Abstract
The primary goal of medical care during the pre-antiretroviral therapy (ART) era was to keep persons living with human immunodeficiency virus (HIV) alive, whereas since the advent of ART, the treatment objective has shifted to decreasing viral loads and infectiousness while increasing CD4+ T-cell counts and longevity. The health crisis, however, is in preventing and managing multimorbidity (ie, type 2 diabetes), which develops at a more accelerated or accentuated pace among aging persons living with HIV. Relative to the general population and age-matched uninfected adults, it may be more difficult for aging HIV-positive persons who also suffer from multimorbidity to improve negative lifestyle factors to the extent that their behaviors could support the prevention and management of diseases. With recommendations and a viable solution, this article explores the impact of negative lifestyle factors (ie, poor mental health, suboptimal nutrition, physical inactivity, alcohol use) on the health of aging individuals living with HIV.
Plain Language Summary
This literature review highlights negative lifestyle factors experienced by aging persons living with HIV, including poor mental health (i.e., depression, cognitive decline), poor nutrition, physical inactivity, alcohol use, sexually transmitted infection, poor sleep quality, and treatment noncompliance as promoting diseases (e.g., type 2 diabetes, hypertension, obesity, cardiovascular disease) and early death. A possible solution to decreasing or eliminating negative lifestyle factors in the aging HIV-positive population is to develop disease prevention-focused teams in clinical and community settings. However, if team development is impractical, primary healthcare providers should routinely assess and monitor lifestyle factors (i.e., cognitive decline) and, if needed, provide appropriate referrals to licensed or certified specialists (e.g., psychologist, clinical dietitian, medical exercise specialist, or health coach).
Introduction
In 2018, more than half (51%) of people in the United States (U.S.) with diagnosed human immunodeficiency virus (HIV) were aged 50 and older, and approximately 1 in 6 HIV diagnoses in 2018 was made in individuals older than 50 years. 1 In 2018, an estimated 1.2 million Americans had HIV, 379,000 of whom were 55 and older. 1 Owing to the advent of antiretroviral therapy (ART), the projected life expectancy of HIV-infected persons has increased. 2 However, in a study linking HIV, ART, and longevity, Althoff et al 2 reported that, even though there was an improvement in life expectancy in persons living with HIV on ART in the U.S. and Canada, based on treatment onset at the age of 20, there were still life-expectancy disparities. In the 2012−2015 period, the life-expectancy disparity in HIV-positive Black versus White men who have sex with men (MSM), respectively, was an additional 50.9 years and 60.3 years; and during that same timeframe, life-expectancy disparity in persons with injection drug use (IDU) versus non-IDU history, respectively, was an additional 39.9 years and 50.3 years. Importantly, Althoff et al 2 further noted that a 20% reduction in drug- and alcohol-related mortality would have the greatest life-expectancy benefit for Black MSM, White women, and people with a history of IDU. Other studies have identified the potential roles of structural racism and stigma, which can reduce access to and retention in health care in the unrelenting racial disparities in Black MSM as well as disparities among those with IDU histories. 2
Irrespective of increased life expectancy and aside from managing HIV, individuals living with the infection are also confronted by and struggling with multimorbidity, especially as they age. 3 Available evidence indicates that multimorbidity—ie, cardiovascular disease (CVD), type 2 diabetes (T2D), and hypertension (HTN)—in aging individuals living with HIV may be accelerated or accentuated by a nexus among HIV infection, biological aging, and immunosenescence.3,4 Many—often mutable—psychological and behavioral lifestyle factors associated with diseases may accelerate or accentuate disease conditions in aging HIV-positive persons. Figure 1 illustrates the interactions between negative lifestyle factors, aging persons living with HIV, and morbidities. The overarching purpose of this article is twofold. First, with recommendations, its objective is to explore the extent to which lifestyle factors, including mental health (ie, depression and cognitive decline), nutrition, exercise (or physical activity), smoking, alcohol use, sexually transmitted infections (STIs), and treatment adherence are explicably linked to the health and well-being of aging individuals living with HIV and multimorbidity. The second objective of this work is to provide a viable solution for mitigating or eliminating those negative lifestyle factors through lifestyle-focused care provided by a client-centered, preventive-clinical, interdisciplinary team.
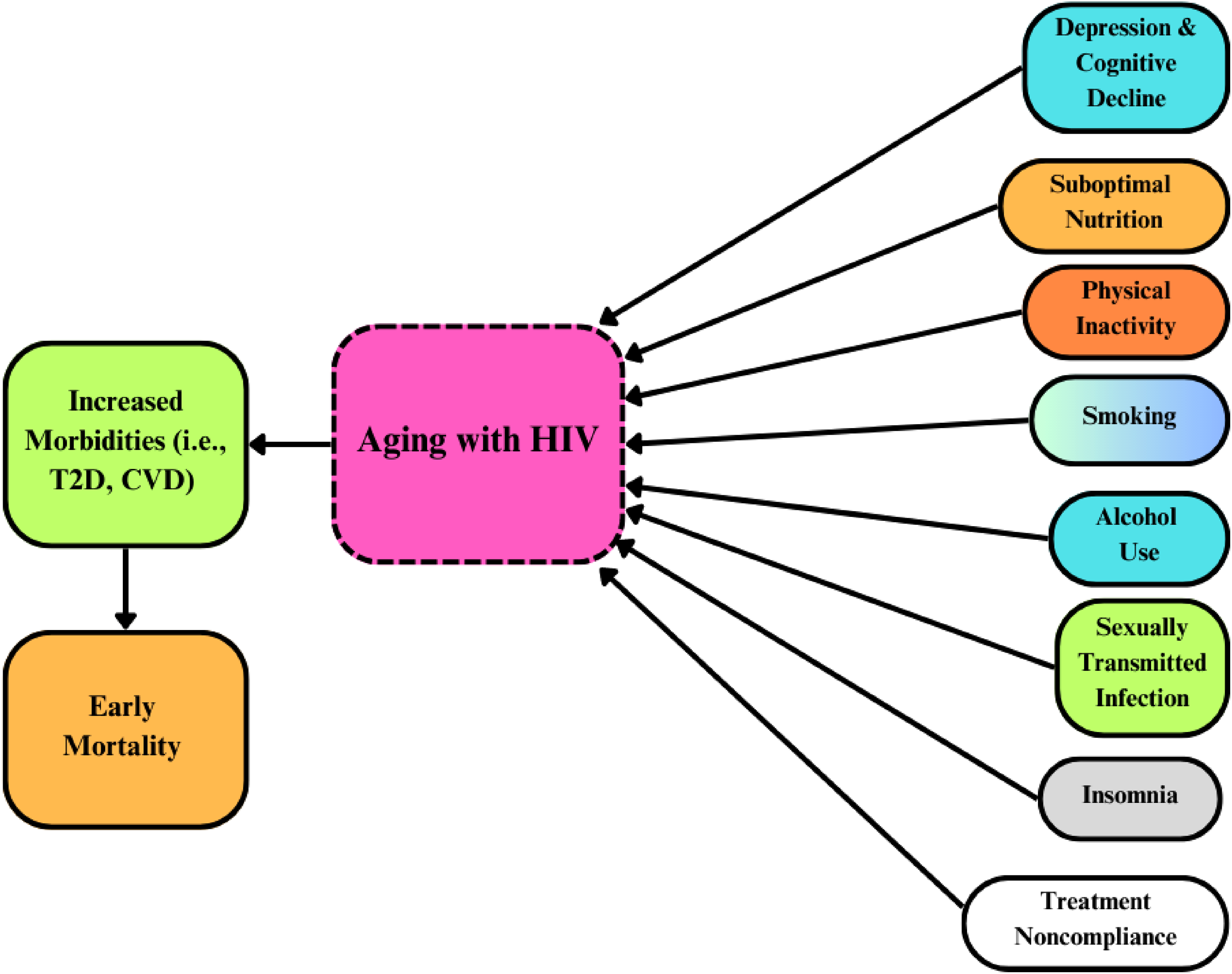
Negative lifestyle factors influence increased morbidity and early mortality in aging persons with HIV. In aging persons with HIV, increased morbidities (ie, T2D, CVD) and early mortality are influenced by negative lifestyle factors such as poor mental health (ie, depression and cognitive decline), suboptimal nutrition, physical inactivity, smoking, alcohol use, sexually transmitted infections, insomnia, and treatment noncompliance.
Mental Health
In the U.S., the encumbrance of depressive symptoms and depression among persons living with HIV is approximately 25%, three times higher than HIV-uninfected persons. 5 As burdensome as depression, 35–50% of adults with HIV in the U.S. exhibit some form of HIV-associated neurocognitive disorder (HAND) ranging from mild asymptomatic neurocognitive impairment to HIV-associated dementia. 6 Depression and cognitive decline in older HIV-positive individuals elevate the probability of increased morbidities (ie, T2D and HTN),7,8 promote negative lifestyle behaviors (ie, treatment noncompliance, smoking, substance use),9–11 as well as fuel an early death. 12
Depression and cognitive decline may not only be a bidirectional sequela for each other but synergistically continue to deteriorate the overall physical and psychological health of older persons living with HIV and multimorbidity. 13 It is crucial to highlight that psychosocial challenges (ie, loneliness and isolation) among HIV-positive older adults with multimorbidity have the potential to mediate and moderate depressive symptoms and depression. As negative manipulators of the health and well-being of older persons living with HIV and multimorbidity, the following discussions explore (a) depression and its connection to psychosocial issues and (b) cognitive decline.
Depressive Symptoms, Depression, and Aging with HIV
Markedly, depression and other psychiatric conditions can overlap with HIV and morbidities in aging persons living with HIV. 14 For example, Chhatre and colleagues 14 explored the intersection of physical (HIV) and mental health (psychiatric) conditions and medical comorbidities in a sample of 218,133,630 (weighted) with .18% (n = 39,264,053) diagnosed with HIV. The researchers identified those with and without psychiatric disorders (ie, mood/depressive disorder, anxiety, post-traumatic stress disorder [PTSD]). The six medical comorbidities included pain, cancer, cardiovascular disease, substance disorders, metabolic disorders, and infectious diseases. Participants were divided into four groups: HIV-positive, psychiatric comorbidity (Group 1); HIV-positive only (Group 2); psychiatric disorder only (Group 3); and non-HIV or psychiatric disorder (Group 4). The average age per group was 48.3 ± 11.7 (Group 1), 46.5 ± 12.9 (Group 2), 48.7 ± 16.9 (Group 3), and 45.9 ± 17.7 (Group 4). Forty-three percent of the HIV-positive group had psychiatric comorbidities, relative to 19% of the non-HIV group. Half of the HIV-positive psychiatric disorder group had at least one medical comorbidity, with pain being the most frequent (23%). Overall, the association between individual medical comorbidity and group status demonstrated that the HIV-positive, psychiatric comorbidity group had the highest odds of presenting with the six types of medical comorbidity.
All-cause mortality in aging persons living with HIV has been identified to be correlated with depressive disorders. For example, So-Armah et al 15 examined depression and all-cause mortality in 129,140 HIV-infected and HIV-uninfected U.S. veterans who participated in the Veteran Aging Cohort Study (VACS). The subjects were over 50; 30% had HIV, and 24% died over a median follow-up of 11 years. Among HIV-positive individuals, there was a 24% increased mortality risk associated with elevated depressive symptoms.
Along with increasing morbidities and mortality among older individuals living with HIV and multimorbidity, depression coalesces with psychosocial issues such as social isolation, as well as the stigma attached to aging with HIV.16–18 Yoo-Jeong and colleagues 18 investigated the extent to which loneliness mediates the effect of HIV-related stigma on depressive symptoms in a sample of 148 older (≥ 50 years) persons living with HIV. The analysis demonstrated that loneliness mediated the association between stigma and depressive symptoms. In another study linked to depression and psychosocial issues, Marg and colleagues 19 assessed successful aging among older persons living with HIV (n = 62) compared with older individuals without HIV (n = 48) in Palm Springs, California. The prevalence of two or more comorbid conditions across the sample was 59.1%, with persons living with HIV being more likely to report depression (35.5% vs 12.5%). Even though a statistical correlation or prediction between depression and loneliness was not assessed, there was a significant association between not working or volunteering and feelings of isolation among aging persons living with HIV, whereas, with older persons living without HIV, there was a significant affiliation between feelings of isolation and not living with someone.
Furthermore, in Marg and colleagues’ findings, 19 there was no noteworthy difference between reported feelings of isolation among persons living with HIV and persons without HIV, which could have been associated with most HIV-positive individuals having disclosed their HIV status to friends and family, indicating relatively high social support among the study sample. Overall, findings suggested that older persons living with HIV experienced successful aging to a similar degree to their peers without HIV. It is important to mention, however, that significant support networks were available for HIV-positive individuals in Palm Springs, including Let's Kick AIDS Survivor Syndrome and Positive Life. Still, the results exemplified that depression and social isolation remained important issues threatening successful aging among older persons living with HIV.
Screening for depression should be a common practice in both clinical and community settings serving aging persons living with HIV and multimorbidity, and, if needed, treatment should be provided or appropriate referrals made. Healthcare providers should ensure adequate social support networks for aging persons living with HIV and multimorbidity, such as community-support organizations. Healthcare providers also must accept assessing and treating depressive symptoms and depression as essential components of providing comprehensive care, whereby supporting successful aging in older persons living with HIV and morbidities.
Cognitive Decline, Aging, and HIV
Cognitive decline is hastened by a deficit in cognitive (or brain) reserve, reflecting the amount of damage that neurons (nerve cells) and neuronal connections can absorb without compromising the support for the physiological functions needed to sustain cognition. 20 Senescence or biological aging is associated with an increased risk for cognitive decline in older adults. 20 Still, it is relevant to emphasize that cognitive aging in older adults can take distinctly different forms depending on various factors (ie, genetics and environment). 21
Based on HAND, HIV affects the immune system, nervous system, and brain, potentially depleting the cognitive reserve in at least two ways. 22 First, HIV can cross the blood−brain barrier and infect and kill glial cells (cells needed to support neuronal health) while producing neurotoxins. Second, HIV is considered a neuroinflammatory disease and has been shown in clinical HIV studies to reduce cognitive reserve and induce cognitive impairments. Furthermore, in older persons living with HIV, cognitive dysfunction may be increased due to the synergistic consequences of biological aging, HIV itself, and comorbid conditions (ie, T2D). 22
Regardless of age and absence of a HAND diagnosis, cognitive decline may be accelerated or accentuated due to HIV infection alone. For example, Sheppard et al 23 explored the cognitive decline trajectory in 146 neurocognitively normal participants of different races and genders. These individuals were assigned into one of four groups based on age (≤40 years and ≥50 years) and HIV status (positive and negative). The results indicated a nearly five-fold risk for developing neurocognitive disorder over approximately one year among HIV-positive individuals, irrespective of age. In older HIV-positive adults, however, lower baseline cognitive reserve, prospective memory, and verbal fluency individually predicted incidents of neurocognitive disorders at follow-up. 23
Aging persons living with HIV and multimorbidity may be at a greater risk of experiencing cognitive decline than their non-HIV counterparts, especially if they do not subscribe to or understand how an enriched environmental paradigm could support positive versus negative neuroplasticity. Positive neuroplasticity refers to the nervous system's ability to develop connections between neurons in response to novel, stimulating, and challenging stimuli, whereas negative neuroplasticity refers to the nervous system's tendency to deteriorate in response to little or no stimulation. 24 Multiple factors are associated with positive neuroplasticity, such as education, intellectual pursuits, physical activity, proper nutrition, adequate sleep, social interaction, and cognitive remediation therapy (ie, playing a computerized game designed to improve visual attention and processing speed).24–31 Conversely, factors linked to negative neuroplasticity may include poor health, low health literacy, a sedentary lifestyle, increased body mass index (BMI) and waist-to-hip ratio (WHR), substance use, poor sleep quality, suboptimal nutrition, depression, anxiety, and loneliness.24,26,27,30,32–34
Data are scant related to older persons living with HIV and positive and negative neuroplasticity. However, Krueger and colleagues 28 explored the association of cognitive activity and current neurocognitive performance in a sample of 176 older HIV-positive persons with a median age = 58.7 years. Based on participation frequency, the cognitive activities assessed as mediators of positive neuroplasticity included reading newspapers, magazines, or books; visiting museums or libraries; and attending a concert or play. Results showed that a higher regularity of activity was linked to better brain health in global cognition, semantic memory, and perceptual speed. Comparatively, loneliness is correlated with poorer cognition (or as a stimulus for negative neuroplasticity). Han et al, 33 for example, explored greater loneliness among older Black adults with HIV as being associated with poorer cognitive function. The sample was composed of 370 participants (177 with HIV, 193 without HIV, age mean = 58.8 years, 73.9% male, 68.9% Black) residing in the U.S. Even though the HIV-positive Black adults reported less loneliness than the HIV-positive White adults, findings suggested that greater loneliness advanced poorer cognition among the HIV-positive Black adults. Linking social, race-related chronic stress with loneliness and poorer cognition, the researchers suggested that older Black adults with HIV relative to older White adults with HIV may experience greater stress from multiple sources acting on neurobiological functioning. 33 In general, aging persons living with HIV must be assessed for cognitive impairment periodically, and, if needed, positive neuroplasticity stimuli should be recommended, and negative neuroplasticity initiators must be identified and remedied.
Nutrition
Even though ART has lessened the burden of wasting syndrome, lean-tissue depletion, hypermetabolic states, malabsorption, and cachexia in HIV-positive individuals,35,36 aging persons living with HIV still experience accelerated or accentuated morbidities or noncommunicable diseases, such as CVD, T2D, osteoporosis, gastrointestinal disorders, and chronic kidney disease.37–39 The diseases experienced by aging HIV-positive individuals are often fueled by suboptimal nutrition, such as vitamin and mineral deficiencies, and consuming foods high in sugar, salt, and saturated and trans fats. 36 Hence, the negative impacts of suboptimal or poor nutrition on micronutrients, body composition changes, and gut dysfunction in HIV-positive older adults will be discussed below.
Micronutrients
Adequate levels of vitamins, minerals, and trace elements are based on Dietary Reference Intake (DRI) levels, and there is growing evidence that routine intake of multivitamins or mineral supplements may improve immune function in older adults. 35 Notably, no conclusive evidence exists supporting the dietary supplement use independently based on improving infectious outcomes in well-nourished, HIV-infected individuals. 35 Nonetheless, it is likely that older adults living with HIV, particularly those with poor diets or who live in poverty, may benefit from regular consumption of multivitamin, mineral, and trace element supplements that contain micronutrients at DRI levels. 35
Micronutrient deficiencies are common in the aging population, but their extent varies from individual to individual, region to region, and country to country. 40 Deficiencies may be modulated by socioeconomic status, food insecurity, mental health (ie, depression, cognitive decline), social isolation, and physical disabilities (ie, loss of teeth and limited mobility).40,41 Furthermore, with aging, optimal nutrition may be impeded by sensory changes, including a decline in sight, smell, and taste acuity. 40 Deviations in smell and taste, for example, may make food less appetizing, leading to decreased nutrient-dense food intake. 41
Based on a comprehensive literature review, Maggini et al 42 indicated that approximately 35% of adults aged 50 years or older living in Europe, the U.S., and Canada have one or more micronutrient deficiencies. Suboptimal micronutrient intake in older adults has been reported in the community (vitamins A, B12, and D, and zinc) as well as in long-term care facilities (vitamins A, D, and E), while lower food intake has been associated with deficiencies in calcium, iron, zinc, B vitamins, and vitamin E. 42 The authors further implied that to maintain immunocompetence, the micronutrients needed include vitamins A, C, D, E, B2, B6, and B12, folic acid, iron, selenium, and zinc. Given the synergism between HIV itself (ie, chronic immune activation and inflammation), accelerated or accentuated immunosenescence (ie, inflammaging), multimorbidity, and ART—older individuals living with HIV may require supplementation (ie, vitamin D and calcium at an earlier stage of life).43,44
Body Composition Changes
Relative to the general aging population, older persons living with HIV are experiencing multiple body composition changes, leading to accelerated or accentuated multimorbidity and mortality.38,45 These changes are driven synergistically by HIV, ART, an obesogenic environment (high-fat diet and sedentary lifestyle) and aging38,45 while simultaneously fueling cardiometabolic diseases (ie, obesity, T2D, CVD, liver disease), osteoporosis, frailty (a syndrome of physiological degeneration in the elderly), 46 increased risk of hospitalizations and death.38,45–47 Body composition changes involve total body weight gain, increased visceral adipose tissue (VAT), muscle loss and function (sarcopenia), and decreased bone mineral density (BMD).38,45,47–49 Brief discussions specific to those changes and how they could impact the health and well-being of aging HIV-positive individuals are as follows.
Weight Gain and Obesity
Research has shown that weight gain after ART initiation was associated with lower mortality among initially underweight and normal-weight persons living with HIV, 50 whereas excessive weight gain and obesity present, exacerbate, accelerate, or accentuate multiple health-related issues in aging HIV-positive individuals. 45 In a review, Bailin et al 45 explored obesity and weight gain in persons living with HIV. The authors indicated that causes of weight gain and obesity are multifactorial among persons living with HIV, involving contemporary ART agents (principally integrase strand transfer inhibitors [INSTIs] and tenofovir alafenamide [TAF]), an obesogenic environment (high-fat diet and physical inactivity), shifting demographics (ie, an increase in the aging population), and older individuals being predisposed to obesity. 45
To gauge the extent to which overweight and obesity increase in persons living with HIV, Mavarani and associates 51 compared body weight, BMI (kg/m2) and WHR (a measure of central adiposity) 52 in persons living with HIV (N = 858) and HIV-negative controls (N = 1170) from the German Ruhr area for over 5 years follow-up; median age at baseline was 55 years. Results showed that persons living with HIV had a lower weight than the controls at baseline but experienced greater increases in weight and BMI after 5 years. After adjusting the analyses for smoking, the individuals living with HIV had an even higher weight difference compared to the controls after 5 years. The investigators noted that the proportion of overweight and obesity in HIV-positive individuals was still much lower than in the controls, illustrating that the BMI shift among the HIV-positive individuals reflected the “return to health” effects of ART. Although, as an indication of weight gain, the observed BMI shifts after 5 years in the HIV-positive persons might have represented the aligning of their BMI with the controls—but not a “return to health.”
In clinical and research settings, the BMI is used most frequently to assess weight status; however, WHR is a much better anthropometric index for indicating cardiometabolic disease risk. 53 Based on Mavarani and associates’ results in the above study, 51 a lower WHR change was demonstrated after 5 years for persons living with HIV compared with the HIV-negative controls, despite a higher WHR at baseline. The researchers 51 indicated that metabolic syndrome, insulin resistance, and liver steatosis were more common in HIV-positive individuals and connected to abdominal obesity,54,55 which may have explained the less than favorable WHR at baseline in the people living with HIV compared to the HIV-negative controls. Generally, healthcare providers must closely monitor weight gain, particularly in the abdominal region, in aging persons living with HIV to deter those changes from fueling morbidities.
Increased VAT, Sarcopenia, and Low BMD
In the aging HIV-positive population, elevated VAT or intra-abdominal fat is associated with an increased risk of cardiometabolic diseases (ie, CVD, T2D)56,57; and the promoters of VAT include current ART drugs (ie, INSTIs and TAF), high-fat diets, sedentary lifestyles, and aging. Whereas sarcopenia or loss of muscle mass and function could lead to frailty, 58 potentially progressing to falls, delirium, hospitalization, institutionalization, and mortality. 59 Like intra-abdominal weight gain, the loss of muscle mass and function may be brought about by ART medications, physical inactivity, poor nutrition, and aging. 60 In a review, data showed that the prevalence of sarcopenia in persons living with HIV was 24.1% in a sample of 2267 participants, and comparatively, HIV-positive persons presented 6.1% greater odds of sarcopenia than people without HIV. 49 Specific to low BMD in aging persons living with HIV, aging itself, lifestyle factors (ie, low calcium and vitamin D intake, physical inactivity), ART drugs (ie, protease inhibitors) are contributors leading to osteoporosis, frailty, fractures, increased risk of hospitalization and decreased quality of life.38,61,62 Notably, osteoporosis can be three times more frequent in HIV-infected individuals than in non-infected persons. 63
When considering instigators of frailty in aging HIV-positive persons, elevated VAT, sarcopenia and low BMD must be collectively factored in. For example, Hawkins and associates 64 investigated abdominal obesity, sarcopenia, and osteoporosis as a collaborative link to frailty in a sample of 399 men, consisting of 199 men living with HIV and 200 men living without HIV, with a median age of 60 years. Results illustrated that frailty prevalence was 16% in men living with HIV relative to 8% in men living without HIV. Specifically, higher waist circumference, VAT, sarcopenia, and femoral neck osteoporosis were associated with increased odds of frailty. 64
Principally, regular aerobic exercise, resistance training, and consumption of a nutrient-dense (ie, adequate protein intake), hypocaloric diet may be recommended to substantially reduce weight, VAT and maintain muscle for the general population and aging persons living with HIV. 65 Specific to BMD, positive lifestyle factors (ie, sufficient calcium and vitamin D intake supported by supplementation and physical activity) and BMD screening are critical to ensuring against frailty, fractures, and hospitalization while improving the quality of life for older HIV-positive individuals. 38 Undoubtedly, this will require a well-organized plan with key health professionals (ie, exercise specialists, dietitians, physical therapists, and orthopedists) to successfully address and manage such issues as VAT, sarcopenia, and low BMD in aging persons living with HIV.
Gut Dysfunction
Individuals with chronic HIV infection who receive ART may display intestinal dysbiosis and increased microbial translocation. 66 Consequently, significant shifts in the composition, diversity, and functional aspects of the intestinal microbiome are often observed in aging persons living with HIV due to normal aging and HIV infection. 67 These changes are manifested by dysbiosis or a disruption of the commensal homeostasis between the host and gut microbiota, 67 which are associated with pathological conditions, including inflammatory bowel disease, obesity, T2D, CVD, and neurodegenerative diseases (ie, Alzheimer's, Parkinson's).68–72
Microbial translocation has been described as a model of immune activation, whereby microbial translocation of bacterial products across damaged mucosal surfaces, primarily in the gut, has been posited to contribute to immune activation. 4 Notably, the GI tract mucosa is a major site of T-cell destruction in the first few weeks following HIV infection, and damage continues throughout all stages of HIV disease. 4 Importantly, the destruction leads to poor epithelial surface integrity, facilitating the translocation of microbial products such as lipopolysaccharide (LPS, endotoxin) across leaky mucosa into the systemic circulation, resulting in endotoxemia. 66
Once LPS enters the bloodstream, it binds to lipopolysaccharide-binding proteins (LBPs) and is then transported to a cluster of differentiation 14 (CD14) and further to Toll-like receptor 4 (TLR4), which activates the innate immune system via monocytes/macrophages. 73 This action results in the release of proinflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNFα). Due to the early loss of gut mucosal integrity, LPS becomes a source of continuous immune activation and a proinflammatory state, 74 further exacerbated by immunosenescence (inflammaging) in older persons living with HIV. 75
The production of short-chain fatty acids (SCFAs) may support greater gut microbiota diversity. 76 SCFAs are crucial in the relationship between host and gut microbiota, explaining the correlation between gut flora alterations and increased frailty in the elderly. 77 Specifically, SCFAs are important metabolites, including acetate, butyrate, and propionate, produced by the fermentation of fiber-rich food or non-digestible carbohydrates that escape digestion and absorption in the small intestine. 78 Studies conducted in the general population show that a diet rich in refined grains and sugars is negatively correlated with fecal concentrations and SCFAs, while consumption of large amounts of fruit and vegetables has a positive correlation with fecal concentrations and SCFAs. 76
There needs to be more literature related to aging persons living with HIV, gut dysbiosis, and diet. Armstrong and colleagues, 79 nevertheless, explored gut microbiome influences on metabolic health in 113 men. The analysis included a typical diet, HIV, ART, and lipodystrophy (LD) status, and other demographic characteristics such as age and BMI. Of note, LD is linked to early ART drugs and is defined as the redistribution of body fat; for example, less fat in the face, arms and legs and more fat in the abdominal region. 54 The cohort included men who have sex with women (MSW; n = 22) and HIV-positive and -negative MSM (n = 91). The HIV-negative MSM participated in activities that put them at high risk of contracting HIV (HR-MSM; ie, having a history of unprotected anal intercourse with two or more male or male-to-female transgender partners). 79 Data have shown that the altered microbiome in HIV-infected individuals 80 and HR-MSM were proinflammatory in vitro and gnotobiotic mice.80,81
Armstrong and colleagues’ findings in the above study 79 indicated a central role of inflammatory processes linked with bacterial translocation, as assessed by LBP and correlated with intestinal microbes, dietary, and demographic attributes such as age and BMI. For example, age was a significant predictor of impaired metabolic health in the cohort and negatively associated with B. adolescentis, and notably, the oldest (median age = 60, ranging from 54-64 years) group in the cohort were the HIV-positive MSM on ART with LD. Specific to B. adolescentis, it has been shown to prevent immunosenescence when fed in a probiotic food to aged rats 82 and to have beneficial effects on barrier function in a murine model. 83 Similarly, BMI was a predictor of reduced metabolic health in the cohort even though obese individuals were excluded from the study, and like age, the highest median BMI, 25.8 (23.0-28.0) kg/m2, was among the HIV-positive MSM on ART with LD. Furthermore, results demonstrated that the genus Prevotella was negatively associated with metabolic disease and positively aligned with dietary fiber, while the genera Coproccus (butyrate-producing bacteria linked with increased SCFAs) and the Dorea species, respectively, correlated with LBP negatively and positively. 79
Compared to the results specific to Prevotella in Armstrong and colleagues’ study, 79 other research has uncovered different health outcomes linked to distinctly different dietary components. For instance, Prevotella copri, the most abundant species of Prevotella, promoted poor glucose response through increased insulin resistance and the production of branched-chain amino acids in mice fed a high-fat diet. 84 Additionally, in a sample of 359 persons living with HIV with a median age = 51 years, results demonstrated that Alcohol Use Disorder Identification Test (AUDIT) scores were positively associated with dysbiosis, intestinal leakage, CD8+ T-cell late differentiation, and the Prevotellaceae bacterial family (including the genus Prevotella). 85 In general, compromised gut health in older persons living with HIV is multifaceted, involving HIV, ART drugs, aging, BMI, and suboptimal nutrition such as a low-fiber diet. Although, in part, a nutrient-dense diet with high fiber can improve gut integrity in the general aging population and older people living with HIV and multimorbidity.
Exercise
Aging adults living with HIV may be at a greater risk of developing accelerated or accentuated frailty because of physical inactivity. In their study, Umbleja and colleagues 86 explored physical function impairment and frailty in a sample comprised of 266 persons living with HIV (median age = 51 years) on ART, 81% of whom were male, 45% were Black, and 28% had HTN. The analyses indicated that older age, Black race, greater BMI, and physical inactivity were associated with physical function impairment, while depression and HTN were linked to prefrailty or frailty, 86 suggesting the relevance of exercise for improving physical and mental health. The following discussions focus on the benefits of habitual exercise as primarily a supporter against frailty and its contributors, such as increased VAT, sarcopenia, low BMD, and poor cardiorespiratory fitness (CRF) in aging HIV-positive adults.
Early frailty, hastened by multimorbidity, in the aging HIV-positive population could increase the risk for falls, worsening mobility, disability, hospitalization, and mortality. 87 Thus, improving functional limitations by engaging in a chronic exercise program should be preventive medicine against early frailty. Shah et al 88 conducted a randomized, controlled clinical trial involving 67 community-dwelling older adults with HIV and mild-to-moderate functional limitations. The participants were divided into physical activity counseling and control groups. The study findings suggested that a physical activity program grounded in the self-determination theory model of behavioral change can improve physical function (ie, gait speed, endurance and strength measures, and physical activity levels). 88
Frailty in old age is posited to increase the risk of dying from chronic inflammatory diseases rather than infectious diseases. 89 As aging persons living with HIV are burdened with increased inflammation precipitated by accelerated or accentuated immunosenescence and HIV-related chronic immune activation, they are more likely to die from an inflammatory disease (ie, CVD, T2D, chronic kidney disease, cancer).4,89 Exercise has been identified to lessen inflammatory states and disease-related outcomes, and this phenomenon specific to HIV-related inflammation was examined by d’Ettorre et al. 90 The authors concluded that the introduction of regular physical exercise in the clinical management of HIV infection could potentially result in a decrease in immune activation, with a concomitant reduction in damage linked to chronic inflammatory conditions, such as CVD and other non-AIDS-defining illnesses (ie, T2D). d’Ettorre and colleagues also posited that regular physical exercise could improve neuropsychological and metabolic (ie, insulin resistance and glucose tolerance) function. 90
As precursors to and interactors with frailty, many individuals living with HIV experience accelerated or accentuated age-related body composition changes, including increased VAT, sarcopenia, and decreased BMD.38,48,49,91,92 To counteract increased VAT, sarcopenia, and low BMD, prescribed and consistent exercise routines, in part, maybe a remedy to body composition change issues for older persons living with HIV. Jankowski et al, 93 for example, compared the effects of prescribed exercise on the body composition of sedentary persons living with HIV (n = 27) and uninfected controls (n = 28) aged 50–75 years. Participants completed 12 weeks of moderate-intensity exercise and were randomized to moderate- or high-intensity exercise for an additional 12 weeks. Results demonstrated that exercise reduced total and visceral fat in older persons living with HIV and controls. Although not significant by serostatus, controls tended to gain more lean mass, which suggested that a greater emphasis on resistance exercise may have been needed to increase muscle in the HIV-positive participants. 93 Furthermore, Ghayomzadeh and colleagues 94 explored the effects of combined resistance and aerobic training (RT + AT) on regional BMD and physical function in persons living with HIV. The study participants consisted of 40 persons living with HIV (20 men and 20 women), who were randomized into the RT + AT group (n = 20; age = 38.3 ± 4.9) or non-exercise control group (n = 20; age = 37.9 ± 5.1). Findings demonstrated that in the exercise group, there was a significant increase in BMD at follow-up for the lumbar spine, femoral neck and 1/3 radius (a skeletal site comprising almost exclusively of cortical bone), 95 and there were no gender differences in the training response between men and women in any of the BMD regions. Also, there were significant improvements in physical function (ie, handgrip strength and gait speed). 94 Even though the sample was relatively young compared to persons living with HIV ≥50 years, the research protocol, if needed, could be adapted to older HIV-positive individuals.
Like increased VAT, sarcopenia and low BMD, poor CRF is a major contributor to frailty and an obstacle to the health and well-being of aging persons living with HIV. 96 Data suggest that CRF has an essential role in reducing CVD and all-cause mortality, as well as incidents of myocardial infarction, hypertension, diabetes, atrial fibrillation, heart failure, and stroke. 97 Adults living with HIV have significantly reduced CRF, which exemplifies the aging phenotype observed in adults with chronic HIV infection. 98 Enduring strategies for exercise training in older adults remain a challenge, with time and motivation as common obstacles. Although with similar or greater CRF gains, a high-intensity interval training (HIIT) program relative to moderate-intensity continuous training may present a viable solution to a sustainable exercise program given less time commitment and motivation among some aging persons living with HIV. 99 HIIT is an aerobic exercise approach that involves periods of high-intensity anaerobic exercise (>85% of heart rate reserve—the difference between the maximum and resting heart rate) alternating with low-intensity continuous training. 100
As a cross-over pilot study, Briggs et al 99 explored the feasibility and effects of HIIT combined with RT on CRF in older adults living with HIV. The research was conducted within a randomized exercise trial with 26 sedentary adults with HIV, median age = 61.5 (± 6.7) years. Initially, participants were randomized to 4 months of continuous high-intensity aerobic exercise (AEX) and RT (3x/week) or control group. Next, the control group completed 4 months of HIIT + RT (3x/week). The clinically significant increases in VO2peak (the highest rate of oxygen that an individual can achieve during physical exertion) assessed during a graded treadmill test after HIIT and AEX represented improvements of 17.1% and 7.7%, respectively. Both groups had improvements in exercise endurance and strength. Overall, the pilot study supported HIIT as a viable solution for delivering high-intensity AEX in improving CRF towards the goal of strengthening cardiovascular health in adults living with HIV. 99
In general, the reviewed research suggests that engaging in a moderate to vigorous aerobic and resistance exercise program at least three times per week can improve body composition and CRF while potentially decreasing frailty in older individuals living with HIV. However, it is imperative to address all the barriers to initiating and maintaining an individualized exercise program, including physical and psychological decline, depression, social influences, reinforcement, lack of self-efficacy, environmental context and resources, negative perceptions of gym culture, and appropriate exercises specific to physical limitations—all ensuring that aging persons living with HIV will attain the benefits of a well-designed exercise program.101,102 To further elevate the success of chronic exercise for older individuals living with HIV and multimorbidity, a clinical exercise scientist or a medical exercise specialist should assess, prescribe, and monitor a tailored exercise program in clinical, behavioral health and home-based settings.
Additional Lifestyle Factors
Smoking
Even though it is estimated that smoking causes one in every five deaths in the U.S. each year, about one in five Americans still smoke cigarettes. 103 At about 40%, cigarette smoking is much higher among persons living with HIV, 104 and aging HIV-positive individuals are less likely to quit smoking than their age-matched uninfected counterparts. 105 Adults with HIV who smoke are more likely to be poor, homeless, previously incarcerated, depressed, and have lower educational attainment than those who do not smoke. 104 Persons living with HIV who smoke are also more likely to suffer from HIV-related infections, such as thrush, airy leukoplakia (white mouth sores), and bacterial and pneumocystis pneumonia. 106 According to the available evidence, in HIV-positive individuals, smoking may lead to chronic obstructive pulmonary disease (COPD), heart disease, stroke, and cancer.4,106 Smoking has also been significantly implicated in treatment noncompliance in HIV-positive populations on ART. 107
In sum, HIV-positive individuals who smoke will lose more life years to smoking than HIV. Max and colleagues, 108 for example, explored deaths and projected deaths from smoking and HIV/AIDS among gay men in California from 2005 through 2050. The data indicated that from 2005 to 2014, smoking caused over 6800 deaths among gay and bisexual men, while nearly 9500 died from HIV/AIDS. Mortality from both causes has been falling, but deaths from HIV/AIDS are falling more rapidly. Projections suggest that in the mid-2040s, more gay/bisexual men will die from smoking than from HIV/AIDS. 108 With the synergism among aging, HIV, multimorbidity (ie, CVD) and smoking, life's trajectory shortens for aging persons living with HIV and multimorbidity; therefore, they should be encouraged to quit smoking. For this purpose, smokers should seek support via their healthcare provider or evidence-based smoking cessation programs, such as those accessible via the Internet.
Alcohol use
HIV and alcohol abuse are linked to a wide range of comorbidities, many of which are typically associated with advanced aging, such as liver, cardiovascular, cerebrovascular, and renal diseases, as well as osteoporosis, cancer, metabolic alterations (ie, increased insulin resistance, decreased glucose tolerance), T2D, and neurocognitive impairment.109,110 Alcohol abuse also leads to medication nonadherence, antiretroviral resistance, and risky sexual behaviors. 111 Overall, alcohol abuse among older persons living with HIV increases the odds of mortality. 112
Hazardous drinking is defined by the quantity and frequency of alcohol consumption and is associated with acute and chronic morbidity 113 and binge drinking is categorized as four drinks for a woman and five drinks for a man over two hours. 114 Comparatively, alcohol use disorder (AUD) is identified as a medical condition characterized by an impaired ability to stop or control alcohol use despite adverse social, occupational, or health consequences. 115 Crane and associates 116 investigated the prevalence of hazardous alcohol use among 8567 persons living with HIV on ART, with a mean age = 46 years, and 3441 (41%) were 50 years or older. Results demonstrated that hazardous alcohol use was reported by 27%, and 34% reported binge drinking. Whereas based on the analysis of the Veterans Aging Cohort Study's (VACS) electronic record data made up of a sample of 32,699 adults living with HIV from urban and large and small rural areas, the predicted prevalence of AUD was between 12–14%. 117
When considering alcohol abuse by older individuals living with HIV, the interrelationship between HIV, aging, and alcohol abuse should be viewed as a negative cumulative effect on immune function. 118 A major link between alcohol, HIV, and immune function lies within the GI tract. As such, alcohol damages the mucosa lining of the intestines, resulting in inflammation, barrier damage, and bacterial leakage, which, in turn, strengthens HIV's hold on the host. 118 HIV infection has been shown to accelerate or accentuate immunosenescence through chronic immune activation and persistent inflammation, 119 while chronic alcohol consumption may hasten immune activation and immunosenescence.85,120
Katz and colleagues 120 tested the hypothesis that hazardous alcohol consumption accelerates immune activation and immunosenescence in chronic binge alcohol (CBA)-consuming simian immunodeficiency virus (SIV)-infected rhesus macaques with and without ART. Their results demonstrated that CD8+ T-cell activation and immunosenescence were increased relative to the baseline level only in CBA-consuming animals not receiving ART. The researchers suggested that CBA consumption and AUD enhance immune activation and immunosenescence in SIV-infected macaques and persons living with HIV. 120 Data published by Maffei et al 85 in a sample of 359 HIV-positive participants with a relatively high ART adherence, median age = 51 years, indicated that AUDIT scores were positively associated with CD8+ T-cell late differentiation, intestinal leakage and dysbiosis—and that alcohol-linked dysbiosis was implicated in CD8+ T-cell senescence. Comparatively, Carrieri et al 121 uncovered that individuals living with HIV on ART who consumed less than 10 grams of alcohol per day had a higher CD4+ T-cell count when compared to abstainers, indicating that the adverse health consequences of alcohol use are dose-dependent.
Aging HIV-positive individuals may experience increased mortality and physiologic injury at lower levels of alcohol use compared with uninfected individuals. 112 In a sample of 18,145 persons living with HIV (76% of whom had undetectable HIV-RNA; median age = 52.5 years) and 42,228 uninfected individuals (median age = 54 years), Justice and colleagues 112 explored the risk of (a) all-cause mortality and (b) physiological injury as measured by VACS Index (which is predicated on biomarkers such as age, CD4+ T-cell count, viral load, hemoglobin, platelet count, creatinine). Specific to HIV-positive individuals, findings indicated an Alcohol Use Disorder Identification Test-Consumption (AUDIT-C) score ≥4 and ≥30 drinks per month were associated with an increased mortality risk. Comparatively, AUDIT-C scores ≥5 and ≥70 monthly drinks among uninfected individuals were associated with an increased mortality risk. AUDIT-C threshold scores of 5−7 and 8 were associated with physiological injury in persons living with HIV and uninfected participants, respectively. Guided by these disparities, the investigators concluded that alcohol limits for HIV-positive individuals should be lowered. 112
Without question, excessive alcohol use can be harmful to the health and well-being of aging persons living with HIV and multimorbidity; thus, healthcare providers should periodically assess hazardous drinking, AUD, and CBA consumption in this aging population and, if needed, provide appropriate interventions. Providers should also assess and deliver support, if needed, for underlying psychosocial determinants (ie, depression, social isolation), which can precipitate or exacerbate alcohol abuse.
Sexually Transmitted Infections
Older HIV-positive persons have sex, and some engage in unprotected intercourse. 122 In a sample of 415 HIV-positive men (45 to 55 years of age at baseline) on ART, Surkan and colleagues 123 identified that 66% (n = 277) reported unprotected insertive anal intercourse, while 72% (n = 299) reported unprotected anal receptive intercourse over six median follow-up visits. In an earlier study, Golub et al 124 explored the prevalence of different sexual behaviors in a sample of 914 older persons living with HIV aged >50 years (640 male, 264 female, 10 transgender) living in New York City. The findings indicated that over one-third of sexually active participants reported unprotected anal or vaginal sex in the past three months, and 18% reported unprotected sex with serodiscordant partners.
Correlates with increased sexual-risk behaviors among aging persons living with HIV, as well as the general aging population, include a lack of knowledge concerning STIs, substance use, and psychosocial issues (ie, depression).125,126 Extant research has also shown several facets of syndemic relationships linking STIs and HIV. Particularly, people with STIs are more likely to become infected with HIV, and those coinfected with HIV and another STI are more likely to transmit HIV. 127 Because of an aging immune system and accelerated or accentuated immunosenescence linked to HIV,75,119 further immunological downregulation may occur in older HIV-positive individuals on ART when coinfected with another STI (ie, human papillomavirus) 128 or other viruses (ie, cytomegalovirus),89,129 thereby increasing the probability of multimorbidity and mortality.46,128
Developing risk-reduction programs for older persons living with HIV may be exceptionally challenging because of their age-related psychological, physical, or sexual stage of life. Nonetheless, they must understand the ease of transmission of other STIs and that, by practicing safer sex, they will protect themselves and their sexual partners against infections. Consequently, healthcare providers should periodically assess sexual-risk behaviors and be able to deliver appropriate safer-sex education as needed.
Insomnia
Insomnia or sleep disturbance is characterized by difficulty initiating and maintaining sleep. 130 Based on a meta-analysis, the estimated sleep disturbances in persons living with HIV is 58%, 131 compared to 10% to 30% in the general population. 132 Although the pathophysiology of sleep disturbances in aging persons living with HIV and multimorbidity is unclear, it may be related to the ability of HIV to infect the central nervous system, polypharmacy, substance use (ie, excessive alcohol consumption), and morbidities (ie, depression, arthritis, digestive disease, renal and urologic diseases, cancer).131–136 Regardless of its etiology, insomnia is clinically important in this population for several reasons, including the development and worsening of negative health outcomes that may be physical (ie, HTN, CVD, T2D, insulin resistance, central weight gain), cognitive (ie, slowed reaction time, poorer problem solving), emotional (ie, depression, inadequate emotional regulation), or immune-function related (ie, dysregulated innate immunity).137–141
Empirical evidence focusing on aging HIV-positive persons with CVD risk is lacking, although data indicate that sleep disturbance increases the risk for CVD in the general aging population. 142 However, Polanka and colleagues 143 explored the HIV−CVD risk in a sample of HIV-infected veterans (n = 3108; median age = 49 years), with a median follow-up time of 10.8 years, during which 267 CVD events occurred. Relative to HIV-infected veterans with no difficulty falling or staying asleep, HIV-infected veterans bothered a lot by insomnia symptoms had an increased risk of a CVD incident, suggesting that highly bothersome insomnia symptoms were significantly associated with CVD incidents in HIV-infected veterans. 143
“Good sleep” is imperative for the physical and psychological health and well-being of aging persons living with HIV and multimorbidity. Healthcare providers should evaluate this aging population's sleep quality regularly and identify and remedy any underlying problems (ie, depression and substance use) that may impede sleep quality. Importantly, providers should also educate these patients on the importance of practicing good sleep hygiene, including (a) following a consistent sleep schedule, (b) avoiding caffeine and alcohol, (c) not consuming large meals too close to bedtime, and (d) exercising daily. 144
Treatment Adherence
Viral replication and drug resistance increase in ART-nonadherent individuals, increasing the chance of morbidities and mortality. 145 Heightened viral replication also results in higher viral load and infectiousness, enhancing the likelihood of transmitting the virus during unprotected sex. 146 Noncompliance with medications prescribed for coexisting conditions, such as HTN and dyslipidemia, may result in negative sequelae (eg, dyslipidemia and HTN to CVD) as well as early death.147,148 Older adults with HIV have a reduced risk for nonadherence to ART than their younger counterparts. 149 Treatment adherence, however, is challenging for aging persons living with HIV and multimorbidity due to several reasons, including decreased medication efficacy associated with age-related physical deterioration, polypharmacy interactions and side effects, and cognitive decline.32,150–152
Prescribed psychotropics are frequently added to the polypharmacy burden of older adults as well as aging HIV-positive persons living with HIV and multimorbidity.153,154 Yet, there is a de-escalation of the use of psychotropics because of their association with increased risk of geriatric syndromes, including falls, fractures, slow gait speed, and impairment of neurocognitive function.155–158 Inappropriate prescribing of psychotropic medications to older patients, including aging HIV-positive persons, is present in institutionalized (ie, assisted living facilities and long-term care living facilities) and non-institutionalized populations.156,159 Specific to aging persons living with HIV, the use of mental health therapeutics, such as antidepressants, may interact with ART, resulting in widened health consequences. 160 In a review, Matt and Gaskill 160 explored the interaction between ART and antidepressants in older adults and uncovered that during the early stage of HIV infection, the association increases virus replication and inflammatory cytokine production. In the later stage, the interface could precipitate neuropathy and create a Parkinson's-like state (ie, slowed movements, rigid muscles, impaired posture and balance) in sensitive individuals. 160
Lastly, among aging persons living with HIV and multimorbidity, there may be clustering and interaction of syndemic (or biopsychosocial) factors (ie, disease, poverty, substance use, depression),161,162 which may adversely affect treatment compliance. Along with the clustering of biopsychosocial determinants, medication noncompliance is multifaceted for aging individuals living with HIV and multimorbidity. It is incumbent upon healthcare providers to monitor treatment adherence closely and, whenever necessary, implement corrective actions to resolve noncompliance.
Viable Solution
Given the alarming findings reported in the preceding sections, the question is: “How do healthcare practitioners effectively address a multitude of negative lifestyle factors associated with numerous conditions faced by aging persons living with HIV while simultaneously delivering high-quality, HIV-related medical care?” In tackling the often integrated, interactive, and multilayered negative lifestyle factors confronted by older persons living with HIV and multimorbidity, the solution may lie in developing an interdisciplinary team or unit comprised of preventive clinical practitioners. The team could work in the community and medical (or clinical) settings and function as a roving unit to reach communities needing this vital service, and it may use telehealth services to support clients in receiving preventive care better.
Importantly, the team's overarching goal will be to promote and implement positive lifestyle factors to improve the health and well-being specific to the aging HIV-positive population; Tables 1 and 2 highlight the benefits of positive lifestyle factors.
Positive Lifestyle Factors Improve the Health and Well-Being of Aging Persons Living with HIV and Multimorbidity.

Abbreviations: T2D, type 2 diabetes; HTN, hypertension; RDI, recommended dietary intake; BMD, bone mineral density; CVD, cardiovascular disease; HIV, human immunodeficiency virus.
Positive neuroplasticity refers to the nervous system's ability to develop connections between neurons (individual cells that make up the nervous system) in response to novel, stimulating, and challenging stimuli, which is associated with education, physical activity, proper nutrition, adequate sleep, and cognitive remediation therapy (ie, playing a computerized game designed to improve visual attention and processing speed). 24
VAT (visceral adipose tissue) is intra-abdominal fat surrounding internal organs. 65
Frailty represents a decreased physiological reserve and function that is more dominant with increasing age. 46
Positive Lifestyle Factors Improve the Health and Well-Being of Aging Persons Living with HIV and Multimorbidity.
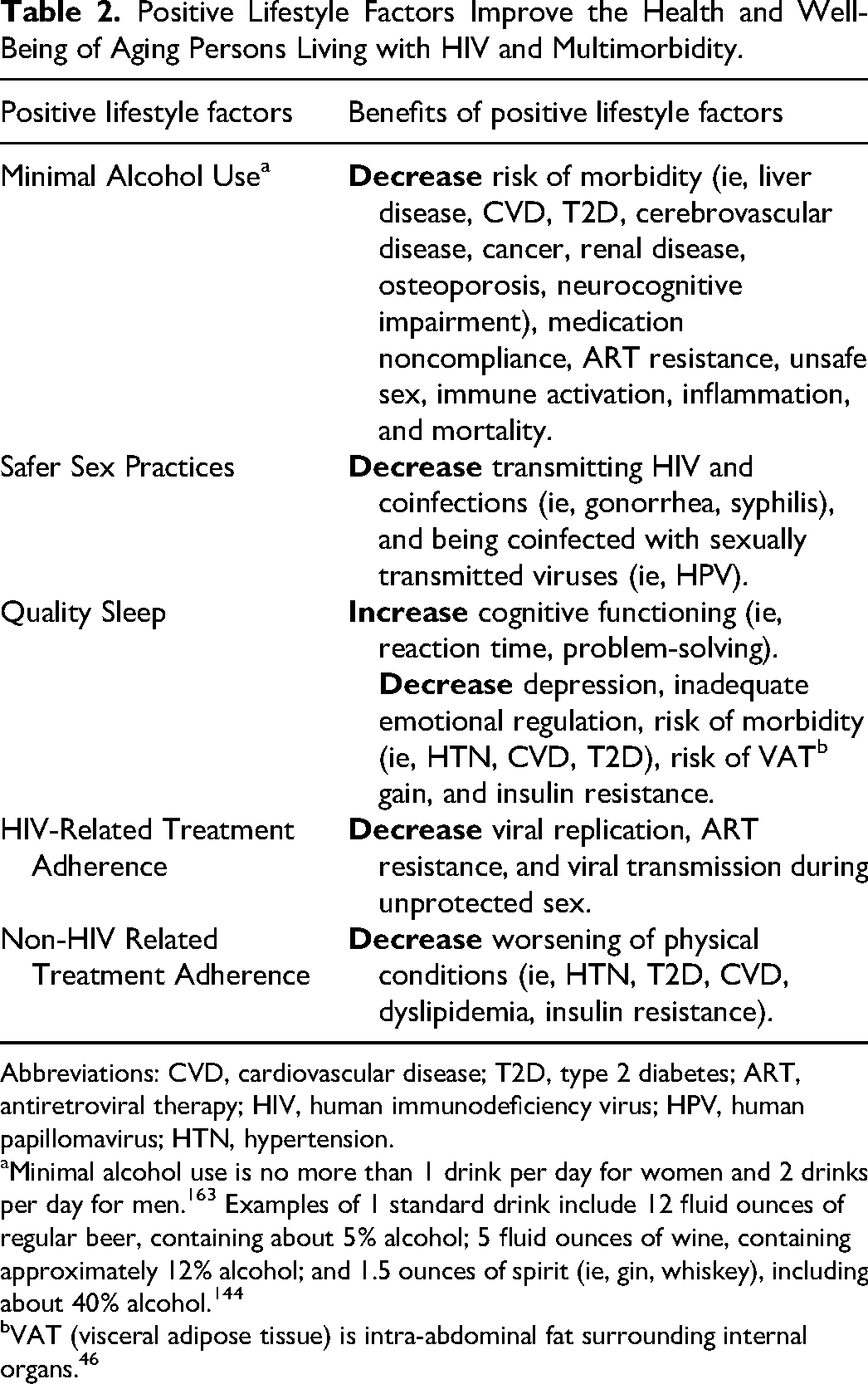
Abbreviations: CVD, cardiovascular disease; T2D, type 2 diabetes; ART, antiretroviral therapy; HIV, human immunodeficiency virus; HPV, human papillomavirus; HTN, hypertension.
Minimal alcohol use is no more than 1 drink per day for women and 2 drinks per day for men. 163 Examples of 1 standard drink include 12 fluid ounces of regular beer, containing about 5% alcohol; 5 fluid ounces of wine, containing approximately 12% alcohol; and 1.5 ounces of spirit (ie, gin, whiskey), including about 40% alcohol. 144
VAT (visceral adipose tissue) is intra-abdominal fat surrounding internal organs. 46
An interdisciplinary preventive-clinical team should be comprised of clinicians with education, training, and experience in preventive and clinical care, with preferable knowledge about and expertise in working with older people living with HIV. Ultimately, the team must also be able to frame and deliver services within the context of primary, secondary, or tertiary prevention constructs. 164 Primary prevention focuses on preventing disease, 165 whereas secondary prevention targets preclinical conditions or potential sequelae (eg, prehypertension to hypertension), 166 and tertiary prevention emphasizes managing an existing disease, focusing on condition improvement and non-worsening of the disease. 167 Disease prevention programs or services should ideally focus on delivering primary and secondary preventions; however, given that older persons living with HIV present with one (ie, HIV disease) to multiple coexisting clinical condition(s), they would require tertiary prevention services.
After forming an interdisciplinary team, all its members should undergo training.168,169 The primary goal of training is to establish respectful and trusting interpersonal relationships among members, with the key understanding that everyone brings unique knowledge and skills to the team, ensuring that high-quality and comprehensive services will be provided to each client. The training should focus on (a) clearly defining the roles and responsibilities of each team member; (b) developing an assessment plan, encompassing identifying and developing instruments; (c) outlining what a client may experience (or client protocol) during and following (follow-up) service; (d) timely and appropriate communication and exchange of information with a client; (e) developing a referral list of health-related resources (ie, foodbanks) and healthcare providers external to the team (ie, occupational and physical therapists); and (f) providing constructive feedback that will foster efficient and effective information exchange between team members. Also, as part of the training, healthcare providers, such as geriatricians who have working knowledge and experience with aging HIV-positive individuals, will be consulted to ensure the team implements best practices. Furthermore, a disease prevention network will be developed through possible referrals made to and received from specialists (ie, geriatricians), primary care providers (PCPs), psychiatrists, physical therapists, occupational therapists, as well as other healthcare providers (ie, dentists, ophthalmologists, optometrists, orthopedists).
A unit specifically tasked with working with aging people living with HIV and multimorbidity could include a medical nutrition therapist (eg, clinical registered dietitian), clinical exercise scientist (eg, certified clinical exercise physiologist or certified medical exercise specialist), mental health therapist (eg, licensed clinical psychologist), pharmacist, client service coordinator (eg, clinical social worker or medical case manager), and certified health coach. The primary role of a certified health coach would be to empower the clients to transform their health via lifestyle and behavioral changes. Evidence indicates that substance use (ie, alcohol abuse) is implicated in the progression of HIV as well as in the development and worsening of comorbid conditions (ie, T2D, osteoporosis, HTN, CVD)37,110,170; therefore, at least one clinician should be certified as a substance abuse counselor. Figure 2 illustrates the proposed client-centered, preventive-clinical, interdisciplinary-team practice model, where the client is in the center and surrounded by clinicians and social support (friends and family).
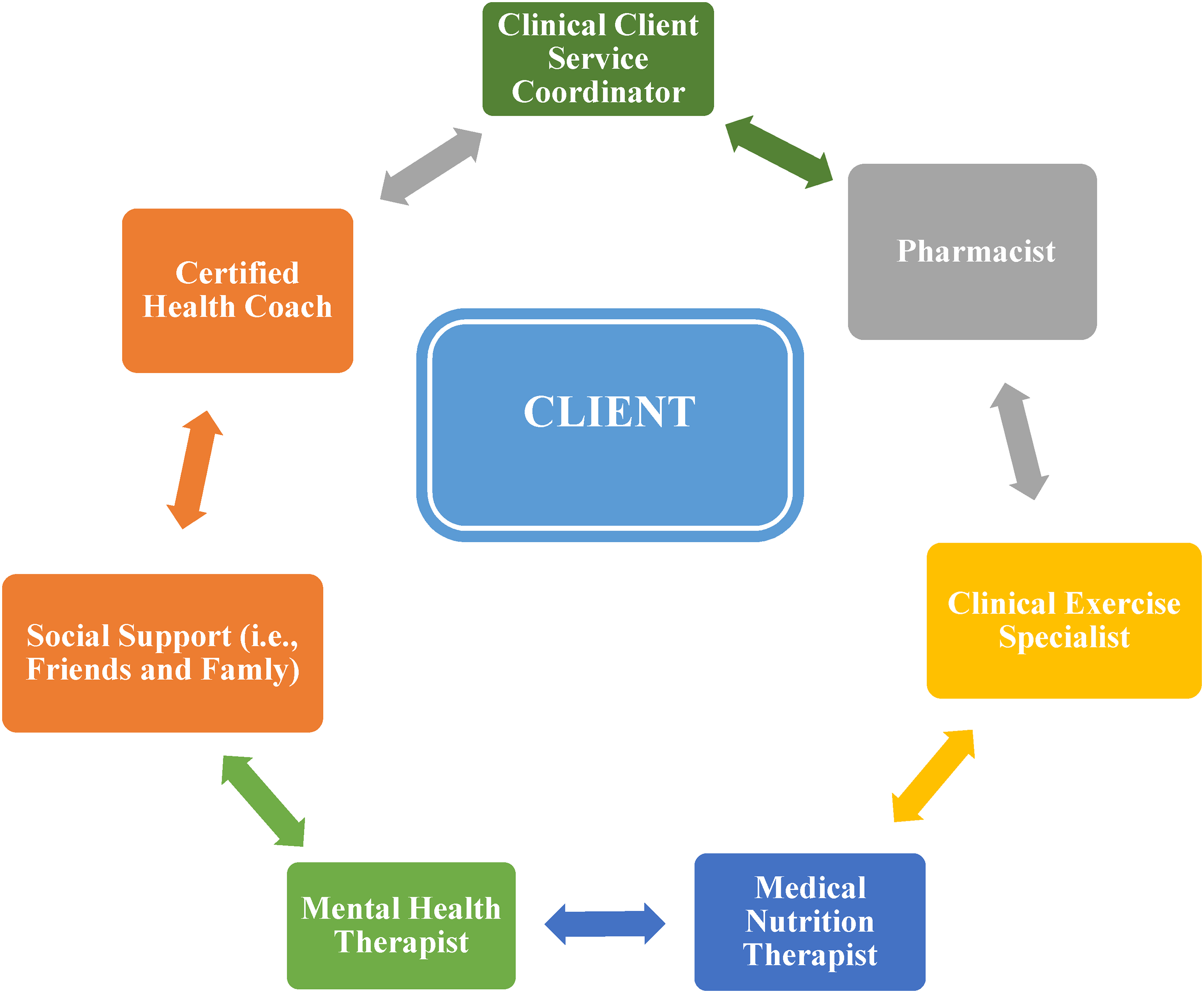
Client-Centered preventive-clinical interdisciplinary-team practice model. The client-centered, preventive-clinical, interdisciplinary-team practice model consists of a) the client as the center and b) the interdisciplinary team (ie, clinical client service coordinator, certified health coach, pharmacist, clinical exercise specialist, medical nutrition therapist, mental health therapist plus social support including friends and family).
As indicated by the model, the role that friends and family could play may be essential to a client's success in adopting positive behavioral changes while accomplishing desirable health outcomes. For this reason, when appropriate, friends and family members should be engaged in the physical and psychological assessments, feedback, education, and training offered to clients. Members of the support system will be guided by respective team members on how to encourage or support a client's behavioral change process, especially without judgment or criticism. Lastly, to support quality services, a strong relationship must be developed via electronic record keeping and information exchange with healthcare providers external to the team, including PCPs, medical specialists (ie, geriatricians, cardiologists), psychiatrists, or physical therapists. The team, for example, will exchange assessment results, treatment plans, and follow-ups via electronic records with external healthcare providers.
Conclusion
It is important to note the limitations in the data reviewed. For example, the exercise research consisted of small sample sizes, which in multiple instances did not control for diet as a possible confounding variable, especially when assessing fat and muscle changes. Irrespective of the review's limitations, the data provided insight into the psychological and physical struggles expressed through negative lifestyle factors that may be experienced by some aging HIV-positive adults with multimorbidity. Even though we are living in an era marked by an emphasis on preventive health, many older adults living with HIV are confronted with a unique set of challenges that may make it more difficult to prevent and manage multimorbidity relative to their aging HIV-negative counterparts. Specifically, when the burden of HIV is added to the physical and psychological decline, social and economic disparities, stigmatization and alienation (ie, based on sexual identity and race), and polypharmacy interactions and side effects, improving psychological and behavioral health and quality of life become increasingly more difficult.
Within the context of preventive medicine, improving psychological and behavioral health in the aging HIV-positive population requires greater focus on preventing and managing negative lifestyle factors such as poor mental health (ie, depression and cognitive decline), suboptimal nutrition, physical inactivity, substance use (ie, smoking and alcohol use), STIs, sleep disturbance, and treatment nonadherence. However, many of the existing wellness programs in clinical- and community-based settings may be ill-equipped to address the negative lifestyle factors experienced by aging persons living with HIV and multimorbidity.The physical and psychological health and quality of life for older individuals living with HIV and multimorbidity could be enhanced by developing a client-centered, preventive-clinical, interdisciplinary-team practice based on well-recognized prevention constructs. Lastly, grounded on a client-centered, preventive-clinical, interdisciplinary team model, longitudinal data collection and analysis should drive program effectiveness and modifications.
Footnotes
Acknowledgments
I greatly appreciate Dave Benedetti, Dr James Davis, Dr Bertina Walker and Daniel Lindquist for their technical support in preparing and finalizing the manuscript.
Contributions
No contributions were made to this review study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Committee
Ethical committee is nonapplicable to this review study.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent is nonapplicable to this review study.
