area, respectively) (Figure 2, F and G). The print length of the operated limbs was reduced at week 4 (P = .003), and this became consistent in weeks 7 and 8 (P = .004 and .04, respectively) (Figure 2H). The print intensity of the operated limbs reduced nearly 30 times from baseline in the first 2 weeks after intra-articular fracture (P = .003) but recovered after 4 weeks postsurgery (P = .8) (Figure 2I).
After intra-articular fracture, the initial contact time (P > .1), paw print width (P > .1), paw angle (P > .09), and swing speed (P > .09) of the operated limbs were not significantly altered from baseline. Stand index of the operated limbs increased from its baseline at weeks 1 (P = .001) and 5 (P = .04).
PTOA Pathology
In week 8, the operated knees demonstrated severe articular destruction, particularly around the fracture site on the medial tibial plateau. Articular cartilage destruction was predominantly in the tibial cartilage but also appeared in the opposite femoral cartilage (Figure 3). Fractured bone by osteotomy was healed but the fracture site could be identified by the destruction of articular cartilage and interruption of the residual growth plate. Around destructed articular cartilage, proteoglycans in the cartilage matrix were depleted and chondrocyte clusters formed. The cartilage lesions were mainly limited in the medial compartment of the knees. The lateral compartment was intact structurally but proteoglycans depleted from some areas. The mean (±SD) Mankin score of the operated knees was 8.8 ± 1.4, which was significantly higher than nonoperated knees (1.4 ± 1.2, P = .00002) (Table 2).

Histology of a rat knee with intra-articular fracture of the medial tibial plateau at week 8. The site of osteotomy was indicated by the visible cuttings in articular cartilage and interruption of the growth plate, although bone fracture was healed. Articular cartilage was extensively damaged on the medial tibial plateau and the opposite femoral condyle. Areas of cartilage were depleted of proteoglycans (safranin O/fast green staining). Arrows, osteotomy site; scale bar = 100 µm.
Modified Mankin Histological Scores

Correlation of Gait with PTOA Pathology
Although the operated limbs had a reduced print length at week 8, this gait feature was not correlated with the Mankin scores of the knees in the same limbs (r = 0.38, P = .39).
Discussion
There are several factors that should be carefully considered when evaluating the clinical relevance of an animal model of PTOA. Animal species and maturity are well-known factors that may influence the healing of the joint. 16 How to produce the joint injury is key to simulating PTOA development in the animals. The joint injury model should also consider clinical factors, such as routine treatments. The procedure of joint injury should be reproducible and practical to be performed on small animals. In this study, a medial parapatellar approach directly exposed the medial tibial plateau. Without the need of extensive tissue dissection, this approach effectively reduced the surgical time and minimized surgical trauma to the animals. The site of osteotomy was determined with easily recognizable anatomical landmarks. The consistency of osteotomy performed on the medial tibial plateau improved the comparability of intra-articular fracture among experimental animals. The surgical tools used for the osteotomy procedure are conveniently available. Since intra-articular fracture was directly visible, there was no need of postoperative imaging for confirmation of fracture and fracture fixation. Most important, histology revealed osteoarthritic pathology in all operated knees, validating the procedure used in this study as a model of PTOA.
This study monitored gait alterations of the operated limbs while PTOA developed in the knees with intra-articular fracture. Indeed, the affected limbs changed in static (maximal contact time, maximal print area, intensity, print length, mean print area, stance time, duty cycle, and stride length) and dynamic (swing and stand index) gait parameters as compared with baseline data collected before surgery (week 0). The study did not compare gait between operated and nonoperated limbs because the nonoperated limbs would compensate for the affected limbs during walking.
In a PTOA model, gait can be influenced by 2 major pathologies in 2 distinct phases: (1) acute injury and (2) joint degeneration. In rodent models, fractures heal in about 2 to 3 weeks and joint degeneration occurs in 3 to 4 weeks. 6 In the acute injury phase, pain and joint dysfunction contribute to gait alteration. A typical pain gait features reduced intensity paw print, stride length, and ground contact time. 4,7,18 In this study, the paw print intensity and stride length of limbs with intra-articular fracture were dramatically reduced in the first 2 weeks after surgery. The gait of arthritic animals exhibited an increased time during a step cycle due to motion restriction. 12 The increased time of maximal contact and stance in the operated limbs in the current study may be relevant to joint dysfunction immediately after intra-articular fracture. Gait alterations in maximal contact time, stance time, intensity, and stride length became insignificant after week 4, indicating the end of the acute injury phase in the joints.
Joint degeneration or PTOA develops in the joints 3 weeks after intra-articular fracture. 6 Compared with baseline data, maximal and mean print area and duty cycle of the operated limbs were decreased from weeks 1 to 5, but the reductions became insignificant by week 6. Gait parameters that were altered in this postoperative period also included an increased time of limb swing from weeks 1 to 5, which returned to baseline by week 6. Since PTOA pathology is progressive in the joints, the normalization of maximal and mean print area and duty cycle of the operated limbs at a late stage of PTOA suggests that they are not specific signs of PTOA. Most likely, this group of gait parameters is related to traumatic joint inflammation, which plays a role in the development of PTOA in both acute and chronic phases.
By week 8, the operated joints were degenerative. The corresponding gait showed a reduced print length. It is unclear whether this was due to a change in paw weightbearing related to joint deformity in the knees. Nevertheless, this gait alteration in the late stage of PTOA did not correlate with the severity of the PTOA pathology quantified with Mankin scores.
A limitation of the current model is that the intra-articular fracture was not produced by high-energy trauma, which is commonly seen in similar joint injuries clinically. The gait analysis provided insight of the functionality of the knees during the development of PTOA. Using this animal model, future studies are necessary to address correlations between PTOA pathology and gait at each stage of PTOA development. Gait alterations found in this study, however, should be interpreted with caution because of the differences in locomotion between humans and animals.
Conclusion
Surgically created intra-articular fracture of the medial tibial plateau consistently produced PTOA in rat knees. This animal model of PTOA is reproducible and minimally invasive. The model and its gait features could be useful for preclinical investigations of therapeutic modalities for PTOA.
Footnotes
Acknowledgment
The authors thank Ivan Gunev, MD, Department of Pathology, MedStar Union Memorial Hospital, for his assistance in histology and Mr Sione Fanua, Microsurgical Laboratory, MedStar Union Memorial Hospital, for his help in animal surgery and care.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution.
