Abstract
Objective
This study assessed the prevalence and factors associated with lipodystrophy and dyslipidemia among 105 children (aged 5 to 15 years) on HAART at the Komfo Anokye Teaching Hospital.
Methods
In this cross-sectional study, data collection included anthropometric measurements, clinical evaluations, dietary assessments, and lipid profile assessments. Logistic regression and students’ t-test were used to identify associations, with significance set at P < .05.
Results
Lipodystrophy prevalence was 11.4%, with lipohypertrophy (10.5%) more common than lipoatrophy (0.9%). Dyslipidemia affected 25.7% of participants. Participants who had entered puberty (as indicated by Tanner staging) were approximately 10 times more likely to have lipodystrophy (OR = 9.91; 95% CI: 1.32-74.23, P = .026). Higher mid-upper arm circumference was associated with significantly lower odds of lipodystrophy (OR = 0.60; 95% CI: 0.36-0.99, P = 0. 046. Participants with lipodystrophy had lower odds of elevated triglycerides (OR: 0.1; 95% CI: 0.00-0.62 P = .029). Previous lopinavir/ritonavir use was significantly associated with reduced odds of dyslipidemia (OR: 0.21; 95% CI: 0.05-0.91, P = .036).
Conclusion
The high prevalence of dyslipidemia and lipodystrophy emphasizes the need for routine lipid and other metabolic assessments among children and adolescents living with HIV.
Introduction
Background
The introduction of the Highly Active Anti-Retroviral Therapy (HAART) has been a remarkable feat in the management of HIV/AIDS, significantly suppressing viral loads, reducing morbidity, and ultimately increasing the life expectancy of persons living with HIV (PLWH).1–3 The frequently used HAART regimens consist of protease inhibitors (PIs), nucleoside reverse-transcriptase inhibitors (NRTIs), and non-nucleoside reverse-transcriptase inhibitors (NNRTIs), which are administered as a combination of 3 or 4 pharmacological agents.3,4 Previously, the World Health Organization (WHO) advocated for a combination of 2 NRTIs and 1 NNRTI or PI to be employed as HAART regimens in resource-limited environments such as Ghana. Contemporary HIV treatment strategies advocate for the replacement of NNRTIs and PIs with integrase strand transfer inhibitors (INSTIs), notably dolutegravir (DTG). The introduction of DTG into HAART regimens in Ghana in 2019 has led to substantial improvements in HIV management, particularly when combined with lamivudine, and tenofovir or abacavir in fixed-dose formulations. 5
Although the use of HAART is crucial in managing HIV/AIDS, its utilization is correlated with metabolic side effects and adverse reactions, which include hypersensitivity rashes, anemia, gastrointestinal disorders, lipodystrophy, dyslipidemia, and irregularities in glucose metabolism. Certain regimens tend to exacerbate these effects; for instance, PI-based treatments have been associated with dyslipidemia, while NRTIs are linked to elevated triglycerides and lipoatrophy.4,6
Lipodystrophy syndrome is a complication usually associated with prolonged use of long-term HAART. It manifests as loss of adipose tissue in the facial area, extremities, and buttocks, accompanied by central adiposity in regions such as the abdomen, breast, and dorsocervical spine. This syndrome consists of 3 forms: lipoatrophy, lipohypertrophy, and a mixed pattern. 7 These physical alterations are frequently observed alongside dyslipidemia, characterized by an isolated elevation in serum triglycerides, cholesterol, or both, in addition to various modifications in cholesterol concentration levels, particularly those of low-density lipoprotein (LDL) or high-density lipoprotein (HDL). Lipodystrophy, dyslipidemia, and glucose dysregulation may occur in adult and pediatric populations receiving HAART. These conditions increase the individual's cardiovascular risk.1,2,8–10 Numerous factors have been linked to the emergence of metabolic disorders in individuals undergoing HAART. Notable factors include the duration of HAART, age, gender, viral load, body-mass index (BMI), the specific HAART regimen utilized, as well as the severity and duration of HIV infection. Additional variables, such as dietary habits, levels of physical activity, and genetic predisposition, have been proposed as potential contributors to this issue.11–13
It is estimated that only a quarter of children under the age of 15 living with HIV in Ghana are on HAART. 14 However, there is a significant lack of local data regarding the adverse effects of these regimens and potential remedies. The scarcity of information on HAART-related lipodystrophy, dyslipidemia, and abnormalities in glucose metabolism is particularly pronounced among the pediatric population. The metabolic alterations associated with long-term HAART can elevate cardiovascular risk during adulthood. The abnormalities in body fat distribution may also adversely affect self-esteem and potentially hinder treatment adherence. 10 This study highlights the prevalence of dyslipidemia and lipodystrophy and identifies associated sociodemographic, nutritional and clinical factors among Ghanaian children on HAART.
Aim
This study aimed to determine the prevalence and determinants of lipodystrophy and dyslipidemia among Ghanaian children on HAART.
Subjects and Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental reference file). 15
Study Design, Subjects and Setting
This was a cross-sectional study with participants recruited between May and August 2024. Participants were eligible for inclusion if they were between 5 and 15 years of age, had been receiving HAART for a minimum of 6 months and had no known metabolic disorders. Patients who did not give informed consent or those who were acutely ill at the time of recruitment were excluded. The study was conducted at the outpatient Paediatric HIV Clinic in the Directorate of Child Health of the Komfo Anokye Teaching Hospital. The Komfo Anokye Teaching Hospital (KATH) is a tertiary hospital located in Kumasi, the second largest city in Ghana and the capital of the Ashanti Region. The hospital has a 1200-bed capacity and serves as a referral center for 13 out of the 16 regions in Ghana. 16
Eligibility Criteria
Children living with HIV (5-15 years) who had been on HAART for at least 6 months, had no metabolic conditions, and were not acutely ill were included in this study. The legal guardians of prospective participants were approached and briefed on the study by a nurse at the facility. The respective children are then recruited upon the guardians’ consent. The convenience sample technique was used.
Ethical Approval
The study was conducted per the principles outlined in the Declaration of Helsinki and the Committee on Publication Ethics (COPE). Ethical approval was obtained from the Institutional Review Board of the Komfo Anokye Teaching Hospital (reference number KATH IRB/AP/053/24). Permission was also obtained from the leadership of the Paediatric HIV Clinic. Written informed consent was obtained from parents and legal guardians of all participants prior to their participation in this study as they were all less than 18 years old. Consent was obtained to collect data from subjects including anthropometric data and blood sample collection to assess lipid profile. Assent was also obtained from participants where appropriate.
Sample Size
The sample size was calculated based on the primary objective of the study, which was to estimate the prevalence of lipodystrophy among children receiving HAART. Using a reported prevalence of lipodystrophy of 8.4%, 11 we applied the Cochran's formula 17 for sample size calculation: n = Z² * p * (1−p) / d², where Z = 1.96 for a 95% confidence interval, p = 0.084 (8.4%), and d = 0.05 (5% margin of error). The minimum required sample size was approximately 118. However, 105 children were included in the study due to recruitment challenges and the application of strict eligibility criteria. Despite this difference, the available sample size was considered adequate for the planned analyses.
Anthropometric Measurements
An Omron bioimpedance scale (HBF-514C, China) was utilized to weigh participants to the nearest 0.1 kg. A Seca 213 stadiometer (14-05-08-237-09G, China) was employed to measure the height of participants to the nearest 0.1 cm. The WHO Anthro Plus software (version 1.0.4) 18 was used to calculate the BMI-for-age z-scores (BAZ) and height-for-age z-scores (HAZ) for each study participant, taking into account their respective weight and height measurements, as well as age and sex.
A mid-upper arm circumference (MUAC) tape (IS Indo Surgicals, 20035, India) and a non-stretchable tape measure were used to assess MUAC and waist and hip circumferences, respectively. A Slim Guide (Creative Health Products, USA) calliper was used to measure skinfold thickness (SFT) following standard procedures. Measurements were taken at triceps, biceps, subscapular, and suprailiac on the right side of the body. This was done with the subject standing in a relaxed position. The anatomical landmark for the specific skinfold site to be measured was located. The skin and subcutaneous fat at the site was pinched with the thumb and index finger, lifting it away from the underlying muscle. The calliper was applied 1 cm below the fingers, perpendicular to the skinfold, and the jaws were released. The measurement was recorded after a few seconds, while ensuring that the calliper remained perpendicular and the skinfold was pinched. Recognizing that measuring SFT in younger children, particularly those around 6 years of age, can be challenging due to smaller limb size, thinner subcutaneous fat layers, and potential difficulty maintaining correct posture or relaxation during measurement, which may introduce variability compared to measurements in older children and adolescents. To minimize this, an experienced nutrition officer performed all the anthropometric measurements, thereby eliminating error due to interobserver variability. Age and gender-adjusted reference tables 19 were used to determine percentiles for SFT among participants. The average of 3 consecutive measurements was recorded as the final measurement for all anthropometric parameters.
Fasting Blood Panel
Before scheduled appointments, caregivers were contacted by a member of the healthcare management team at the Paediatric HIV Clinic via telephone. During this communication, the project was explained, and interested caregivers were asked to instruct their children to maintain an overnight fast in preparation for blood sampling for fasting lipid profile during recruitment. A qualified phlebotomist drew 5 ml of venous blood samples from the arms of study participants and subsequently transferred them into well-labelled gel tubes. Participants were provided with breakfast immediately after the blood was drawn. The samples were transported to the laboratory on ice. At the laboratory, the samples were centrifuged at 3000 rpm for 10 min. The blood sera were then pipetted into appropriately labelled Eppendorf tubes and stored at −4 °C until the day of analysis. The lipid profile test was conducted according to the instructions provided by the reagent manufacturer (Medsource Ozone Biomedicals Pvt. Ltd, India).
Clinical History
The medical history of each participant was obtained from hospital records. The information obtained included the previous and current regimen of participants, treatment start dates, HIV RNA viral load and stage of disease based on the WHO classification. 20 Participants were categorized into pubertal stages according to the Tanner Staging system, with prepubertal children classified as Stage 1.21,22
Dietary Assessment
For each participant, a comprehensive 24-h dietary recall of all foods consumed in the 24 h preceding the data collection day was conducted. Based on this recall, dietary diversity scores were evaluated according to the 10 food groups delineated by the Food and Agriculture Organization (FAO). 23 A score below 5 out of 10 was classified as “inadequate,” whereas a score of 5 and above was classified as “adequate.” 24 Household handy measures and food models were employed to assist caregivers in estimating the portions of food consumed by participants. These portions were then converted to grams and entered into the RIING Nutrient analysis template, developed by the University of Ghana, to ascertain the nutrient composition of the foods. 25 The sufficiency of macronutrient intake was gauged by the Acceptable Macronutrient Distribution Range (AMDR) for children (protein: 10%-30%, carbohydrate: 45%-65%, and fat: 25%-35% of total daily calorie intake). 26 Of the 105 children initially included in the study, nutrient intake estimation was successfully performed for 85 children. The remaining 20 were excluded due to challenges in accurately estimating their dietary intake, primarily because a substantial portion of their meals were consumed outside the home, thereby limiting caregivers’ ability to provide precise information.
Definitions7,27–30
Some key definitions essential to this manuscript are presented below. Lipodystrophy refers to abnormal changes in body fat distribution. Lipohypertrophy is defined as subscapular and suprailiac SFT measurements that exceed the 97th percentile (approximately +2 standard deviations) for the corresponding age and gender, indicating fat accumulation. Lipoatrophy is characterized by bicep and tricep SFT measurements at or below the 3rd percentile for the corresponding age and gender. The mixed subtype refers to the presence of 1 or more signs of both lipoatrophy and lipohypertrophy in the same individual. Dyslipidemia denotes abnormal levels of total cholesterol, triglycerides, HDL, and/or LDL. Hypercholesterolemia is defined as total cholesterol levels equal to or greater than 200 mg/dL. Hypertriglyceridemia is characterized by triglyceride levels equal to or greater than 150 mg/dL. A low HDL level is defined as HDL cholesterol less than 35 mg/dL, while high LDL refers to LDL cholesterol levels greater than 130 mg/dL.
Statistical Analysis
Collected data were entered into a Microsoft Excel pre-designed database. This was cleaned regularly before being exported to SPSS software (version 27.0.1) for analysis. Descriptive statistics, such as frequencies, means, and standard deviations, were used to summarize the characteristics of the study participants. For the analyses of factors associated with lipodystrophy, a multivariable binary logistic regression model was applied, considering lipodystrophy status (present/absent) as the dependent variable. Independent variables included fasting blood lipid levels, anthropometric indices, Tanner stage, clinical stage, and gender. Similarly, factors associated with dyslipidemia were assessed using binary logistic regression with dyslipidemia (present/absent) as the dependent variable. The predictors were dietary diversity score, macronutrient consumption, and exposure to specific antiretroviral medications. The results were reported in odds ratios (ORs) with 95% CIs. Variables with P-values less than .05 were deemed statistically significant. Listwise deletion was applied where variables included in the model had any missing data.
Results
Participant Characteristics
Table 1 presents a summary of the sociodemographic, anthropometric, nutritional, and clinical characteristics of participants in this study. The mean age of participants was 9.7 (± 2.6) years. The mean MUAC was 18.5 (± 2.5) cm. Nutritional indicators showed a mean of BAZ of −0.9 (± 1.0) and HAZ of −0.9(± 1.2). Lipid profile values were generally within normal ranges with mean total cholesterol of 4.3 (± 0.9) mg/dL and triglycerides 0.9 (± 0.4) mg/dL. The participants had been on antiretroviral therapy for an average of 5.3 (± 3.0) years with a mean dietary diversity score of 4.9 (± 1.2).
Participant Characteristics.
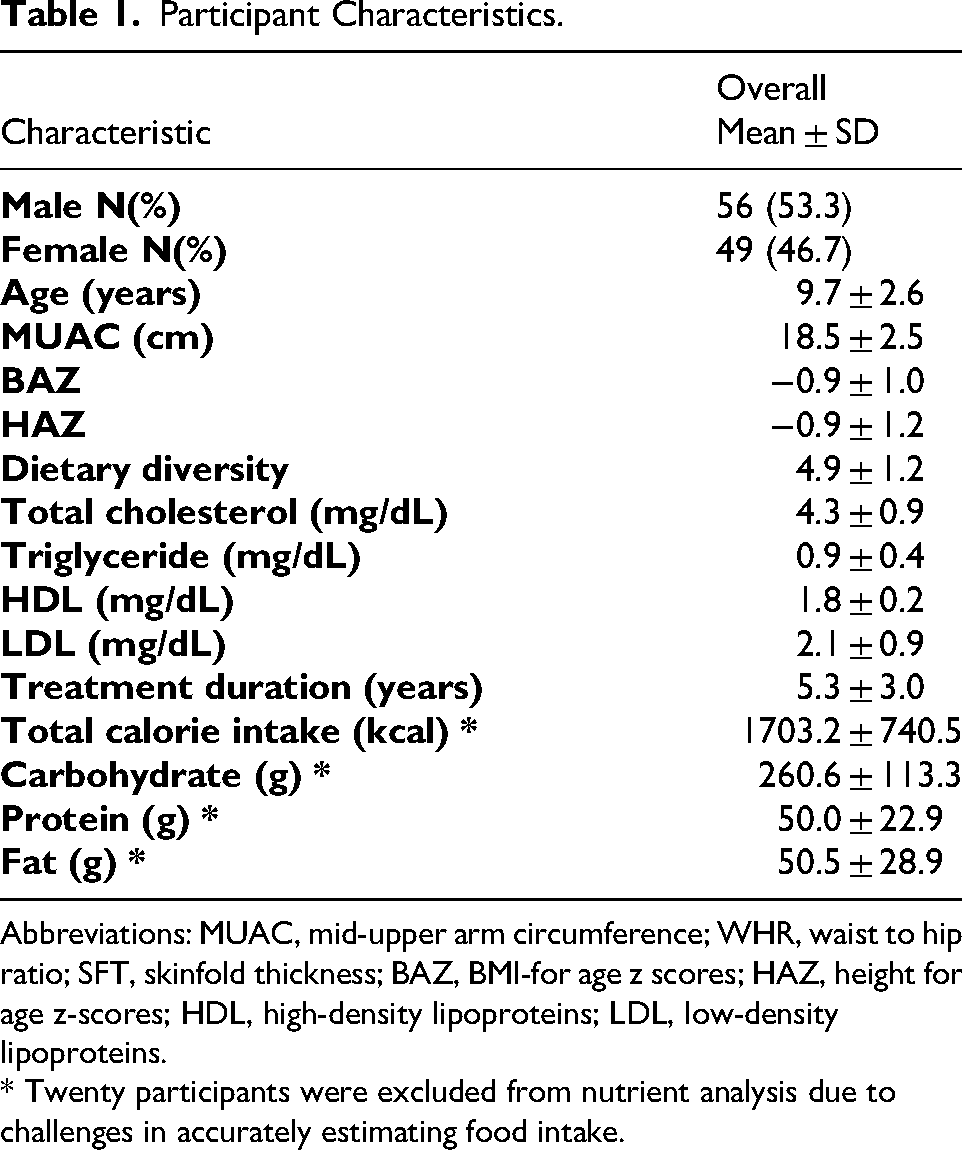
Abbreviations: MUAC, mid-upper arm circumference; WHR, waist to hip ratio; SFT, skinfold thickness; BAZ, BMI-for age z scores; HAZ, height for age z-scores; HDL, high-density lipoproteins; LDL, low-density lipoproteins.
* Twenty participants were excluded from nutrient analysis due to challenges in accurately estimating food intake.
Clinical Characteristics of Participants
Table 2 presents the clinical characteristics of the participants. The majority (47.6%), commenced treatment at WHO Stage 3, had achieved viral suppression at the time of this study (53.3%), and were identified as pre-pubertal (44.8%). The Ghana national treatment protocol changes accounted for 78.8% of regimen modifications to DTG. Previous use of zidovudine (ZDV) among participants was recorded at 70.5%, whereas previous use of efavirenz (EFV) and lopinavir/ritonavir were 24.8% and 29.5%, respectively. The regimens most and least utilized among participants were zidovudine + lamivudine + dolutegravir (41%) and efavirenz + tenofovir + emtricitabine (1%), respectively.
Clinical Characteristics of Participants.

Abbreviations: ZDV, zidovudine; 3TC, lamivudine; DTG: dolutegravir; EFV, efavirenz; TDF, tenofovir; FTC, emtricitabine; ABC, abacavir.
*WHO Clinical stage. 20
Nutritional Status of Participants and Nutrient Intake Among Participants
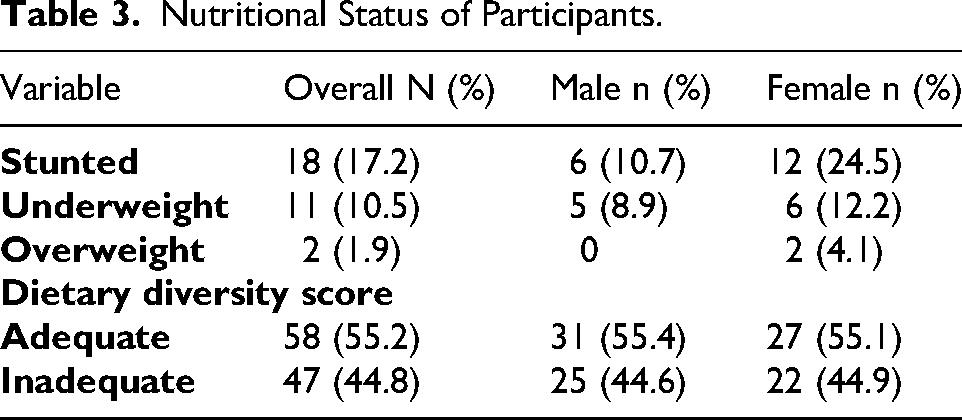
Table 3 illustrates the prevalence of stunting (HAZ ≤ −2 SD), overweight (BAZ ≥ + 2 SD), and underweight (BAZ ≤ −2 SD) among the participants. The corresponding prevalence rates are 17.2%, 1.9%, and 10.5%, respectively. Figure 1 presents the macronutrient intake of participants in accordance with the AMDR. A significant proportion of participants (71.8%) consumed adequate protein, whereas 28.2% did not meet the minimum protein requirements. Additionally, a considerable percentage of participants (44.7%) failed to meet the minimum fat requirement, while 23.5% exceeded this requirement. Estimation of nutrient intake was done for only 85 participants due to challenges in accurately estimating their dietary intake, primarily because a substantial portion of their meals were consumed outside the home, thereby limiting caregivers’ ability to provide precise information.

Adequacy of Macronutrient Intake Among Participants.
Nutritional Status of Participants.
Prevalence of Lipodystrophy and Dyslipidemia Among Participants
Table 4 illustrates the prevalence of lipodystrophy and dyslipidemia, alongside their identified forms within the study population. Lipodystrophy was observed in 12 participants, accounting for 11.4% of the total participants. The most prevalent form of lipodystrophy was lipohypertrophy, which constituted 10.5% of the participants, whereas lipoatrophy represented the least prevalent form, at 0.9% (1 participant). None of the participants in this study exhibited the mixed form of lipodystrophy. Further, dyslipidemia was observed in 27 participants, constituting 25.7% of the total. The prevalence among children was noted at 24%, whereas the rate among adolescents was documented at 27.6%. Elevated total cholesterol levels were reported in 24 participants, representing 22.9%, while the prevalence of hypertriglyceridemia and elevated LDL levels were recorded at 3.8% and 10.5% of participants, respectively. It is noteworthy that none of the participants exhibited low HDL levels.
Prevalence and Forms of Lipodystrophy Among Participants.

Lipoatrophy: bicep and/or tricep skinfold thickness < 3rd percentile; Lipohypertrophy: suprailiac and/or subscapular skinfold thickness > 97th percentile.
Fasting cholesterol ≥ 200 mg/dL.
Triglyceride levels ≥150 mg/dL.
LDL levels > 130 mg/dL.
Factors Associated With Lipodystrophy
Table 5 presents the results of a multivariable binary logistic regression analysis concerning the factors associated with lipodystrophy. This model incorporated variables such as fasting blood lipid levels, anthropometric indices, Tanner stage, clinical stage, and gender. Participants who had entered puberty (as indicated by tanner staging) were approximately 10 times more likely to have lipodystrophy than those who had not (OR = 9.91;95% CI: 1.32-74.23). Higher MUAC was associated with significantly lower odds of lipodystrophy (OR = 0.60; 95% CI: 0.36-0.99). Participants with lipodystrophy had lower odds of elevated triglycerides (OR: 0.1(95% CI: 0.00-0.62).
Factors Associated With Lipodystrophy.

Abbreviations: TAR, trunk-arm ratio; WHR, waist-hip ratio; BMI, body mass index; MUAC, mid-upper arm circumference; LDL, low-density lipoprotein cholesterol; HDL, high-density lipoprotein cholesterol.
The P-values in the table represent statistical significance of the association between each variable and the odds of having lipodystrophy. B: beta coefficient or the regression coefficient (log odds), representing the direction and strength of the association. SE: standard error of B, showing the precision of the estimate. The values in bold p-values that are less than 0.05.
Factors Associated With Dyslipidemia
Table 6 presents the results of a multivariable binary logistic regression analysis concerning factors related to dyslipidemia. The model utilized for the analysis incorporated the dietary diversity score, macronutrient consumption, and exposure to specific antiretroviral medications. The use of lopinavir/ritonavir (0.036) was significantly correlated with a decreased likelihood of dyslipidemia; however, treatment duration, the use of EFV, the use of ZDV, macronutrient intake, and the dietary diversity score exhibited no statistically significant associations with dyslipidemia.
Factors Associated With Dyslipidemia.

Abbreviations: ZDV, zidovudine; EFV, efavirenz; LVP/r, lopinavir/ritonavir.
The P-values reported here represents the significance of association between each dependent variable and the odds of having dyslipidemia while controlling for carbohydrate, fat and protein intake, dietary diversity, zidovudine, efavirenz, and lopinavir/ritonavir exposure and treatment duration. B: beta coefficient or the regression coefficient (log odds), representing the direction and strength of the association. SE: standard error of B, showing the precision of the estimate. The values in bold p-values that are less than 0.05.
Discussion
This study aimed to examine the prevalence of lipodystrophy and dyslipidemia among Ghanaian children receiving HAART, and to identify associated sociodemographic, nutritional, and clinical factors. Our findings revealed that while most anthropometric and nutritional factors fell within expected ranges, a notable proportion of participants were stunted, underweight, had lipodystrophy and dyslipidemia. Females consistently showed significantly higher SFT compared to males in multiple sites (S1). Prior exposure to lopinavir/ritonavir was linked to dyslipidemia. These results highlight the metabolic and nutritional challenges faced by children on long-term ART in this setting and demonstrate the need for continuous monitoring and tailored interventions.
The prevalence of lipodystrophy within this cohort was 11.4%, which is consistent with the lower rates reported in comparable settings. Other studies have identified a wide prevalence range of lipodystrophy among children on HAART, ranging from 8.4% to 60%.30,31 For instance, an investigation conducted in India among 320 children between 2 and 18 years reported a prevalence rate of 60%, whereas a study in Kinshasa reported that 16.3% of the 80 participants on HAART presented with lipoatrophy, and 5% with lipohypertrophy.10,31 In contrast, the current study measured rates of lipohypertrophy at 10.5% and lipoatrophy at 0.9%, which may reflect disparities in diagnostic methodologies. The Kinshasa study utilized clinical signs such as a prominent zygomatic arch and buffalo hump, which, although valuable, are generally more subjective compared to the more objective SFT measurements employed in this study. 32 Although the exact etiology of lipodystrophy remains elusive, specific NRTIs, such as stavudine and, to a lesser extent, ZDV, have been implicated in the development of lipoatrophy, whereas lipohypertrophy has been associated with PIs.7,33 Thus, the HAART regimen of the study population can also affect the prevalence and type of lipodystrophy. The administration of stavudine likely influenced the higher prevalence rates reported in earlier studies; over 70% of participants in the Indian study conducted 30 were treated with stavudine, in contrast to the current cohort, where none were administered this medication as it is no longer part of national treatment protocols.
Dyslipidemia is increasingly being reported in PLWH. Pathogenic mechanisms include the effects of the virus itself, the effects of antiretroviral drugs on key metabolic pathways, and drug-associated adipose repartitioning, leading to the subsequent development of insulin resistance and associated metabolic derangements. 34 Numerous studies have also documented a markedly elevated prevalence of dyslipidemia among children on HAART, with low levels of high-density lipoprotein cholesterol (HDL-c) and hypertriglyceridemia identified as the predominant manifestations. A research undertaken in Indonesia indicated that 54.2% of children undergoing HAART experienced dyslipidemia, with a substantial proportion of these individuals prescribed second-line therapy incorporating PIs. 35 In a research conducted in Ethiopia, 72% of children exhibited reduced HDL-c levels, whereas 21.3% presented with elevated triglycerides. 36 A Tanzanian study reported that 22.7% of participants had abnormally low HDL-c levels, with 7.7% and 12.3% displaying high levels of low-density lipoprotein cholesterol (LDL-c) and triglycerides, respectively. 37 These observations contrast with the findings of the present study, which recorded a prevalence of 25.7% for dyslipidemia; 22.7% for elevated total cholesterol levels, 3.8% and 10.5% for elevated triglyceride and LDL levels respectively. None of the participants had a low HDL. These discrepancies may arise from variations in the metabolic responses of individuals across diverse populations or from the fact that, unlike the aforementioned studies, no participants in this study were currently undergoing any regimen based on PIs. Older PIs, mainly when boosted with ritonavir, are more likely to cause dyslipidemia. 34 The prevalence of dyslipidemia in this research (25.7%), despite being relatively low compared to findings from comparable contexts, remains a significant concern, as atherosclerosis—an essential precursor to cardiovascular disease—commences during childhood and progressively advances into adulthood. 37
A significant proportion of participants (47.6%) commenced treatment at WHO Clinical Stage 3, and only 6.7% of participants were classified in Stage 1. This indicates a tendency towards late presentation, a phenomenon that correlates with observations in low- and middle-income countries (LMICs), where delayed diagnosis frequently occurs due to limited healthcare access and the stigma associated with HIV. The late initiation of treatment heightens the risk of complications related to HIV and diminishes the advantages of early HAART initiation. Some studies have also reported dyslipidemia in ART-naive patients.10,38 An advanced stage of HIV infection and a longer duration of HIV infection, in tandem with a fall in CD4 cell count and a high viral load, are strongly associated with the development of lipodystrophy syndrome.38,39 These factors may play a contributory role in the onset of lipodystrophy in patients who have not previously been exposed to antiretroviral therapy.
Therefore, enhancing early diagnosis and establishing connections to care are imperative for improving health outcomes. Despite showing promise, the proportion of participants achieving viral suppression (53.3%) remains lower than the national and UNAIDS target. 5 Research within LMICs has similarly reported challenges related to viral suppression, attributed to factors such as inadequate adherence, drug resistance, and suboptimal treatment regimens.
All participants in this study, except one, were currently receiving DTG-based first-line HAART, reflecting strong adherence to current WHO and national guidelines, which prioritize DTG for pediatric populations due to its superior efficacy and safety profile. None of the participants was receiving stavudine or any PI at the time of the study. The most common regimen, zidovudine/lamivudine/dolutegravir (ZDV/3TC/DTG) was used by 41.0% of participants while the utilization of TDF-based regimens, such as TDF/3TC/DTG (29.5%) and tenofovir/emtricitabine/efavirenz (TDF/FTC/EFV) (1%), was notably less common, as TDF is generally initiated for children whose weight exceeds 30 kg, in line with WHO recommendations. Although no significant correlation was observed between specific HAART regimens and lipodystrophy in this study, the presence of lipodystrophy highlights the complex metabolic effects of long-term antiretroviral therapy. The percentage of change in therapy due to the alteration of national protocol, which stands at 78.8%, reinforces the consistency of clinical practices with the National AIDS Control Programme (NACP) and WHO guidelines. The adjustments in treatment due to anemia and renal injury, comprising 6.1%, indicate that adverse effects of HAART are indeed present within the study population and are duly monitored. In this study, exposure to ZDV and EFV did not demonstrate a significant association with lipodystrophy and dyslipidemia (P > .05). This finding is aligned with the research conducted in Uganda and Zimbabwe, 8 and Uganda and Zambia, 40 which also reported no increased risk among participants utilizing these medications. Conversely, prior use of lopinavir/ritonavir (LPV/r) was significantly correlated with dyslipidemia (P = .036). However, this seemingly protective relationship (OR = 0.21) contradicts the already established fact that LPV/r exposure predisposes individuals to dyslipidemia.35,40 This may reflect probable confounding by subsequent ART regimens or selection bias (as children with known metabolic conditions were excluded from the study), rather than an actual protective effect of LPV/r. Further, a likely explanation for this paradox is temporal bias as LPV/r is no longer commonly used since the introduction of pediatric formulations of DTG. Participants who previously used LPV/r may have had different clinical trajectories or been transitioned to newer regimens due to side effects or availability. Our results highlight the challenges of studying metabolic complications in HIV, where treatment changes over time can hinder causal inferences.
A majority of participants in this study (55.2%) achieved the minimum dietary diversity score of 5 out of 10, with a mean score of 4.85. This is an improvement when compared to the findings from a study conducted among Ghanaian school children, where the average dietary diversity score was recorded at 3.8 out of 10, with 84.7% failing to meet the minimum diversity score. 24 Notably, CLWH face a 10% to 30% increase in nutritional demands compared to their uninfected counterparts. Despite these increased needs, 44.8% of participants failed to achieve the minimum diversity score, putting them at a heightened risk of micronutrient deficiencies and anaemia. 41 Furthermore, this study demonstrated higher mean intakes of energy (1703.2 vs 1318 kcal), protein (50.0 vs 40.6 g), fat (50.5 vs 31 g), and carbohydrates (260.63 vs 227 g) in comparison to the findings reported by another Ghanaian study. 42 Both studies indicated a high level of carbohydrate adequacy (91.7% in this study and 91% in the other study). However, a greater percentage of children in this study (71.8%) met their protein requirements compared to 15% in that study. 42 The prevalence of excessive nutrient consumption was greater in the present study, with 38.4% of participants exceeding carbohydrate and 23.5% surpassing fat requirements, as opposed to 11.4% and 4.2%, respectively, in a South African study. 43 The lower rates of stunting (17.2%) and underweight (10.5%) observed in this study contrasts with the findings from another Ghanaian study 42 (28% stunting, 16% underweight, and 13% wasting) 42 and a study conducted in Chennai, India (52.4% stunting, 58% underweight, and 17% wasting), 44 suggesting that overall nutritional outcomes are improved in this group, aligning with the reported higher mean intake of energy and macronutrients. Nevertheless, the prevalence of overweight (1.9%) within this study population points to a potential risk of over nutrition. 42 Given that 70.1% of participants were on DTG-based regimen, it is noteworthy that DTG use in children and adolescents with HIV has been associated with some weight gain. Current evidence, however, considers this as healthy weight gain consistent with improved growth and viral control. Despite this, monitoring is still important, particularly during adolescence, to ensure appropriate weight trajectories and overall health outcomes. 45
Dietary diversity and dietary fat did not demonstrate a significant association with lipodystrophy or dyslipidemia, which is in agreement with the findings of Innes et al. 43 Nevertheless, some research indicates that a diversified diet, particularly one akin to the Mediterranean diet, may mitigate cardiovascular risks of patients on HAART. 7
There was no significant association between stunting, BAZ, lipodystrophy, and dyslipidemia. Nevertheless, an Ethiopian study identified that participants with a BMI-for-age percentile of less than 5% were twice as likely to exhibit dyslipidemia. 35 This discrepancy may be attributed to the differences in BAZ between participants with and without dyslipidemia in their study cohort and the lack of such differences within this cohort. Both studies, however, noted no difference in HAZ between participants with and without dyslipidemia. The significant relationship between MUAC and lipodystrophy is consistent with a Tanzanian study, which indicated that MUAC was significantly associated with lipoatrophy (a form of lipodystrophy) in children whose weight-for-height z-scores were less than 1. 28 Specifically, in our study, higher MUAC was significantly associated with lower odds of lipodystrophy, suggesting a protective relationship between nutritional status and the development of lipodystrophy. Given that MUAC reflects peripheral and muscle stores, this association is biologically plausible because undernourished individuals may be susceptible to abnormal fat distribution and metabolic dysfunction. These findings highlight the importance of routine nutritional screening as part of lipodystrophy risk management. Further studies should also be conducted into the possibility of MUAC being used as a screening tool for lipodystrophy, especially in resource-limited settings.
Gender was not significantly associated with either lipodystrophy (P = .325) or dyslipidemia (P = .90), a finding consistent with that of the Ugandan study, 9 but divergent from the Indian study, 30 which reported a significant association between female gender and lipodystrophy. Additionally, another study indicated that males exhibited - slightly higher odds of experiencing lipodystrophy. 49 These conflicting outcomes necessitate further investigation into the relationship between lipodystrophy and gender. Consistent with this study, Lamesa et al 37 also reported no significant association between gender and dyslipidemia.
Hormonal factors during puberty significantly affects body fat distribution in males and females differently. Males tend to gain more fat-free mass and less fat mass whereas females gain more fat mass with increased overall body fat level. 46 Pubertal stage showed significant association with lipodystrophy. This finding is consistent with that of similar studies, which identified notable associations between sexual maturation and lipodystrophy.8,30 Conversely, this outcome is in contradiction to the findings reported by research conducted in India. Both studies similarly found that sexual maturation does not represent a significant factor in the development of lipodystrophy among children living with HIV.47,48,49 This discrepancy necessitates further investigation into the relationship between sexual maturation and lipodystrophy.
The duration of treatment in this study pertains to the length of time participants have been on HAART. Logistic regression analysis did not demonstrate a significant association between treatment duration and lipodystrophy (P-value of .096). However, previous studies, such as those conducted in Uganda and India,9,31 indicated that prolonged exposure to HAART heightens the risk of lipodystrophy in pediatric patients, potentially attributable to the cumulative metabolic effects arising from prolonged HAART use. Additionally, both viral suppression (VL < 50 cp/mL) and WHO clinical stage did not show significant correlation with lipodystrophy and dyslipidemia. This result parallels research conducted in Indonesia, which identified that clinical stage and immunosuppression were not risk factors for lipodystrophy 35 In this study, the presence of lipodystrophy was associated with a significantly lower odds of hypertriglyceridemia suggesting a potential inverse association. This finding contrasts well-documented evidence that lipodystrophy, particularly among HIV-infected adolescents or those on ART is associated with dyslipidemia especially hypertriglyceridemia and low HDL. 36 Several factors may explain this unexpected relationship; a smaller number of lipodystrophy cases as well as a limited statistical power. Further Investigation with a larger cohort may help understand this association.
Strengths of the Study
This study, to the best of our knowledge, is the first study on HAART-related lipodystrophy and dyslipidemia among Ghanaian children living with HIV, filling a critical gap in local and regional research. It focuses on a pediatric population, a group often underrepresented in HIV-related metabolic studies, thereby addressing an important but neglected public health issue. This study also adopted a comprehensive approach, assessing both the prevalence and determinants of metabolic abnormalities by integrating nutritional, clinical, and sociodemographic variables. This multifaceted analysis enhances the robustness and relevance of findings. This study also holds substantial clinical value, as its results can directly inform patient management strategies, guide nutritional counselling, and help traitor HAART regimens to mitigate risks. Moreover, the findings have potential policy implications, supporting the integration of routine metabolic monitoring into pediatric HIV care protocols and national treatment guidelines.
Limitations of Study
Despite its contributions, this study has several limitations. The cross-sectional design restricts causal inference, as it captures data at only one point in time and does not allow for assessment of temporal changes in body composition or metabolic outcomes. This limits causal inferences between HAART regimens and lipodystrophy. The sample size (n = 105) was slightly lower than the calculated requirement (n = 118), this may have reduced the statistical power to detect significant association and moderate effect sizes. Further, the inability to reach data saturation may limit the ability to detect smaller effects and slightly affect generalizability of the findings. The diagnosis of lipodystrophy relied on SFT measurements (biceps, triceps, subscapular, and suprailiac), which, while suitable for low-resource settings, lack the precision of imaging techniques like DEXA scan. Additionally, the dietary assessment was limited to a single 24-h dietary recall, which has a low respondent burden but may not fully reflect participants’ dietary patterns, potentially missing dietary factors that may influence metabolic profile. The use of retrospective data, such as previous regimen and WHO clinical stage at the start of treatment, poses the risk of confounding attrition bias due to missing data. In addition, the absence of a control group of HIV-uninfected children limits our ability to directly attribute the observed body composition patterns to HIV or antiretroviral therapy. Future studies including matched control groups are warranted. This study did not include interviews with individuals living with lipodystrophy, which limited insights into the personal and psychosocial impacts of the condition. Future research should incorporate patient perspectives to better understand quality-of-life and self-esteem challenges. Lastly, another limitation worth noting is the fact that this study involved multiple statistical analyses across 2 primary outcomes and several predictors. Although these analyses were hypothesis-driven and clinically relevant, the number of statistical tests increases the risk of Type 1 error (false positive findings). Formal adjustments for comparisons were not applied; therefore the results should be interpreted with caution.
Conclusion
This study highlights a notable burden of lipodystrophy and dyslipidemia among Ghanaian children in long-term HAART, despite adherence to WHO-recommended regimens. Prior exposure to lopinavir/ritonavir was linked to dyslipidemia while better nutritional status (MUAC) appeared protective against lipodystrophy. These findings align with global concerns about metabolic effects of pediatric ART and highlight the need for routine metabolic monitoring, early diagnosis, integrated nutritional support, and context-specific interventions, particularly in resource-limited settings.
Recommendations
To mitigate metabolic complications in pediatric HIV care, we recommend integrating routine metabolic monitoring into national treatment protocols, including standardized lipid profiling and anthropometric assessment (eg, SFT), particularly for children with prolonged HAART exposure or advanced HIV disease at presentation. Nutritional support should be strengthened through dietary counselling and micronutrient supplementation to ensure better treatment outcomes. These clinical measures must be supported by operational improvements in early diagnosis through expanded family index, along with targeted adherence interventions to address suboptimal viral suppression rates. These recommendations, framed within SDG 3 and UNAIDS 95-95-95 target, provide a roadmap for optimizing the health outcomes of children living with HIV as they transition to adulthood, with particular relevance for resource-limited settings implementing standardized DTG-based regimens. Implementation will require coordinated efforts between HIV programs, nutritional services, and policymakers to ensure sustainable integration into routine care.
Supplemental Material
sj-docx-1-jia-10.1177_23259582251397741 - Supplemental material for The Prevalence and Determinants of Dyslipidemia and Lipodystrophy in Children on Highly-Active Antiretroviral Therapy at a Ghanaian Teaching Hospital: A Cross-Sectional Study
Supplemental material, sj-docx-1-jia-10.1177_23259582251397741 for The Prevalence and Determinants of Dyslipidemia and Lipodystrophy in Children on Highly-Active Antiretroviral Therapy at a Ghanaian Teaching Hospital: A Cross-Sectional Study by Gifty Araba Adadzeba Yawson, Mary Amoako, Charles Martyn-Dickens, Kenneth Ofori-Panyin, Serwah Bonsu Asafo-Agyei, Jessica Ayensu and Anthony Enimil in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Abbreviations
Acknowledgments
The authors are grateful to the staff, patients, and caregivers of the Paediatric HIV Clinic at KATH for their immense support and cooperation during this study.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the guidelines of the Declaration of Helsinki and the COPE. The Institutional Review Board of KATH reviewed all procedures and granted ethical clearance (KATH IRB/AP/053/24, dated 22nd May 2024) for this study, and permission was obtained from the leadership of the Paediatric HIV Clinic. All participants were below 18 years and written informed consent was obtained from their parents/legal guardians. Consent was obtained to collect data from subjects including anthropometric data and blood sample collection to assess lipid profile.
Author Contributions
GAAY, MA, and CMD conceptualized and designed the study, participated in analysis and interpretation of the data, and drafted the initial manuscript. GAAY, MA, CMD, JA, KO-P, SBA, and AE contributed to the methodology, and reviewed the manuscript drafts. All authors reviewed, read, and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the KNUST Grasag Research Fund (GRef).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The data sets used and /or analyzed during this current study are available from the corresponding author on reasonable request.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
