Abstract
Background
HIV self-tests (HIVSTs) have been promoted as one way to increase testing.
Methods
We extracted data from the GetaKit study for October 11, 2023 to June 30, 2025, focusing on participants to whom we co-offered an HIVST and serology.
Results
We co-offered HIVST and serology to 3611 persons; 71.9% agreed to go to a lab and 19.4% opted for only the HIVST. Participants who were Black, Indigenous, or Persons of Color were less willing to attend a lab; participants who were men who have sex with men or reported injection drug use or sex work were more willing to attend a lab. First-time testers opted for the HIVST at a higher rate. HIVST did not yield new diagnoses.
Conclusions
HIVSTs were an entry point to testing for some but were not the preferred modality for most. Promoting HIVSTs too broadly would not align with patient preference.
Plain Language Title
Comparing the uptake of HIV self-testing to HIV serology in the GetaKit study
Plain Language Summary
HIV testing is the first step in getting treatment and prevention. HIV self-tests may help people do testing. We wanted to see people's real-world uptake of HIV self-tests compared to serology when both were made equally available to them. We found that people who were Black, Indigenous, or Persons of Color were less willing to attend a lab for testing, but that gay, bi, and other men who have sex with men were more willing to attend a lab. People who reported sex work and injection drug use were also more willing to attend a lab for testing. First-time testers were more likely to opt for the self-test. No one tested positive through the self-test, suggesting that the self-test might be better suited for targeted use to identify people who could use prevention strategies.
Keywords
Introduction
In recent years, HIV prevention has advanced dramatically. Antiretroviral medications can eliminate sexual transmission when an HIV-positive person has an undetectable viral load (ie, undetectable equals untransmittable, or U = U). 1 Antiretroviral medications can also be given to HIV-negative persons as pre-exposure prophylaxis (PrEP), with preventive effects exceeding 99%. 2 The first step toward U = U and PrEP, however, is testing. 3 Persons must believe they are at risk for HIV and then must complete screening. Individuals must also feel safe accessing testing and be comfortable with follow-up. Research 4 shows that among the populations most affected by HIV, these factors can impede testing—and, by extension, undermine the aims of U = U and PrEP.
Other research5,6 suggests that HIV self-tests (HIVSTs) may overcome some barriers to testing, leading some authors to have presented HIVSTs as the key to “reaching the undiagnosed.” 7 This excitement about HIVSTs has resulted in mass deployments of these devices in many settings and studies, 8 and data from these observational projects have often shown good uptake among first-time testers and members of communities with higher rates of HIV.9,10 Positivity rates from HIVST projects have also usually matched or exceeded those from serologic testing in clinics.11,12
In contrast, a systematic review and meta-analysis showed increased rates of testing with HIVSTs, but no changes in diagnosis rates. 13 That is, more testing was done but without increases in case finding. Similarly, while our prior publications14,15 on HIVSTs in Ontario, Canada initially showed higher positivity rates for HIVSTs compared to laboratory testing, these differences began to dissipate when clinical services re-opened after the COVID pandemic. Our recent analyses 11 also identified that HIVSTs only accounted for a small fraction of HIV testing being performed in our region. Other work we have completed, as a discrete choice experiment 16 and the provision of COVID self-tests, 17 furthermore identified that, while a subset of participants reported a preference for self-tests, a different and sizeable group preferred standard healthcare services, including the certainty of results provided by laboratory-based testing. Stated differently, this latter group had a preference against self-tests (all types). Some data thus support HIVSTs, while other findings suggest they have utility in facilitating screening for some persons, but with caveats.
Missing from current understandings about if and how HIVSTs may promote U = U and PrEP uptake are data about the use of HIVSTs versus serology in the real-world. That is, what occurs when persons are given information about HIVSTs and HIV serology and are then provided equal access to both tests? Such information is important because there can be discrepancies between what people say they would do (when given a choice in a survey) and what they do (when given the choice to make in real life). Findings from studies involving only HIVSTs could also inadvertently inflate the perceived preference for HIVSTs, confusing availability for choice. It may not be that participants in these studies and projects had a true preference for the HIVST; it could simply be that they wanted HIV testing and took the only option offered to them. To help inform future HIVST implementation, real-world data from head-to-head comparisons of the uptake of HIVSTs and serology are required to understand testing preferences when patients are given the option for both modalities.
To generate some such information, we extracted data from GetaKit.ca, which is a research website that allows persons in Ontario, Canada to obtain HIVSTs and laboratory-based testing for sexually transmitted infections (STIs), including HIV, syphilis, hepatitis C, gonorrhea, and chlamydia. Through GetaKit, we offer both an HIVST and HIV serology to persons who (1) have risk factors for HIV acquisition, including being sexually active, engaging in sex work, or using or sharing drug equipment and (2) belong to the groups with higher rates of HIV in Ontario, 18 including gay, bi, and other men who have sex with men (gbMSM), members of Indigenous communities, persons of African, Caribbean, and Black (Black) ethnicities, and persons who use injection drugs. Herein, we report on the data from participants to whom we co-offered an HIVST and HIV serology, focusing on how many consented to any form of testing, how many of those who consented opted for the HIVST and/or serology, HIV testing completion and reporting rates, and test results. These data add to our prior publications on GetaKit, which detailed operations of the website, 19 uptake among populations,20–22 diagnostic outcomes, 11 and overall uptake and use. 15
Methods
At the time of publication, GetaKit was an ongoing prospective open cohort study in Ontario, Canada—a province of ∼16 million people. GetaKit launched as a pilot for HIVSTs in Ottawa in July 2020 and expanded province-wide in April 2021. HIV serology became available on October 11, 2023. For persons who were eligible and opted for serology, requisitions were provided that expired 8 weeks after the date the participant requested testing. Participants could obtain testing from GetaKit at the frequencies recommended in Ontario guidelines. 18 Anyone with expired requisitions could re-order at their convenience to obtain clinically indicated testing.
To receive testing from GetaKit, persons had to live in Ontario, be ≥17 years old for laboratory-based testing, and ≥18 years old for HIVSTs. To enroll, persons had to create an account on GetaKit.ca, complete a risk assessment, review pretest information, which included details about test performance (Box 1), opt into testing, and complete testing. Reporting of HIVST results was not required and reporting options included positive, negative, invalid, and prefer not to report. Reported HIVST results and laboratory results triggered tailored messaging (Box 2).
Pre-Test Information for HIVST and Serology.

Information Provided for Test Results.

HIV Testing Modalities
Laboratory testing was completed when participants visited a local specimen collection center (henceforth, “lab”), where they had blood collected. The lab then sent this blood sample to the Public Health Ontario Laboratory for analysis using the 4th generation Abbott Architect® HIV test, with a turnaround time of 2-6 business days. The window period for serology was 6 weeks. Self-tests were mailed out for participants to perform when and where they wanted, with results available in less than 5 min. We used was the Biolytical INSTI® HIVST, a qualitative flow-through fingerstick blood test with an estimated sensitivity of 99.9% and specificity of 99.5%. 23 This HIVST is equivalent to a third generation antibody-only serologic test for window periods (12 weeks). This test is not diagnostic and requires confirmatory serologic testing to rule out false positive results.
Data Collection
All data were collected through the study website. Persons completed a registration form (containing their name, date of birth, and contact information), filled in a demographic form (about ethnicity, sex/gender, sexual orientation), and answered a risk assessment survey (about sex and drug use practices, prior testing and diagnoses). Risk assessment questions were the same for every participant regardless of gender, ethnicity, or risk factors and were completed with each testing request. As part of this assessment, participants also reported if they would go to a lab for testing. Those who indicated they would not go to a lab were not offered lab-based testing. See Supplemental file 1 for a complete list of questions in the risk assessment survey.
Statistical Analysis
We extracted data from GetaKit.ca for October 11, 2023 to June 30, 2025 (ie, date of launching HIV serology until data extraction). Our unit of analysis was orders, not unique participants, meaning that one participant may have appeared more than once in the dataset. Once extracted, we restricted the dataset to orders when participants were offered both an HIVST and serology (ie, ≥ 18 years old and a member of the HIV risk groups with HIV acquisition risk factors). We removed all orders when only one form of HIV testing was offered (eg, only serology or only HIVST) or when a participant declined all forms of HIV testing.
We completed descriptive data analysis (counts, frequencies) and performed chi-square testing to determine if there were differences in the characteristics of participants for orders when persons opted for HIVST but declined serology. Specifically, we reviewed willingness to attend a lab and preference for HIVST versus serology (with or without the HIVST) and did so by ethnicity and reported risk practices (ie, gbMSM, sex work, injection drug use), and prior HIV testing history. For ethnicity, based on the literature which highlights experiences of stigma by those who are Black, Indigenous, or Persons of Color (BIPOC), 24 we compared persons who were BIPOC to white. We selected an alpha of 0.05 to determine statistical significance. We calculated 95% confidence intervals using Wilson score interval with continuity correction.
We lastly compared results for participants who opted for both the HIVST and serology, looking at test results and completion rates. Completion was defined as a reported result for the HIVST and the receipt of a lab result for serology. For missing test results in the database, HIVSTs were classified as “not reported” and incomplete HIV serology were classified as “not received.” We also reported on positive test results for other STIs among participants who went to the lab.
Results
For our analytic period (October 11, 2023 to June 30, 2025), participants sought testing via GetaKit a total of 14 132 times, with 97.0% (n = 13 702/14 132) of these requests being eligible. From eligible orders, 79.6% (n = 10 912/13 702) included HIV testing and 33.1% (n = 3611/10 912) involved an order for both an HIVST and serology. Of these 3611 orders, the majority were from participants who were white (46.2%, n = 1669), cis male (67.6%, n = 2441), and gbMSM (49.2%, n = 1775). One-third (33.0%) reported this was their first time completing testing (see Table 1). Notably, 81.0% (n = 2924/3611) of orders were from unique participants.
Characteristics of Participants who Were Eligible for a Self-Test & Serology (n = 3611).

Abbreviations: gbMSM = gay, bisexual, and other men who have sex with men.
Regarding testing preferences, in orders when participants were eligible for both the HIVST and serology and some form of HIV testing was completed (n = 3611), 28.1% (95%CI [26.6%, 29.6%], n = 1014/3611) included an indication that the participant would not go to a lab. Among the 71.9% (95%CI [70.4%, 73.3%], n = 2597/3611) of orders when participants reported a willingness to go to a lab, 19.4% (95%CI [18.0%, 21.0%], n = 505/2597) included a selection of the HIVST but no serology, 15.1% (95%CI [13.8%, 16.6%], n = 393/2597) opted for serology but declined the HIVST, and 65.4% (95%CI [63.6%, 67.3%], n = 1699/2597) opted for both tests. In orders when participants were willing to go to the lab and they selected the HIVST but declined HIV serology, in 59.6% (95%CI [55.2%, 63.9%], n = 301/505) of these orders, participants declined all serology (but completed other lab testing), and in 40.4% (95%CI [36.1%, 44.8%], n = 204/505) of these orders, participants completed serology for syphilis and/or hepatitis C but not HIV (see Figure 1).

Participant Willingness to Go to a Lab and Use an HIVST or Serology.
Figure 1 shows that, in orders when participants agreed to lab testing, 19.4% (95%CI [18.0%, 21.0%], n = 505/2597) were for the HIVST only, whereas 80.6% (95%CI [79.0%, 82.1%], n = [393 + 1699]/2597) included HIV serology. Coupled with the group of orders when participants responded that they would not go to a lab (n = 1014), a total of 42.1% (95%CI [40.5%, 43.7%], n = 1519/3611) of our orders which involved participants who were eligible for both an HIVST and serology exclusively ordered the self-test, whether due to an outright selection preference or due to a preference to not visit a lab.
We compared these groups to determine if there were significant differences, starting with orders when participants would versus would not attend a lab. We found less willingness to visit a lab in orders from participants who identified as BIPOC (67.2%, 95%CI: 65.0%, 69.3%), compared to white (78.3%, 95%CI: 76.2%, 80.3%) (x2 = 54.74, P < .001). This difference persisted when we compared the reported rates of willingness to attend a lab among all ethnicity groups to the rate reported by white participants (78.3%, 95%CI: 76.2%, 80.3% willingness). For this analysis, the lowest willingness to attend a lab was in participants who identified as South Asian (willingness = 49.6%, 95%CI: 43.4%, 55.8%) (x2 = 97.92, P < .001) followed by (in ascending order) South / East Asian (willingness = 58.5%, 95%CI: 52.9%, 63.9%) (x2 = 56.71, P < .001), Arab (willingness = 66.1%, 95%CI: 60.0%, 71.6%) (x2 = 19.55, P < .001), and Black (willingness = 74.3%, 95%CI: 71.0%, 77.4%) (x2 = 4.60, P = .03). In contrast, we found greater willingness to visit a lab in participants who reported injection drug use (89.9%, 95%CI: 86.2%, 92.7%), compared to those who denied this practice (69.8%, 95%CI: 68.2%, 71.4%) (x2 = 65.47, P < .001). There was also more willingness to attend a lab by participants who reported sex work (81.3%, 95%CI: 76.4%, 85.5%), compared to those who denied sex work (71.0%, 95%CI: 69.4%, 72.5%) (x2 = 14.51, P < .001). Not being willing to attend a lab was thus more common in BIPOC participants, but willingness was higher among those who reported injection drug use and sex work.
Next, among orders when participants reported being willing to attend a lab, we sought to determine if there were differences between those who opted for the HIVST only, compared to those who opted for serology with or without the HIVST. For these groups, we found higher rates of HIVST-only use among participants who were first-time testers (24.9%, 95%CI: 21.8%, 28.3%), compared to repeat testers (17.3%, 95%CI: 15.7%, 19.2%) (x2 = 18.76, P < .001). We also found lower rates of HIVST-only use in orders from participants who reported injection drug use (14.6%, 95%CI: 11.1%, 19.0%), compared to those who denied this practice (20.2%, 95%CI: 18.6%, 21.9%) (x2 = 5.70, P = .017). We also identified lower rates of HIVST-only uptake among orders from participants who reported sex work (12.7%, 95%CI: 8.9%, 17.7%), compared to those who denied sex work (20.2%, 95%CI: 18.6%, 21.9%) (x2 = 7.84, P = .005). Otherwise, we did not find any differences regarding a preference for HIVST-only versus serology by ethnicity (white at 19.0% [95%CI: 16.9%, 21.2%], compared to BIPOC at 19.8% [95%CI: 17.6%, 22.1%], x2 = 0.26, P = .612), sexual orientation (heterosexual at 20.2% [95%CI: 17.8%, 22.9%], vs gbMSM at 19.5% [95%CI: 17.5%, 21.8%], x2 = 0.17, P = .681), or sex (male at 20.0% [95%CI: 18.2%, 21.9%], vs female at 18.4% [95%CI: 15.7%, 21.6%], x2 = 0.75, P = .385).
For test results, for the HIVST, there were 2204 tests ordered, with results reported for 53.8% (95%CI [51.7%, 55.9%], n = 1185/2204). For these 1185 reported results, 0.08% (95%CI [0.004%, 0.54%], n = 1/1185) was positive, 63.6% (95%CI [60.8%, 66.4%], n = 754/1185) were negative, and 33.6% (95%CI [30.9%, 36.4%], n = 398/1185) were invalid. For serology, 51.5% (95%CI [49.1%, 54.0%], n = 821/1593) of the testing requisitions that were ordered had results, for which 0.12% (95%CI [0.006%, 0.79%], n = 1/821) was positive, 0.1% (95%CI [0.006%, 0.79%], n = 1/821) was a false positive, 99.4% (95%CI [98.5%, 99.8%], n = 816/821) were negative, and 0.4% (95%CI [0.09%, 1.16%], n = 3/821) were cancelled by the lab. Serology confirmed the positive HIVST as false positive and one invalid HIVST result as positive. Participants completed the HIVST for 29.5% (95%CI [26.2%, 33.0%], n = 213/722) of the serology specimens that were not completed. For these HIVSTs results, 39.0% (95%CI [32.5%, 45.9%], n = 83/213) were invalid, 57.3% (95%CI [50.3%, 64.0%], n = 122/213) were negative, and 3.8% (95%CI [1.8%, 7.5%], n = 8/213) were “prefer not to report.” For the HIVSTs that did not have reported results, 8.9% (95%CI [7.4%, 10.8%], n = 106/1185) had serology results, all of which were negative. Taken together, we obtained test results for 61.4% (95%CI [59.5%, 63.3%], n = 1595/2597) of our participants who were eligible for both an HIVST and serology (see Table 2 for completion rates by ethnicity, gender, and risk factors).
Comparison of Participants who Completed Versus did not Complete any HIV Testing.
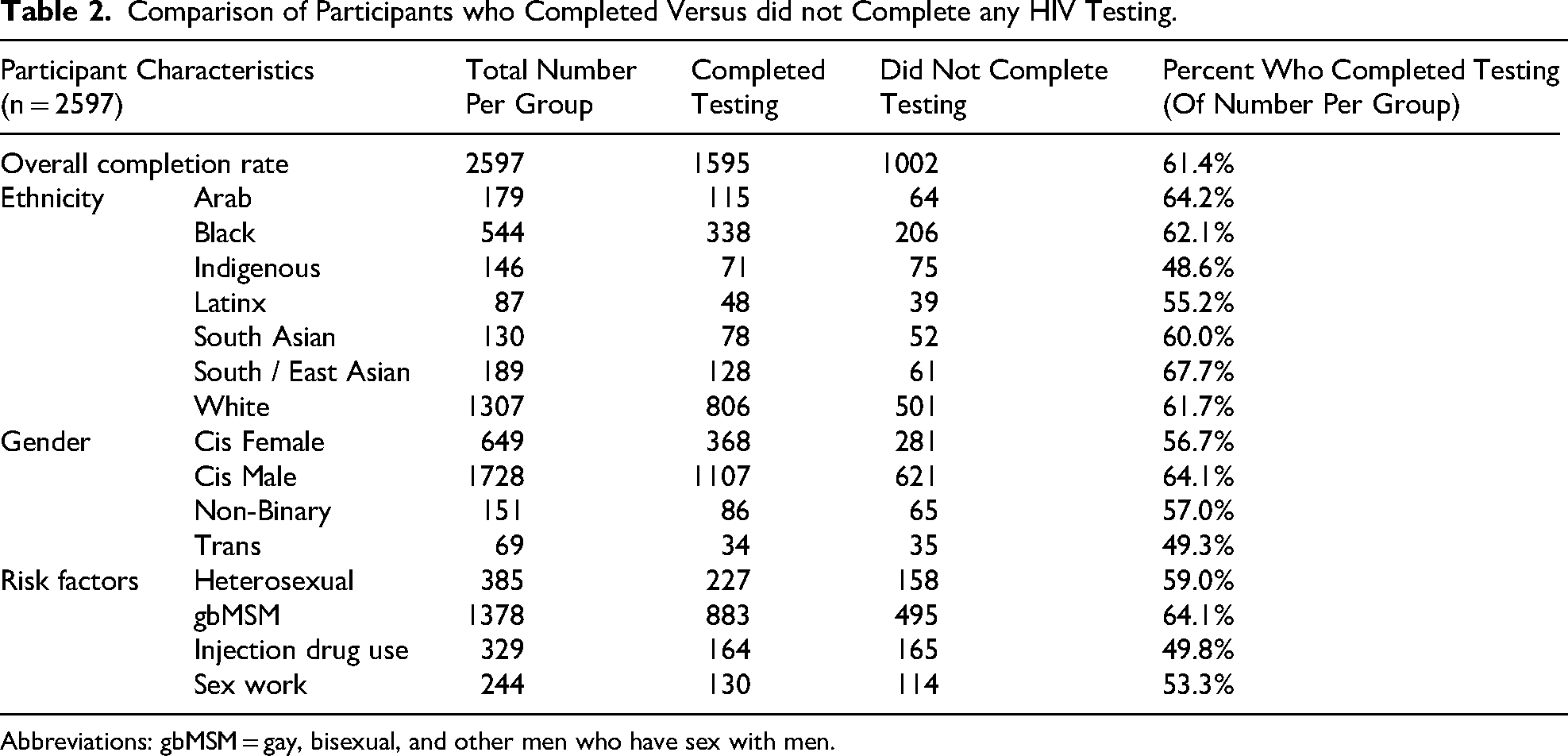
Abbreviations: gbMSM = gay, bisexual, and other men who have sex with men.
Lastly, among those who agreed to go to the lab (n = 2597), irrespective of their choice for HIV testing, 92.0% (95%CI [90.9%, 93.0%], n = 2389/2597) completed other testing (eg, swabs or urine for gonorrhea, chlamydia, or serology for syphilis or hepatitis C). A total of 4.3% (95%CI [3.5%, 5.2%], n = 103) of these 2389 testing episodes identified a positive STI test result.
Discussion
In this paper, we reported on outcomes from the GetaKit study in Ontario, Canada, focusing on orders from participants to whom we co-offered an HIVST and serology and (1) willingness to visit a lab, (2) uptake of HIVST versus serology, and (3) test completion and results. From October 11, 2023 to June 30, 2025, we co-offered HIVST and serology in 3611 orders, of which 28% involved a participant reporting an unwillingness to attend a lab for testing. Among orders from those who would visit a lab, nearly one-fifth involved an order for the HIVST only. Among orders when HIV serology was declined, three-fifths declined serology for syphilis and hepatitis C as well (but completed other lab testing), suggesting the barrier was serology and not HIV testing per se. Moreover, orders from participants who were BIPOC had lower rates of reported willingness to visit a lab, while orders from participants who reported injection drug use or sex work had higher rates of willingness to visit a lab. Among orders from those willing to go to the lab, there were higher rates of HIVST-only uptake among first-time testers, but lower rates among gbMSM and persons who reported injection drug use or sex work. Results reporting was just over half for the HIVST and about the same for serology completion at a lab. Combined, the HIVST and serology yielded test completion for roughly two-thirds of orders when we co-offered these tests. These results raise a few points for discussion.
First, it appears that HIVSTs might be a good option for a niche group of persons who have objective risk factors for HIV acquisition but, due to personal or systemic barriers,4,24 may not obtain testing otherwise. One such group in our study was first-time testers, who opted for HIVSTs at higher rates than repeat testers. Based on our findings, the HIVST was a “hook” to link these individuals to sexual health services, including HIV/STI screening and HIV prevention (when results were negative), and perhaps connected people to testing in a stepwise way that supported patient autonomy as they progressively engaged with care. This stepwise engagement is reinforced by findings from our earlier work implementing a urine drop-off box for chlamydia and gonorrhea screening in gay bathhouses in Ottawa, Canada. 25 In this earlier project, not one participant tested positive for either chlamydia and gonorrhea, but we did find an 8% incidence for infectious syphilis diagnoses among first-time testers in this project who subsequently accessed clinical services for additional screening. Offering discrete testing options, such as the HIVST, may be one way to facilitate engagement in HIV testing and other sexual health services (including prevention or care) for first-time testers with specific risk factors for HIV acquisition.
Second, the use of HIVSTs as an engagement strategy does not, and should not, apply to all persons seeking HIV testing. While certain groups in our study were less willing to attend a lab (and thus more likely to use the HIVST), nearly three-fifths of participants still opted to complete HIV serology, with or without an HIVST. Similarly, for BIPOC participants, while we observed less willingness to attend a lab, over two-thirds (67.2%) overall still opted into and completed lab-based testing for HIV. This reduced willingness, while statistically significant—just crossed the threshold of significance—with Black participants indicating the highest willingness to attend a lab (at 74.3% willingness) among all BIPOC participants. Considering the reported barriers that exist for members of Black communities in accessing HIV testing,4,24 the fact that three-quarters were willing to attend a lab suggests that HIVSTs may not be the only solution to promote screening. Such willingness to attend a lab was also high among participants who were gbMSM or who reported sex work or injection drug use—with these being three groups disproportionately affected by HIV in our jurisdiction. 18 Lastly, among those who were willing to attend a lab for testing and opted for the HIVST only, three-fifths opted out of all serology, suggesting that many participants who declined HIV serology may have had some underlying aversion to serologic testing, rather than a preference for HIVSTs.
One potential explanation for our findings is that, at its core, what our participants wanted was access to sexual health services. In other words, most participants wanted HIV testing, without any preference regarding modality. Our findings suggest that results from studies involving HIVSTs only (where serology was unavailable) should be interpreted with caution. Did participants in those studies actually prefer HIVSTs, or did they simply accept these devices because that was all they could obtain? Without a direct comparison between HIVST and serology uptake, it is hard to interpret findings from such research. While our previous publications which reported on data from when we only offered HIVST did show good uptake for these devices, much of this usage diverted to serology—at participants’ request—once we began co-offering this modality. It thus appears that our participants wanted access to gold-standard HIV testing, not to HIVSTs. Another possibility is that the information we provided on the performance of the HIVST and serology (Box 1) influenced decision-making regarding the type of testing selected. That is, pre-test information may have affected participant conduct. Even if this effect was small, this is important to consider because it highlights the ethical requirement of giving information about the sensitivity, specificity, and invalid rates, and diagnostic and public health reporting requirements of different types of tests. Researchers and ethics boards should consider this point moving forward.
A third point for discussion is that by co-offering HIVSTs and serology, we increased testing but without corresponding increases in diagnoses. While these findings differ from our earlier findings14,15—which showed decent HIV diagnosis rates—our previous findings emerged during the COVID pandemic when HIV testing clinics were restricted or closed and when serology was not yet available through GetaKit. Since we began offering serology as part of GetaKit, HIVST uptake has decreased and our only diagnoses have arisen from serology, whereas the HIVST only yielded false positive results (as confirmed by serology). In fact, in the case of one participant with a positive serology result, linkage to care efforts were unintentionally delayed due to an initial invalid result on the HIVST. 1
This point about diagnosis rates furthermore aligns with a randomized controlled trial from the UK 26 and a meta-analysis, 13 which similarly found that HIVSTs led to a larger volume of testing but no additional HIV diagnoses. From a resource perspective, findings from our work, the UK study, 26 and the meta-analysis 13 highlight that focused distribution of HIVSTs might be the best strategy for these devices; eg, to first-time testers or those who outright will not go to the lab and have objective risk factors for HIV acquisition. In such instances, testing is still provided to members of affected groups and diagnoses will occur, as we have observed before. This approach, as noted, may also be a way to link these individuals to other testing and prevention services.
Having a low positivity rate, as we observed in this study, however, is not inherently an issue. While the goal of testing is usually case finding, there is a secondary goal for HIV testing: to identify people who are HIV-negative so they can be offered preventive services, such as PrEP. 2 While much focus on HIVSTs to date has been on how they can help achieve the (UNAIDS) 95-95-95 targets, 27 perhaps the best use of these devices—at least within higher income regions—is to rapidly identify those who are good candidates for PrEP. The so-called goal of “reaching the undiagnosed” might not be the best use of these devices. In fact, this strategy may be misguided and might be an outright waste of resources, especially considering the possibility of invalid test results (which occurred at a rate of 33.6%-39.0% in this analysis, and for the 1 true new HIV diagnosis that we identified from the study sample). In these instances, people remained untested for HIV and unable to be linked to prevention or care.
Our data consequently suggest a key point to consider when designing HIVST interventions: use HIVSTs as a “hook” to care. Gone are the days of repeatedly testing people until they do not need screening any further because either they no longer have risk factors for HIV acquisition, or they test positive and need treatment. The approach now is to ensure direct PrEP linkages 3 at the outset of testing and upon receipt of results, and each time a person requests testing again. These pathways need to be developed for both those who are willing and who are unwilling or unable to attend a lab. Information about risk also needs to be communicated to members of these two groups. For example, among those who attend the lab, additional STI testing was completed during 92% of orders. In our procedures, information about HIV risk was communicated to everyone who tested HIV-negative (whether by serology or HIVST), but very specific information was given to those who tested positive for rectal gonorrhea and/or chlamydia or infectious syphilis (see Box 2). This latter point is important because these infections correlate strongly with HIV acquisition 28 —and it is essential that study participants and patients know this information. In our study, HIVSTs did not “reach the undiagnosed,” but these devices were a good entry point for linking people into sexual healthcare services, including test information and direct linkages for PrEP, in a way that felt most accessible to them. In other words, the HIVST was a “hook” for subsequent engagement in care for a subset of participants. That nearly 5% of those went to a lab were diagnosed with a different STI demonstrates that risk was present in our cohort; now prevention is needed. We feel this is the best way forward with these devices.
Limitations
Our results must be interpreted considering certain limitations. First, HIVST ‘completion’ was based on participant reporting. Likely, more people did the test than reported results and there may have been positive results among those who did not report. Second, our system was web-based, meaning that our participants were comfortable and able to use technology. Our results might not apply to persons who cannot or will not use an online system. Third, our study occurred in only one jurisdiction, which had free access to testing, but also historically high rates of criminalization for BIPOC persons. This sociopolitical context likely affected the results we observed. Fourth, the rationales for why we observed our results were speculative. Interviews are required to help us understand the patient/participant perspective. Fourth, we used a blood-based HIVST, which was the only licensed device in Canada at the time this study occurred. Preferences for, and uptake of HIV testing between HIVST and serology may have differed had saliva-based HIVST been available or offered to our participants. Taken as a whole, these limitations may affect the generalizability or transferability of our results to other jurisdictions. New studies need to evaluate uptake in different jurisdictions and using different modalities for obtaining testing.
Conclusion
In this paper, we reported on findings from GetaKit about the uptake of HIVSTs and serology among participants to whom we co-offered these tests. We found that some participants were unwilling to attend a lab, and that this was more pronounced among BIPOC participants. Conversely, there was a greater willingness to attend a lab among participants who were gbMSM or who reported sex work or injection drug use. Among those who were willing to attend a lab, most opted for serology, either with or without an HIVST. The outcome of co-offering an HIVST and serology was increased HIV testing, but without additional diagnoses from the HIVST. We interpreted these findings to mean two things. First, projects which offer HIVSTs should also offer serology, as there was a strong preference for it overall in our study. While the end goal would be that people should be offered serology whenever they are given access to an HIVST, a starting point would be to at least ensure people have full information regarding their testing approach, including details on the performance of the HIVST compared to serology, and how and where to obtain serology. Many barriers impede HIV testing, and these are compounded for many members of BIPOC communities. We must not efface our responsibilities by simply offering HIVSTs instead of fixing fundamental flaws in healthcare. 29 Second, a key function of HIVSTs seems to be a first step in engaging persons with risk factors for HIV acquisition, but who otherwise may not be ready or able to complete lab-based testing. In these instances, HIVSTs aided in identifying persons who tested negative and would therefore benefit from PrEP. With advancements in prevention (PrEP and U = U), we think such a re-orientation of the role of HIVSTs may best position it within our HIV prevention armamentarium. From our findings, to promote these devices as the ideal strategy for diagnosis for all populations is misguided and would waste precious resources. A secondary role has emerged for them though, and it is time that we lean into this success.
Supplemental Material
sj-docx-1-jia-10.1177_23259582251393357 - Supplemental material for Comparing the Uptake of HIV Self-Testing to HIV Serology: Findings from GetaKit—A Prospective Open Cohort Study in Ontario, Canada
Supplemental material, sj-docx-1-jia-10.1177_23259582251393357 for Comparing the Uptake of HIV Self-Testing to HIV Serology: Findings from GetaKit—A Prospective Open Cohort Study in Ontario, Canada by Patrick O’Byrne, Lauren Orser, Alexandra Musten and Jennifer Lindsay in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582251393357 - Supplemental material for Comparing the Uptake of HIV Self-Testing to HIV Serology: Findings from GetaKit—A Prospective Open Cohort Study in Ontario, Canada
Supplemental material, sj-docx-2-jia-10.1177_23259582251393357 for Comparing the Uptake of HIV Self-Testing to HIV Serology: Findings from GetaKit—A Prospective Open Cohort Study in Ontario, Canada by Patrick O’Byrne, Lauren Orser, Alexandra Musten and Jennifer Lindsay in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgment
The team would like to acknowledge the participating health units in the GetaKit for their contributions to this work. Patrick O’Byrne would like to thank the OHTN for his chair in HIV Prevention and Public Health.
Ethical Considerations
The Research Ethics Board at the University of Ottawa approved this study (H-12-20-6450). All participants provided digitally signed informed consent to participate in this study. At each order, participants had to opt in and consent to testing.
Informed Consent
Upon registration, all participants provided digitally signed informed consent to participate in the study. At each order, participants had to opt in and consent to testing. This study did not involve minors (participants had to be at least 18 years of age to register), so consent from a legally authorized representative was not applicable.
Author Contributions
POB: design, study implementation and operations, data analysis, writing, editing, submission.
LO: design, study implementation and operations, data analysis, writing, editing.
AM: study implementation and operations, review, editing.
JL: study implementation and operations, review, editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was obtained from the Ontario HIV Treatment Network (OHTN), Public Health Ontario (PHO), the Ontario Government Ministry of Health, and the Public Health Agency of Canada (PHAC). Funders were not involved in the study or in this publication. The reporting of this study conforms to the STROBE statement 30 [Supplemental file 2].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
