Abstract
Polygamy is the practice of marriage to multiple partners. Approximately 6-11% of households in Uganda and 4-11% of households in Kenya are polygamous. The complex families produced by polygamous marriage customs give rise to additional considerations for healthcare providers and public health messaging around HIV care. Using 27 in-depth, semi-structured qualitative interviews with participants in two studies in rural Kenya and Uganda, we analysed challenges and opportunities that polygamous families presented in the diagnosis, treatment and prevention of HIV, and provider roles in improving HIV outcomes in these families. Overall, prevention methods seemed more justifiable to families where co-wives live far apart than when all members live in the same household. In treatment, diagnosis of one member did not always lead to disclosure to other members, creating an adverse home environment; but sometimes diagnosis of one wife led not only to diagnosis of the other, but also to greater household support.
Plain Language Summary
Polygamy is the practice of marriage to multiple partners. Approximately 6-11% of households in Uganda and 4-11% of households in Kenya are polygamous. The complex families produced by polygamous marriage customs give rise to additional considerations for healthcare providers and public health messaging around HIV care.
Background
Polygamy is the practice of marriage to multiple partners; marriage to multiple women (polygyny) is the most common form of polygamy worldwide. 1 Though rare in the global north, polygamy is common throughout the world, being legal in 58 out of nearly 200 countries including many in sub-Saharan Africa. In East Africa, where HIV prevalence is 4–5% of the adult population – and in the Lakes region (specifically around Lake Victoria) is between 22% and 29% – approximately 6–11% of households in Uganda and 4–11% of households in Kenya are polygamous. Technically polygyny is the dominant form of polygamy in these regions; polyandry, or marriage to multiple men, is quite rare and not legal in Kenya and Uganda.2–4 Therefore, in this manuscript, the term polygamy refers to heterosexual polygynous relationships.5–9 In Uganda, polygamy is most common among people aged 35–44 and 45–54 years, at 15% of all marriages in these age cohorts. It is most common in the Karamoja region in northeastern Uganda, where up to 24% of households are polygamous. In Kenya, polygamy is notably practiced by Muslims along the coast, with an average of 15% of marriages in Kilifi and 16% in Kwale being polygamous; in Turkana (27% of marriages); and among the Luo around Lake Victoria (Migori: 26%; Homa Bay: 23–25%; Kisumu: 15%). 10 In particular, the Luo of both Kenya and Uganda are known for the practice of levirate marriage, sometimes generically referred to as ‘wife inheritance’. Wife inheritance is a custom in which if a woman's husband dies, one of the husband's brothers marries her, even when that brother already has a wife. Historically, the widow is duty-bound to accept this, though she could sometimes choose a specific man within the lineage to marry. 11 This practice stems in part from the wife being part of the homestead of the extended family, not simply related by marriage to the husband alone. Wife inheritance preserves this extended family relationship, but also carries risks for HIV.11–14
Early HIV prevention messaging, often developed by funders in the global north, assumed monogamy as the standard or ideal practice for married relationships.15–17 These messages often neglected the complicated realities of polygamous families and were mismatched to the lived experiences of people living with HIV (PLHIV) in many parts of the world. Indeed, up to the present day, few HIV prevention interventions or HIV treatment programs incorporate polygamous families in any meaningful way, despite recognition of this need.13,18,19 Marriage has been identified as a risk factor for HIV acquisition and polygamy may play a part in the spread of HIV among married couples.14,20–27 One study found that at an individual level, men in polygamous unions have triple the rates of HIV than men in monogamous marriages, while women in polygamous marriages have twice the rate of HIV compared to their monogamous counterparts; for people in monogamous unions, rates of HIV were about the same for both men and women. 28 At a community cluster level among the Luo, however, researchers found “hardly any difference in HIV prevalence between those in polygynous and monogamous unions,” yet the presence of polygyny as a community custom is associated with approximately three-times higher HIV prevalence than in communities where polygyny is absent as a tradition. 28 Despite the widespread practice of polygamy and the abundant literature on the topic from both legal and social science perspectives, little has been written on how it affects clinical HIV care and practice. Often, polygamy receives just a passing reference in many clinical publications as well as programmatic recommendations.
The complex families produced by polygamous marriage customs give rise to additional considerations for clinicians, providers, STI testing, and public health messaging around sexual and reproductive health. Although certain negative consequences particularly affect women in polygamous relationships, ranging from interpersonal violence to lower mental and economic health, positive influences are also present, especially around testing and treatment of HIV.29–34 Thus, while polygamy can challenge HIV prevention, diagnosis, and care, it also offers potential opportunities for the engagement of wider families in preventing or managing HIV together. 14 These positive influences have often, though not always, been neglected in the literature.35,36
This paper, drawn from interviews with participants in two HIV treatment and prevention studies in Kenya and Uganda, seeks to examine these dynamics. We begin with a brief look at how providers engage these relationship contexts among their patients, before going on to examine diagnosis experiences, treatment, and support within polygamous families. Next, we turn our attention to HIV prevention negotiation in polygamous families. We present challenges in clinical care but also opportunities for engaging polygamous families in both HIV prevention and care.
Methods
This paper draws on data from two separate qualitative studies within the Sustainable East Africa Research in Community Health (SEARCH) consortium: the ‘Strategic antiretroviral therapy and HIV testing for youth in rural Africa’, or SEARCH-Youth study (NCT03848728), and ‘A Multisectoral Strategy to Address Persistent Drivers of the HIV Epidemic in East Africa, or SEARCH-SAPPHIRE study (NCT04810650). Both studies were conducted in rural Kenya and Uganda.
SEARCH-Youth was a cluster-randomized controlled trial in 28 clinics in western Kenya and southwestern Uganda, that aimed to evaluate whether a combination intervention using a life-stage approach would improve viral suppression among youth living with HIV aged 15–24 years. In the study, patients in intervention clinics received life-stage-based assessment at routine visits, flexible clinic access, and rapid viral load feedback. Providers had a secure mobile platform for consultation with one another. The intervention was found to be effective at increasing viral suppression, especially among those re-engaging in care (RR 1·60, 95% CI 1·00-2·55; p = 0·025). 37 We conducted a qualitative study among a subset of SEARCH-Youth participants (n = 83) from both intervention and control arms, family members (n = 33), and providers (n = 20), conducting in-depth interviews at baseline in 2019, and follow-up at one and two years (2020 and 2021) to identify the mechanisms of action, barriers, and facilitators of study implementation. Interview questions with youth explored social support systems, experiences of HIV care, and HIV status disclosure, while provider interviews sought their perspectives on the needs and barriers to care engagement among youth, as well as perceived challenges to care delivery to this population.
The ongoing SEARCH-SAPPHIRE Study in rural Uganda and Kenya is evaluating whether a community precision health approach to delivering multidisease patient-centered testing, prevention and care services can reduce HIV incidence and improve community health. The first published results of the Study, based on pilot trials in four communities in the same rural settings as SEARCH-Youth, showed that the “Dynamic Choice HIV Prevention (DCP) intervention, which provided structured patient choice in HIV prevention product (eg pre-exposure prophylaxis/PrEP, post-exposure prophylaxis/PEP, condoms), HIV testing modality, and location delivery preferences over time, increased HIV prevention coverage by 28% to 40% (absolute effect size) compared to standard of care when delivered in three settings (ante and postnatal care clinics, outpatient clinics, and community settings via village health teams).38–41 An additional pilot trial of a “dynamic choice treatment” (DCT) intervention for mobile participants living with HIV showed an improvement in ART possession and retention, but no impact of viral suppression. 42
The qualitative study within SEARCH-SAPPHIRE involved in-depth interviews with purposively-selected participants from the intervention arms of these 4 pilot trials, who received HIV prevention services using the ‘dynamic choice prevention’ model (n = 19 in antenatal clinics, n = 19 in outpatient clinics, n = 18 in the community); or HIV treatment services using the ‘ dynamic choice treatment’ model (n = 18); plus providers in these communities (n = 26). The interviews, conducted from November 2021 through March 2022, explored experiences with the intervention, preferred prevention and treatment options, patient-provider interactions, life and community contexts, and discussions with peers about HIV, HIV-prevention methods, and HIV-related stigma. The narratives sometimes also yielded information about past experiences with health service providers, some of which occurred prior to the start of the SEARCH-SAPPHIRE study.
We purposefully selected a subset of participants from each of these studies to participate in in-depth, semi-structured interviews. These participants were contact by phone. A team of two female (CA, AO) and three male (LO, FA, TOA) trained qualitative researchers with native fluency in the local languages (Luganda, Dholuo, English, Swahili) conducted the interviews. The interviewers maintained confidentiality by conducting interviews in private spaces convenient for the participants. Interviews lasted between 60 and 90 min and participants received transport reimbursement and approximately US$5 in appreciation of their time. The team audio-recorded the interviews, created a brief memo of the interview, then transcribed and translated them into English. This procedure was followed for both SEARCH-Youth (between 2019 and 2021) and SEARCH-SAPPHIRE (in 2022).
An eight-person team (CA, FA, TOA, JJP, JL, IM, AO, LO) then coded data using Dedoose software, following codebooks developed from both a priori and inductive codes. 43 We derived inductive codes from a set of transcripts the team individually coded, whose codes were then collated and discussed. This not only functioned to blind team members while double-coding the selected transcripts, but later permitted the team to resolve any discrepancies or questions about which codes to apply and when before actual coding. 44 During the coding process, in case of new questions, we also kept memos to discuss with team members about which codes to apply.
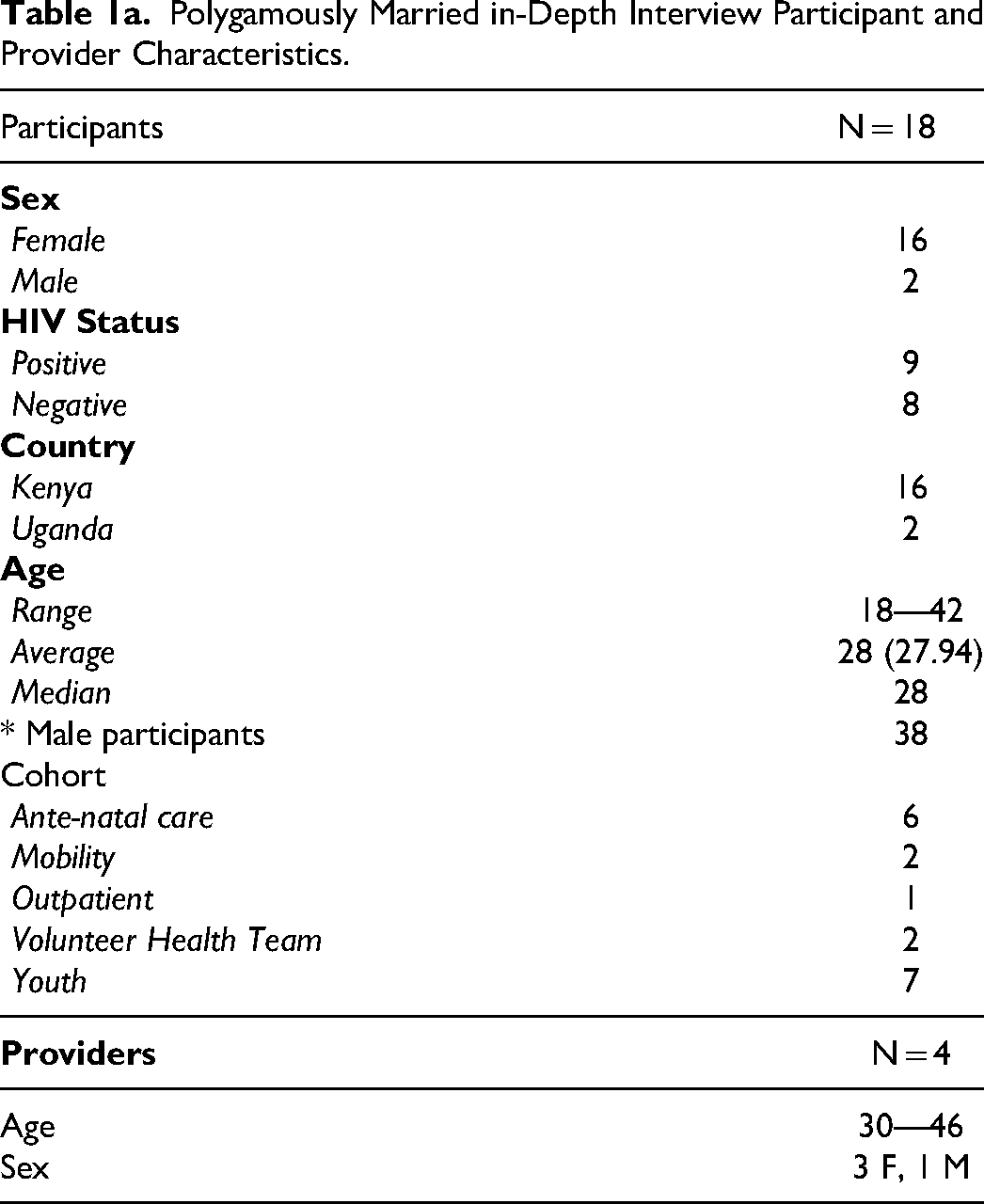
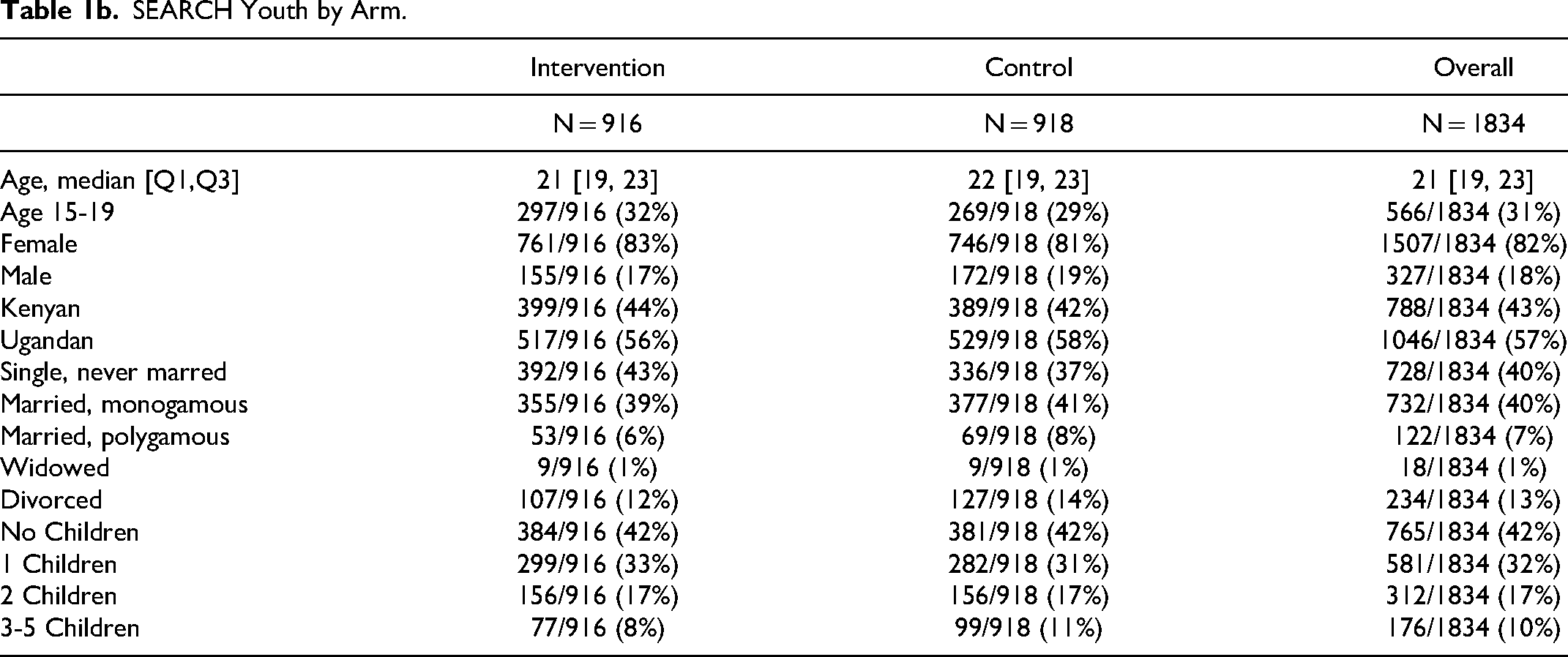
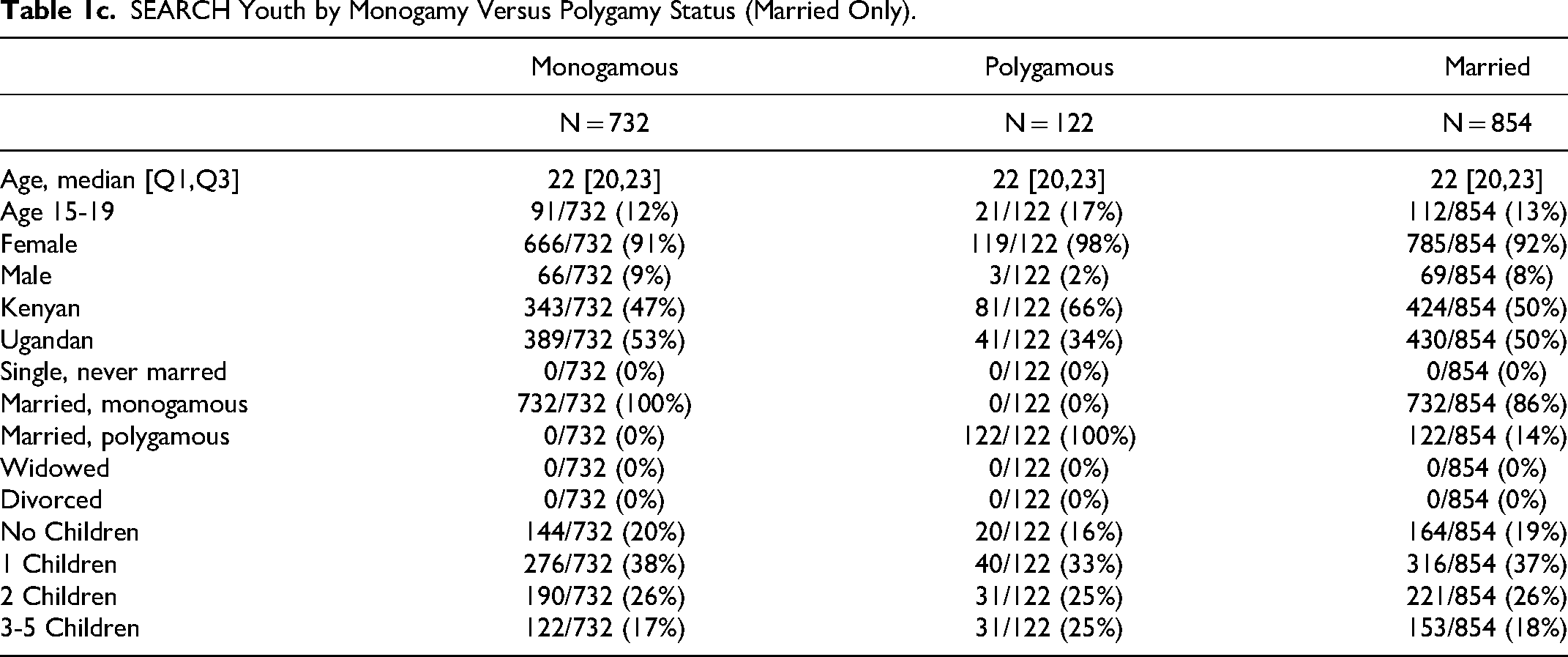
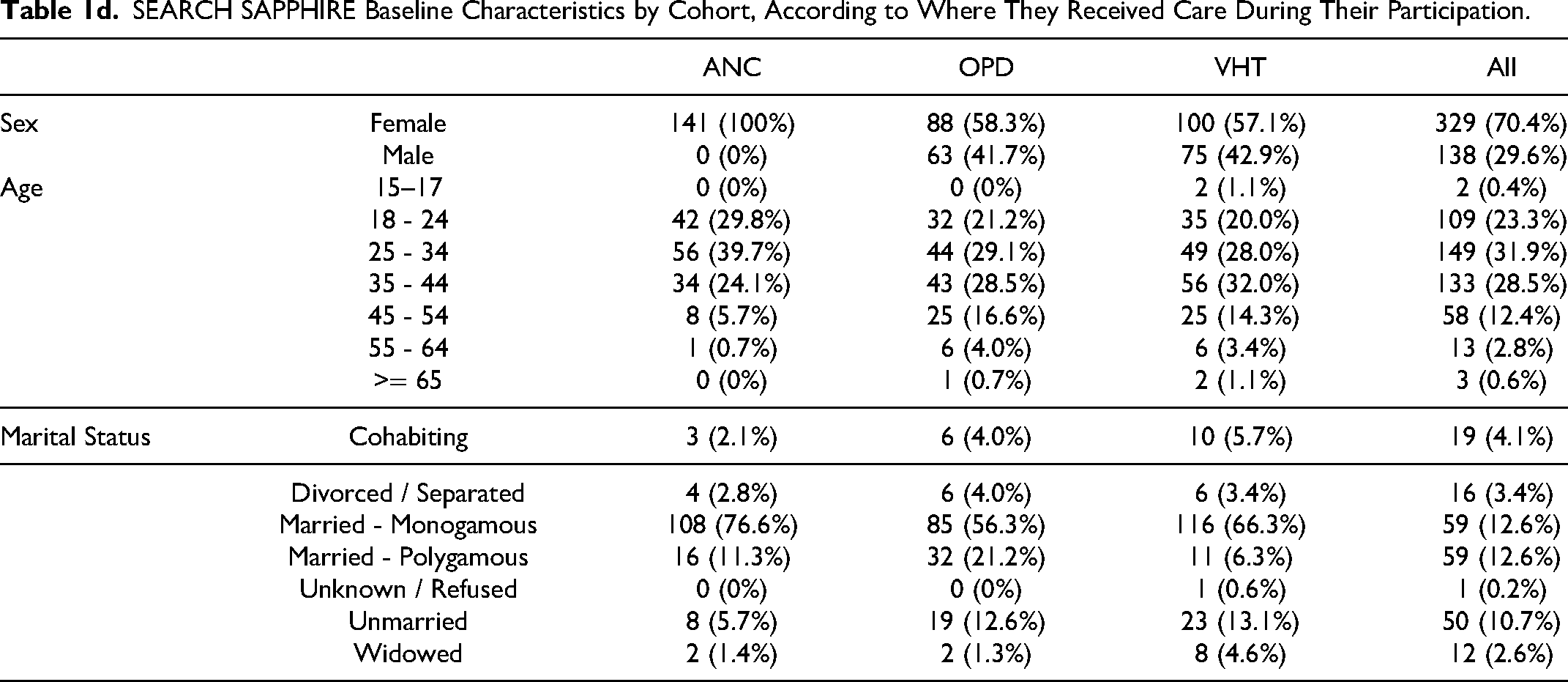
After coding, we used a thematic analysis approach45–47 to generate analytic memos across several broad themes. Among these themes, the challenges particular to people in polygamous families in the SEARH-Youth study stood out, but the data set was deemed too slim. We therefore subsequently made note of interviewees who mentioned being in polygamous families as we coded SEARCH-SAPPHIRE interviews. After determining from the SEARCH-SAPPHIRE interviews that we would have enough data for deeper exploration, we went back to the SEARCH-Youth transcripts and searched for the terms, ‘co-wife’, ‘co-wives’, ‘another wife’, and ‘polygamous’ to re-identify relevant transcripts. Thus, in these qualitative interviews, persons self-defined whether they were in polygamous relationships. The definition of polygamous is therefore not related to having multiple sex partners, but rather to socially understood, and sometimes legally defined marriages. For absolute numbers from the overall study, we used quantitative results from a questionnaire answered by all participants (or a household informant) that defined polygamous marriage as, “If a woman, this means your husband has other wives aside from you; if a man, this means you have more than one wife.” (See Tables 1b, 1c, and 1d.) We then read through all SEARCH-Youth and SEARCH-SAPPHIRE transcripts involving polygamous families (n = 27) in full, creating memos and identifying additional emergent themes. The final numbers are reported in Table 1a. The study was conducted and presented in accordance with the COREQ statement. 48 Here we report the major themes that were most salient for clinical prevention and care delivery.
Polygamously Married in-Depth Interview Participant and Provider Characteristics.
Ethical Approval and Informed Consent. The SEARCH-Youth and SEARCH-SAPPHIRE studies were both approved by the Kenya Medical Research Institute (KEMRI, 3834 & 4173), the Makerere University School of Medicine Research and Ethics Committee (REC REF 2019-014 & 2020-029), the Uganda National Council of Science and Technology (UNCST, HS 2542 (SEARCH-Youth)), and the University of California San Francisco (UCSF) Committee on Human Research (18-25703 & 20-32144). All study participants provided written informed consent to be interviewed. For our youth participants, both Kenyan and Ugandan national guidelines do not require parental consent for youth over the age of 12 and 14 respectively, to participate in research if they have a sexually transmitted disease, including HIV.
Results
In the SEARCH-Youth Study, among participants with HIV aged between 15 and 19 years nearly 60% were married, of whom 14% (6.7% of the total cohort) were in polygamous marriages. In the SEARCH-SAPPHIRE study, 12.6% of interviewed participants reported being in polygamous marriages at baseline, with 11.3% in the antenatal clinics, 21.2% in outpatient clinics, and 6.3% of participants served by village health teams reporting being polygamously married later in the study. (Tables 1b through 1d) These numbers are slightly higher than the country averages for both Kenya and Uganda. Apart from the outpatient clinics, however, the numbers were slightly lower than average in the counties where SEARCH recruited its participants in Kenya (Migori and Homa Bay), and about average for the Ankole subregion of Uganda, where the SEARCH studies were located, and where 11% of marriages are polygamous. 7
Likewise, the proportion of our transcripts mentioning polygamy was slightly higher than the quantitative data for the study as a whole would have suggested: for the SY study, 8.4% mentioned polygamy in the qualitative dataset to 7% in the quantitative; for SEARCH-SAPPHIRE: 14.9% of qualitative interviews mentioned polygamy compared to 12.6% reported in the quantitative data. In the ANC cohort, however, 31.6% of transcripts mentioned polygamy, more than twice the quantitative average for all trials within the SEARCH-SAPPHIRE study. In contrast, while we interviewed 46 providers, we only quoted four (=8.7%) with respect to polygamy, closer to the national (though not regional) averages for Kenya and Uganda.
The motives for taking or becoming a second wife varied. Participants mentioned a range of reasons, from a desire to have more children to the custom of wife inheritance should a married brother die. Below we describe some emergent findings as they pertain to providers, diagnosis, treatment, prevention and agency within polygamous families. We have summarized some of these themes in Figure 1.
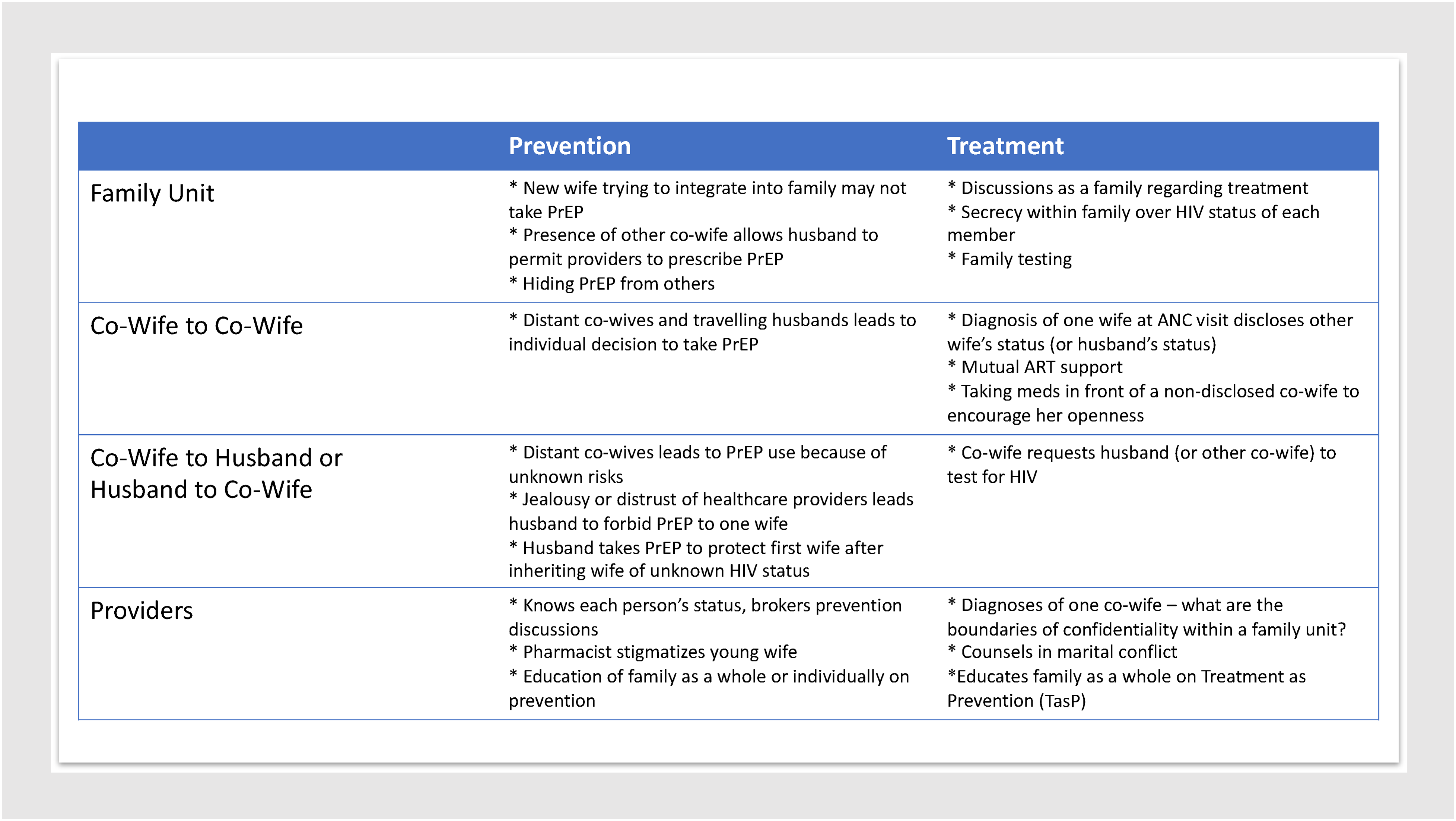
Thematic elements pertaining to treatment and prevention stratified by relationship.
Polygamous Families and Providers’ Roles
Due to the trusted position that providers hold, patients often reveal problems to them which go beyond the immediate biomedical issues which bring those patients into the clinic. This puts providers in the position of not only being responsible for educating patients about health and wellness, but at times also involves them as mediators in familial conflicts which affect individual patient health. Polygamous families present an additional element in such mediation, as the provider might mediate between a husband and an individual wife, co-wife and co-wife, or between both co-wives and the husband. “At times patients share about their social issues, especially marital conflicts, and I have to offer counselling. … Some also present to us a lot of family problems and I’m able to counsel accordingly. … Some have issues and they are very categorical that they don’t want to involve anyone else; in such cases I have to always find a way of helping because they have full trust in me and do not want anything like a referral. There is one case of a woman who used to engage in a lot of fights with a co-wife, but I managed to settle her through counselling and [they] no longer fight.” – 35 y.o. male provider (PrC Coordinator), Oyani (Kenya) “…Right now, she is married in a nearby homestead but at the time of enrollment, the same people where she is married, I tested the man and the first wife because she has been married as the second wife. I know their status but right now, I do not know their status whether they are still okay or not because we were done with enrollment [in the study]. She cannot continue taking PrEP in this homestead because she is newly married and the husband may perceive her as a cheat.” – 30 y.o. female provider (CO), Ogongo (Kenya)
While PrEP might be useful in this case, the challenge was knowing everyone's status at any given time. Similarly, a third provider, commenting on provider perceptions about PrEP and those who use PrEP, shared the following observation: “…we know that we have very young girls in Sena and they get married when they are still young. You find someone who is 15 or 16 years is already married as a third wife and they do not know the status of the husbands. When they come and tell you about it and you tell them about PrEP, they will say ‘I am willing to take that in order to protect myself.’ Unfortunately, you randomize and they fall in the control arm and you send them to the pharmacy and the pharmacist chases them away. You find that the next time they come they may already be HIV positive, and you could have prevented it.” – 46 y.o. female provider (PrC Research Assistant), Sena (Kenya)
HIV Diagnoses in Polygamous Families
The HIV diagnosis of one person in a polygamous family presents both opportunities and challenges for clinicians. Since both co-wives are part of the same family, some providers disclose to the other co-wife at the time of diagnosis, as they belong to the same family unit. This seems especially the case when the co-wife has accompanied the other wife on a clinic visit. “Interviewer (I): Have you shared your HIV test results with anyone? You’ve mentioned that your co-wife accompanied you to the hospital, right? … Can you tell me how you disclosed to her about your status? Participant (P): When I was tested, she suspected something and then she went to the nurse and asked. Then the nurse showed her. I: She showed her your results. P: Yes, then she also requested to be tested, though she cried at first. Then she accepted to be tested. I: Uh now, the nurse is the one who disclosed to her about your status, and why was she suspicious? P: She was suspicious because when people were tested, mine was put aside. Then she suspected that there is something wrong.” – 19 y.o. female, second wife, Oyani (Kenya) (Intervention)
At other times, one co-wife discovers her HIV status on her own, leading her to surmise the source of her infection was from the husband, possibly via the other co-wife. This HIV-status discovery can happen when a second wife comes to her first ante-natal visit, where expectant mothers are routinely tested for HIV. “He has always told me that he is HIV-negative but would not agree to come for couples testing. I believed that he was negative because our first child whom I bore him while still in school was negative, which was in 2014. If he would have been positive at the time, my child would have been born positive and I would have tested positive when I went for testing because I used to test regularly. I still do not know [his status].” – 20 y.o. female, second wife, Kitare (Kenya) (Control) “I learned of my status last year. You know I have lived with them [my husband and co-wife] before; when I was still going to school. Then I went back to school in 2016 to 2019. When I was done with school in 2019, I stayed home and that is when I conceived. So at some point, I had symptoms of malaria and when I went to the clinic, I was first tested for HIV and the results came back positive.” – 20 y.o. female, second wife, Kitare (Kenya) (Control)
In this instance, the wife learned of her HIV-status at the ante-natal clinic where she went during her second pregnancy, thinking she might have malaria. She was able to estimate the time of her seroconversion and deduce that she was infected by her husband. The chain of transmission – whether the husband was infected by the co-wife, and then infected the participant, or whether the husband infected both wives – remained unclear, as neither had disclosed their status to her, the husband refused to go for couples’ testing, and the participant was left to rely on their word (or lack thereof).
In some locations, couple's testing is an option. For polygamous families, this means testing all spouses together: “I personally come from a polygamous family – we are two wives. I may be aware of my partner's status and in fact, we have all tested together the three of us, but this does not give me the leeway to monitor each one's movement and any of them may mess up [have an extra-marital affair] at any time. That is why I opted to use PrEP.” – 24 y.o. female, (wife order unknown), Sena (Kenya)
Treatment and Support Among Co-Wives and Husbands
The addition of another spouse to a previously dyadic relationship means not only the addition of two more relationships (husband to second wife, co-wife to co-wife), but also the possibility of either greater support or increased challenges to ART adherence due to secrecy and suspicion.
After diagnosis, co-wives can become excellent household supporters of one another in adhering to treatment and engaging in care. “I: Please tell me about your decision to share your test results with your husband, when you came from the hospital, how did you disclose to him? P: We [both of us wives] went and sat him down then we narrated everything that transpired at the hospital – that we were tested HIV-positive. Then we were supposed to be on care though I was the one who accepted to start care immediately. He then said that, ‘There is no problem it has happened, just take your medications.’” – 19 y.o. female, second wife, Oyani (Intervention) “I am free to disclose my status; and even when we had a meeting here [when] they asked a question about HIV I had to raise my hand because I was happy because of how far I have come, and I would answer the questions. I am free – even my spouse cannot disclose to you. And looking at my co-wife and how she looks…I cannot tell where she receives her HIV care but I know they have the infection but they would not wish for you to know. I am free and I tell her ‘I am on HIV care, and God has been good because He gave me HIV-negative children.” – 35 y.o. female, second wife, Sena (Kenya)
In this instance, one co-wife was free with disclosing her HIV status and sought to encourage the co-wife who had not yet disclosed to her, either to make her comfortable in disclosing or to encourage her to engage in care, though apparently without much success.
Even when the patient's husband supports a wife's medication use, he may not admit his own status: “I used to urge him to come for HIV counselling and testing but he denied. I then consoled myself that maybe they (my husband and the eldest wife) both knew of their status since there was a time my co-wife fell seriously sick. She became completely emaciated and when I asked the husband, he told me that it was just a stomachache. She was taken to Migori. I just consoled myself that they might have known what she was suffering from and I could also not just silently suffer after knowing my status. After knowing my status, I was directly enrolled for care without considering my husband and co-wife's status.” – 21 y.o. female, second wife, Oyani (Intervention)
Prevention, Negotiations Among Co-Wives, Husbands, and Larger Familial (In-Law) Contexts
Similar dynamics to treatment support or discouragement come into play where HIV-prevention is concerned. For example, mutual support can be seen when women come for prevention options, and the other wife's presence can smooth over any potential domestic violence that might arise from the husband's suspicion the wife is carrying on an affair with the provider or CHV (community health volunteer), an unfortunately not uncommon misperception. “I had a challenge because of my spouse, he is still young like the provider and whenever you leave he thinks you have gone to hangout; and if anyone that he does not know comes, he thinks that they are your partners. The challenge I had is that sometimes the provider would take me home on a motorbike but when he leaves my spouse would start a fight saying they are my friends. I was bothered by that the first time he took me home. Luckily enough, when we got home we found my co-wife and my spouse also came home and into the house and saw me. The provider then introduced himself, ‘I work with KEMRI-SEARCH and I have enrolled this lady into the study, so please feel free.’” – 35 y.o. female, younger wife, Sena (Kenya)
In this instance, the husband's jealousy seemed to have been attenuated because the other wife was also present when the CHV dropped the participant off. This gave the CHV time to introduce himself and provide an opening for discussion around HIV prevention.
HIV prevention, when wives live in proximity to one another, can create challenges, but notably, our data suggest that when co-wives live apart, the decision to engage in preventive care is easier. This extends not only to the co-wife's individual decision, but the husband may also choose to initiate PrEP: “I: What reason did you explain to him [your husband] about your PrEP use? P: When he cornered me, I told him that ‘I may trust you as my husband but I may not trust my co-wife since she stays away’ and he felt satisfied with that.” – 24 y.o. female, (wife order unknown), Sena (Kenya)
The risk and prevention-literacy of husbands can also create awareness of HIV-prevention options for their wives, as in the following example: “He [my husband] is knowledgeable about the risk of HIV because there was a time I was staying with him in Nairobi while the other wife was here at home, and then he came with those medications [PrEP]. By then I was still not aware about them [HIV prevention medications]. Then I was like ‘Are you on care?’ then he replied, ‘Your co-wife refused to go for HIV testing and I could not risk my life because I did not know her status.’” – 33 y.o. female, (wife order unknown), Sena (Kenya)
In the above instances, the spouses are familiar enough with one another that they can have these discussions, and notably the husbands who take an active role do so in the context of PrEP rather than in the context of HIV treatment. A different challenge in initiating prevention options like PrEP, however, comes when a second wife is new to the marriage. Thus, a man who inherited a wife responded to an interviewer's question,
“I: Why do you think you have never opted to open up to your inherited wife about your PrEP use? P: Not in bad faith, we are not that old in that relationship and because she has never opted to disclose to me about her status, I have to take all the precautions. I have used the drugs while with her and so I assume she knows that I am using those drugs.” – 39 y.o. male, Sibuoche (Kenya) “I have had a case with one of our participants. The client I told you about, she stays with the guardian who is a maternal aunt and currently she has been married to a nearby homestead as a second wife. Once she was ready and willing to take PrEP, but the mother [ie mother-in-law] did not want her to take PrEP because she perceived her to be sleeping around. … She cannot continue taking PrEP in this homestead because she is newly married and the husband may perceive her as a cheat.” – 30 y.o. female provider (CO), Ogongo (Kenya) “I: Do you think your husband thinks he is at a high risk of contracting HIV or he is not worried at all? R: I just told him I would guard my health; if he wanted to die then he dies alone. But he didn’t seem bothered. I: Does he think he is at risk of HIV or…? R: He knows he is at risk because at times he says cheating leads to HIV. And [he] continues to say he worries about even getting it from his wives but I tell him he doesn’t have to worry about me but maybe the wife in the village because she may cheat on him.” – 29 y.o. female, (wife order unknown), Bwizibwera (Uganda)
On the other hand, one participant who enrolled in the dynamic choice prevention arm of the SEARCH-SAPPHIRE study acknowledged that sero-difference among the partners is a potential source of problems – not because one wife is HIV-positive and the other HIV-negative, but rather because the problems would arise if the husband should seroconvert. P: Yes, at some point I may seroconvert. As I said earlier, I am a polygamous man, and above all one of my wives is HIV positive. If I happen to seroconvert, the other wife who is still HIV negative might tend to be away from us [ie refuse to be intimate or leave the marriage]. I think that is the challenge, which I may foresee. I: Why do you think that the other wife would also turn away from the two of you in case you seroconvert? P: She would be like, “My husband is cheating on me [ie deceiving me] that he is using protection yet he is having unprotected sex with my co-wife.” – 37 y.o. male, Ogongo (Kenya)
Agency and Individual Engagement in Care
Overall, our participants exhibited very realistic attitudes towards the sexual behaviour of their co-wives and husbands. Many acknowledged that despite one's best efforts, one can slip up – or can knowingly continue to engage in high-risk behaviour. “I decided to join this research … because of my husband. I have a husband that has another wife and yet he goes to different places far away to work because he is a builder. In that case, I don’t trust him; hence I joined to get these [HIV prevention] services. Six months after joining the research, my husband had a long journey and went to Sudan so I also stopped the medication but would come in for tests.” – 28 y.o. female, second wife, Itojo (Uganda) “They first offered me HIV testing and the result came out negative. Then they requested me to enroll in PrEP so that I may be able to prevent HIV infection. I said, ‘No, I cannot trust my husband wherever he is, just give me PrEP to take. They might help me in case I could be exposed.’ [Later]: My husband is away in town yet I am at home, so I would need to plan well. When he is away, I may not know how many partners he is having out there.” – 33 y.o. female, (wife order unknown), Sena (Kenya)
Agency, however, was sometimes constrained by fear of the husband and the power that in-laws wield, especially over new wives. In these cases, the wife may enlist the support of a CHV to help persuade her husband or mother-in-law to let her take PrEP. At other times, the husband may himself be curious about what the CHVs have to say: “I was not afraid at first, but my concern was only one: because I enrolled into the study without my husband's knowledge – so I thought he might feel that I am having an affair outside marriage and that is why I am taking PrEP. However, when I shared with him, he welcomed the idea and he was like, ‘Why didn’t you tell the providers to come home?’ Then I told him to relax; time will come and they will come home. By good luck, after some time, they came home and he was around as well. When they informed him about the study, he was impressed and after explaining everything to him, he agreed to enroll in the study. Since that time, we have been interacting very well with the providers.” – 42 y.o. female, second wife, Oyani (Kenya)
Discussion
The findings of our two related studies in rural Kenya and Uganda, where nearly one-fifth of participants ranging in age from 15 to over 65 were in polygamous marriages, highlight that polygamy is a lived context that clinicians are navigating in their provision of HIV treatment and prevention and sexual and reproductive health services.
We found that an HIV diagnosis of one person in a polygamous family presents particular challenges and opportunities for clinicians regarding both treatment and prevention for the entire family unit. Challenges to HIV prevention and care arise in four main areas: ascertaining risk, partner testing, disclosure, and managing family dynamics. Positive influences include trust among co-wives and between husband and co-wives, openness about status, and sharing health knowledge.
Most literature surrounding polygamy and HIV is epidemiological in nature; we were unable to find much qualitative literature about the clinical implications of HIV care for polygamous families.12,22 Similar to findings by Nwoye, we discovered that co-wife relationships are not necessarily competitive or acrimonious, but can also be supportive; this contrasts with both Bove and al-Krenawi, who found more competitive relationships in a variety of sub-Saharan contexts and among Bedouin Arabs in Israel and Jordan.34,36,49 This may perhaps stem from the custom of wife inheritance, a common form of polygamy in our region, being ‘interventive’ and designed to support the widow, rather than the ‘affluent’ polygamy that signals a husband's economic status. 36 Like Bove, however, we did find evidence of ‘cooperative conflict’ and poor communication, which raises the risk of HIV transmission or non-adherence. Several articles noted discussions within the Luo community over whether levirate marriage is a cause and consequence of the spread of HIV, or whether it has the potential to check it.11–13 In our admittedly small sample, Luo participants expressed interest in HIV preventive therapy specifically because of this possibility – in effect, preserving the custom while mitigating its potential health risks. 11 Likewise, our informants did not indicate whether their decisions resulted from changes in broader risk communication targeting the Luo practice of wife inheritance in particular. 13 Since our qualitative sample did not include any polygamous men from Uganda, we cannot confirm or deny Muldoon's findings around higher sexual risk behaviour among that population. 22 However, our findings do concur with Bove's argument that strengthening relationships within polygamous families may lead to lower HIV transmission or defaulting from HIV care – findings which have been known since the 1990s, though in a very different HIV treatment and prevention landscape. 49
Recommendations. The findings have important implications for clinical practice in populations who practice polygamy similar to what we report for Kenya and Uganda. Providers find themselves responsible for HIV education and general mediation between family members, including between multiple co-wives, between the husband and co-wife (or husband and both co-wives), and occasionally between a co-wife and her in-laws. In this context, providers represent a credible and trusted outside source for communication around HIV. 13 Several recommendations for providers follow, concerned with risk assessment, family (partner) testing, and ARV treatment.
Ascertaining risk is a central clinical concern for providers working with patients from polygamous households. Our data show different prevention dynamics when co-wives live together compared to when they are far apart, with families where co-wives live distant from one another more accepting of taking up PrEP (and justified in so doing in the eyes of their spouses). For husbands with more than one wife, providers can ascertain the husband's reasons for taking on an additional wife -- for example, whether the wife was inherited, or whether the husband wanted more children -- and then guide the conversation around HIV risk and prevention from there. Importantly, assessing the husband's knowledge not only of each wife's status, but also their character, and his relationship with each separately and together, can inform these discussions. The provider may also want to make available risk-assessment counselling for all members of the polygamous family to attend at the same time. Such counselling might also be useful in the case of a younger second wife entering into a larger family unit where no one has disclosed their status to one another.
Partner testing involves different dynamics where polygamous families are concerned. How partner testing is conducted follows from the larger dynamics around decision-making within the family, which providers should seek to discover. In families where each member makes their own medical decisions, partner testing as a family may not be warranted, while in households where collective decision-making is the norm, all partners may want to test together. Since family medical decision-making sometimes includes in-laws, which we have discovered from both transcripts and ethnographic work, whether the mother-in-law is responsible for the daughter-in-law's medical decisions or not can be an important factor to consider in these contexts. 50 In this way, partner testing can also account for the different types of polygamous marriages (e.g., inherited, arranged, same compound, distant), and crucially, the personalities involved – since some husbands are discerning and peaceable, and others are jealous and coercive. Similarly, some co-wives view one another as sisters, others as rivals.
Providers also navigate the ethics of disclosure and confidentiality. Participants reported breaches of confidentiality when nurses or VHTs disclosed their HIV status to co-wives. Disclosing HIV status to other co-wives may or may not be justified, given the particular contours of confidentiality within family units and cultural groups. At the very least, the provider is positioned to help broker disclosure conversations with the people most involved. According to the Kenyan testing guidelines, the provider should give the client a chance to disclose their status to their partners and may assist in disclosure. 51 The providers are only allowed to disclose to the other partners when the client has refused to disclose, and the other partners are at risk. Providers should plan with clients how much time is needed to disclose, or what patient needs exist around assisted disclosure. This can extend to making a plan to facilitate partner or co-wife testing (when the relationships permit) or suggest co-wife testing at ANC visits if a co-wife usually accompanies the new mother.
Providers can leverage positive co-wife relations to encourage treatment and adherence. When one member of the family is open about their status, initiation onto PrEP or ART adherence seems smoother. For husbands who are supportive of their wives and care for their health, openness about taking PrEP and encouraging one another to do so, especially when co-wives do not live near one another, helps maintain HIV prevention. In supportive co-wife homesteads, providers can leverage those relationships to encourage the co-wives to attend the clinic together or to pick up medications for one another. If possible, providers can plan slightly more time for participants from polygamous relationships to uncover the degree of support co-wives provide one another, and to decide whether this warrants follow-up with a VHT when VHTs are available and trusted. Since VHTs are more familiar with the issues people have in their homes, a VHT could also assist with disclosure, testing, counselling, and treatment encouragement. The challenge is whether people would be comfortable with the VHTs themselves and discussing these issues with them.
Hand in hand with greater transparency, sharing health knowledge also improves care. Importantly, trusting the self-knowledge of one's patients is essential: they often know what their husbands or co-wives may be up to, and they can discuss what options are realistic given their living situation and household relationships. Providers are sometimes called upon to manage these family dynamics, especially as HIV prevention meets hierarchical relationships between younger co-wives getting established in the family, older co-wives or mothers-in-law who may have considerable influence on the younger co-wife's medical decision-making, and potential intimate partner violence. Long-acting injectable PrEP may provide a useful workaround in some cases, as pill bottles and tablets would not be a source of disclosure. 52 Probing a bit deeper to come up with workable plans, co-creating a ‘therapeutic agenda’ may pay dividends for the entire family.
From clinic to community. Shifting from a clinic-specific perspective to community-wide public health, our findings support the movement away from ABC (Abstinence, Be Faithful, Condoms) messaging that was common in Kenya and Uganda until recently. Not only were these messages especially directed to women, rather than men (thus neglecting half the population), people did not always understand what ‘be faithful’ meant, 25 especially in the context of polygamous families. These messages further fell flat in areas where monogamy was not the default option for sexual and domestic partnerships. Strangely, in the 2005 version of Uganda's HIV testing guidelines polygamous families were mentioned, but the most recent versions leave them out.53,54 Additionally, condom use can be problematic in marriages, especially when men marry a second wife in order to have more children. Yet most people are still not aware of prevention apart from condoms; nor are they often aware of the husband's status, or their co-wife's status.
In fact, one important finding regards the difference in HIV-prevention and HIV-treatment initiation and disclosure by husbands. We discovered that HIV-negative men in polygamous relationships seemed willing to take HIV-prevention medications, while co-wives reported that husbands they suspected of being HIV-positive seemed unwilling either to discover their status (through testing), take medication to treat HIV, or have honest discussions about the topic. Since men are a key vector in HIV transmission in this region, it appears that sensitizing married men specifically to Treatment as Prevention (TasP) remains needed.
Our participants have highlighted additional opportunities for broader community messaging. Cultural leaders such as imams and community elders can promote a normative standard of knowing everyone's status in a polygamous family. They can persuade the community that it is the responsibility of the whole family to protect one another and foster their health, as has been done with voluntary medical male circumcision among the Luo, and with pre-marital HIV testing among Muslims in Northern Nigeria.55–57 Respected community leaders can also discuss HIV prevention and treatment with mothers-in-law so as to minimize or eliminate PrEP stigma towards their daughters-in-law. As people who can speak to the community and family's values, such leaders are also well-positioned to encourage all partners to know their status and be open with one another about it. They may also play a role in normalizing conversations about testing before marriage and discuss HIV treatment and prevention within polygamous families. Such encouragement may help people engage in either treatment or prevention practices as necessary to ensure their own health, and when children are desired, the birth of HIV-negative children, especially given the recent commitment of African leaders to end HIV in children by 2030. 58
Limitations
These study findings have strengths and limitations. The two studies were not designed to explore the emergent findings concerning polygamy that are presented here, thus our sampling design did not include purposive sampling for polygamous families; the limited number of Ugandan relative to Kenyan participants and men relative to women with findings related to polygamous marital status precluded more detailed analysis by region or gender. In tandem, we did not interview cultural authorities or leaders in this research, and their perspectives may have contributed useful insights. However, a strength of our research was that the Kenya-based investigators and research team shaped this study so that local voices and perspectives guided study design and implementation. Furthermore, including both women and some men in polygamous relationships as well as the voices of providers adds depth and allows for an exploration of challenges and opportunities for engaging polygamous families in HIV prevention and care. Our data drew from two different studies with two different ways of reporting statistics due to the different foci of each study. The two datasets therefore could not be seamlessly integrated for stratification. This has prevented us from making more granular analyses. However, the general thrust of our findings, which are directed towards clinicians should be sufficient to highlight the challenges particular to polygamous families when it comes to HIV treatment and prevention.
We quoted only two polygamous husbands, due in part to the proportion of men in each study: while 18% of SY and 30% of SS is male (Table 1b), only three male participants in the entire SY trial were polygamous at baseline (Table 1c). Since we are reporting an emergent finding, we had not purposefully selected to interview those three men. Similarly, while 59 people in SS reported being in a polygamous marriage, male/ female stratification within that category is unavailable (Table 1d).
SEARCH Youth by Arm.
SEARCH Youth by Monogamy Versus Polygamy Status (Married Only).
SEARCH SAPPHIRE Baseline Characteristics by Cohort, According to Where They Received Care During Their Participation.
The IDIs were conducted in rural areas where the prevalence of HIV is higher than the regional estimate; its intersection with polygamy is thus specific to our informants and these findings are not generalizable to all contexts. Nonetheless, while it is possible that those in urban settings would express different sentiments, just over a quarter of the population in Kenya and Uganda is rural, making the experiences of this group especially meaningful for offering future programs. 59 Future research aimed at better including polygamous families in HIV prevention and care could benefit from engaging community leaders to gain insight on how to harness cultural practices and values and better integrate polygamous families in HIV services.
Finally, two findings warrant specific study in their own right. The first is the aforementioned discrepancy between husbands being more willing to take HIV-prevention medication than HIV-treatment medication, and to have frank discussions around health with their younger co-wives. The second picks up on the salience of wife order, which our findings suggest may influence HIV prevention discussions and initiation, but for which we really do not have enough data to draw even tentative conclusions. In such research, if comparing experiences between monogamously and polygamously married women, it would be useful to compare the experiences of a first wife to a monogamous wife, then compare the experiences of a second wife to a monogamous wife separately, or to focus exclusively within the polygamous family itself, to compare the experiences of the first wife with those of latter wives.34,60,61
Conclusion
In some areas of intensified HIV prevention and treatment outreach, polygamous families are common, accounting for up to 20% of the family population. Polygamous partners have very different HIV prevention and treatment needs compared to monogamous couples. When more partners are involved, chances for HIV acquisition and transmission increase. Importantly, because polygamous families present with many variations in internal dynamics, just as monogamous and single-parent families do, no single approach is appropriate for clinicians to take. Yet, providers are well-positioned to provide tailored care to individuals in complex family units, as trusted authorities on health and wellness for polygamous families.
Footnotes
Authors’ Contribution
JJP led analysis and with AO and SAG wrote the manuscript, with contributions to analysis from CA, FA, AO, TOA and LO. TOA, CA, FA, AO and LO conducted data collection, and along with JJP conducted curation and coding of the transcripts. MRK, MLP, DH, LB, TR, JA and DVH conceived the parent study, provided field-based oversight and revised critically. CSC conceived the larger qualitative project. All authors have read and approved the final version.
Acknowledgements
The authors gratefully acknowledge the contributions of study participants and communities, advisory boards, and the contributions of all team members in the SEARCH Collaboration to this research. We thank the Ministries of Health of the governments of Kenya and Uganda, the Infectious Disease Research Collaboration (IDRC), and the Kenya Medical Research Institute (KEMRI) for their collaboration.
Data Availability Statement
The data on which the findings of this study are based can be made available on request from the corresponding author. The data are not publicly available due to ethical considerations around confidentiality and HIV status.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Research reported in this manuscript was supported by the U.S. National Institute of Allergy and Infectious Diseases (NIAID), the National Heart, Lung, and Blood Institute (NHLBI), and the National Institute of Mental Health (NIMH) and co-funded under award number U01AI150510 (Havlir). Funding for the SEARCH-Youth study was provided by the National Institutes of Health/National Institute of Child Health and Human Development, grant number UG3 HD96915-01.The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Ethical Approval and Informed Consent
The SEARCH-Youth and SEARCH-SAPPHIRE studies were both approved by the Kenya Medical Research Institute (KEMRI, 3834 & 4173), the Makerere University School of Medicine Research and Ethics Committee (REC REF 2019-014 & 2020-029), the Uganda National Council of Science and Technology (UNCST, HS 2542 (SEARCH-Youth)), and the University of California San Francisco (UCSF) Committee on Human Research (18-25703 & 20-32144). All study participants provided written informed consent to be interviewed. For our youth participants, both Kenyan and Ugandan national guidelines do not require parental consent for youth over the age of 12 and 14 respectively, to participate in research if they have a sexually transmitted disease, including HIV.
