Abstract
To compare the characteristics, presentation, investigations, and outcomes in tuberculous meningitis (TBM) patients with and without human immunodeficiency virus (HIV) coinfection. A retrospective cohort study was conducted on adult (age > 18 years) patients whose final diagnosis was TBM and who were treated at Vajira Hospital, Navamindradhiraj University, Thailand, between January 2005 and December 2016. A final total of 174 individuals were included in the study. Of these, 97 (55.75%) were HIV positive. Treatment was successful in 53 (30.5%) individuals. In HIV-infected TBM patients, there were higher proportions of patients who were younger in age (≤40 years), patients with a low body mass index, history of previous tuberculosis infection, or hepatitis C virus coinfection. A successful treatment outcome rate was lower in HIV-infected TBM patients than in HIV-uninfected TBM patients. Since HIV infection decreases the chance of successful treatment outcomes of TBM patients, future studies are needed to determine the clinical indicators for poorer survival outcomes in HIV-positive TBM patients.
Introduction
An estimated 10 million people fall ill with tuberculosis (TB) annually, and 1.4 million TB patients die, of which 208,000 are human immunodeficiency virus (HIV) positive. 1 An estimated 44% of people who developed TB in 2019 were in Southeast Asia. Despite a decline in the TB incidence rate worldwide, 1 it is still a leading cause of death, especially in resource-limited countries. Thailand is one of the 22 countries in the world with a high incidence of TB. In 2019, the incidence rate of TB in Thailand was estimated to be 105,000 cases annually.1,2 Ten percent of TB-infected patients in Thailand had HIV coinfection. 2 Tuberculosis can infect many organs, such as pulmonary, lymph node, gastrointestinal system, and central nervous system (CNS). Tuberculous meningitis (TBM) is one of the most serious extrapulmonary involvements of Mycobacterium tuberculosis (MTB). Although TBM accounts for only 1% of TB cases, 3 morbidity and mortality can be as high as 50% in those affected.4-6 The incidence of TBM was estimated to be >100,000 cases annually. 7 Death occurs in ∼20% of HIV-uninfected and ∼40% of HIV-infected patients, even when receiving the appropriate anti-TB drug treatment.7,8
Regarding diagnosis, TBM is usually diagnosed on the basis of clinical presentations, physical examination, neuroimaging assessment, and cerebrospinal fluid (CSF) profiles. Many proposed criteria for the case definition of TBM are available.9-11 Confirmation of TBM requires identification of acid-fast bacilli in CSF, isolation of MTB from the CSF culture, or molecular identification. Because of the lower organism load in CSF compared with that of other specimens (i.e. sputum), confirmation of TBM can be difficult to determine. In most cases, the diagnosis remains unconfirmed. Recently, a scoring system for TBM diagnosis was developed by Thwaites et al. 12 and Marais et al. 13 Studies found that clinical scoring systems were quite sensitive but not specific when used to distinguish TBM from bacterial meningitis, especially in HIV-uninfected adults.14,15
Recent data suggest that mortality in TBM patients is associated with many factors, such as HIV infection, older age, multidrug resistance, neurological deficit, and low numbers of CSF white cell counts.16-20 HIV-infected individuals are more susceptible to developing severe disseminated forms of TB, included CNS involvement, 21 and mortality in these patients can exceed 60%.9,22
Because of the devastating mortality rate of TBM, especially in patients with HIV coinfection, we conducted a study to compare the clinical characteristics, presentation, laboratory investigations, and outcomes of TBM patients with and without HIV coinfection.
Materials and Methods
Study Population
We performed a retrospective cohort study of adults (age > 18 years) whose final diagnosis was TBM and who were treated at Vajira Hospital, Navamindradhiraj University, between January 1, 2005, and December 31, 2016. Vajira Hospital is a 900-bed university hospital in Bangkok, Thailand. This hospital receives more than 700,000 outpatient visits, and approximately 30,000 inpatients are admitted annually. Cases were identified by reviewing electronic and paper-based medical records and the TB registry. Classification of cases as definite, probable, or possible TBM was based upon the diagnostic criteria described by Thwaites et al. 10 Briefly, definite TBM was diagnosed if there was evidence of the microbiological confirmation of MTB, that is, a positive acid-fast bacilli stain, positive MTB culture, or positive molecular test (PCR for MTB) of the CSF. Probable TBM was diagnosed if patients presented with any symptom of meningitis, and there was evidence of MTB infection in other organ(s) to exclude other causes of meningitis. Possible TBM was diagnosed if patients had any symptom of meningitis and abnormal CSF examination findings suggestive of TBM and that excluded other causes of meningitis, without the isolation of mycobacterium. We excluded patients with incomplete medical records and patients whose final diagnosis was not TBM.
Treatment Protocol and Measurements
We recorded the medical history, clinical characteristics including presenting symptoms, laboratory results, radiological examination, information related to HIV/TB diagnosis, and treatment outcome as documented by the evaluating physician. The treatment outcome was defined as the result of TB treatment according to the World Health Organization “Definitions and Reporting Framework for Tuberculosis–2013 revision” (updated December 2014). 23 Briefly, treatment success was based upon the sum of cured patients and treatment completed. Treatment failure was the sum of patients with failed treatment, patients who were lost to follow-up, and patients who had died. Cutaneous adverse drug reactions were diagnosed according to the US National Cancer Institute's Common Terminology Criteria for Adverse Events grading criteria. 24 Hepatitis was defined by the case definition and phenotype standardization for drug-induced liver injury. 25 Deaths were included in all-cause mortality data if they occurred within the routine 12-month follow-up period. The total follow-up time was calculated from the date of the initiation of anti-TB drugs to the date of discharge from the TB clinic, and the result of TB treatment outcome was recorded.
Statistical Analysis
Categorical variables were presented as the number (n) and percentage (%). Numerical variables were presented as the mean and standard deviations (mean ± SD) or medians with interquartile range (IQR), depending on the normality of the variable. To compare the clinical characteristics and outcomes among HIV-infected and uninfected TBM patients, Chi-square or Fisher's exact test was performed for univariable analysis of categorical data, and Student t test or Mann–Whitney U test was performed for continuous data. Variables with a P value less than 0.1 in the univariable analysis were included in a multivariable model. Multivariable logistic regression was performed using a backward stepwise elimination approach to construct the final model. Time to treatment failure was analyzed using the Kaplan–Meier curve and Cox's regression. Data analysis was performed using the Statistical Package for Social Sciences software, version 22.0 (SPSS Inc, Chicago, IL). A P value less than 0.05 was statistically significant in all analyses.
Ethical Approval and Informed Consent
Study approval was provided by the Vajira Institutional Review Board (COA43/2560).
Results
From January 1, 2005, to December 31, 2016, a total of 238 individuals with a provisional diagnosis of TBM were included in the study; 64 were excluded from the analysis (34 individuals had a final diagnosis other than TBM: spinal TB [n = 9], tuberculous myelitis [n = 8], bacterial meningitis [n = 8], cryptococcal meningitis [n = 7], and CNS lymphoma [n = 2]; and 30 individuals were excluded because there were insufficient patient data for further analysis). A final total of 174 individuals with a diagnosis of TBM remained in the study, with a total follow-up time of 74 person-years (mean follow-up time of 155 days/person). HIV infection was diagnosed in 97 (55.75%) of the 174 individuals. The study flow is provided in Figure 1.

Study flow.
The characteristics of the 174 individuals are provided in Table 1. TBM diagnosis was classified as definite TBM, 10 (5.75%) individuals; probable TBM, 87 (50%) individuals; or possible TBM, 77 (44.25%). The mean age was 43.14 ± 14.12 years, and 60.92% were males. A prior history of TB infection was reported in 15 (8.6%) individuals. There were 21 (12.1%) individuals who had active TB infection and were receiving anti-TB treatment with subsequent development TBM. Isolated TBM was diagnosed in 102 (58.6%) cases. There were 72 (41.4%) individuals who had more than one site of TB involvement, of which 63 (87.5%) included pulmonary involvement. There were 104 (59.8%) and 100 (57.5%) individuals who had a serologic test for viral hepatitis B and C, respectively, of which 10 (9.62%) were reactive for hepatitis B surface antigen (HBsAg) and 19 (19%) were reactive for anti-hepatitis C virus (HCV).
Characteristics of 174 Individuals.

HIV: human immunodeficiency virus; TB: tuberculosis; IQR: interquartile range; HCV: hepatitis C virus; BMI: body mass index; SD: standard deviation; HBV: hepatitis B infection; HT: hypertension; DM: diabetes mellitus; COPD: chronic obstructive pulmonary disease.
There were 104 and 100 individuals who had a serologic test for viral hepatitis B and C, respectively.
Other; thalassemia = 2, hepatoma, cervical cancer, multiple myeloma, SLE, thyrotoxicosis, rheumatoid arthritis, gout, BPH, cirrhosis, Addison's disease, each = 1.
HIV infection was diagnosed in 97 (55.75%) individuals, of which 24 (24.7%) were receiving antiretroviral treatment when TMB was diagnosed. The median CD4+ cell count and the percentage at the time TBM diagnosis were 89 (IQR: 45.5-167.5) cells/mm3 and 10% (IQR: 5%-14%), respectively. Most patients (52 individuals, 53.6%) had CD4+ cell counts ≤100 cells/mm3 prior to the initiation of TBM treatment. Antiretroviral therapy (ART) was initiated before TBM was diagnosed for 30 (30.9%) individuals, of which ART treatment was discontinued for 6 (6.2%) due to a loss to follow-up. The remaining 67 (69.1%) were ART naive.
Regarding the treatment outcome, treatment was successful in 53 (30.5%) patients. Among the remaining 121 patients for whom the treatment outcome was not successful or was unknown, 32 (18.4%) cases were lost to follow-up, 5 (2.9%) cases were transferred out, and 84 (48.3%) cases were deceased, of which 44 (52.4%) cases of death occurred in less than 30 days after TBM diagnoses. After anti-TB drugs were initiated, drug-induced hepatitis and/or rash occurred in 36 (20.7%) patients.
Univariate and multivariate analyses of the estimated odds ratio (OR) and 95% confidence interval (CI) of the patient's characteristic in terms of HIV status are shown in Table 2. For baseline characteristics, univariate analysis revealed that HIV-infected patients were significantly more likely to be of younger age (mean ± SD 38.91 ± 9.48 vs. 48.48 ± 16.98 years; P < 0.001) and have a lower body mass index (BMI; mean ± SD 19.45 ± 2.67 vs. 22.28 ± 4.31 kg/m2; P = 0.009) compared to HIV-uninfected patients. HIV-infected patients also had higher rates of HCV coinfection (OR 6.47, 95% CI 1.4-29.86, P = 0.008). In terms of treatment outcome, univariate analysis revealed that HIV-infected patients were significantly less likely to be treated successful (OR 0.487, 95% CI 0.25-0.94, P = .03) than were HIV-uninfected patients. There were no differences in the rate or duration of fever or headache, rate of seizure, alteration of consciousness, or stiffness of neck on clinical presentation. Additionally, there was no difference in the proportion of abnormal radiological findings among TBM individuals with or without HIV infection, which included results for leptomeningeal enhancement, tuberculoma, and hydrocephalus status. Multivariate analysis revealed that in HIV-infected TBM patients, there were higher proportions of patients of a younger age (≤40 years) and patients with a low BMI, history of previous TB infection, or HCV coinfection. A successful treatment outcome rate was lower in HIV-infected patients than in HIV-uninfected patients (OR 0.4, 95% CI 0.20-0.81, P < 0.05). Mortality was observed in 54 (55.7%) of 97 of HIV-infected patients compared with 30 (39%) of 77 of HIV-uninfected patients (OR 1.97, 95% CI 1.07-3.62, P = 0.033). HIV infection is an independent risk factor for an unsuccessful treatment outcome.
Univariate and Multivariate Analysis to Estimated Odds Ratio and 95% Confidence Interval (CI) of Patient's Characteristic and Treatment Outcome in Terms of HIV Status.
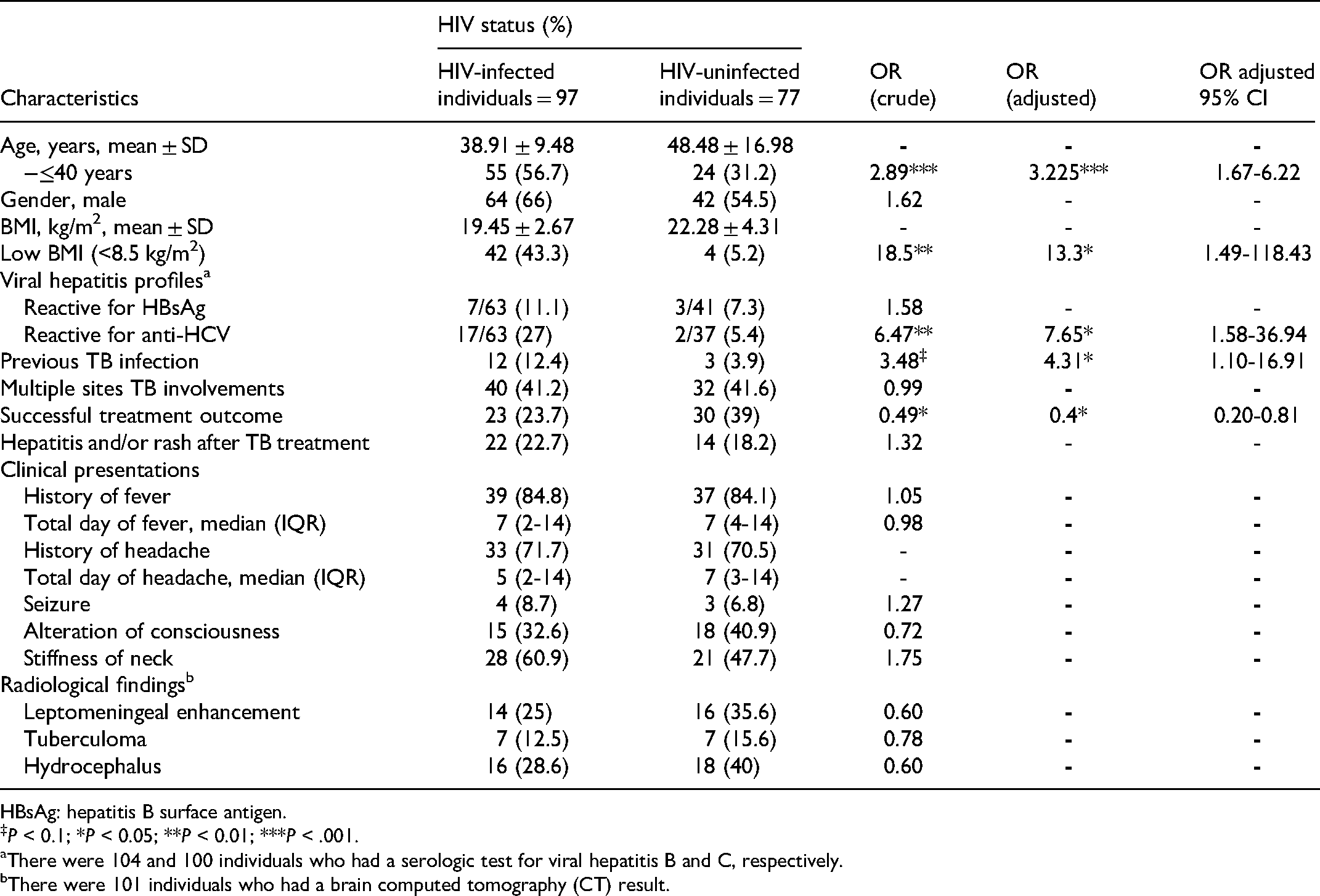
HBsAg: hepatitis B surface antigen.
P < 0.1; *P < 0.05; **P < 0.01; ***P < .001.
There were 104 and 100 individuals who had a serologic test for viral hepatitis B and C, respectively.
There were 101 individuals who had a brain computed tomography (CT) result.
Univariate analysis of the patient's initial laboratory investigations of CSF profiles in terms of HIV status is shown in Table 3. Compared to HIV-uninfected patients, HIV-infected patients were significantly more likely to have a lower hematocrit score (mean ± SD 31.09% ± 6.83% vs. 35.23% ± 5.9%, P < 0.001); lower total white blood cell (WBC) count and absolute lymphocyte count (median total WBC count and absolute lymphocyte count was 5800 vs. 9200 cells/mm3, P < 0.001, and 902 vs. 1009 cells/mm3, P = 0.007); and higher serum globulin level (mean ± SD 4.52 ± .91 vs 3.74 ± .89 g/dL, P < 0.001). The CSF protein was significantly higher in HIV-infected patients (median CSF protein 318 vs. 231 mg/dL, P = 0.034) than in HIV-uninfected patients. There were no statistical differences in the CSF open/close pressure, CSF glucose/blood glucose ratio, CSF WBC count, and CSF absolute lymphocyte count between HIV-infected and uninfected patients.
Univariate Analysis of Patient's Initial Laboratory Investigations and CSF Profiles in Terms of HIV Status.

HIV: human immunodeficiency virus; IQR: interquartile range; WBC: white blood cell.
Kaplan–Meier curves of the time to TBM treatment failure stratified by HIV status are provided in Figure 2. The log-rank test revealed a difference in the time to treatment failure among HIV-infected patients compared with HIV-uninfected patients (P = 0.041).

Kaplan–Meier curves of time to tuberculous meningitis (TBM) treatment failure stratified by human immunodeficiency virus (HIV) status.
Discussion
We reported the results of a retrospective cohort study in 174 adults with TBM. The overall mortality rate was 84 (48.3%) of 174 patients, in which more than 50% of deaths occurred in less than 30 days after TBM diagnosis. This finding is consistent with a previous study by Vinnard et al., 5 in which mortality was reported in 183 (56.5%) of 324 patients. Regarding HIV status, our cohort and their cohort had a comparable proportion of HIV-infected patients (55.75% patients in our cohort compared with 63% patients in their cohort). A lower mortality rate was shown in previous studies from Christensen et al. 6 and Soria et al. 20 in which mortality was 32.7% and 30.4%, respectively. This could be explained by the low proportion of HIV-infected patients in their cohorts. In our cohort, the rate of mortality for HIV-infected patients was almost 15% higher than that for HIV-uninfected patients with an OR of 1.97.
In our cohort, a higher proportion of low BMI and lower age was observed in HIV-infected patients, which could be explained by advanced-stage HIV infection in our cohort, as our median CD4+ cell count at the time of TBM diagnosis was only 89 cells/mm3. In addition, more than 50% of patients had CD4+ cell counts of ≤100 cells/mm3 prior to initiation of TBM treatment. The advanced stage of HIV infection leads to wasting and malnutrition. These findings were consistent with previous studies from Thwaites et al., 26 which reported that patients with HIV-associated TBM were significantly more likely to be younger and weigh less. Moreover, the advanced stage of HIV infection reflects a high degree of systemic immune suppression, which could explain the lower hematocrit score, WBC count, and absolute lymphocyte count in HIV-infected TBM patients in our cohort. Compared to other chronic meningitis, such as cryptococcal meningitis, there was a study by Lee et al. 27 reported that HIV-infected patients with cryptococcal meningitis also have younger age compared to HIV-uninfected patients. This finding is similar to our study. Another study from Qu et al. 28 compared the clinical features of cryptococcal meningitis and TBM in HIV-uninfected patients. This study revealed that younger age, lower CSF WBC counts, and total protein were observed in cryptococcal meningitis compared to TBM patients.
From our findings, serologic evidence of HBsAg and anti-HCV in HIV-infected TBM patients were 19% and 9.62%, respectively. Our study found that a higher proportion of HIV-infected patients had HCV coinfection and/or a previous history of TB infection, which is consistent with previous epidemiological studies regarding the pattern of HIV infection in the populations of Thailand29-31 and other parts of the world.19,20,26 Regarding treatment outcomes, HIV-infected TBM patients in our study were associated with poor prognosis, higher likelihood of treatment failure, and/or death. This finding is similar to the previous studies in TBM patients.5,17,19,20,26,32,33
Our study found no difference in clinical presentations, including a history of fever, headache, total days of fever and headache before admission, seizure, alteration of consciousness, and stiffness of the neck, among patients in our study cohort. This is consistent with a previous study from Berenguer et al. 33 These results indicate that HIV does not alter the presenting neurological symptoms of TBM.
CSF examination in TBM patients with and without HIV infection revealed similar profiles of abnormalities. Our study confirmed other previously reported20,26 findings that found no significant differences in CSF characteristics (CSF opening/closing pressure, CSF glucose/blood glucose ratio, CSF WBC, and lymphocyte count) between TBM patients with and without HIV infection, except for the CSF protein level, which was higher in HIV-infected patients than in HIV-uninfected patients, which differed from previous studies.19,20,26 A study by Berenguer et al. 33 reported that a normal CSF protein concentration was found more frequently in HIV-infected TBM patients, although the difference did not have a statistical significance. With regard to CSF protein, another study by Cecchini et al. 34 also reported the lower CSF protein levels in HIV-infected patients with TBM compared to HIV-uninfected patients. In their study, the median CSF protein in overall HIV-infected patients with TBM was 77 mg/dL (IQR 37-145 mg/dL). When stratified by the level of CD4+ cell counts, HIV-infected patient with higher CD4+ cell counts (>50 cells/mm3) was reported a higher median CSF protein more than HIV-infected patients with lower CD4+ cell counts (<50 cells/mm3) with the median CSF protein of 122 (IQR 66-199) mg/dL compared to 65 (IQR 24-127) mg/dL, respectively. In our study, there were 71 (73.2%) of 97 patients with CD4+ cell counts of more than 50 cells/mm3. This could explain the higher CSF protein level in HIV-infected patients with TBM in our study.
The strengths of our study include a large sample size of our cohort that represented a wide range of disease severity. Moreover, the inclusion criteria and outcome definitions for TB diagnosis were comparable with those used by other studies and strictly adhered to the international guidelines of TB diagnosis and outcome definitions.23-25 However, our study had limitations. First, this study was conducted in a single hospital, and whether the Thai TBM patients in our cohort are representative of TBM patient populations of other geographic areas are unknown. Second, because of the retrospective nature of the study, incomplete data collection may have occurred. However, the missing data in most of these cases were not the main variables of interest. To minimize this effect, we used multiple data sources (electronic, paper-based medical records, and the TB registry book). Mortality in our study was defined as all-cause mortality because the ability to determine TBM-related death was limited by the retrospective nature of the study. Third, accurate TBM diagnosis is limited by a lack of sensitive diagnostic techniques. Thus, it is possible that some cases of TBM were misdiagnosed. Fourth, data may have been insufficient for evaluating the effect of corticosteroids on the treatment outcome owing to the variation in dose and frequency of dexamethasone administration. The prospective data collection could give us more accurate data and decrease these biases.
In conclusion, the results of the present study indicate that HIV infection does not alter neurological features, CSF profiles (except higher CSF protein in HIV-infected patients), or radiological findings of TBM. Therefore, further studies are needed to determine the clinical characteristics that lead to poor prognosis of HIV-infected TBM patients our findings help eliminate clinical factors that did not differ among HIV-infected and HIV-uninfected TBM patients. In terms of outcome, HIV infection decreases the chance of successful treatment outcome and survival. We recommend early diagnosis and treatment as a strategy for improving the treatment outcome of TBM. This finding could help physicians develop early intervention strategies and more effective treatment approaches for TBM patients, especially in HIV-infected patients, to improve the quality and outcome of TBM care.
Footnotes
Acknowledgments
The authors would like to thank all of the staffs of the TB Clinic and Infectious Diseases Division in Vajira Hospital, Navamindradhiraj University, Bangkok, Thailand, for their help and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Navamindradhiraj University Research Fund (grant number COA43/2560).
