Abstract
Background:
The rollout of antiviral therapy in Low and Middle Income Countries (LMICs) has reduced HIV transmission rates at the potential risk of resistant HIV transmission. We sought to predict the risk of wild type and antiviral resistance transmissions in these settings.
Methods:
A predictive model utilizing viral load, ART adherence, genital ulcer disease, condom use, and sexual event histories was developed to predict risks of HIV transmission to wives of 233 HIV+ men in 4 antiretroviral treatment centers in Maharashtra, India.
Results:
ARV Therapy predicted a 5.71-fold reduction in transmissions compared to a model of using condoms alone, with 79.9%, of remaining transmissions resulting in primary ART-resistance.
Conclusions:
ART programs reduce transmission of HIV to susceptible partners at a substantial increased risk for transmission of resistant virus. Enhanced vigilance in monitoring adherence, use of barrier protections, and viral load may reduce risks of resistant HIV transmissions in LMIC settings.
What Do We Already Know About the Topic?
Antiretroviral therapy in LMIC settings substantially reduces transmission rates of HIV to sexual partners.
How Does Your Research Contribute to the Field?
This research models the likelihood of HIV transmission events resulting in primary resistance in the context of LMIC antiretroviral therapy programs.
What Are Your Research’s Implications Toward Theory, Practice, or Policy?
This results of this research suggest that more frequent monitoring of sexually active HIV-infected patients for evidence of laboratory evidence of virologic failure or suboptimal medication adherence may help reduce the potential for sexual transmission of resistant HIV in the LMIC setting.
Introduction
The concept of “treatment as prevention” 1 has gained primacy in addressing HIV/AIDS. The rollout of free or low cost antiretroviral therapy (ART) programs to treat HIV-1 infections worldwide has not only improved the quality and longevity of the lives of persons living with HIV (PLHIV), but it has also resulted in a 50% reduction in AIDS incidence transmission rates, as shown in 25 low and middle income countries (LMICs) from 2001 to 2011. 2 The implementation of antiretroviral therapy programs have led to significant reduction of transmission to seronegative heterosexual partners. 3,4 However, a significant subset fail to show sustained reductions in viral load after initiation of ART due to pharmacologic issues related to resistance, and/or dosing inconsistencies, resulting in transmission risk to seronegative sex partners. Antiretroviral therapy can be associated with development of ART resistant variants, which may not only lead to treatment failure in the index patient, but may be transmitted to other partners, limiting response to ART for the newly-infected partner. 5 Substantial increases in prevalence of drug resistance since initiation of ART rollout programs in LMICs have been noted since the initiation of ART programs. 6 In LMICs, the frequent use of ART medications, characterized by a low genetic barrier to resistance, has significant potential to lead to antiviral resistance in an individual’s circulating viral isolates, with subsequent potential for sexual transmission of ART-resistant virus to the partner. A 6-year project focused on alcohol-consuming men on ART in Mumbai generated clinical data and reported behaviors to develop a model of risk of seroconversion among the wives of sexually active study participants.
Methodology
Data for this paper were collected as a part of the NIAAA-funded, Indo-U.S. research and intervention project entitled “Alcohol and ART Adherence: Assessment, Intervention and Modeling in India,” clinical trial # NCT03746457, listed at ClinicalTrials.gov. The project involved a formative qualitative and quantitative data collection stage followed by interventions at the individual, group, and community levels. Inclusion criteria were 18 years of age and above, treatment with ART for 6 months or more, and having consumed alcohol at least once in the last 30 days. Project research staff administered an anonymous screener to establish eligibility to male PLHIV waiting to receive care and medication pickup in the 5 centers. A total of 9,954 patients were screened to generate a sample of 940 alcohol consuming male PLHIVs, 188 in each of the 5 ART centers. When PLHIV met the criteria, they were asked to participate in the project and provide written consent. They were then administered the baseline survey and viral load testing (not a part of the standard operating procedure for ART programs in India).
The baseline survey instrument included sociodemographic variables, patient-reported serostatus of their wives, ART adherence, and sexual transmission risk factors including numbers of sexual exposures, use of condoms and presence or absence of self-reported sexually transmitted diseases including genital ulcer disease (GUD). HIV-1 Viral load was measured using Roche Amplicor PCR (Roche, Indianapolis, Indiana, USA). Of the 940 male PLHIVs, 419 reported ongoing behaviors potentially leading to transmission to wives (Figure 1).

Enrollment.
Ethical Approval and Informed Consent
The study was approved by the University of Connecticut Health Center Institutional Review Board (reference #IE-13-0270-2), the India Council for Medical Research, the Health Ministry Screening Committee, National AIDS Control Organization, and the institutional review boards of all collaborating agencies and hospitals. Written informed consent in Hindi or Marathi was obtained from all participants.
Analytical Process
Three hypothetical scenarios were developed to characterize potential HIV viral transmission to susceptible wives. We developed a mathematical model that incorporated self-reported frequencies of coital activity, condom use, medication adherence, presence of genital ulcer disease, as well as measured viral load data. A baseline / no intervention scenario calculated expected HIV transmissions without condom use and without ART-related viral suppression. Reported condom use was added to the model to predict HIV transmission rates in the setting of condom use but in the absence of antiretroviral therapy programs. Finally, medication adherence and ART-related viral control were then added to model transmissions of ART-sensitive and ART-resistant virus to model the effect of ART programs on viral transmission characteristics. Each serodiscordant couple’s viral transmission characteristics were calculated as an annualized risk of HIV transmission within that couple, and then further characterized with the likelihood of transmission of an ART sensitive / wild type virus or ART resistant virus. Calculated transmission rates were adjusted to a standard population measurement and reported as an expected incidence rate. Calculated incidence rates from the ART + condoms intervention were compared to the condoms-only scenario to calculate a Number Needed to Treat with ART to prevent one HIV transmission event. For seroconcordant couples (e.g. HIV+ wife), exposures with the potential to result in HIV superinfection with ART-resistant virus were noted.
Defining Key Variables
Whereas
r = risk of transmission per coital event
c = hazard ratio per coital event imparted by condom use
g = hazard ratio, per coital event, imparted by presence or absence of GUD
eu = annualized number of sexual events when a condom was not used
ec = annualized number of sexual events when a condom was used
Risk per coital event was calculated using the metanalysis of Boily et al, 10 defining the risk of transmission of male to female vaginal penetration at 0.003 (CI 0.0014-0.0063) per event in developing countries. Condom effectiveness in preventing HIV transmission was calculated at 95% on a “per event” basis using data by Pinkerton et al. 11 Impact of GUD was modeled using Boily et al 10 suggesting a 5.3-fold (CI 1.4-19.5) HIV transmission risk per act risk when GUD was present.
For modeling purposes, subjects with serum viral loads of <1000 copies/mm3 were defined as unlikely to transmit HIV to the partner regardless of reported adherence. To model transmission of resistant virus, a husband was presumed to harbor resistant virus as the predominant quasi-species, and thus would be at risk of transmitting a primary resistant isolate, when adherence was reported at more than 50% in the setting of detectable viremia of >1000 copies/ml.
Results
Demographic Characteristics and Key Variables
Table 1 presents the baseline characteristics of the sample of male PLHIV, their wives, and potential HIV sexual transmission behaviors (N = 419). About half of the wives of HIV+ men were either seronegative or sero-unknown. Of wives who were HIV positive, 82.5% were receiving ART. Men reported a high level of adherence, yet 38% had detectable HIV viremia within the preceding 3 months, with 20% having detectable viremia of at least 1000 copies/µl. Husbands reported an annualized rate of 15,288 coital events with their wives, of which 70.4% employed a condom. There was no significant difference in condom use between couples that were serodiscordant and seroconcordant (X2 = 0.36, p > 0.05)
Baseline Characteristics (n = 419).

Transmission to Seronegative Wives
Table 2 presents calculated transmission rates using reported behaviors based on scenarios of baseline risk, condom utilization, and condom utilization plus antiviral therapy roll-out among 233 HIV-negative wives.
Modeling ART Sensitive and ART Resistant Virus Transmission.
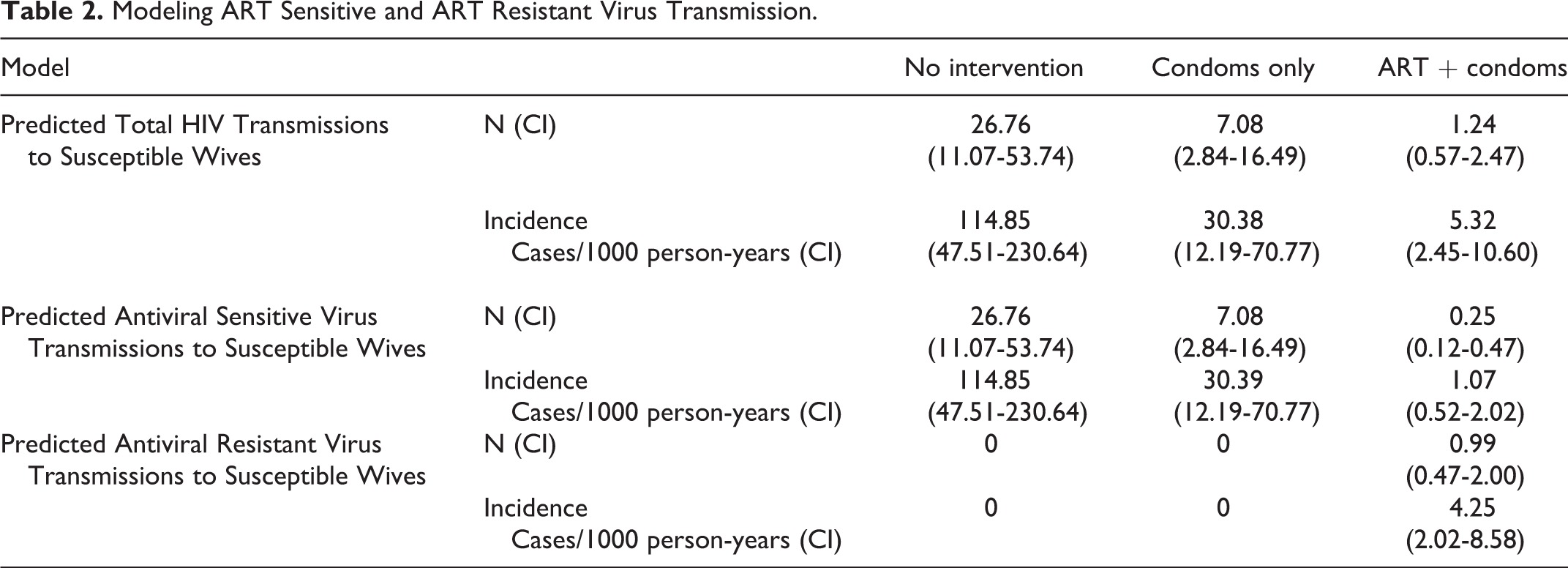
The predicted incidence rate of HIV infection in susceptible wives was 114.85 cases/1000 person-years without use of condoms or ART. Use of condoms at the reported rate of 70.4% of coital events would lead to a 3.78-fold reduction in transmissions to 30.39 cases/1000 patient years. The rollout of ART services leads to an additional 5.71-fold reduction in transmissions to 5.32 cases/1000 person-years. The Number Needed to Treat with antiviral therapy to prevent one transmission to a wife was 35.11. Coincident with the reduction due to ART is the recognition that of the remaining transmissions, there would be an incidence of 4.25/1000 person-years of primary ART-resistant transmissions, representing 79.9% of these transmission (Figure 2). Over the 3 year study, only one wife was reported to have seroconverted (incidence rate of 1.43 / 1000 person years). Virologic data on primary resistance is not available.

Modeled incidence of HIV transmission.
Superinfection of Seropositive Wives
We identified a significant number of exposures that could potentially lead to superinfection of HIV+ wives. Of seropositive wives, 82.8% were taking HAART and 17.9% of husbands met the case definition for harboring resistant virus. Because rates of HIV superinfection have not been well established, with reports ranging from none 12,13 to 5% per year, 14 the current analysis was restricted to serodiscordant couples.
Discussion
We have presented a model for predicting the impact of an ART program in an urban setting in a LMIC using a cumulative infection risk calculation based on individual partner characteristics and behaviors. ART programs have led to substantial reductions in morbidity and mortality in LMICs. Other studies have evaluated the impact of ART programs on morbidity and mortality, 15 -19 population-based incidence rates, 20 or have focused on the cumulative effect of transmission behaviors measured over time. 21 This study applies previously-demonstrated metrics to the behavioral and biological characteristics of individual HIV-discordant partnerships to predict risks of HIV transmission to individuals and transmissions within the cohort as a whole, including reductions in the rate of HIV transmissions. However, these reductions are associated with an increased risk of transmitting a virus that would be resistant to antiviral therapy in the newly-infected partner.
A baseline scenario modeled transmissions without benefit of the public health interventions of condom promotion or ARVs. This scenario predicted HIV transmission rates similar to prospective cohorts measuring male-to-female transmission in discordant couples in other settings including Uganda, 22 -24 Tanzania 25 and China. 26 The addition of condom promotion without ARV therapy to the model predicted a rate of transmission consistent with other observed cohorts with high rates of condom utilization. 27
Reduction in viral burden to levels below detection lead to transmission rates approaching zero even in the absence of condoms. 4 In the LMIC settings, condom use and ART-mediated viral suppression are imperfect, and are likely to reduce overall transmissions but increase the rate of resistant virus transmission. There is limited data and much less discussion of the transmission of superinfection to seropositive HIV wives and regular partners in the LMIC setting.
As with any modeling technique, accurate definition of the variables is necessary for overall accuracy of the model. A wide range of risk has been attributed to factors such as viral load, CD4 count, circumcision, GUD and other sexually transmitted diseases, age, pregnancy status, as well as specific sexual behavior. 8 Host susceptibility through individual immune responses 28 and viral factors 29 may also play a role. In this cohort, detailed information on frequency and type of sexual contact, use of condoms, viral load were used with a case definition for determining if resistant virus was likely to be transmitted was employed. Other factors, including STDs other than self-reported GUD, circumcision, and CD4 count were not included in the present model.
Antiviral therapy may lead to altered risk behaviors. Because of improvements in morbidity and general health, PLHIV may feel healthy enough to be more sexual active. 30 More than 96% of the men reported active employment, indicating the impact of HAART on maintaining functional status. The impact of ART on serum viral load and subsequent reduction of transmissibility may lead a sense of security about being non-infective, leading to more risky transmission behaviors 31,32 and subsequent STI incidence. 33
Superinfection between hetero-concordant partners may lead to drug resistance, virulence, or altered cell tropism that can compromise the effectiveness of antiviral therapy, 34 leading to subsequent virologic rebound 35 and clinical deterioration of the recipient partner. 36 -38 Modeling superinfection may be complex 39 and not fully understood. Thus the potential for superinfection from husbands to HIV+ wives and vice-versa, were not modeled in this cohort. However, a large number of exposure events that could potentially lead to transmission of resistant virus between HIV-positive spouses was identified.
The case definition of resistance was developed assuming that a significantly detectable viral load despite adequate medication adherence would predict the presence of a resistant viral quasi-species. However, detectable viremia may be due to other factors not addressed in this evaluation, including medication absorption, drug interactions, genetics, viral factors, and other factors. In LMICs, antiviral resistance may be fostered by regimens with a low genetic barrier to resistance, drug-drug interactions, interruption of drug supplies, poor retention in care, and infrequent viral load testing. 40 In this evaluation, a cut point of ≥ 50% adherence was arbitrarily chosen as a threshold above which enough drug pressure was likely to be in place to promote resistance. Most subjects reported either very high or very low adherence. The majority (n = 352, 84.0%) of participants reporting perfect adherence, with only 24/419 (5.8%) reporting adherence between 50 and 75% of doses taken.
HIV is more likely to be transmitted with higher degrees of plasma viremia. 41 Reductions in viral load through the use of ART reduces viral transmission 42,43 from males to females primarily through reduction of HIV in semen. 44,45 A dose-response effect of viral load was seen with increasing viral load in male to female transmissions with no transmissions seen at < 1094 copies/ml3 and <1500 copies/ml3 in other LMIC settings. 23 However transmission from patients with non-detectable plasma viremia have been described. 46 Viral load “blips” are not associated with a high risk of clinical progression or virologic or immunologic deterioration. 47 Because of the potential that a single viral load represents a “blip” when <1000 copies/ml, and cohort studies indicating limited, if any, transmission at this level, a value of 1000 copies/ml was chosen in the model as the cut point for determining transmissibility.
These study results have limitations. The sample consists of men who have consumed alcohol in the last 30 days and should not be generalized to non-alcohol consuming PLHIV in India. Rates of sexual exposure to nonwives and regular partners (commercial sex partners, MSM, single night encounters) is very low in this cohort as compared to HIV positive individuals in other States of India and other countries. The study group was from a large urban area and should not be generalized to rural PLHIV. This project was over-represented by a single religious background (Hindu), and thus may not be reflective of ethno-religious groups from other communities. Factors affecting transmission such as viral load, medication adherence, sexual activity and use of condoms may vary over time. This analysis used a self-report of 1 month’s reported behaviors at single time point to extrapolate risk longitudinally.
Risk assigned to differing types of sex acts, GUD, and efficacy of condoms has been variable across studies and may not appropriately reflect actual risk in this specific cohort of women married to HIV+ men in Mumbai, India. In addition, data on other factors known to impart risk of transmission, such as GUD, pregnancy status, oral contraceptive use and specific sexual behaviors were not elicited in the survey and therefore could not be incorporated into the model. Infectiveness in this model was characterized dichotomously as “transmissible” or “nontransmissible” based on a single viral load determination with a specific cut point. Genetic resistance data were not collected routinely with detectable viral loads and were thus not available for modeling the transmission of resistant virus. Antiviral resistance was imputed based on measured detectable viral loads and reported medication adherence. Wives were not able to be formally assessed for HIV serostatus, phylogenetically-linked transmissions or resistance.
Implications for Intervention
Antiretroviral therapy has a profound public health impact by reducing transmission rates of HIV in LMIC settings. However, given the enhanced potential for transmission of primary ART resistance as a direct result of ART exposure, continued education regarding condom use should be emphasized to patients. More frequent viral load monitoring of patients in LMICs would better identify increases in viremia that may result in transmission, and allow a more timely approach to develop individual strategies for re-suppression including adherence counseling and ART regimen modifications. More rapid identification of viremia despite high levels of adherence will also help clinicians address antiviral resistance testing through more tailored individualized therapy. Clinicians in LMICs should also focus attention on the serostatus of wives and regular partners and be more active in promoting condom use with both seronegative and seropositive partners in order to prevent new HIV infections as well as superinfections that may impact the partner’s health.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant support for the research was supported by National Institute on Alcohol Abuse and Alcoholism (NIAAA) Grant No. U01AA021990-01, Alcohol and ART Adherence: Assessment, Intervention and Modeling in India, S. Schensul, J. Schensul, N. Saggurti, A. Sarna, MPIs; 2014–2019.

