Abstract
Background
The burden of advanced HIV disease remains a significant concern in sub-Saharan Africa. In 2015, the World Health Organization released recommendations to treat all people living with HIV (PLHIV) regardless of CD4 (“treat all”) and in 2017 guidelines for managing advanced HIV disease. We assessed changes over time in the proportion of PLHIV with advanced HIV and their care cascade in two community settings in sub-Saharan Africa.
Methods
Cross-sectional population-based surveys were conducted in Ndhiwa (Kenya) in 2012 and 2018 and in Eshowe (South Africa) in 2013 and 2018. We recruited individuals aged 15-59 years. Consenting participants were interviewed and tested for HIV at home. All participants with HIV had CD4 count measured. Advanced HIV was defined as CD4 < 200 cells/µL.
Results
Overall, 6076 and 6001 individuals were included in 2012 and 2018 (Ndhiwa) and 5646 and 3270 individuals in 2013 and 2018 (Eshowe), respectively. In Ndhiwa, the proportion of PLHIV with advanced HIV decreased from 2012 (159/1376 (11.8%; 95% CI: 9.8-14.2)) to 2018 (53/1000 (5.0%; 3.8-6.6)). The proportion of individuals with advanced HIV on antiretroviral therapy (ART) was 9.1% (6.9-11.8) in 2012 and 4.2% (3.0-5.8) in 2018. In Eshowe, the proportion with advanced HIV was 130/1400 (9.8%; 8.0-11.9) in 2013 and 38/834 (4.5%; 3.3-6.1) in 2018. The proportion with advanced HIV among those on ART was 6.9% (5.5-8.8) in 2013 and 2.8% (1.8-4.3) in 2018. There was a significant increase in coverage for all steps of the care cascade among people with advanced HIV between the two Ndhiwa surveys, with all the changes occurring among men and not women. No significant changes were observed in Eshowe between the surveys overall and by sex.
Conclusion
The proportion with advanced HIV disease decreased between the first and second surveys where all guidelines have been implemented between the two HIV surveys.
Plain Language Summary
We examined changes over time in the proportion of people living with HIV (PLHIV) with advanced HIV and their care cascade in two community settings in sub-Saharan Africa: Ndhiwa (Kenya) and Eshowe (South Africa). In 2012 and 2018, a total of 6,076 and 6,001 individuals were included in Ndhiwa, and 5,646 and 3,270 individuals were included in Eshowe in 2013 and 2018, respectively. In Ndhiwa, the proportion of PLHIV with advanced HIV decreased from 11.8% in 2012 to 5.0% in 2018. The proportion of individuals with advanced HIV on antiretroviral therapy (ART) decreased from 9.1% in 2012 to 4.2% in 2018. In Eshowe, the proportion PLHIV with advanced HIV decreased from 9.8% in 2013 to 4.5% in 2018. Among those on ART, the proportion of PLHIV with advanced HIV decreased from 6.9% in 2013 to 2.8% in 2018. The results also showed a significant increase in coverage for all steps of the care cascade among people with advanced HIV in Ndhiwa in 2018 compared to 2012, with these changes observed only among men and not women. No significant changes were observed in Eshowe between the surveys, both overall and when comparing by sex.
Introduction
The burden of advanced HIV disease remains a significant concern in low- and middle-income countries particularly in sub-Saharan Africa, although morbidity and mortality associated with HIV have dropped over the past decade due to increased access to antiretroviral therapy (ART). About 30% of people living with HIV (PLHIV) continue to present for care with advanced HIV disease some of whom are returning to care after treatment interruption. 1 This group contributes to high morbidity and mortality.2-5 Moreover, individuals with advanced HIV disease may have a greater risk of viral transmission because of unsuppressed viral load.
To further reduce HIV-related mortality and the risk of new infections, in 2015, the World Health Organization (WHO) released treatment guidelines with recommendations for countries to start ART treatment for every person living with HIV regardless of CD4 count (“treat all”). 6 These were complemented in 2017 with guidelines for managing patients with advanced HIV disease and their initiation into care. 7 Published data showed varying differences in changes in the proportion of advanced HIV disease among people presenting for care at clinics in South Africa with some little or no change8,9 while others reported substantial decline over the years.10-12 A larger proportion of people with advanced HIV are ART experienced 2 suggesting that the majority of these seek care late when their health has already deteriorated although the proportion of late presenters and those initiating ART with advanced HIV is slowly decreasing11,13 with mixed results in others. 12
Médecins Sans Frontières (MSF) had been working in two high HIV-burden areas in Ndhiwa (Kenya) and Eshowe (South Africa) supporting the ministries of health of the respective countries to implement programs that promote early HIV diagnosis through improved access to voluntary counseling and testing services. South Africa has the highest number of PLHIV in the world. South Africa's HIV prevalence was estimated at around 19.5%, and incidence of 12.0 per 1000 population in 2021 among adults aged 15-49 years old. 14 HIV prevalence in Kenya is estimated at 4.9% among adults 15-64, however, geographical location plays a major role in the distribution of HIV in the country with prevalence ranging from as low as <0.1% in some areas to as high as 19.6% in others. 15 Both Ndhiwa and Eshowe had above average country-level HIV prevalences which prompted MSF to open HIV projects and programs some of which specifically targeted PLHIV with advanced HIV to help the respective ministries contain the disease.16,17 To identify PLHIV with advanced HIV, MSF had also been supporting the implementation of point-of-care CD4 cell count testing. Between 2012 and 2013, MSF carried out its first HIV surveys to collect baseline data on HIV burden and for use in the evaluation of the program's impact at the population level at a later stage. We previously analyzed data from those surveys and showed the distribution of advanced HIV disease in these populations. 18 In 2018 MSF carried out follow-up surveys in the same areas to evaluate the impact of its programs. Using data from the HIV population surveys, we aimed to assess changes over time in the proportion of PLHIV with advanced HIV disease and, among them, the proportion who know their status, the proportion on ART, and the proportion with suppressed viral load in both settings in Ndhiwa and Eshowe.
Methods
Study Design
In this analysis, we used secondary data from four population-based cross-sectional HIV surveys which were conducted between September and November 2012 (survey 1) and from October 2018 to January 2019 (survey 2) in Ndhiwa and between July and October 2013 (survey 1) and August to December 2018 (survey 2) in Eshowe. All surveys used similar study designs and methods which have been described in detail elsewhere.19-21 Briefly, a two-stage sampling design was used. At the first stage, the probability proportion to size (PPS) of the cluster was used to select clusters that followed demarcations of the national population and housing censuses conducted before the surveys. Then, from each selected cluster, we randomly selected 25 households (20 households for the Ndhiwa 1 survey), making the sample selection self-weighting. All residents or visitors who had spent the previous night in the study area who were aged between 15 and 59 years old were eligible for inclusion.
Between the first and second surveys, WHO introduced major policy recommendations for ART eligibility (universal treatment for all PLHIV regardless of CD4 count) 6 and guidelines for managing PLHIV with advanced HIV disease and their initiation in care. 7 During the first surveys, in both countries ART eligibility criteria for PLHIV were CD4 ≤ 350 cells/µL or WHO Stage 3 or 4 but differed with respect to prevention of mother-to-child transmission guidelines with Kenya using “Option A” (ART when CD4 ≤ 350 cells/µL for pregnant and breastfeeding women and antiretroviral prophylaxis only if maternal CD4 > 350 cells/µL) while South Africa was on “Option B” (ART for all pregnant and breastfeeding women until cessation of breastfeeding). South Africa implemented the 2013 WHO guidelines 22 which had recommended starting PLHIV on ART if they had CD4 ≤ 500 cells/µL in January 2015. When the WHO recommended starting every person living with HIV on treatment regardless of CD4 count in 2015, both countries adopted the recommendation with South Africa implementing in September 2016 and Kenya in July 2016. Following the introduction of guidelines for the management of people with advanced HIV in 2017, MSF in 2018 started implementing the screening and care management of advanced HIV individuals at primary health facilities in Eshowe and in Ndhiwa they started focusing on treatment failure and advanced HIV by introducing a dedicated laboratory and point-of-care testing for inpatients to facilitate faster treatment of those in need. For our study, advanced HIV was defined as CD4 < 200 cells/µL a definition similar to what other studies have used. 23
Data Collection Methods
Consenting individuals were interviewed at their households and a rapid HIV test was conducted if they gave consent to be tested. Detailed methods for HIV rapid testing and other laboratory tests carried out for the earlier surveys are reported elsewhere. 24 For recent surveys, HIV testing was done either on site at the participants’ dwelling, or at a laboratory for participants willing to participate in the study but not willing to undertake an HIV test on site. In both recent surveys, in Ndhiwa and in Eshowe, an HIV rapid test was done using the Determine test kit as a screening test which was followed if positive by the Unigold Rapid HIV test kit for confirmation. A positive test was considered confirmed if both tests were positive. For discordant results, Western blot (Bio-Rad GS HIV-1 Western Blot run on the Autoblot 3000, Med tech Biolab Equipment) was used as a tiebreaker in the South African survey while the Kenyan survey used polymerase chain reaction tests as a tiebreaker. For both recent studies, venous blood was collected from participants who tested positive for further laboratory tests. CD4 tests were done at a laboratory for Eshowe (South Africa) while for Ndhiwa they were done using the PIMA™ CD4 point-of-care test (Alere, Germany) machine. We used structured questionnaires to collect information on demographics, HIV status awareness, history of HIV testing, and ART among other variables. For minors younger than 18 years, consent was not required from the parent/guardian as children between the ages of 12-17 in South Africa25-27 and 15-17 in Kenya 28 do not require parental/guardian consent for HIV testing and treatment. Data were double entered using EpiData and checked for inconsistencies before analysis.
Statistical Methods and Analysis
Our hypothesis was that due to the implementation of universal ART (ART for all HIV positive) together with other interventions to increase diagnosis and linkage to care, the proportion of PLHIV with advanced HIV would decrease between the first and second surveys and, among those with advanced HIV, a greater proportion would have previously started ART at the time of the second survey compared to the first. To answer our hypothesis, we pooled together HIV population level data from the four surveys. Two repeat cross-sectional surveys were conducted in each survey area. We assumed independence of the surveys as (1) the time that had passed between the first and second surveys was relatively long (about 5 years), (2) for each survey, the participants were selected randomly, and (3) it would be difficult to know if any individuals were selected and participated in both surveys. Using chi-square tests, we compared the distribution of demographic characteristics by survey period for each setting. This allowed us to assess if the populations sampled were similar between the first and second surveys at each site. We then assessed the characteristics of PLHIV with and without advanced HIV disease using chi-square tests. As no clinical data were collected during the surveys, we considered all PLHIV with CD4 count <200 cells/µL as having advanced HIV disease, as in our previous article. 18 Only data collected during the surveys were used for this analysis.
We used both methods of monitoring the HIV cascade of care, the prevalence method with the denominator being all PLHIV (measures coverage of HIV indicators) and the 90-90-90 UNAIDS targets for 2020 (where the denominator changes for each step of the cascade) which states that by the year 2020, we should have 90% of all PLHIV know their positive status, of these 90% should be retained on ART and 90% of those on treatment should have suppressed viral load. Using chi-square tests, we compared the prevalence-based cascade of care (diagnosed [those who knew they were HIV-positive], linkage to care, on ART, virologically suppressed) and the 90-90-90 UNAIDS targets for all PLHIV with advanced HIV disease for each survey setting by sex. Results are presented in bar graphs with 95% confidence intervals (CIs).
To measure the effect of time on advanced HIV disease between the two survey periods, we created a variable “survey period” distinguishing the two survey periods which became the main variable of interest. Using logistic regression, we assessed the crude association between age, sex, education, marital status, occupation, ART status, HIV status awareness, viral suppression, and advanced HIV disease for each of the four surveys. We repeated the same process with pooled data and assessed the association between survey period and advanced HIV disease. The inclusion of the variable “survey period” in the model helped us measure the magnitude of change in advanced HIV disease between the two periods and meant that we took into consideration the non-independence of the surveys clustered by time the same way a random effect model would, had we not assumed independence of surveys. We also undertook a random effects regression model, and the results were similar. We used the stepwise technique in model building. Survey period, age, and sex were a priori explanatory variables. Other variables were included one by one starting with the variable most strongly associated with advanced HIV disease in the crude pooled univariate analysis. Variables not associated with advanced HIV disease and those deemed to be collinear with variables already added in the model were not included in the final model. ART status and a priori variables were therefore included as explanatory variables for advanced HIV disease, but HIV status awareness and viral load were not included in the final model because there was some evidence of correlation with ART status. Missing values for each variable were reported but were not included in the analysis. Statistical significance was ascertained with 95% confidence intervals or a P value of <.05. We used STATA 15 (Stata Corporation, College Station, TX) for data analysis and graphs were drawn in MS Excel.
Ethical Approval and Informed Consent
For all surveys, ethical approval was sought at both local and international levels. For Ndhiwa, local approvals were obtained from the Kenya Medical Research Institute Ethical Review Committee (KEMRI). For Eshowe, local approvals for both surveys were obtained from the Human Research Ethics Committee and the Health Research Committee of the Health Research and Knowledge Management Unit of the Kwazulu-Natal Department of Health. International approvals for the first survey were obtained from the Comité de Protection des Personnes d’Ile de France and a different Ethics Review Board for the second survey. Survey participants were given sufficient information about the aims of the surveys and provided written consent for their inclusion in the study.
Results
The survey included 6076 participants, 2321 (38.2% male) (Ndhiwa 1), 6001 participants, 2509 (41.8% male) (Ndhiwa 2), 5646 participants, 2131 (37.7% male) (Eshowe 1) and 3270 participants, 1116 (34.1% male) (Eshowe 2) (Table 1). HIV prevalence among adults 15-59 years old in Ndhiwa 2 was lower than Ndhiwa (survey 1) (16.9%, 95% CI: (16.0-17.9)) versus 24.1% (22.7-25.6) (survey 2) but was similar in both Eshowe surveys 25.2% (23.6-26.9) (survey 1) versus 26.4% (24.9-27.9) (survey 2).
Distribution of Demographic Characteristics by Country and Survey Period Among 12 077 Individuals in Ndhiwa (Kenya) and 8816 Individuals in Eshowe (South Africa) Enrolled in the Study.
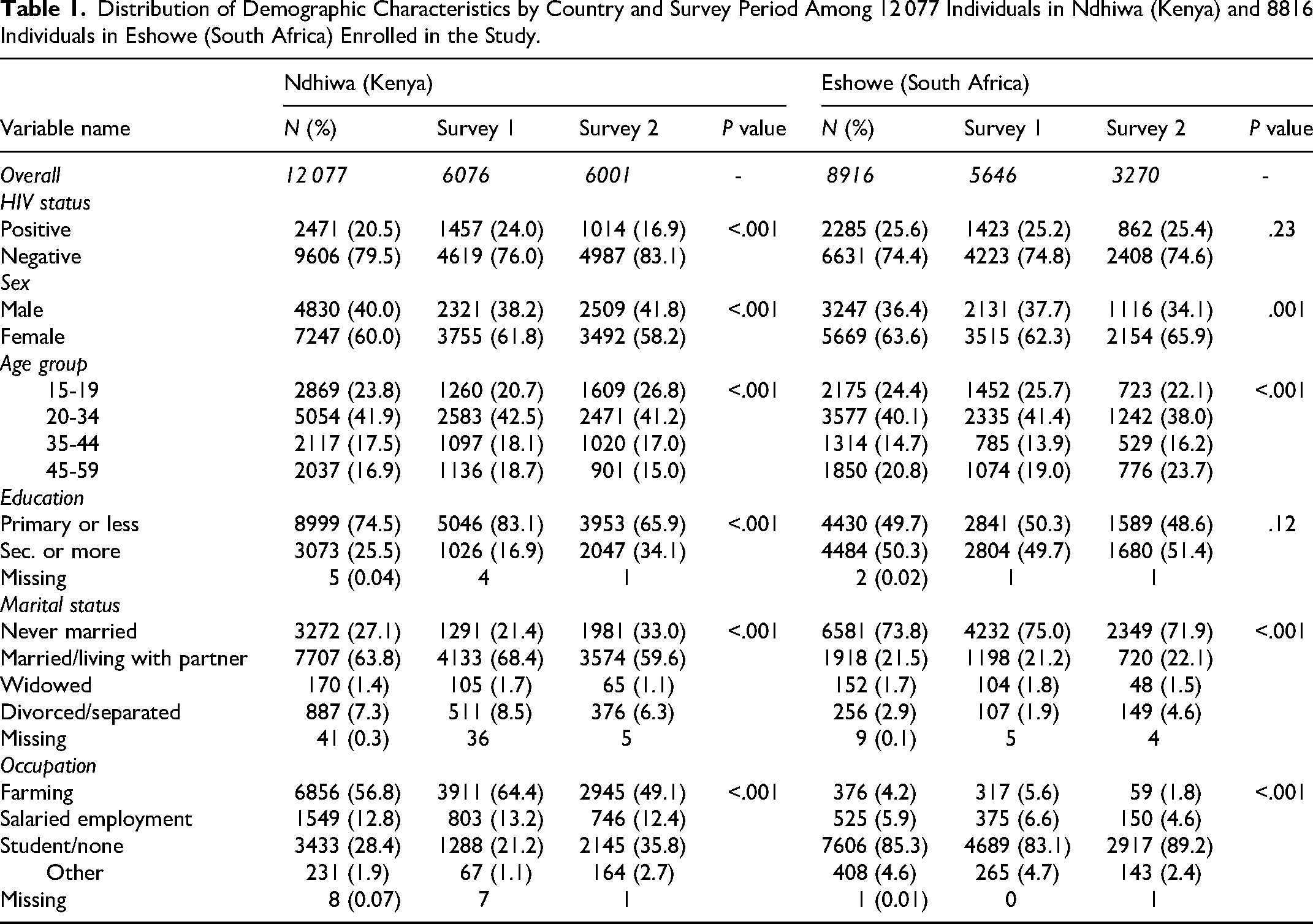
Overall, in Ndhiwa 95 (3.8%) individuals, 14 (14.7%) of which came from the second survey, were missing CD4 results. In Eshowe 51 (2.2%) individuals, 28 (54.9%) of which were from the second survey, had missing CD4 results. The proportion of people with advanced HIV disease in Ndhiwa 1 was 159/1376 (11.8%; 9.8-14.2) and 53/1000 (5.0%; 3.8-6.6) in Ndhiwa 2. In Eshowe, the proportion of advanced HIV diseases was 130/1400 (9.8%; 8.0-11.9) in the first survey and 38/834 (4.5%; 3.3-6.1) in the second survey.
In Ndhiwa 2, we found fewer PLHIV, more males, and more with secondary and post-secondary education compared to Ndhiwa 1. In Eshowe, however, we found no difference in the proportion of PLHIV between the first and second surveys, and that individuals were equally educated but there were fewer individuals aged 15-19 years old in the second survey compared to the first survey (Table 1).
The distribution of demographic characteristics among HIV-positive individuals comparing the first and second surveys in Ndhiwa was similar by sex, but there were fewer individuals in those aged 20-44 years and more people with secondary or more education in the second survey. Similarly, in Eshowe, there was no difference in the distribution by sex and there were fewer individuals aged 20-34 years in the second survey. However, there was no difference in the level of education between the first and second surveys (Table 2).
Distribution of Demographic Characteristics Among People Who Tested Positive for HIV by Country and Survey Period.
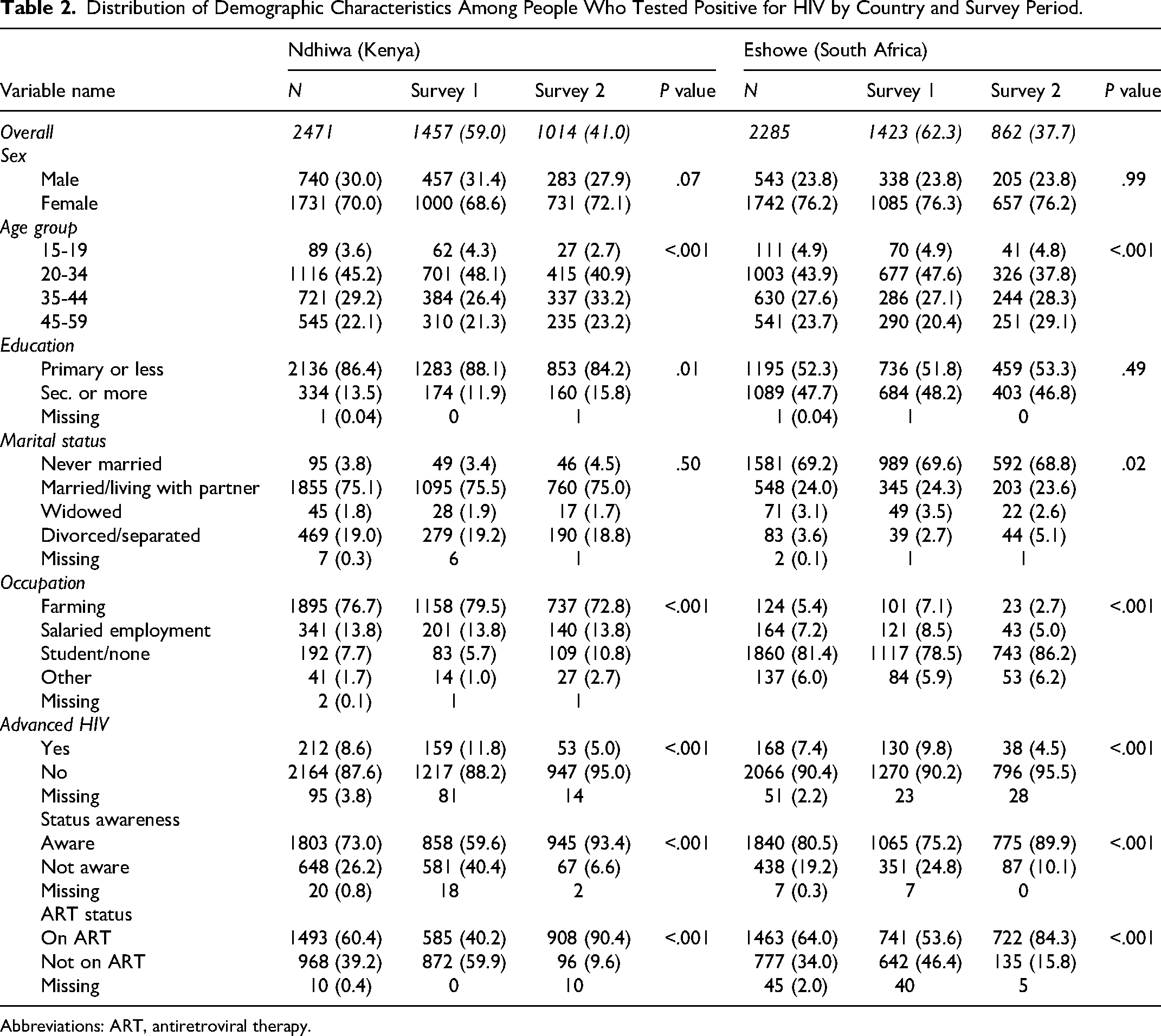
Abbreviations: ART, antiretroviral therapy.
Figures 1a and 1b show the HIV cascade of care for PLHIV with advanced HIV disease for each of the survey periods by site. Overall results in Ndhiwa (survey 1 vs survey 2) showed that 59.0% (51.2-66.4) versus 83.5% (67.7-91.4) were diagnosed (aware of their HIV-positive status), 50.5% (41.7-59.4) versus 79.6% (64.7-89.3) were linked to care, 31.7% (24.5-40.0) versus 74.2% (60.0-84.7) were on ART and 21.2% (14.6-30.0) versus 51.5% (37.4-65.3) had suppressed viral load, respectively. Sex-specific results showed that men registered a big improvement in all steps of the HIV cascade of care comparing the first and second surveys while there was no change between the surveys for women. Among PLHIV on ART, the proportion of individuals with advanced HIV disease was 9.1% (6.9-11.8) in 2012 and 4.2% (3.0-5.8) in 2018 in Ndhiwa. The cascade of care for PLHIV in the general population in Ndhiwa 2 was; status awareness 93.4% (91.7-94.8), those linked to care 91.8% (90.0-93.4), on ART 90.4% (88.5-92.1) and those with suppressed viral load 88.3% (86.1-90.1). 16
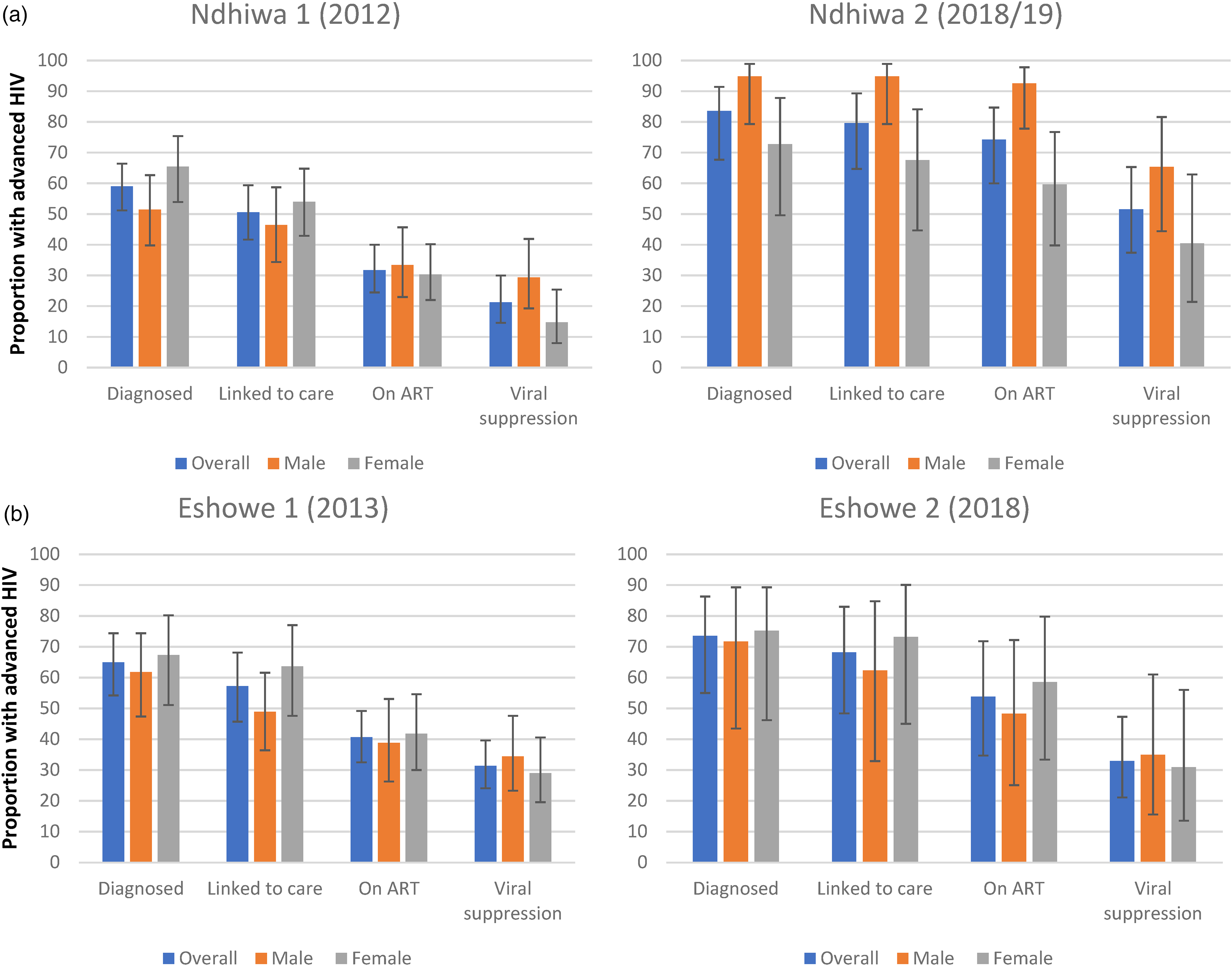
(a). Cascade of care graphs for Ndhiwa (Kenya) comparing surveys 1 and 2 among PLHIV advanced HIV. (b). Cascade of care graphs for Eshowe (South Africa) comparing surveys 1 and 2 among PLHIV advanced HIV.
In Eshowe 1, among PLHIV with advanced HIV the cascade of care results for survey 1 versus survey 2 showed that 64.9% (54.2-74.4) versus 73.5% (55.0-86.3) were diagnosed, 57.2% (45.7-68.1) versus 68.2% (48.4-83.0) linked to care, 40.6% (32.5-49.2) versus 53.8% (34.7-48.3) on ART and 31.3% (24.1-29.6) versus 32.9% (21.1-47.3) had suppressed viral load, respectively. For Eshowe, the cascade of care sex-specific results for PLHIV with advanced HIV were similar between the first and second surveys. Among PLHIV on ART, the proportion with advanced HIV disease was 6.9% (5.5-8.8) in Eshowe 1 and 2.8% (1.8-4.3) in Eshowe 2. In the general population in Eshowe 2, the cascade of care for PLHIV was; status awareness 89.9% (87.7-91.8), those linked to care 88.6% (86.3-90.5), on ART 84.3% (81.7-86.5) and viral suppression 83.8% (81.1-86.1). 17
Figures 2a and 2b show results of the 90-90-90 UNAIDS targets among those with advanced HIV disease. Our results from Ndhiwa show improvement in the first and second 90 comparing the first and second surveys. Overall, the proportion who were diagnosed (first 90) were 59.0% (51.2-66.4) in the first survey compared to 82.5% (67.7-91.4) in the second survey and those who were on ART among those diagnosed (second 90) were 53.8% (42.9-64.3) in the first survey compared to 90.0% (74.8-97.6) in the second survey. However, viral suppression among those who were on ART (third 90) was not statistically different; 55.2% (40.6-68.9) in the first survey compared to 63.8% (47.6-77.5) in the second survey. Men made gains in the first and second 90 but no change in the third 90 while there was no significant change between the first and second surveys in all the three 90s among women. For Eshowe, we found no significant differences in all three 90 targets between the first and second survey overall and even when the results were stratified by sex; however, the 95% CIs were wide. The overall results were 64.9% (54.2-74.3) versus 73.5% (55.0-86.3) (first 90), 65.2% (54.6-74.4) versus 74.1% (51.1-88.7) (second 90) and 79.2% (64.7-88.8) versus 56.7% (39.0-72.8) (third 90).
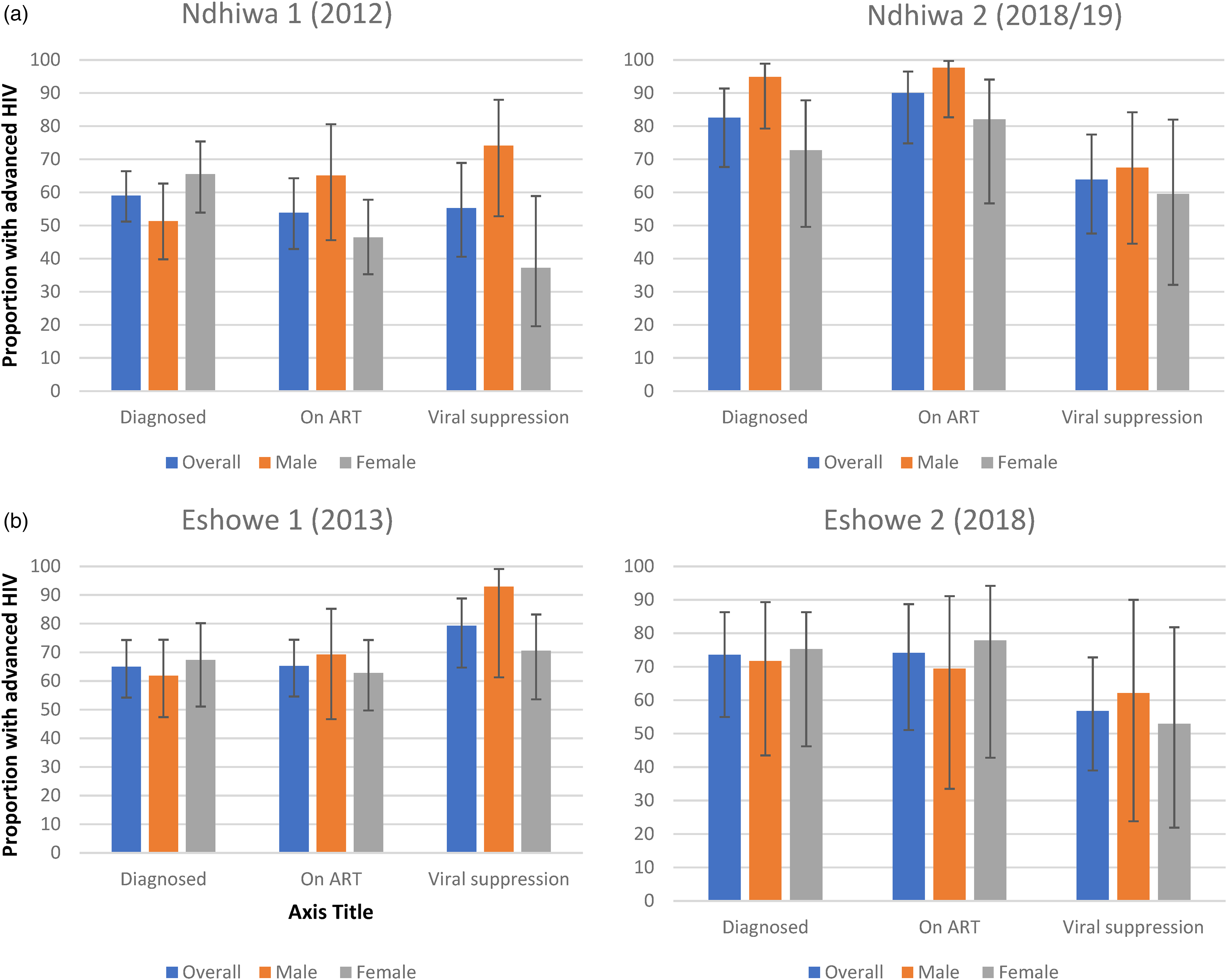
(a). 90-90-90 Graphs for Ndhiwa (Kenya) comparing surveys 1 and 2 among PLHIV advanced HIV. (b). 90-90-90 Graphs for Eshowe (South Africa) comparing surveys 1 and 2 among PLHIV advanced HIV.
The results of the regression analysis are presented in Tables 3 and 4. In Table 3, we present survey-specific results. In Ndhiwa, we found that in both surveys, sex, ART status, education, and viral suppression were associated with advanced HIV disease, but HIV status awareness was associated with advanced HIV disease in the Ndhiwa second survey only. In Eshowe, sex, ART status, knowledge of HIV-positive status, and viral suppression were associated with advanced HIV disease in both surveys. Results of the pooled analysis by survey site are presented in Table 4. In multivariable regression, we found that in Ndhiwa, the risk of advanced HIV disease was 40% less in the second survey compared to the first survey (adjusted odds ratio [aOR] 0.6; 95% CI: 0.4-0.9), 2.3 (95% CI: 1.7-3.1) times more likely in men than in females and 1.9 (95% CI: 1.3-2.7) times higher among those not on ART compared to those on ART. Similarly, in Eshowe, the risk of advanced HIV disease was 40% less in the second survey compared to the first survey (aOR 0.6; 95% CI: 0.4-0.9), 2.3 (95% CI: 1.7-3.2) times more likely in males than in females and 2.0 (95% CI: 1.4-2.9) times more likely in those not on ART compared to those on ART. No differences were observed by age group in Ndhiwa but in Eshowe, individuals in the age group 35-44 years were 2.0 (95% CI: 1.2-3.3) times more likely to have advanced HIV compared to the older age group of 45-59 years old.
Results of Crude Logistic Regression of the Association Between Demographic and HIV Characteristics and the Outcome of Advanced HIV for Each of the Demographic Surveys.
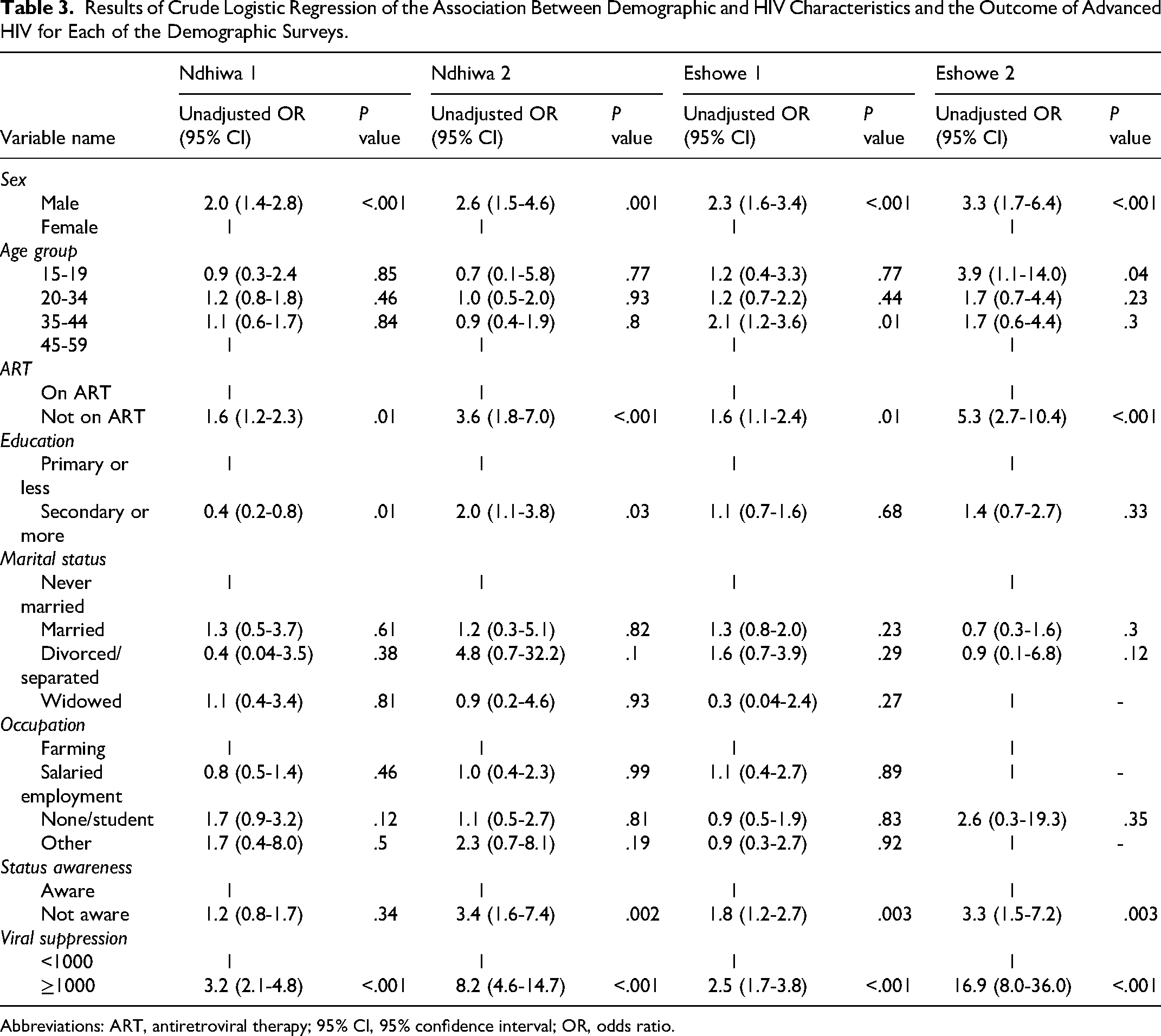
Abbreviations: ART, antiretroviral therapy; 95% CI, 95% confidence interval; OR, odds ratio.
Results of Crude and Adjusted Logistic Regression of the Association Between Survey Period (First or Second Survey) in Ndhiwa, Kenya, and Eshowe, South Africa, and Having Advanced HIV Disease.
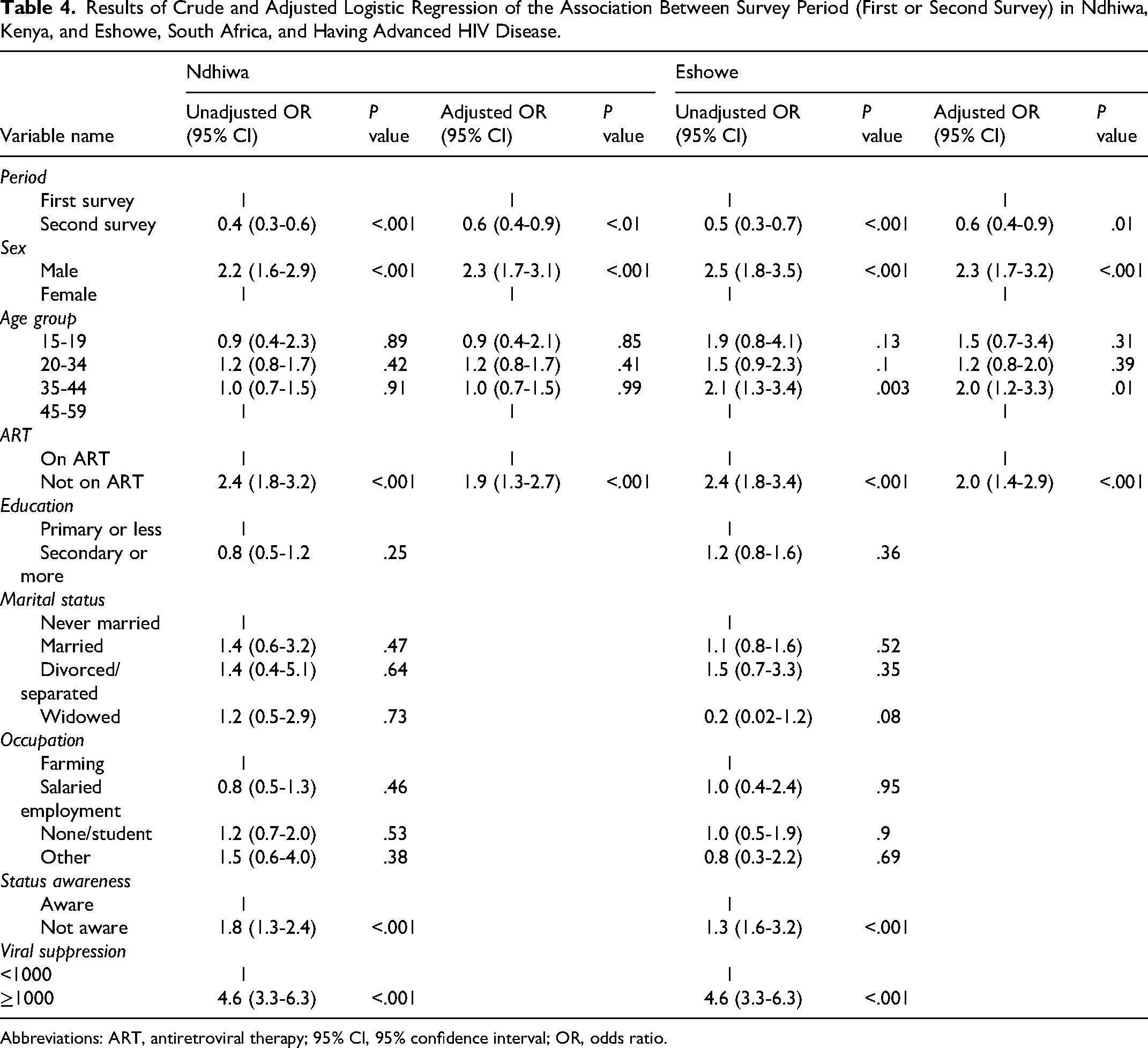
Abbreviations: ART, antiretroviral therapy; 95% CI, 95% confidence interval; OR, odds ratio.
Discussion
Our results in both settings show that there has been an overall decline in the proportion of PLHIV with advanced HIV disease by 6.8% in Ndhiwa (Kenya) and 5.3% in Eshowe (South Africa). The risk of PLHIV with advanced HIV dropped by about 40% over the past 5 years comparing the first with second surveys in both areas. A significant drop in the proportion with advanced HIV disease was also noted among those on ART. However, the risk of advanced HIV remains high among males compared to females and individuals not on ART compared to those on ART at the time of the survey. The decline was accompanied by significant improvements in all stages of the cascade of care (status awareness, linkage to care, those on ART, and viral suppression) in Kenya but not in South Africa.
A study 23 that looked at trends and proportions of advanced HIV between 2012 and 2016 in four high HIV-burden sub-Saharan countries (Cameroon, Mozambique, Uganda, and Zimbabwe) reported a 3% significant drop in the proportion of advanced HIV disease between the two periods. The study, however, used data sourced from cross-sectional CD4 records collected from a variety of medical facilities across countries that participated. The bigger drop in our study, however, could be because, by the time we conducted the repeat surveys, both countries had already implemented the WHO treat all guidelines allowing all people with HIV to be initiated on ART. Kenya implemented the treat all guidelines in July 2016 while South Africa implemented in September of the same year 2016 representing at least 2 years of implementation before the second surveys were conducted, Implementation of these guidelines could have allowed more PLHIV to be initiated on treatment early. In addition, the impact of treat all could be manifested in the population first while the clinics may continue to receive the sick people with advanced HIV disease who did not seek care early.
Our findings, do not align with several other studies that have looked at trends in advanced HIV disease or CD4 cell count at enrollment into ART care. Most of these studies indicated little or no change in the proportion of advanced HIV disease over relatively the same study period as our study. Two studies in South Africa,8,9 one using data from nationwide laboratory CD4 count data between 2004 and 2016 to determine entry into HIV care, and the other using a cross-sectional cohort of individuals receiving CD4 tests from public sector laboratories between 2008 and 2017 found that the proportion of individuals with advanced HIV disease and very advanced HIV (CD4 < 50 cells/μL) remained relatively unchanged over the last 5 years of the period of which data was analyzed. These results are despite increased coverage of ART over the years, suggesting late treatment initiation, poor retention, or adherence to treatment. 29 The second study also found that a higher proportion of those with advanced HIV disease were ART experienced, a result supported by another study that looked at data from two sub-Saharan African countries of Kenya and the Democratic Republic of Congo. 2
Reassuringly the decline in the proportion with advanced HIV disease concurs with successes in viral suppression in the cascade of care for the general population in both survey settings. In both settings, more people knew their HIV status, were on ART, and had suppressed viral load. We would therefore expect the majority with suppressed viral load to have improved immune function and clinical status as evidence suggests that CD4 rises in the majority of patients who initiate ART and much more quickly if treatment is initiated early 30 resulting in the decline in the levels of advanced HIV disease in both settings. Similar to the findings of our study, several other studies have also found the risk of advanced HIV disease to be higher among men than women8,31,32 highlighting the important need to intensify targeting men to get them tested through innovative methods of testing, getting them into care and ensuring that they remain in care through male friendly differentiated models of care. We, however, also noted an improvement in all steps of the cascade of care comparing the first and second surveys in Ndhiwa among men.
Another important finding of our study is that although we found an improvement in the cascade of care coverage and 90-90-90 UNAIDS targets between the first and second surveys more especially in Kenya, we cannot claim the same when we compare cascade of care and the 90-90-90 UNAIDS target results for individuals with advanced HIV disease with that of the general population. The general population has succeeded in achieving the targets 1 year ahead of time in both settings16,17,33 which is why we need to put more effort into identifying those PLHIV with advanced HIV through testing, initiating them on care and ART, and ensuring that they are retained. This suboptimal coverage of the cascade of care in those with advanced HIV risks derailing the efforts of reducing HIV-related mortality and morbidity which is high among people with advanced HIV disease. In this era of “treat all” we must intensify testing with innovative methods to ensure that more PLHIV know their status, routine CD4 testing for PLHIV should be encouraged so that individuals with advanced HIV disease are identified and given special care to ensure they remain and respond to treatment. Nonetheless, we found a lower proportion of those on ART with advanced HIV, and the overall decline in advanced HIV is supported by significant improvements in coverage of all steps of the HIV cascade of care among individuals with advanced HIV especially in Kenya. In Eshowe, however, while there were no significant differences in any individual step of the HIV cascade of care between the first and second survey, in general, there was an improvement at each step of the HIV cascade of care, likely explaining the overall reduction in the percent with advanced HIV disease.
Our study compares changes that have taken place in the same population between two time periods. While the results show successes between the periods, the lack of yearly data to show the yearly changes makes it difficult to have a true picture of what happened in between and to accurately assess the success of those interventions. The differences between clinical cohort studies and population surveys indicate that it is difficult to have a proper estimate of prevalence or trends in advanced HIV disease using a single source of data. Population-based surveys are representative of the general population but for the study of advanced HIV, they may underestimate the prevalence as they will exclude those who are very sick and admitted to the hospital. While this proportion may be low for the population, it is a high proportion of those in hospital. Furthermore, it is difficult to capture clinical data for the WHO clinical definition of advanced HIV disease in population surveys. Combining all these, our study may have underestimated the prevalence of advanced HIV. The fact that our data comes from four large population-based surveys with less missing data for the main outcome variable and a good participation rate makes our results more reliable. We, however, suggest future advanced HIV prevalence studies to consider participants' clinical data and to include hospital patients within the study catchment area.
Conclusion
This study demonstrated a decrease over time in the proportion of PLHIV with advanced HIV disease in these two countries. This finding is encouraging for other nations aiming to reduce HIV-related mortality and morbidity, which is largely due to the high prevalence of advanced HIV disease. Nonetheless, increased investment in HIV and CD4 testing is crucial to reach and identify individuals at the highest risk of advanced HIV for prompt and appropriate care.
Footnotes
Authorship
All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
