Abstract
Background:
The implication and clinical significance of low-level viremia (LLV) in HIV patients are still not clear. This study aimed to characterize the clinical outcomes and to evaluate whether LLV could predict future virological failure in a well-defined cohort of HIV-infected Omani patients attending a large HIV clinic.
Methods:
Patients on regular antiretroviral therapy (ART) for at least 12 months, and had at least 2 HIV RNA measurements 1 year after starting ART, were prospectively enrolled in a cohort study. LLV was defined as plasma HIV RNA between 50-200 copies/mL that persists after at least 2 consecutive measurements after 12 months of ART. Multivariate Cox proportional hazards regression model was used to measure the association among virological failure, LLV and potential predictors.
Results:
After 12 months of starting ART, 60 patients (40%) had undetectable viral load (UVL) < 50 copies/mL, while 37 patients (24%) had LLV and 53 patients (35%) had primary virological failure > 200 copies/mL. The incidence rates of subsequent secondary virological failure for UVL and LLV groups, were 3 and 7 cases per 1000 patient-months, respectively. Compared to UVL group, LLV group had increased risk of subsequent secondary virological failure with hazard ratio of (4.437 [95% CI, 1.26-15.55]; p = 0.02). Age, duration of HIV infection, pretreatment HIV RNA level, pretreatment CD4+ cell count, and ART adherent were associated with subsequent secondary virological failure.
Conclusion:
Collectively, Omani HIV patients with LLV were at a higher risk for HIV virological failure, and should be monitored closely. Further studies are need to assess whether ART modification in LLV patients would lower the risk of virological failure.
What Do We Already Know about This Topic?
The clinical implications and future directions for management of HIV-infected patients with low-level viremia (LLV) are still unclear in the Arab Gulf states context.
How Does Your Research Contribute to the Field?
We assessed the implication and factors associated with LLV in a well-defined cohort of HIV-infected Omani adult patients attending a large HIV clinic.
What Are Your Research’s Implications toward Theory, Practice, or Policy?
Our findings indicate that patients with LLV were at increased risk of subsequent virological failure, and designing efficient strategies for optimizing HIV treatment outcomes in these patients are highly needed.
Introduction
Human immunodeficiency virus (HIV) infection remains one of the top 10 causes of death in young adults. Untreated and uncontrolled HIV infection continued to firewood HIV pandemic. Therefore, HIV incidence among young adults is increasing and its mortality rate is plateauing. 1,2 Despite the recent advances in HIV treatment strategies, there is a gap in the medical care provided even in rich resources countries. For instance, among HIV-infected patients only one-third are taking antiretroviral therapy (ART), and of those only one-fourth have suppressed HIV viral load. 3 Unsuppressed HIV viral load during ART therapy is often due to ART noncompliance, infection with drug-resistant HIV strains or taking an inappropriate ART. 4 Poorly controlled HIV infection increases the risk of death, transmission of the HIV infection and emergence of HIV drug resistance.
Currently, HIV RNA level is the most important indicator for HIV treatment outcomes. World Health Organization (WHO) and US guidelines define HIV treatment failure as a confirmed HIV RNA ≥ 1000 and ≥ 200 copies/ml, respectively. 2,5 Moreover, HIV treatment success is defined in most guidelines as HIV RNA below the level of detection, depending on the assay used, usually HIV RNA < 20 to 75 copies/mL. However, low-level viremia (LLV) between 50-200 copies/mL is common among treated HIV-infected patients and elite controllers. 6,7
To date, the implications and clinical significance of LLV are still not clear. Additionally, there is no consensus on how to manage those patients with LLV. Therefore, it is crucial to examine the effect of LLV on patients’ clinical outcomes. Herein, we aimed to illustrate LLV clinical outcomes and to explore whether LLV could predict future HIV virological failure in a well-defined cohort of HIV-infected Omani patients attending a large HIV clinic.
Patients and Methods
Study Population and Data Collection
This was a prospective cohort study conducted in outpatient infectious diseases clinic at Sultan Qaboos University Hospital (SQUH), a tertiary care center in Oman. This clinic provides free of charge medical care and ART for HIV patients. We included HIV-infected patients older than 18 years, who were taking ART for at least 12 months and had at least 2 HIV RNA measurements 1 year after starting ART. We excluded patients who were not taking ART, died, lost follow-up or discontinued ART during first year of starting ART. For each patient, the socio-demographic characteristics, clinical and laboratory data, treatment details and outcomes were collected using a special case report form. ART Adherence was measured by pharmacy-refill method at the most recent clinic visit as previously reported. 4
Outcome Measurement
The primary study endpoint was virological failure, which was defined as 2 consecutive measurements of HIV RNA level of ≥ 200 copies/mL by or after 12 months of starting ART, as defined by United States Department of Health and Human Services. 5
Definition Viral Suppression
Virological suppression was defined as a confirmed HIV RNA level below the level of detection. Virological failure was defined as the inability to achieve or maintain suppression of viral replication to an HIV RNA level < 200 copies/mL by or after 1 year of starting ART. The LLV was defined as plasma HIV RNA between 50-200 copies/mL that persists after at least 2 consecutive measurements after 12 months of ART.
Plasma HIV Determination
Plasma HIV viral load was measured by COBAS TaqMan 48 (Roche, France), as previously reported. 4 The lower limit of HIV RNA detection was 20 copies/mL.
Lymphocyte Subsets Determination
Lymphocyte subsets were determined on fresh blood samples, routinely at 3 months apart during the follow-up visits. Two sets of 4-color monoclonal antibody combinations were used (Cyto-Stat TetraChrome, Beckman Coulter, USA) to assess lymphocytes subsets using flow cytometry, as previously reported. 4
Ethical Consideration
The study was approved by the institutional review board at Sultan Qaboos University, Oman.
Statistical Analysis
Normally distributed continue variables were calculated as mean and standard deviation, otherwise reported as median and interquartile range. Categorical variables were calculated as proportions. Comparisons were performed by Pearson chi square and Fisher exact tests in case of categorized variables and student t-test for continues normally distributed data. In case of non-parametric variables Wilcoxon Rank-Sum test was used. Virological failure was investigated as a time-dependent variable. Patients was entered survival analysis at 12 months of ART if their viral load remained <200 copies/ml. Patients were followed until virological failure occurred or censored at the time of the most recent clinic visit. Multivariate Cox proportional hazards regression model was used to measure the association between virological failure and level of viremia. Schoenfeld residual tests for time, log and rank were used to assess the proportional hazards assumption test. Data was analyzed using STAT software version 13. A P-value of < 0.05 was considered statistically significant.
Results
A total of 153 patients were enrolled in this study, where 3 patients were excluded as they lost clinic followed-up visits. The median duration of follow-up of this cohort was 48 months, interquartile range (IQR) (30-72) months. Table 1 shows the demographic characteristics of the study population. The mean age of the study cohort was 44 years, and (59%) of them were males. The median duration of HIV infection was 7 years and IQR was (4-10) years. The median pretreatment CD4+ cell count was 171 cells/mm 3 and IQR was (48-385). The median pretreatment HIV RNA was 4.439 log10 copies/mL with IQR (3.19-5.21) logs copies/mL. All enrolled patients were on ART that included 2 non-nucleoside reverse transcriptase inhibitors (NNRTIs) plus either protease inhibitor (PI) or non-nucleoside reverse transcriptase inhibitor (NNRTI). The most common backbone was Tenofovir plus Emtricitabine (36%) followed by lamivudine plus zidovudine (28%). Only (5%) patients were on integrase strand transfer inhibitor (INSTI).
Baseline Characteristics of Study Population.

IQR: interquartile range; SD: standard deviation; HIV: Human immunodeficiency virus; RNA: ribonucleic acid; ART: active antiretroviral therapy; NNRTIs: Non-nucleoside reverse-transcriptase inhibitors; NRTIs: Nucleoside reverse-transcriptase inhibitors; PI: Protease Inhibitors; INSTI: Integrase strand transfer inhibitor.
After 12 months of starting ART, 60 patients (40%) had undetectable viral load (UVL) < 50 copies/mL, while 37 patients (24%) had LLV between 50 to 200 copies/mL and 53 patients (35%) had virological failure > 200 copies/mL. The incidence rates of subsequent secondary virological failure for UVL and LLV groups, were 3 and 7 cases per 1000 patient-months, respectively. The incidence rate difference was 0.004 (95% CI, [−0.001 to 0.009], p = 0.06).
The univariate analysis of variable associated with subsequent (secondary) virological failure for UVL and LLV groups, is shown in Table 2. Of note the secondary virological failure was defined as the inability to achieve or maintain suppression of viral replication to an HIV RNA level <200 copies/mL after initial complete suppression of HIV RNA < 50 copies/ml. Age, duration of HIV infection, pretreatment HIV RNA, pretreatment CD4+ cell count, ART adherent and taking NNRTI were found to be associated with subsequent secondary virological failure.
Patients with Undetectable and Low-Level Viremia after 12 Month of Antiretroviral Therapy.
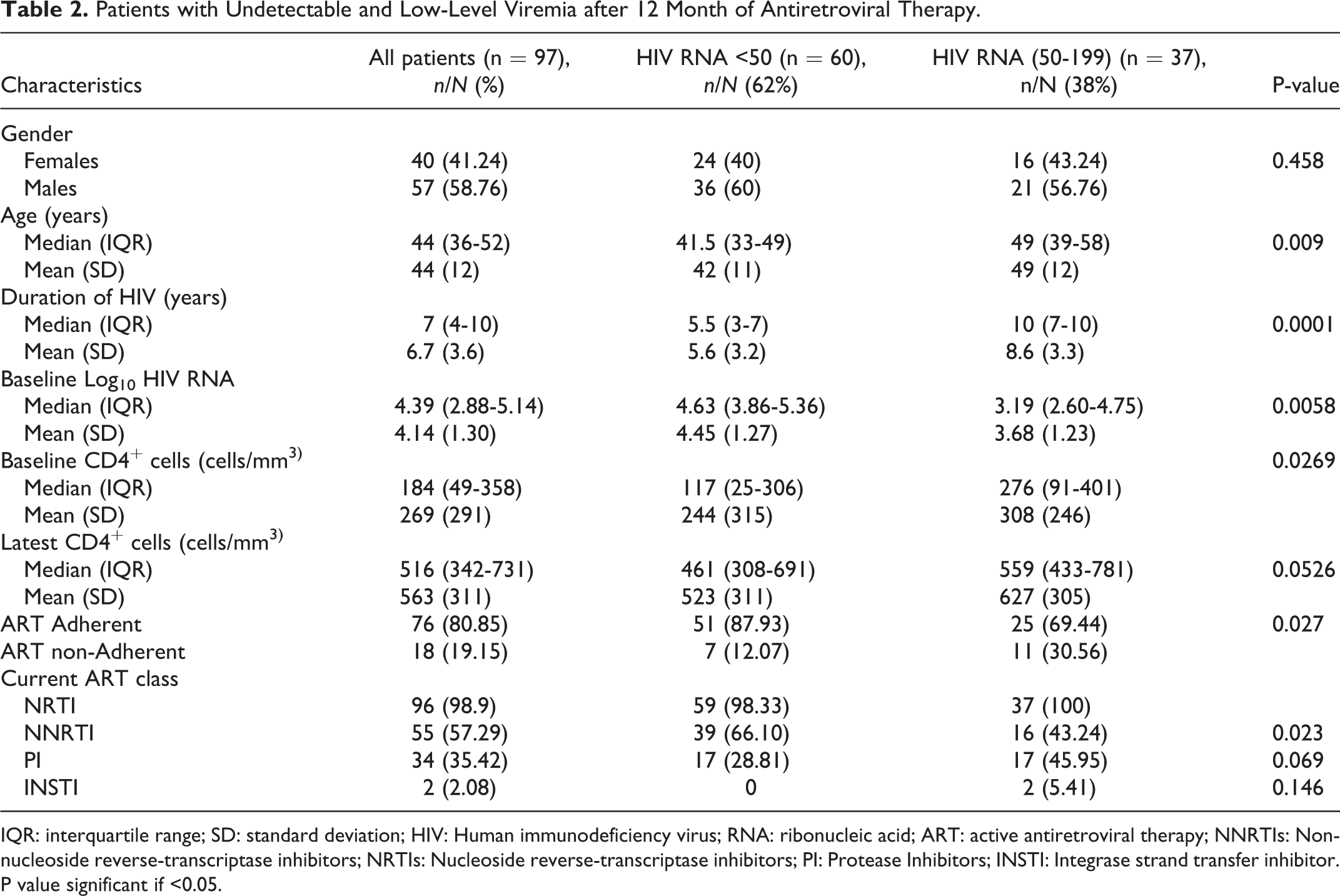
IQR: interquartile range; SD: standard deviation; HIV: Human immunodeficiency virus; RNA: ribonucleic acid; ART: active antiretroviral therapy; NNRTIs: Non-nucleoside reverse-transcriptase inhibitors; NRTIs: Nucleoside reverse-transcriptase inhibitors; PI: Protease Inhibitors; INSTI: Integrase strand transfer inhibitor. P value significant if <0.05.
In the adjusted Cox proportional hazards analysis of patients with UVL and LLV groups, the median survival analysis time was 36 IQR (24-66) months. Compared to UVL group, LLV group had increased risk of subsequent secondary virological failure with hazard ratio (HR) of (4.43 [95% CI, 1.26-15.55]; p = 0.02). In the same model, higher pretreatment HIV RNA level increased the risk of subsequent virological failure with a HR of 1.005 [95% CI, 1.002-1.008] compared to lower pretreatment HIV RNA level (p = 0.003).
Discussion
As the association between LLV and treatment failure is still unclear, this study explores whether LLV predicts virological failure among HIV-infected Omani patients attending a large HIV clinic. Our data revealed that patients who have LLV between 50-200 copies/mL were at a higher risk for HIV virological failure compared to patients with undetectable viremia of less than 50 copies/mL. Our findings are in line with a number of recent studies showing that LLV may increase the risk of virological failure, although these studies differ in terms of demographic characteristics of the participants and their racial and ethnic origin. 7 -10 Thus, our findings confirm and support the evidence that LLV should be considered an early warning sign for virological failure. One possible mechanism by which LLV leads to virological failure is that it stimulates the appearance of HIV resistant strains. This assumption is supported by several studies showing that persistent LLV predisposes to virological failure through emergence of HIV resistant strains. 11 -13
However, our results are at variance with those reported recently by Bernal et al., where patients having a LLV (50-199 copies/mL) were not associated with a higher risk for HIV treatment failure. 14 These conflicting results could be attributed to a number of reasons. Firstly, the two studies differ significantly in the criteria used to classify the study participants. In their study virological failure was defined as at least two consecutive viral loads of more than 500 copies /mL, which is higher than the value used in our study, and even higher than the value that defines virological failure in most guidelines. Secondly, they used different HIV RNA assays, which may have caused discrepancies in quantification of LLV. Another study also found a weak association between LLV (50-199 copies/mL) and virological failure. 9 This study had a relatively short follow-up period, and it defined virological failure as viral load >500 copies/mL.
Currently, there is no consensus on how to manage patients with LLV. Many HIV treatment guidelines consider LLV insignificant, and it is not considered indication for ART modification. 2,5 However, emerging evidences continue to support the impact of LLV in imminent HIV treatment failure. Moreover, LLV and residual viremia cause chronic immune activation, which is associated with increased morbidity and mortality in HIV-infected patients. 15,16 Although the source of LLV is still not well understood, it was postulated it origins from lymphatic tissues reservoirs. 17
In the past, LLV management was hindered by the limited options for effective tolerable ART, and the uncertainty of HIV resistance testing performance for a viral load <1000 copies/mL. Currently, available ART are more tolerable and effective and recent studies support using HIV resistant testing at a viral load lower than 1000 copies/Ml. 11,18 Altogether, these studies suggest and call for an improvement in LLV management. Performing HIV resistant testing in LLV and optimizing of ART seem to be a solution for LLV. Cautiously, ART adjustment may be ineffective in reducing LLV, which may originate from lymphatic tissue reservoirs or other sources. 19 Importantly, LLV may be even suppressed without ART, as evidence suggests that (6.7%) of newly diagnosed HIV patients have viral load of < 400-500 copies/mL 5 years after HIV seroconversion. 7
This study has several limitations. The HIV resistant testing was not available to detect HIV resistant strains as a cause of LLV. Consequently, whether LLV is due to ART noncompliance or HIV drug resistant needs to be further studied. The current study cohort did not include enough number of patients who were taking integrase inhibitors, which may have different effects of LLV. However, it is unlikely that the above mentioned two factors have affected our study outcomes. Lastly, our sample size was relatively small as the study was conducted in low-incidence HIV area. Nonetheless, the results were significant despite the small cohort size.
In conclusion, our results indicated that Omani HIV-infected patients with LLV were at a higher risk for HIV virological failure, and should be monitored closely. Further studies are need to assess whether ART modification in LLV patients would lower the risk of virological failure.
Footnotes
Authors’ Note
The datasets generated and/or analyzed during the present study are not publicly available due to SQU regulations, but are available from the corresponding author on reasonable request.
Acknowledgments
We are thankful to all participants and also to the HIV Clinic Nurses for providing assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported in part by a grant from the Sultan Qaboos University (grant no. IG/MED/ MEDE/16/01).
