Abstract
Black men bear a disproportionate burden of HIV infection. These HIV inequities are influenced by intersecting social, clinical, and behavioral factors. The purpose of this analysis was to determine the combinations of factors that were most predictive of HIV infection and HIV testing among black men in Toronto. Classification and regression tree analysis was applied to secondary data collected from black men (N = 460) in Toronto, 82% of whom only had sex with women and 18% whom had sex with men at least once. For HIV infection, 10 subgroups were identified and characterized by number of lifetime male partners, age, syphilis history, and perceived stigma. Number of lifetime male partners was the best single predictor of HIV infection. For HIV testing, the analysis identified 8 subgroups characterized by age, condom use, number of sex partners and Chlamydia history. Age (>24 years old) was the best single predictor of HIV testing.
What Do We Already Know about This Topic?
Individual behavior, social economic status, and HIV-related stigma have been identified as factors influencing HIV infection risk and are consistently linked to delays in HIV testing and negative HIV care continuum outcomes among African, Caribbean, and Canadian black men.
How Does Your Research Contribute to the Field?
To identify the combinations of social, clinical, and behavioral factors that are most predictive of HIV testing and infection in a sample of black men who only have sex with women and men who only have sex with men in Ontario, Canada. The findings of this study could highlight the link between risk factors and HIV transmission which relate to the issue of ending HIV transmission in Canada in the next 10 years.
What Are Your Research’s Implications toward Theory, Practice, or Policy?
The understanding of the combinations of social, clinical, and behavioral factors that could predict HIV infection and testing can help inform strategies for optimizing HIV prevention and care continuum outcomes among black men in Canada.
Introduction
In recent decades, there have been major advances in the prevention of HIV and sexually transmitted infections (STIs). This had led to significant reductions in new infections in high-income countries, such as Canada and the United States. In 2011, Canada’s HIV prevalence rate was 208 per 100 000 people (0.002%). 1 Despite the low HIV prevalence in Canada, some communities are heavily impacted by high rates of HIV infection. For instance, the black community in Ontario comprise only 3.9% of the total population but represent approximately 22.5% of HIV infections in the province. 1,2 African, Caribbean, and Canadian black (ACB) men bear a disproportionate burden of health inequities increasing their vulnerability to HIV. For example, the HIV prevalence among ACB men who do not use intravenous drugs is 6 times higher than white men who do not use intravenous drugs in Ontario (Canada’s most populous province). 3,4 In order to optimize early detection of HIV infection and treatment initiation among black men, it is important to identify the predictors of HIV infection as well as characteristics associated with HIV testing.
There is a growing recognition that although behavioral factors play a major role in the transmission of HIV, they have limited explanatory utility for inequities in HIV infection. 5,6 The social determinants of health (SDOH) framework provides a useful tool for understanding various factors in the environment that contribute to health inequities. Social determinants of health include social, economic, environmental, and personal factors that impact an individual’s vulnerability and exposure to health-compromising conditions. 7 The 5 categories of SDOH are psychosocial factors, material circumstances, behaviors, biological factors, and the health system. For example, HIV-related stigma is a psychosocial stressor that has been consistently linked to delays in HIV testing and negative HIV care continuum outcomes among black men who have sex with men (MSM) and numerous other populations. 8 -11 Psychosocial determinants interact with the material and behavioral determinants to produce health inequities.
There are a range of material circumstances that contribute to HIV inequities among black men in North America, including the availability of resources to access health care services and safe housing conditions. Housing vacancy (a proxy measure of poverty and neighborhood distress) is positively associated with HIV diagnosis rates among black men. 12,13 Low educational attainment is also correlated with higher HIV diagnosis rates among black men. 12 Although this pattern is seen within both MSM and non-MSM populations, MSM are more likely to be living in areas comprised of residents with lower levels of education, which represents a structural impediment that limits employment opportunity. 12
Individual behavior has been shown to have damaging or protective influence on health. Inconsistent condom use and concurrent partnering have been identified as factors influencing HIV infection risk. 14 Among black men who only have sex with women (MSW), low rates of condom use are associated with beliefs that condom use diminishes pleasure, interferes with erection, and symbolizes infidelity. 14 Condom nonuse among black men is also influenced by attitudes regarding the perceived risk of disease exposure with different types of sexual partners, such as new or casual partners. 14,15 Condoms may be used less frequently after multiple sexual encounters with a given partner. Some men feared that their partners would be suspicious of infidelity if they reintroduced condoms into their sexual relationship. 14 Black MSM and black men who have sex with men and women have also echoed similar beliefs that condomless sex is more sensually exciting and gratifying and that condoms reduce sexual pleasure. 16 Black MSM are more likely than white MSM to have received HIV testing within the last year 17,18 ; therefore, the observed HIV inequities among black MSM are unlikely to be isolated to individual-level health care seeking behavior as a single explanatory factor.
The period between HIV infection and development of clinical signs and symptoms is characterized by very high viral replication and increasing risk of transmitting HIV to others. 19,20 Delays in HIV diagnosis and treatment initiation impede viral suppression, which increases HIV transmission. 21 Today, it is widely acknowledged that HIV treatment and viral load suppression are fundamental to HIV prevention (ie, “Treatment as Prevention”), 22 STIs also predispose individuals to sexual transmission of HIV 4,23 through 2 key mechanisms: increased HIV viral load in genital secretions and higher number and density of HIV-susceptible mucosal cells in the sex partner without HIV. 24,25 These underlying mechanisms are unrelated to ethnicity and do not account for the HIV inequities seen between black and other communities.
Currently, little is known about how the range of social, behavioral, and clinical factors intersect to influence the risk of HIV infection among black men in Canada. This study aims to identify the combinations of social, clinical, and behavioral factors that are most predictive of HIV testing and infection in a sample of black men in Canada. The knowledge from the effect of these factors can help to inform strategies for optimizing HIV prevention, testing, and linkage to care.
Methods
Design and Setting
We conducted a secondary analysis of data collected in The Kali (Swahili for “fierce” or “hot”) Study, a cross-sectional descriptive epidemiological study with black men from Toronto, Canada, between 2011 and 2013. The main goals of the Kali Study were to determine the distribution of HIV/STI coinfection among black men and to assess whether current STIs predicted likelihood of having an undiagnosed HIV infection. 26 The KALI study was conducted at 3 community health center (CHC) sites across Toronto. These 3 sites were selected based on their geographic locations so as to offer participants multiple options to access the study. The study was approved by the University of Toronto HIV Research Ethics Board.
Recruitment and Data Collection
Black men and women in the Greater Toronto Area were hired as peer recruiters and trained to engage in community-based outreach to identify other black men to participate in the study. Peer-based, venue-based, and self-referral methods were used as part of a multipronged recruitment strategy. All data collection took place within one of the 3 designated CHC study sites. Survey data were collected using an audio computer-assisted self-interview. Study participants also provided biological specimens for clinical diagnostics. Detailed descriptions of the recruitment and data collection methods for the parent study are published elsewhere. 26,27
Measures
Social variables
Participants reported various sociocultural and demographic characteristics including age, sexual orientation, country of birth, current residential information, educational attainment, religion, language(s) fluently spoken, family and social history, immigration status, employment, and income. Those born in the Caribbean or Africa provided information on their immigration status upon arrival to Canada, current immigration status, as well as parents’ and grandparents’ country of origin. All participants, regardless of birthplace of origin, were prompted to provide the number of trips and date of most recent trip to the Caribbean and/or Africa. Sexual orientation was self-reported based on a 5-item measure (“straight/heterosexual,” “gay/homosexual,” “bisexual,” “two-spirited,” and “transgender”) with “other,” “don’t know,” and “refuse to answer” options.
HIV stigma was measured based on validated indicators used by United Nations Programme on HIV/AIDS and United States Agency for International Development as described in our previous study. 27 HIV stigma was captured utilizing a 10-item, 5-point Likert scale with answer options ranging from “strongly disagree” to “strongly agree.” Each item on the scale included “don’t know” and “refuse to answer” options. Questions inquired about participants’ desire to keep friends’ and/or family members’ HIV status a secret, willingness to care for love one’s of people living with HIV, disclosure, and responses to people living with HIV during everyday encounters (ie, attending or teaching school and eating at a restaurant whose cook is living with HIV). 28,29 Higher HIV stigma implied increased level of stigmatization toward HIV infection or people living with HIV.
Behavioral variables
Participants were asked about their sexual history, including age of sexual debut, total number of lifetime sexual partners (female, male, or both), and the number of sexual partners in the past 6 months. They were also asked about various characteristics of their partners, including whether they were a casual or a regular partner, and what geographic region of the world they were from. Condom use measures included use at last intercourse, frequency of condom use with male and female partners, and condom use during different types of intercourse. Features of higher risk sexual activity were explored, including whether participants have had sex under the influence of alcohol, ever paid or received payment for sex, or had unprotected sex with a partner known to be living with HIV or have an STI.
Clinical variables
Clinical data were collected using both self-report and biomarker-validated measures. Participants self-reported their past history of STIs by indicating whether a health care professional had ever informed them that they had HIV, gonorrhea, chlamydia, syphilis, genital herpes, human papillomavirus, or genital ulcers. They were also given the option of reporting that they “forgot the name of the STI” or if they had any STIs other than the ones presented in the list. Participants were also asked whether they had any STI symptoms in the past 6 months and when they first had an STI. General health behavior characteristics were discussed, such as smoking and alcohol use history. Participants self-reported if they ever had an HIV test, as well as when were their first and most recent tests, their total number of HIV tests, and the geographic location and type of setting in which they had their first and most recent HIV tests. Serum enzyme immunoassays were performed for HIV and syphilis. All reactive HIV results were confirmed by Western blot. A urine nucleic acid amplification test was used to detect Neisseria gonorrhea and Chlamydia trachomatis infection.
Statistical Analysis
Analyzed data included all men who had sexual experience. Continuous variables, including age, number of years married, age of first sexual encounter, number of sexual partners (lifetime and past 6 months), were summarized using mean, median, and interquartile range. All other demographic data were summarized using proportions. The distribution of social, behavioral, and clinical variables were summarized using proportions (Tables 2-4). The classification and regression tree (CART) analysis, 30 -32 an empirical technique based on recursive partitioning analysis, was then applied to identify the best predictors of HIV infection and HIV testing. The CART analysis is well suited to stratify risk groups and it does not require parametric assumptions. The CART method involves the segregation of different values of classification variables through a decision tree composed of progressive binary split, and the optimal split is selected based on the complexity parameter that corresponds with the lowest cross-validation error. As a result, CART analysis produces decision trees that are simple to interpret and readily used for risk stratification. The “rpart” package in R was used to fit the CART models for the 2 outcomes of interest on 26 potential predictors of interest in this sample. 30
Ethical Approval and Informed Consent
This study did not need ethical approval because the secondary data set used has been stripped of all identifying information and could not be linked back to the individuals from whom it was originally collected.
Results
Demographics
Demographic data are presented in Table 1. The mean age of the men in the study was 36 years (standard deviation [SD] = 13.75). For those men born outside of Canada (n = 315; 64.68%), their average age of arrival in Canada was 19.58 years (SD = 12.48 years) and they had lived in Canada for an average of 23.4 years (SD = 13.66 years). Most men in the sample (n = 395; 82%) have only ever had sex with women with a smaller proportion of the sample (n = 86; 18%) reporting that they had sex with another man at least once in their lifetime. Most MSM in the sample self-identified as homosexual (n = 82; 95%). A significantly higher proportion of MSM than MSW (37% versus 11%) were refugee claimants on first arrival to Canada, while more MSW (41% versus 21%) were landed/permanent residents on first arrival in Canada. A summary of variables representing the social, clinical, and behavioral variables entered in the CART analysis are presented in Table 2 (social), Table 3 (clinical), and Table 4 (behavioral).
Summary of Selected Demographic Characteristics.a

Abbreviations: SD, standard deviation; STI, sexually transmitted infection.
a “Decade of arrival in Canada” only includes men who reported that they were born outside of Canada.
Distribution of Social/Structural Characteristics.a

Abbreviation: SD, standard deviation.
a Paid employment—past week excludes data from men who reported that they were full time students in college or university, unable to work because of long-term illness of disability, or retired.
Distribution of Clinical Characteristics.
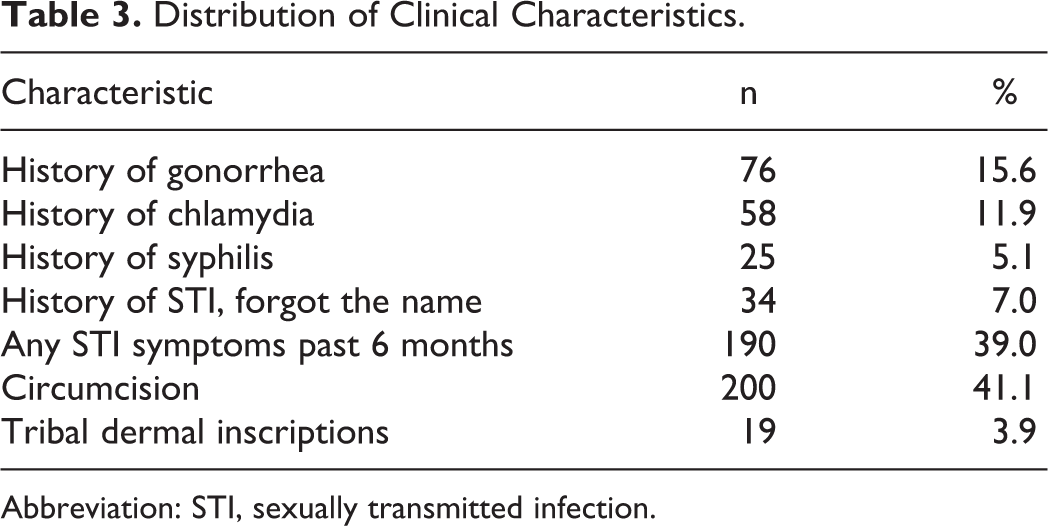
Abbreviation: STI, sexually transmitted infection.
Distribution of Behavioral Characteristics.

Abbreviation: SD, standard deviation.
HIV Infection
The rate of HIV infection was 2.91%, 42.11%, and 36.59% for those who reported sex with women only, men only, or both, respectively. Of the 26 variables evaluated, the CART method identified the number of male partners (6 or higher) as the best single predictor of current HIV infection. Among those with 6 or more male partners (n = 69), age (≥24 years old) was the next best predictor. For those who were 24 years or older, an HIV stigma scale score of 17.5 or higher further predicted HIV infection compared to those with lower HIV stigma scores (54% versus 33% probability). Among those with less than 6 male partners (n = 376), self-reported history of syphilis was the next best predictor of HIV infection. For those without a history of syphilis, an HIV stigma score less than 12 further predicted HIV infection (22% versus 1% probability). Figure 1 depicts the final tree resulting from the CART analysis. The branches of the regression tree stratify patients into 10 subgroups (Table 5) that represent low (3 groups), moderate (2 groups), high (4 groups), and very high (1 group) probabilities of HIV infection.

Predictors of HIV infection.
Probabilities of HIV Infection Distributed by Subgroup Stratification.a
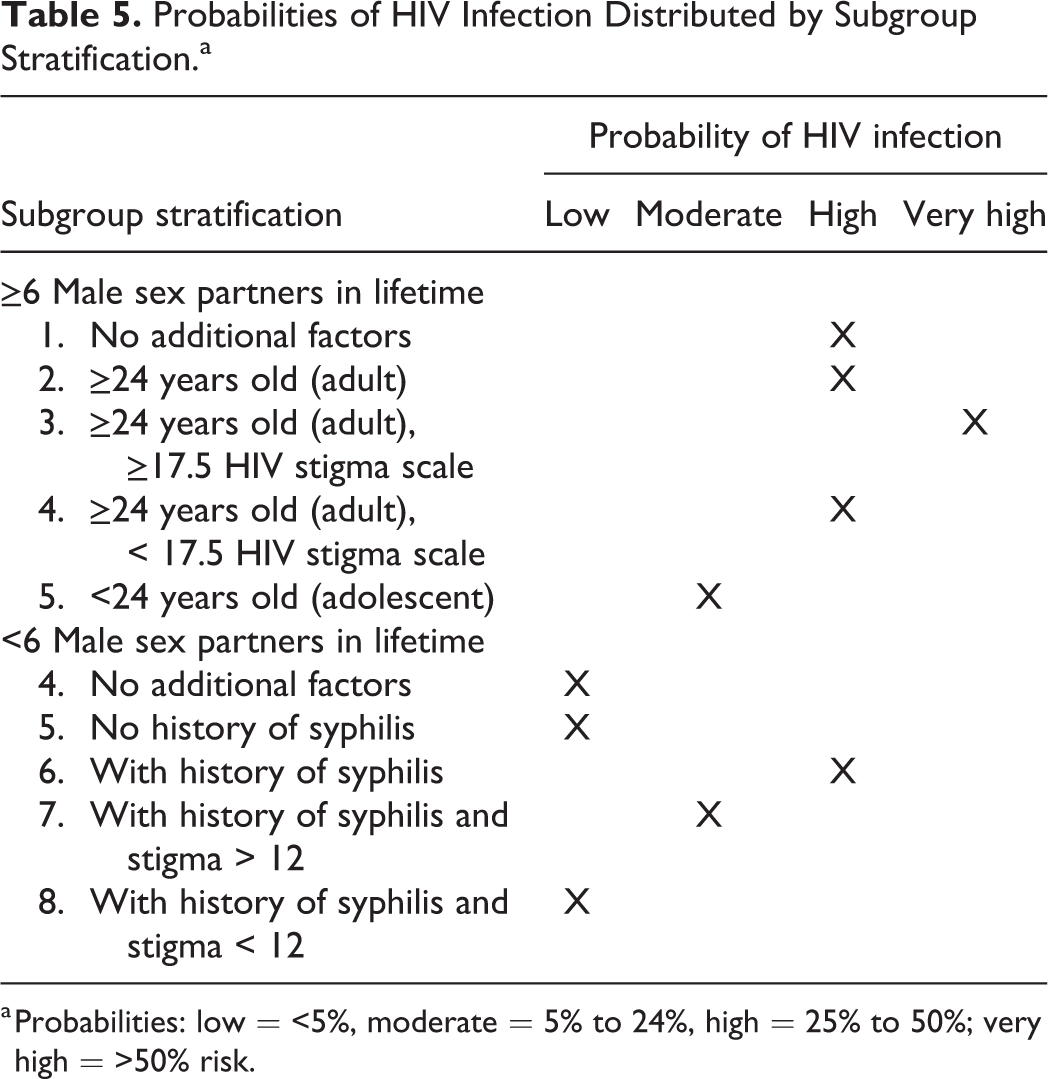
a Probabilities: low = <5%, moderate = 5% to 24%, high = 25% to 50%; very high = >50% risk.
HIV Testing
Of the same 26 variables evaluated using CART, age was also the best discriminator between those who had been tested for HIV and those who had not. Men who were 24 years or older with a recent history of condomless sex (n = 188) had a high probability of having prior HIV testing (85%). Men without recent condomless sex and with more advanced age (>60 years old) were less likely to have had HIV testing than those 60 years or younger (70% versus 20%, Figure 2). For men younger than 24 years old, a lifetime history of 2 or more male sex partners was the best predictor of having received testing for HIV. Among men younger than 24 years who had fewer than 2 but at least 1 male sex partners in their lifetime, having a history of chlamydia infection was the next best predictor of having tested for HIV. In those without a history of chlamydia, HIV testing was predicted by having a lifetime history of 2 or more female sex partners. Figure 2 depicts the final tree resulting from the CART analysis. The branches of the regression tree stratify the sample into 8 subgroups that have low (2 groups), moderate (4 groups), and high (2 groups) probabilities of ever having received an HIV test. Table 6 displays the 8 subgroup categorizations characterized by their probability of having ever received an HIV test.

Predictors of HIV testing: not infected individuals.
Probabilities of Ever Testing for HIV Distributed by Subgroup Stratification.

Discussion
The primary goal of this study was to identify social, clinical, and behavioral factors that could best predict HIV infection and testing in a community-recruited, nonprobability sample of black men in Toronto, Ontario, Canada. To identify the best predictors of HIV testing and infection, we used the CART analysis which is an empirical technique based on recursive partitioning analysis. 30 -32
As illustrated in Figure 1, we found that having 6 or more lifetime male sexual partners was associated with a 42% likelihood of current HIV infection. For MSM, an important behavioral risk factor for HIV infection is a high number of sexual partners. Studies have shown that compared to heterosexual men, MSM report a higher number of sexual partners. 33,34 Koblin and colleagues reported, in a study investigating risk factors for HIV infection among MSM, that having 4 or more partners accounted for the largest percentage of HIV infections with an attributable risk of 32.3%. 35 In our study, among MSM with more than 6 lifetime male sexual partners, age older than 24 years was associated with 46.7% likelihood of current HIV infection. In a study of risk behaviors among young and older men in Montreal, Canada, disparities among MSM’s sexual risk behavior did not exist between age groups (MSM younger than 30 years compared to MSM older than 30 years). 36 In the Unites States, however, men aged 26 years or older reported engaging in unprotected anal intercourse with HIV-infected partners more so than their younger counterparts. Older MSM also had an increased likelihood for sex with 10 or more partners over a 6-month period, similar to results in the present study. 37
Furthermore, we found that risk of HIV infection was elevated among men older than 23 years with perception of high HIV stigma (53.8%). We also found that syphilis diagnosis was the best predictor of HIV infection among men who had less than 6 male partners in their lifetime. This finding is consistent with previous research that suggests syphilis predicts HIV diagnosis among men. 38 -40 An analysis of New York City public health department data between 2000 and 2010 found that men had a 1 in 30 chance (3.6% incidence) of being diagnosed with HIV within a year of diagnosis with primary or secondary syphilis. 39 In our previous analysis of Kali Study data, we also found that there were increased odds of HIV infection for men with histories of syphilis (odds ratio = 6.48, P < .01). 26 The current analysis builds on the previous report by indicating the relative contribution of syphilis to HIV risk among other influencing factors. 38 Although syphilis screening has become more routine in STI screening with MSM, our CART findings indicate that syphilis may also be a main predictor of HIV among black MSW. This highlights the importance of routine screening and treatment for syphilis among black men which could reduce risk of HIV acquisition among people not living with HIV. A diagnosis of syphilis is also a key opportunity for health care providers to present ACB men with the option of HIV pre-exposure prophylaxis (PrEP). Despite the availability of PrEP in Ontario, it has not reached wide coverage within black Canadian communities. Culturally tailored care-coordination interventions that address social barriers to PrEP use and adherence may be useful to optimize scale-up among black men and scale-out across other subpopulations in black Canadian communities. 38,41
Stigma emerged as one of the primary factors associated with HIV infection among black men. Higher HIV stigma implied increased level of stigmatism toward HIV disease or people living with HIV. Our study shows that perception of HIV stigma increased the risk of HIV infection among participants reporting no history of syphilis. Several studies have indicated that HIV-related stigma increases vulnerability to HIV infection by reducing access to HIV prevention, testing, and presenting barriers to treatment, care, and support for people living with HIV. 42 Stigma, in this context, has been defined as prejudice, discounting, discrediting, labeling, stereotyping, and discriminating directed at people who are perceived to have or be at risk of HIV infection and which manifest to loss of status, unfair and unjust treatment, and social isolation of individuals or groups. 43 Stigma often arises from misconceptions about how HIV is transmitted and from judgmental attitudes toward those social groups that are disproportionately affected by HIV, particularly for people from endemic countries/ethnic minorities. 44 HIV/AIDS has been under the microscopic lens of morality and deeply rooted stigma that has generated assumptions that HIV/AIDS is an epidemic brought to Canada by the “outsiders”/”foreigners” and therefore an African and black people’s disease; associating the epidemic to stigmatized sexual behaviors such as sexual partner concurrency, extramarital sex, sex work, and drug use. 45 Furthermore, a study found that lack of knowledge about the HIV epidemic in Canada, cultural norms, and beliefs associated with heteronormativity and masculinity impacted HIV disclosure and uptake of HIV prevention tools such as condom, facilitating HIV infection. 46
Our findings show age, the lifetime number of male partners, nonuse of condoms, history of chlamydia, and the lifetime number of female partners to be the main predictors of HIV testing. Thus, age emerged as the best discriminator of lifetime HIV testing and major predictor for HIV testing among participants not diagnosed with HIV. Testing for HIV infection was more likely among those aged 24 years or younger who reported having 2 or more male sexual partners with 89% likelihood for testing for HIV. This finding is somewhat consistent with previous findings on HIV testing among gay, bisexual, and other MSM (GBMSM) in Vancouver, Canada. Armstrong et al found that older GBMSM were less likely to have received an HIV test in the past 2 years. 47 Furthermore, the finding that younger MSM with a greater number of male partners were more likely to have been tested for HIV is consistent with findings from an analysis of HIV testing behavior and HIV risk reduction strategies based on HIV risk categories (low, medium, high risk) among MSM in Vancouver. 48 Bogowicz and colleagues found that participants who were categorized as medium and high risk were more likely to have been tested for HIV in the previous year than men who were categorized as low risk (lower 46.5%, medium 54.6%, higher 67%; P < .001). 48 Thus such findings are supportive of clinical recommendations for health care practitioners on HIV testing provided by the US Centers for Disease Control and Prevention (CDC) 49 and Public Health Agency of Canada (PHAC). 49 -51 The CDC and PHAC suggest testing MSM who are sexually active for HIV at least once annually. More frequent testing is encouraged among MSM who have additional risk factors, such as unprotected sex. Collectively, the clinical practice guidelines and efforts taken up by public health agencies to decrease seroconversion rates among MSM broadly may contribute to increased testing rates, particularly among younger MSM. For participants older than 24 years, the likelihood of HIV testing increased to 85% for those who reported having unprotected sex. This is in line with other studies showing similar findings and the reason why CATIE, a Toronto-based AIDS Service Organization, provided clinical recommendations for health care practitioners that suggest HIV testing for MSM, as well as any person who has engaged in unprotected sex. 52 -54 Furthermore, there was an elevated HIV testing likelihood (69%) among participants younger than 23.5 years with less than 2 male sexual partners with history of chlamydia. Participants who had no history of chlamydia and reported having 23 female partners or more had a 63% likelihood of HIV testing. On the other hand, likelihood of HIV testing among participants who reported less than 22.5 female sexual partners was further stratified by a high perception of HIV stigma. Participants with a high perception of HIV stigma also showed an elevated HIV testing likelihood with 52% chance of HIV testing compared to a 13% chance of HIV testing among those who did not report high levels of HIV stigma. Our results are at odds with most of the studies in the literature suggesting that, due to stigma, ACB communities may be discouraged from taking advantage of testing, early care, and treatment, facilitating onward transmission of HIV. 55 -57 The reason for higher HIV testing with higher perceived stigma is probably due to some yet unmeasured cofounding variables. The research is consistent with evidence in the social psychology literature which indicates that controlling motivations (eg, fear, shame, stigma) can effectively provide impetus for engaging in a target behavior such as HIV testing but that the behavioral enactment is achieved at the experience of heightened distress, internalizations of stigma, and decreased psychological well-being. 58 -61 The associations of stigma with HIV testing should not be viewed as an advance, but a serendipitous consequence of an oppressive social process that ultimately leads to poor health outcomes for black men.
The ending of HIV transmission in the next 10 years has become a huge issue in the United States, Canada, and internationally, but there are still a lot of questions about what that will mean for black communities in Canada and the United States (especially black gay men) and similarly minority communities in other places. 62 The findings of this study highlight the link between risk factors and HIV transmission which relate to the issue of ending HIV transmission in Canada. Furthermore, the results could inform the identification and monitoring of HIV transmission clusters as well as HIV prevention and intervention with linkage and retention to HIV PrEP and care. 63
Conclusion
We identified several social, clinical, and behavioral factors correlated with current HIV diagnosis and lifetime HIV testing in a sample of black MSW and MSM in Ontario, Canada. Our findings are in line with results from previous studies that assert probability for HIV seroconversion among men increases with (1) increasing number of sexual partners, (2) increasing age, (3) higher levels of perceived stigma, and (4) previous diagnosis of syphilis. Increased risk factors (ie, higher number of sexual partners, condomless anal intercourse, history of syphilis diagnosis) were among the variables that yielded the highest correlations with lifetime HIV testing. Yet, serendipitous findings including a high correlation of lifetime HIV testing with high levels of perceived HIV stigma emerged. Based on our findings, black men in Canada may benefit from being presented with the option of HIV PrEP, particularly during routine HIV/STI screenings. Therefore, a diagnosis of syphilis could be a key opportunity for health care providers to present ACB men with PrEP. Future research should be conducted to identify how culturally tailored sexual health interventions may be effectively implemented. Additionally, widespread longitudinal epidemiologic efforts should be conducted to further explore and identify the mechanics of structural, social, and behavioral factors impacting HIV and STI infections. Understanding which combinations of social, clinical, and behavioral factors are most predictive of current HIV infection and testing will help improve HIV prevention strategies and care continuum outcomes among black men in Canada.
Footnotes
Acknowledgments
The authors posthumously acknowledge Dr Robert Remis’s leadership in the development and implementation of this study. The authors are also grateful to Jamie Thomas-Pavanel for managing the overall project and to Juan Liu for her support in managing the study data and to Anu Rebbapragada and Sanja Huibner for their expert performance of laboratory assays and management of laboratory-related logistics components for the study. The authors wish to thank Women’s Health in Women’s Hands Community Health Centre, Rexdale Community Health Centre, and Taibu Community Health Centre for the support in conducting the study at their sites. Finally, the authors express our sincere gratitude to the entire KALI Study team of recruiters and volunteer whose time and efforts led to the successful completion of this research project. This publication/presentation/grant proposal was made possible through core services and support from the University of Rochester Center for AIDS Research (CFAR), an NIH-funded program (P30 AI078498) and the Yale Center for Interdisciplinary Research on AIDS (CIRA) an NIH-funded program (P30 MH062294).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Canadian Institute of Health Research grant #HET-85518, Ontario HIV Treatment Network (OHTN) Knowledge, Translation and Exchange grant and OHTN HIV Research Chair in Implementation Science with Black communities (AHRC#1066).
