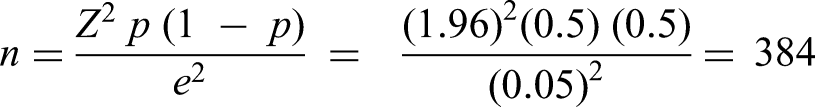Abstract
Background
This study assessed treatment outcomes, medication adherence, and associated factors among human immunodeficiency virus (HIV) patients receiving dolutegravir-based antiretroviral therapy (ART) at Tikur Anbessa Specialized Hospital, Ethiopia.
Methods
A hospital-based retrospective study was conducted from February to May 2024 using medical records of patients treated between 2018 and 2020. A total of 425 records were selected through random sampling. Sociodemographic, clinical, and treatment-related data were collected using a structured tool and analyzed with SPSS.
Results
Among participants, 58.8% were female, and 76.0% were aged 31 to 60 years. Overall, medication adherence was 85.7%, and viral load suppression was achieved in 99.8% of patients. All patients were screened for tuberculosis, and 60% received isoniazid preventive therapy. Adherence was significantly associated with World Health Organization clinical stage, follow-up status, advanced HIV disease, and antiretroviral dose days.
Conclusion
Dolutegravir-based ART showed excellent virologic outcomes and high adherence levels, highlighting key factors influencing treatment success.
Plain Language Summary
Dolutegravir-based antiretroviral therapy (ART) is now widely used to treat people living with human immunodeficiency virus (HIV) because it is effective and well-tolerated. However, the success of HIV treatment depends not only on the medicine itself but also on how consistently patients take their medication and remain engaged in care. This study looked at treatment outcomes, medication adherence, and factors linked to poor adherence among HIV patients receiving dolutegravir-based ART at Tikur Anbessa Specialized Hospital in Addis Ababa, Ethiopia.
Researchers reviewed the medical records of 425 adult patients who had been taking a dolutegravir-based ART regimen for at least 6 months between 2018 and 2020. Information on patient characteristics, treatment history, tuberculosis screening, preventive therapy, and medication adherence was collected and analyzed.
The findings showed that treatment outcomes were very strong. Nearly all patients achieved viral load suppression, meaning the amount of HIV in their blood was reduced to very low levels. Most patients (about 86%) were taking their medications as prescribed. All patients were screened for tuberculosis, and the majority received preventive treatment with isoniazid. However, some patients had difficulty maintaining good adherence. Lower adherence was more common among patients with advanced HIV disease, those who had restarted treatment after interruption, and those classified as clinically unstable. Adherence was also affected by the HIV disease stage and how ART services were delivered. Patients who received longer medication refills and were enrolled in less frequent clinic visit models showed much better adherence.
Overall, this study shows that dolutegravir-based ART is highly effective in clinical settings. It also highlights the importance of providing tailored care, early disease management, and supportive service delivery models to help patients maintain consistent medication use and achieve the best possible health outcomes.
Introduction
Human immunodeficiency virus (HIV) remains a major global public health challenge, affecting millions of individuals worldwide. In 2023, an estimated 39.9 million people were living with HIV globally, with approximately 1.3 million new infections and 630 000 deaths attributed to Acquired Immunodeficiency Syndrome (AIDS)-related illnesses. 1 Substantial regional disparities persist, with sub-Saharan Africa bearing the greatest burden of the epidemic. These variations are influenced by socioeconomic conditions, cultural practices, and disparities in access to healthcare services. 2 In Ethiopia, HIV/AIDS continues to be a significant public health concern. According to the Joint United Nations Program on HIV/AIDS, an estimated 690 000 people were living with HIV in 2019, with an adult prevalence of 0.9% among individuals aged 15 to 49 years. 3
The widespread scale-up of combination antiretroviral therapy (ART) has been the primary driver in reducing AIDS-related mortality, with deaths declining by 51% from a peak of 1.9 million in 2004 to 0.94 million in 2017. The expansion of ART access marked a transformative milestone in HIV care, shifting the disease from a fatal condition to a manageable chronic illness by restoring immune function and suppressing viral replication to undetectable levels. This transition has also played a critical role in preventing onward transmission of the virus.4,5 Furthermore, the introduction of highly active antiretroviral therapy (ART) revolutionized HIV management by effectively suppressing viral replication and significantly improving patient prognosis. 6
Advances in antiretroviral pharmacotherapy have further enhanced treatment efficacy, safety, and tolerability. Dolutegravir (DTG), a potent integrase strand transfer inhibitor with a high genetic barrier to resistance, has demonstrated superior efficacy in suppressing viral replication and improving clinical outcomes. Since April 2019, Ethiopia has been transitioning to DTG-based regimens as the preferred first-line therapy due to their enhanced viral suppression and favorable resistance profile.7–9 Despite these advantages, comprehensive evidence on long-term outcomes, including medication adherence, virologic suppression, and broader epidemiological impacts, remains limited in the Ethiopian context. 10
Although DTG exhibits strong antiviral activity, emerging evidence indicates that some patients experience adverse effects, including insomnia, weight gain, and neuropsychiatric symptoms, which may compromise treatment adherence.11,12 For instance, a study conducted in Kenya reported greater weight gain among patients receiving DTG compared to those on efavirenz-based regimens, raising concerns about potential long-term metabolic complications. 13 Similarly, other studies have shown that, despite its overall tolerability, a subset of patients discontinue DTG due to intolerable side effects. 14 These findings highlight the importance of continuous patient monitoring and individualized treatment approaches to optimize outcomes.
Medication adherence remains a critical determinant of ART success. Evidence from South Africa indicates that, although DTG-based regimens have improved rates of viral suppression, adherence challenges persist, particularly among socioeconomically disadvantaged populations. Factors such as stigma, limited social support, and financial constraints continue to hinder optimal adherence.15,16 In addition, despite DTG's high genetic barrier to resistance, cases of resistance have been reported, especially among patients with prior exposure to suboptimal ART regimens. Studies from Uganda have demonstrated an increased risk of DTG resistance in individuals with previous treatment failure, underscoring the need for vigilant monitoring and effective treatment management strategies.17,18
The transition to DTG-based regimens in Ethiopia, while promising, has not been without challenges. Implementation barriers, including supply chain interruptions, gaps in healthcare provider training, and limited patient awareness, have affected the effective rollout of the regimen. 19 Therefore, there is a need to evaluate treatment outcomes, medication adherence, and associated factors in specific settings such as Tikur Anbessa Specialized Hospital (TASH). Generating context-specific evidence will help identify barriers and facilitators to optimal HIV care, inform targeted interventions to improve adherence and manage adverse effects, and support the successful scale-up of DTG-based ART in Ethiopia. Ultimately, such evidence will contribute to national and global efforts to control HIV/AIDS through informed, evidence-based strategies.
Methods
Study Design and Setting
A hospital-based retrospective study was conducted at the ART clinic of TASH in Addis Ababa, Ethiopia. TASH is one of the largest referral and teaching hospitals in the country, serving as a training center for undergraduate and postgraduate health science students. The hospital has more than 800 inpatient beds and provides healthcare services to over 500 000 patients annually. The ART clinic at TASH offers comprehensive HIV care services free of charge, including ART initiation and follow-up, management of opportunistic infections, HIV testing and counseling, care for HIV-exposed infants, family planning services, adolescent psychosocial support, and adherence counseling for adults. The study was conducted over the period from February 1 to May 30, 2024. The study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines (STROBE) for cross-sectional studies 20 (see Supplemental materials).
Study Population and Sample
The source population comprised all people living with HIV/AIDS receiving care at TASH. The study population included adult patients (aged ≥18 years) who had been receiving a DTG-based ART regimen for at least 6 months at the hospital during the study period. The sample size was determined and calculated using Cochran's formula:
To account for potential incomplete records, a 10% contingency was added to the calculated sample size, resulting in a final target of 425 patient records. A probability sampling approach was employed to ensure representativeness.
Initially, a comprehensive sampling frame was developed by identifying all eligible patients aged ≥18 years who had been receiving a DTG-based ART regimen for at least 6 months between 2018 and 2020, using the SMART-CARE database. Each eligible patient record was assigned a unique identification number. From a total of 1290 eligible patients, a sample of 425 was selected using systematic random sampling. The sampling interval (k) was determined by dividing the total number of eligible patients by the required sample size (1290/425 ≈ 3). After selecting a random starting point, every third patient record was included in the study sample.
Regarding the eligibility criteria, adults (≥18 years) living with HIV who had been receiving a DTG-based ART regimen for at least 6 months at TASH were included. Patients younger than 18 years, those on a DTG-based regimen for less than 6 months, and individuals with incomplete medical records were excluded.
Data Collection and Management
Data were collected through a retrospective review of patient medical records documented in the SMART-CARE system. A pretested, structured data extraction tool developed in Microsoft Excel was used to collect relevant information. The tool was reviewed by experts to ensure content validity and comprehensiveness. Data on sociodemographic and clinical characteristics including age, sex, World Health Organization (WHO) clinical stage, cluster differentiation 4 (CD4) count, body mass index (BMI) at ART initiation, current ART regimen, time from HIV diagnosis to ART initiation, duration on ART, presence of opportunistic infections (including tuberculosis [TB]), regimen changes, and treatment outcomes were extracted from patient records. Laboratory data, including CD4 cell counts and viral load measurements, along with information on opportunistic infection screening status and medication adherence, were extracted from patient records. The operational definition of viral suppression is a viral load ≤50 copies/mL. Accordingly, an undetectable viral load, typically defined as <50 copies/mL, depending on the sensitivity of the assay, is regarded as virological suppression in accordance with the Ethiopian national ART treatment guidelines. 21
Data collectors received training on the study objectives, data collection procedures, and the use of the extraction tool. Ongoing supervision and support were provided by the principal investigator throughout the data collection process. A pretest was conducted on 5% of the sample prior to the main data collection, and necessary revisions were made to the tool based on the findings.
Data Analysis
The collected data were checked for completeness, cleaned, and exported to SPSS statistical software for analysis. Descriptive statistics were used to summarize sociodemographic and clinical characteristics of the study participants. Multivariate logistic regression analysis was performed to identify independent predictors of treatment outcomes and medication adherence. A P-value of less than .05 was considered statistically significant.
Ethical Considerations
Ethical approval was obtained from the Institutional Ethics Review Committee. A letter of support was submitted to the hospital administration to secure permission for data access. The study was conducted in accordance with the principles of the Declaration of Helsinki. Confidentiality of patient information was maintained throughout the study by anonymizing data and restricting access to authorized personnel only.
Results
Sociodemographic and Clinical Characteristics of the Study Participants
Among the 425 HIV/AIDS patients receiving DTG-based ART at the TASH follow-up clinic, the majority were female (58.8%, n = 250). Most participants (76.0%, n = 323) were aged between 31 and 60 years. Regarding the DTG-based ART regimen distribution, 9.2% (n = 39), 59.3% (n = 252), and 30.6% (n = 130) were on the first, second, and third rounds, respectively. The majority of participants had been on DTG-based ART for 48 months (57.2%, n = 243), followed by 60 months (33.4%, n = 142). In terms of tuberculosis preventive therapy (TPT), 60.0% (n = 255) received isoniazid (INH) preventive therapy, while 36.5% (n = 155) did not receive any TPT. Nearly all patients (98.4%, n = 418) had uninterrupted ART follow-up (Table 1).
Sociodemographic and Clinical Characteristics of the Study Participants on DTG-Based ART Regimen, at Tikur Anbessa Specialized Hospital.

Abbreviations: DTG, dolutegravir; ART, antiretroviral therapy; CD4, cluster differentiation 4; VL, viral load; TB, tuberculosis; CCA, cryptococcal antigen; AHD, advanced HIV disease; WHO, World Health Organization; ARV, antiretroviral; TPT, tuberculosis preventive therapy; INH, isoniazid; MMD, multimonth dispensing; HEP-CAG, health extension professional-led community adherence group; ASM, appointment spacing model; DSD, differentiated service delivery.
Viral load suppression was achieved in 99.8% (n = 424) of participants, and 81.2% (n = 345) were categorized as clinically stable. CD4 count data were largely unavailable, with 98.8% of records lacking this information. All patients were screened for TB, with 99.8% testing negative. Recent viral load measurements indicated that 95.8% of patients had undetectable viral load levels, and overall viral suppression was achieved in 99.8% of the cohort. Cryptococcal antigen screening was performed in 53.6% of patients. Advanced HIV disease (AHD) was identified in only 0.2% of participants, while 99.8% had no documented opportunistic infections. The vast majority of patients (99.5%) were classified as WHO clinical stage I (Table 1).
Regarding ART dispensing, 80.5% of patients were on a 180-day refill schedule, while 19.5% were on a 90-day schedule. At enrollment, 92.0% of participants were categorized under the appointment spacing model (ASM)/6-month multimonth dispensing (6MMD), 6.6% under 3 multimonth dispensing (3MMD), and smaller proportions under AHD/adolescent differentiated service delivery (DSD) and health extension professional-led community adherence groups (HEP-CAG). At the time of assessment, 80.5% remained in ASM/6MMD, 16.7% in 3MMD, and the remaining participants were distributed across the AHD, HEP-CAG, and adolescent DSD categories (Table 1).
Factors Influencing Adherence
Several factors were found to be significantly associated with adherence to DTG-based ART regimens (P < .05). These included WHO clinical stage (P = .046), follow-up status (P = .03), type of TPT (P = .004), presence of AHD (P = .014), DSD category at enrollment (P < .001), recent assessment category (P < .001), recent DSD category (P < .001), and ART dispensing duration (P < .001). Higher adherence rates were observed among patients classified as stable recent assessment (99.1% adherent) and those enrolled in the ASM/6MMD DSD category (99.1% adherent). In contrast, patients categorized as unstable recent assessment and those in the 3MMD DSD category demonstrated substantially lower adherence, with nonadherence rates of 72.5% and 69.0%, respectively. Additionally, patients receiving 180-day ART refills exhibited markedly higher adherence (99.1%) compared to those on 90-day refills (30.1%) (Table 2).
Cross-Tabulation of Variables Influencing Adherence Among HIV Patients on DTG + 3TC + TDF at TASH.
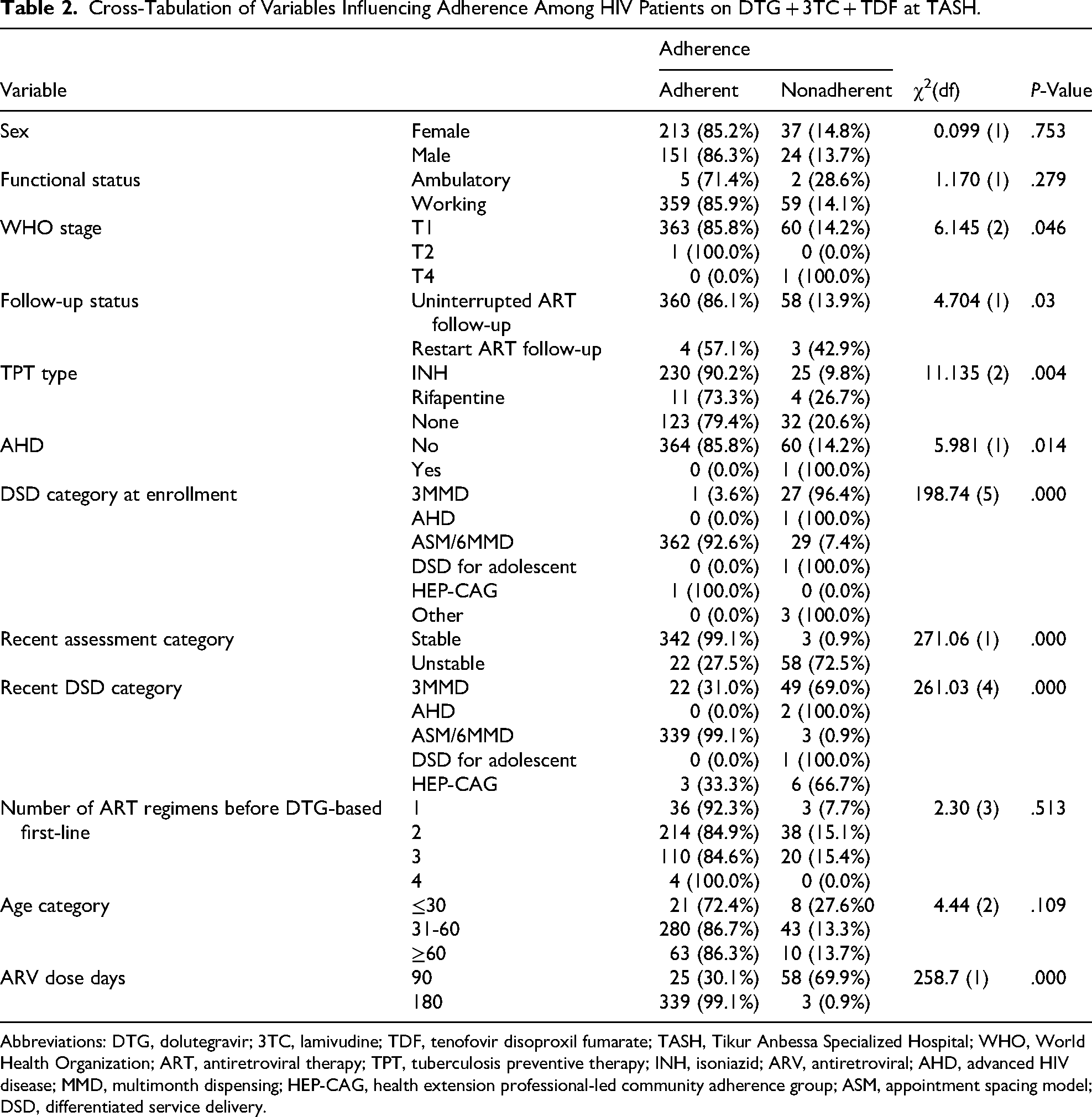
Abbreviations: DTG, dolutegravir; 3TC, lamivudine; TDF, tenofovir disoproxil fumarate; TASH, Tikur Anbessa Specialized Hospital; WHO, World Health Organization; ART, antiretroviral therapy; TPT, tuberculosis preventive therapy; INH, isoniazid; ARV, antiretroviral; AHD, advanced HIV disease; MMD, multimonth dispensing; HEP-CAG, health extension professional-led community adherence group; ASM, appointment spacing model; DSD, differentiated service delivery.
Nutritional Status of Study Participants
Assessment of nutritional status revealed that 50.1% (n = 213) of participants had a normal BMI. However, 31.5% (n = 134) were classified as overweight and 10.8% (n = 46) as obese. A smaller proportion of patients was identified as having mild, moderate, or severe malnutrition (Figure 1).

Nutritional status of HIV/AIDS patients on DTG + TDF + 3TC at Tikur Anbessa Specialized Hospital. Abbreviations: DTG, dolutegravir; TDF, tenofovir disoproxil fumarate; 3TC, lamivudine.
Adherence Status of Study Participants
According to the registry data, 85.7% of participants demonstrated good adherence to ART. In contrast, 8.9% and 5.4% exhibited fair and poor adherence, respectively. When adherence was dichotomized, 85.7% of participants were classified as adherent (good adherence), while 14.3% were considered nonadherent (combining fair and poor adherence).
Discussion
This study, conducted at TASH, evaluated treatment stability and factors influencing adherence among people living with HIV receiving a DTG-based regimen combined with lamivudine and tenofovir disoproxil fumarate. The findings revealed that the majority of participants were female (58.8%) and aged between 31 and 60 years (76.0%). This demographic distribution is consistent with findings from other African settings. For example, a study conducted in South Africa reported a similar predominance of female patients (approximately 60%) and a comparable age distribution among individuals receiving ART. 22 This pattern is often attributed to higher HIV testing uptake and better healthcare-seeking behavior among women. 15
Clinically, most participants had previously received 2 ART regimens (59.3%) and had been on their current DTG-based regimen for 48 months (57.2%). The overall adherence rate observed in this study (85.7%) was below the recommended optimal adherence threshold (≥95%) for ART. 23 However, the target viral load suppression rate was successfully achieved. These findings are comparable to those reported in a study from Nigeria, which also demonstrated high viral suppression despite suboptimal adherence levels. 24
Nearly all participants were classified as WHO clinical stage I (99.5%), with very few in advanced stages. Early-stage HIV infection is generally associated with better treatment outcomes, which likely contributed to the high rate of viral suppression observed in this cohort. Similar findings have been reported in studies from Spain, where patients receiving DTG-based regimens achieved viral suppression rates exceeding 95%. 25 The effectiveness of DTG-based regimens is well established, largely due to their high genetic barrier to resistance, which plays a critical role in sustaining viral suppression.26,27
All participants in this study were screened for TB, and 60% received INH TPT prophylaxis, while a smaller proportion (3.53%) received rifapentine-based prophylaxis. This coverage is lower than that reported in a study conducted in Addis Ababa, 28 possibly due to differences in study design, population, or sample size. The use of INH preventive therapy has been shown to significantly reduce the incidence of TB among people living with HIV, as well as mitigate complications associated with TB treatment initiation. 29 Routine screening for opportunistic infections, particularly TB, remains essential in high-burden settings such as Ethiopia. Evidence from South Africa highlights that routine TB screening in HIV clinics improves early detection and management of TB–HIV coinfection. 30
The nutritional assessment indicated that a considerable proportion of participants was overweight (31.5%) or obese (10.8%). This finding is consistent with studies from India, which have reported similar trends of weight gain following ART initiation. 31 These observations underscore the growing importance of addressing metabolic complications in HIV care and integrating nutritional counseling into routine clinical management. 32
This study identified several factors significantly associated with ART adherence, including WHO clinical stage, follow-up status, type of TPT, AHD status, DSD model, recent assessment, and ART dispensing duration. These findings showed the multifactorial nature of ART adherence, and both clinical and programmatic factors contribute to maintaining optimal adherence levels among ART patients. One of the most common findings of this study was the association between recent assessment and adherence rates. Patients classified as clinically stable demonstrated significantly higher adherence (99.1%) compared to those categorized as unstable, among whom 72.5% were nonadherent. This is consistent with the existing literature indicating that clinical stability is a prerequisite for enrollment in less intensive DSD models, which are designed to enhance adherence by reducing patient burden and improving convenience.33,34 The stability of a patient often reflects better immunological recovery and fewer opportunistic infections, thereby reducing the likelihood of missed doses due to illness or side effects.
Similarly, the DSD model at the time of assessment had a substantial impact on adherence. Patients enrolled in the 6MMD model, particularly within the ASM, demonstrated very high adherence (99.1%), whereas those in the 3MMD model exhibited significantly lower adherence, with 69.0% classified as nonadherent. This finding supports evidence that extended ART refill intervals, when clinically appropriate, improve adherence by reducing the frequency of clinic visits, transportation costs, and time burdens, especially in resource-limited settings.35,36
Adherence also varied by ART dispensing duration. Patients receiving a 180-day supply had markedly higher adherence (99.1%) compared to those receiving a 90-day supply (30.1%). While this may partly reflect selection bias, whereby more stable patients are eligible for longer dispensing intervals, it aligns with global recommendations advocating for multimonth dispensing for stable patients. 34
The association between the WHO clinical stage and adherence (P = .046) suggests that patients with advanced disease may face greater challenges, including medication side effects, comorbidities, and psychosocial barriers. This is consistent with findings by Angelo et al, who reported lower adherence among patients with WHO stage III/IV disease. 37 Similarly, patients with AHD (P = .014) may experience reduced adherence due to increased clinical complexity and susceptibility to opportunistic infections.
Follow-up status was also significantly associated with adherence, with patients retained in care demonstrating better adherence outcomes. This finding aligns with evidence identifying retention in care as a critical determinant of sustained ART adherence. 38 Additionally, the type of TPT was associated with adherence (P = .004), suggesting that regimen complexity or duration may influence adherence behavior. Further studies are needed to elucidate the mechanisms underlying this relationship.
This study has some limitations that may constrain the generalizability of its findings. First, its retrospective design limited the availability and completeness of some patient data. Second, as the study was conducted in a single tertiary hospital, the findings may not be generalizable to other settings in Ethiopia. Third, incomplete documentation in medical records may have affected data quality. Furthermore, the exceptionally high viral suppression rate may reflect selection bias, as most participants were clinically stable, enrolled in DSD models, and on ART for at least 6 months. This may limit generalizability and overestimate true population-level viral suppression. Despite these limitations, the study provides valuable insights into the importance of individualized HIV care and the role of DSD models in improving adherence and treatment outcomes. Ensuring clinical stability before transitioning patients to less intensive care models and extending ART refill intervals can significantly enhance adherence. Moreover, close monitoring of patients with advanced disease or unstable conditions remains essential to prevent poor outcomes.
Conclusion
A substantial proportion of patients receiving DTG-based ART achieved optimal viral load suppression and demonstrated high levels of treatment adherence. TB screening was implemented for all patients living with HIV, and the majority of patients received INH preventive therapy. Several factors including WHO clinical stage, follow-up status, type of TPT, presence of AHD, DSD model, assessment category, and ART dispensing duration were significantly associated with adherence. These findings underscore the critical role of both clinical and programmatic factors in shaping adherence and treatment outcomes. Strengthening patient-centered care, optimizing DSD models, and ensuring continuous monitoring of high-risk patients are essential to further improve ART adherence and sustain treatment success.
Supplemental Material
sj-xlsx-1-jia-10.1177_23259582261446464 - Supplemental material for Treatment Outcome, Medication Adherence, and Associated Factors After the Introduction of Dolutegravir-Based Antiretroviral Therapy (ART) Regimen Among HIV/AIDS Patients in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia
Supplemental material, sj-xlsx-1-jia-10.1177_23259582261446464 for Treatment Outcome, Medication Adherence, and Associated Factors After the Introduction of Dolutegravir-Based Antiretroviral Therapy (ART) Regimen Among HIV/AIDS Patients in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia by Zenebe Negash, Yonatan Fekadu, Mengistu Girma Tirore, Zelalem Tilahun Tesfaye and Malede Berihun Yismaw in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582261446464 - Supplemental material for Treatment Outcome, Medication Adherence, and Associated Factors After the Introduction of Dolutegravir-Based Antiretroviral Therapy (ART) Regimen Among HIV/AIDS Patients in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia
Supplemental material, sj-docx-2-jia-10.1177_23259582261446464 for Treatment Outcome, Medication Adherence, and Associated Factors After the Introduction of Dolutegravir-Based Antiretroviral Therapy (ART) Regimen Among HIV/AIDS Patients in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia by Zenebe Negash, Yonatan Fekadu, Mengistu Girma Tirore, Zelalem Tilahun Tesfaye and Malede Berihun Yismaw in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank all data collectors, participants, and other staff of the Tikur Anbessa Specialized Hospital. The authors would also like to thank all those who helped facilitate the research and provided constructive feedback.
Ethical Considerations
The research was conducted after obtaining approval from the ethics review board of the School of Pharmacy, Addis Ababa University (Ref. No. ERB/SOP/577/16/2024). The study was performed in accordance with the Declaration of Helsinki.
Consent to Participate
Information was obtained from patients’ medical records; hence patients’ consent is not applicable as historical data was evaluated.
Consent for Publication
Information was obtained from patients’ medical records; hence patients’ consent is not applicable as historical data was evaluated.
Author Contributions
ZN, YF, MGT, ZTT, and MBY made major contributions to the reported work, including the concept and methodology of the study, execution, data collection, analysis, and interpretation; involved in the drafting, revision, or critical review of the manuscript; gave final approval for the version to be published; agreed on the journal to which the study was submitted; and accepted responsibility and accountability for the contents of the article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.