Abstract
Although drug-resistant HIV variants are considered to be less fit than drug-susceptible viruses, replication competence of these variants harbored by patients has not yet been elucidated in detail. We herein assessed the replication competence of strains obtained from individuals receiving antiretroviral therapy. Among 11 306 participants in a drug resistance surveillance in the Philippines, 2629 plasma samples were obtained from individuals after a 12-month treatment with zidovudine (ZDV)/lamivudine (3TC)/nevirapine (NVP). The replication competence of HIV isolates was then assessed by reinoculation into seronegative peripheral blood mononuclear cells in the absence of drugs in vitro. The drug resistance rate was estimated to be 9.2%. Drug-resistant strains were still a minority of closely related strains in a phylogenetic cluster. Among the available 295 samples, 37 HIV strains were successfully isolated. Progeny viruses were produced at a wide range (5.1 × 106 to 3.4 × 109 copies/mL) in primary culture of peripheral blood mononuclear cells. The viral yields were higher than the corresponding plasma viral load (1300 to 3.4 × 106 copies/mL) but correlated with those (r = 0.4). These results suggest that strains with higher intrinsic replication competence are one of the primary targets of newly selected drugs at the increasing phase of the plasma viral load during antiretroviral therapy.
What Do We Already Know About This Topic?
Replication competence of drug-resistant HIV variants harbored by patients has not yet been elucidated in detail.
How Does Your Research Contribute to the Field?
We herein assessed the replication competence of strains obtained from individuals after a 12-month treatment with zidovudine (ZDV)/lamivudine (3TC)/nevirapine (NVP) in the Philippines.
What Are Your Research Implications Toward Theory, Practice, or Policy?
The results of this study suggest that strains with higher intrinsic replication competence are one of the primary targets of newly selected drugs at the increasing phase of the plasma viral load during antiretroviral therapy.
Introduction
The global incidence of HIV peaked at 3.3 million in 1997, then decreased to 1.8 million in 2016. Although its incidence was declining, the total number of individuals living with HIV increased and reached 36.7 million in 2016 according to Joint United Nations Programme on HIV and AIDS (http://www.unaids.org). HIV vaccines are considered to be necessary for the better control of this epidemic, and a new vaccine candidate was reported with improved efficacy (up to 31% protection), thereby offering encouragement for future research. 1 However, it is still far from clinical use. In contrast, advances have been achieved in the use of antiretroviral drugs for the treatment and even prevention of HIV infections. 2 New drugs that combine excellent potency with greater convenience, safety, and tolerability make lifelong viral suppression achievable. 3
However, the marked increase in antiretroviral drug use has resulted in more prevalent drug resistance. Drug-resistant variants are frequently produced due to the error-prone nature of HIV-1 reverse transcriptase and recombination. 4 The levels of these strain variants occasionally increase to detectable levels in the presence of selective drug pressure. 4 Although drug-resistant variants are considered to be less fit than drug-susceptible viruses, 5 the acquired level of replication competence by drug resistance strains has not yet been elucidated in detail. If high replication competence is likely to be retained after the emergence of these drug-resistant variants, these high replicative variants need to be effectively detected and preferentially extinguished in a clinical setting in order to prevent the development of AIDS.
In the Philippines, HIV prevalence is still less than 1%. However, a marked increase has occurred in the number of new infections over the past few years. It is the fastest growing HIV epidemic in Southeast Asia. 6 The fastest spread of HIV was reported among injecting drug users, and a wide variety of HIV strains were found to be harbored by men who have sex with men. 7 A total of 22 413 individuals living with HIV were receiving antiretroviral therapy (ART) in this country as of August 2017 (HIV/AIDS & ART Registry of the Philippines). Among the regimens recommended by the World Health Organization (WHO), 8 the Department of Health of the Philippines decided to use the antiretroviral drugs zidovudine (ZDV), lamivudine (3TC), and nevirapine (NVP) as the preferred first-line regimen. Ninety-five percent of the individuals living with HIV were on first-line regimens according to the HIV/AIDS and ART Registry of the Philippines.
In the present study, the emerging rates and patterns of antiretroviral drug resistance were estimated after a 12-month treatment with ZDV/3TC/NVP. The replication competence of HIV isolates from those patients was assessed for the intrinsic replication competence of the isolates, using a primary culture system of peripheral blood mononuclear cells (PBMCs) in vitro.
Methodology
Participants and Samples
In the drug-resistant surveillance (Department of Health, the Philippines), 11 306 ART cases treated with ZDV/3TC/NVP (one of the WHO-recommended regimens 9 ) were noted in 2015 to 2016, respectively. Of these, 2629 samples were collected after a 12-month treatment and sent to the National Reference Laboratories, the Philippines. The HIV viral load (>1000 copies/mL, lowest detectable limit) was detected in 11% of samples (295/2629). Of the 295 samples, the pol nucleotide sequences (1306 bases) of 127 samples were successfully elucidated. In addition, a drug-naive HIV-positive population from an annual serological surveillance conducted between 2008 and 2016 was used as the control population (n = 224).
Plasma Viral Load Measurements
The commercially available kit, Cobas Taqman HIV-1 Test v2.0 (Roche Molecular Systems Inc, Pleasanton, California), was used according to the manufacturer’s instructions.
RNA Extraction, Reverse Transcription, and Polymerase Chain Reaction
HIV-RNA in plasma and culture medium (140 µL) was extracted using the QIAamp viral RNA mini kit (QIAGEN, Tokyo, Japan). The HIV-1 pol region (HXB2 position, 1827-3528) was reverse-transcribed and amplified in the first round of the polymerase chain reaction (PCR) with F1849/R3500 10 using the SuperScriptIII One-Step Real-Time (RT) PCR System with Platinum Taq DNA Polymerase (Thermo Fisher Scientific, Tokyo, Japan). Two HIV-1 pol regions (2074-2716 and 2388-3425) were amplified in the second round of PCR with DRPRO5/DRPRO2L 10 and DRRT1L/DRRT4L 10 using the AmpliTaq Gold 360 Master Mix (Thermo Fisher Scientific). Annealing temperatures and elongation times were 57°C and 90 seconds for the first PCR and 55°C and 60 seconds for the second PCR, respectively. 10
Nucleotide Sequences and Phylogenetic Analyses
Polymerase chain reaction products from HIV-RNA in plasma were subjected to direct sequencing using the BigDye Terminator v3.1 cycle sequencing kit (Thermo Fisher Scientific). The primers of DRPRO2L, DRRT1L, and RT2882 11 were used to elucidate the 2 nucleotide sequences amplified with the primer pairs DRPRO5/DRPRO2L and DRRT1L/DRRT4L. Sequencing results were aligned with subsequent inspections and manual modifications using MEGA 7 software. 12 Neighbor-joining and maximum composite likelihood were used to build a phylogenetic tree, and 1000 bootstrap replications were performed for its reliability estimation.
Assessment of Drug Resistance Strains
HIV nucleotide sequences were subjected to the HIV Drug Resistance Database at Stanford University (http://hivdb.stanford.edu/). 13
HIV Isolation
Of 295 samples from drug-treated patients with a detectable HIV load (>1000 copies/mL), 142 had sufficient sample volumes for HIV isolation. These 142 samples were exposed to Viro-adembeads (AdemTech, Pessac, France) and used for inocula to phytohemagglutinin (PHA)-activated human peripheral blood mononuclear cells (PHA-PBMC, 1.5 × 106 cells, 500 µL) obtained from HIV-seronegative volunteers. RPMI1640 complete medium supplemented with interleukin 2 (IL-2, 10 ng/mL; Genzyme, Cambridge, Massachusetts) and 10% fetal bovine serum was used for the culture. On day 3 of the culture, half of the culture medium was replaced with complete medium. On day 7, half of the cell suspension was replaced with fresh PHA-PBMC (500 µL of 1.5 × 106 cells/mL culture). The 7-day process was repeated 3 more times, and the entire process was completed in 4 weeks. The HIV antigen in the culture medium was assayed using an HIV antigen capture kit (Alere HIV Combo; Alere Medical, Tokyo, Japan). The culture supernatant was stored in a deep freezer after centrifuging at 800g for 5 minutes.
Assessment of Replication Competence of HIV Isolates
Phytohemagglutinin-activated human peripheral blood mononuclear cells (1.5 × 106 cells) were pelleted at 800g for 5 minutes, resuspended with an HIV preparation (106 HIV-RNA copies in 100 µL), and incubated at 37°C for 1 hour. After the addition of complete medium with IL-2 (900 µL), a culture (1 mL) was conducted for 1 week. Half of the culture medium was replaced on day 3 of this procedure. The culture supernatant on day 7 was subjected to RNA extraction and quantitative 1-step RT-PCR as described below. One of the isolates (DR1505-87) was used as a control to evaluate primary culture conditions.
Quantitative Reverse-Transcribed PCR
GagB-1F (5′-AGTGGGGGGACATCAAGCAGCCATGCAAAT-3′, 1359-1388) and GagB-1R (5′-TGCTATGTCACTTCCCCTTGGTTCTCT-3′, 1500-1474) were synthesized after a slight modification of the primers, SK462 and SK431. 14 Using those primers, a gag fragment was amplified and cloned into the pCR2.1-TOPO vector (Thermo Fisher Scientific) to construct an HIV-1 RNA transcription vector. After digestion of the HIV-1 RNA transcription vector with HindIII, a fragment including the gag sequence was transcribed using Riboprobe In Vitro Transcription Systems (Promega, Madison, Wisconsin). After the removal of the template DNA, the concentration of RNA transcripts was measured using the Qubit 2.0 fluorometer (Thermo Fisher Scientific) and calculated using the following equation: RNA (copies/mL) = [Measured RNA (g/mL)] × [6.02 × 1023 (copies/mol)]/[272 × 340 (g/mol)]. The values in this equation were as follows: 6.02 × 1023 (copies/mol), Avogadro constant; 272 (bases), the length of DNA from +1 to the Hind III site in the RNA transcription vector; and 340 (g/mol), the average molecular weight of a ribonucleotide monophosphate.
The amount of HIV-RNA was assayed using the SuperscriptIII Platinum SYBR Green One-Step qRT-PCR kit (Thermo Fisher Scientific) and assessed by relating the PCR signal to a standard curve (encompassing 102 to 108 copies/reaction of in vitro transcribed RNA transcript) after a manufacturer’s protocol.
Statistical Analysis
Rates of drug resistance among the naive and 12-month treatment groups were analyzed by Fisher exact test using JMP software version 9 (SAS Institute Japan, Tokyo, Japan).
Ethical Approval and Informed Consent
This study was conducted as a collaborative research project under the approval and control of the institutional review boards of San Lazaro Hospital, the Philippines (No. SLH RERU 2014-024), and the Faculty of Medicine, Tottori University, Japan (No. 1982), and were conducted after obtaining informed consent from the patients participating in these laboratory surveillances in an unlinked anonymous manner. All procedures were performed according to the principles of the Declaration of Helsinki.
Results
HIV Strains Resistant to Protease and Reverse Transcriptase Inhibitors
Among the referred samples (n = 2629), 295 showed a detectable HIV load with a wide variety: 1.3 × 103 to 3.4 × 106 copies/mL. The percentage of cases with detectable HIV over the total number of referred cases was 11% (295/2629). The drug-resistant rate of the analyzed cases was 84% (107/127; 70/79 and 37/48 in 2015 and 2016, respectively). Therefore, the drug-resistant rate of referral cases was estimated to be 9.2%.
The drug-naive control population infected by drug-resistant strains was observed (2.7%; 6/224 in 2008-2016). Drug-sensitive strains (n = 8, denoted by open circles) clustered closely with a drug-resistant strain collected from a drug-naive patient at the time when an HIV-positive individual was scarcely found (2010, N; inset A of Figure 1). The 7 drug-resistant strains from individuals receiving ART (closed circles) clustered closely together. However, other neighboring strains were still sensitive to antiretroviral drugs (inset B of Figure 1). The result of phylogenetic analysis also suggested that “transmitted drug-resistant” strains had spread limitedly in the Philippines.

A phylogenetic tree of the HIV-pol region of HIV isolated in the Philippines. The nucleotide sequences of the HIV-pol region (1306 bp, 2096-3401 of HXB2, accession no. K03455) were elucidated, and a phylogenetic tree was produced using the neighbor-joining method. Symbols denote drug-resistant (•) and drug-sensitive (ˆ) strains. In insets A and B, strains of interest are indicated with year and T/N. T and N denote strains collected from individuals with (T) or without (N) treatment history. Bootstrap values (88% and 80%) are indicated at the basal nodes of clusters A and B. The sequence of the simian immunodeficiency virus, isolate 239 (accession no. M33262), was used as an outgroup.
High Replication Competence Retained in Drug-Resistant HIV Isolates
One hundred forty-two plasma samples collected from individuals treated with AZT/3TC/NVP for 12 months were available after viral load measurements and subjected to HIV isolation. The HIV antigen was detected in 37 cultures at the end of the fourth week of the culture (26%, 37/142). The cutoff value for the antigen detection system was estimated to be within the range of 106 to 107 copies/mL using our in-house HIV detection system. The HIV amount produced during the 7-day culture period of PHA-PBMC inoculated with these 37 strains at 106 copies/mL ranged between 5.1 × 106 and 3.4 × 109 copies/mL (mean: 3.9 × 108 copies/mL; Figure 2). Throughout all assessments of HIV replication competence, an isolate (DR1505-87) was employed for the quality control of cells. This isolate produced a constant amount of progeny viruses in a 7-day culture (N = 14, range: 1.8 × 107 to 5.4 × 107, mean: 3.4 × 107, standard error: 2.9 × 106 copies/mL). The plasma HIV load in each case was lower than the production level in PHA-PBMC in vitro, without any exception (range: 1.3 × 103 to 3.4 × 106 copies/mL, mean: 4.1 × 105 copies/mL; Figure 2).
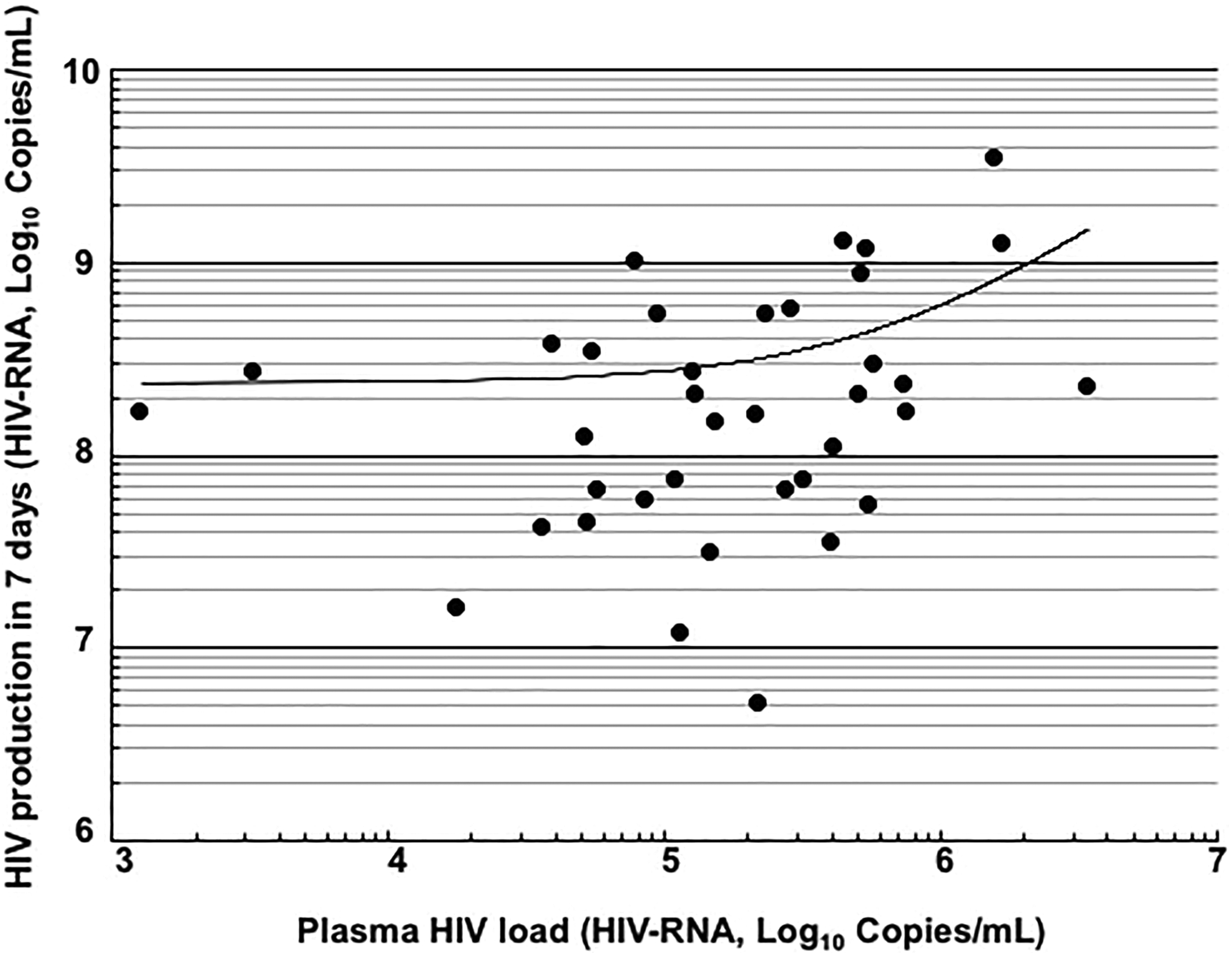
Relationship between plasma HIV loads and replication potencies of HIV strains. A correlation analysis was performed between plasma HIV loads and the amounts of HIV produced from peripheral blood mononuclear cells inoculated with HIV (HIV-RNA: 106 copies/1.5 × 106 cells/mL) in 7 days (n = 37). Pearson correlation coefficient (r) was 0.375 (P < .05).
HIV production for 7 days in PHA-PBMC inoculated with 5 representative strains is shown in Figure 3. After the inoculation (106 HIV-RNA copies to 1.5 × 106 cells), high replication potent strains showed rapid growth and produced high amounts of HIV on day 7.

HIV production and levels attained in peripheral blood mononuclear cell cultures. Peripheral blood mononuclear cells (1.5 × 106/mL) were exposed to 5 representative strains (106 copies) and cultured for 7 days. Drug resistance profiles are shown in Table 1, except for DR1509-397. This strain (DR1509-397) did not have a drug resistance-related amino acid variant, although it had a detectable plasma viral load (>1000 copies/mL). Symbols denote the mean values of HIV production; bars represent standard error.
Strain-Specific Infection Efficiency
HIV production levels were dependent on the inoculum size, and even a strain with low replication competence grew to some extent (Figure 4A). However, strains with lower replicative capacity, such as DR1606-505, showed limited growth: 2-fold that of the inoculum size (Figure 4B), whereas highly replicative strains produced progeny viruses at amounts that were more than 10-fold that of the inoculum size (Figure 4).

Relationship between inoculum sizes and production levels. Three different inoculum sizes were used for infections with 5 strains (A). The production levels attained for the strain (DR1606-505 with the lowest replication competence in this study) after its exposure to 106, 107, and 108 (copies/mL) inoculum sizes to peripheral blood mononuclear cells in vitro (B). Bars (A) and symbols (B) denote the mean values of HIV production; error bars depict the standard error.
Reproducible HIV Production Levels from Different Virus Lots
Virus passage was performed in order to assess reproducible HIV production levels. Four different strains produced on day 7 in the first round of the culture were exposed again to newly prepared PHA-activated PBMCs. The viral yields in the first and second rounds of the culture showed a less than 1 log difference (data not shown).
Amino Acid Sequences with Drug Resistance Mutations
The profiles of the drug resistance mutations of HIV in plasma are shown in Table 1, with a focus on 10 strains harboring the higher and lower replication competence of the 37 strains assessed for replication competence. However, no apparent difference in amino acid substitution patterns was detected from 2 different HIV populations in plasma (Table 1).
Drug-Resistant Mutations of the Strains with Lower and Higher Replication Competence.

Abbreviations: NVP, nevirapine; 3TC, lamivudine; ZDV, zidovudine; L, Leucine; R, Arginine; E, Glutamic acid; F, Phenylalanine; C, Cysteine; A, Alanine; I, Isoleucine; S, Serine; and V, Valine.
a Assessed by HIV production levels (copies/mL) in a peripheral blood mononuclear cells culture activated by phytohemagglutinin (PHA) for 7 days.
b Interpreted using Stanford HIV database.
c Numbering positions in HIV relative to HXB2 (accession no. K03455).
d Plasma (pre-passage): An HIV-RNA in plasma samples was used for Reverse transcription polymerase chain reaction (RT-PCR), and the products were subjected to nucleotide sequence determination.
e HIV isolate (passaged 4 times in 4 weeks): A patient plasma was exposed to anti-HIV-negative PHA-activated peripheral blood mononuclear cells and cultured for 28 days. An HIV-RNA in the culture medium was used for RT-PCR and resultant determination of the HIV-nucleotide sequence.
Drug resistance mutations in the pol region of 5 HIV isolates with higher replicative competence were also examined (see the lines of “HIV isolate” in Table 1) and were mostly identical to those in the HIV of plasma samples, except in 2 cases: DR1509-477 and DR1510-726 (Table 1). The isolates of DR1959-477 and DR1510-726 appeared to have 2 quasi-species as major drug-resistant variants.
Discussion
The rate of individuals harboring drug-resistant HIV strains was estimated to be 9.2% (2015-2016) in the Philippines. The phylogenetic analysis supported the limited spread of drug-resistant strains. HIV variants with drug resistance after treatment with ZDV/3TC/NVP for 12 months retained high replication competence. The preferred first-line regimen was changed in 2017 from the ZDV/3TC/NVP to tenofovir (TDF)/3TC/efavirenz (EFV) following the guidelines on the use of antiretroviral drugs (issued by WHO in 2016, https://www.who.int/hiv/pub/arv/arv-2016/en/).
Previous studies reported that the rates of “transmitted drug resistance” were constant at 8.3% and 20.5% in Europe 15 and Washington DC, 16 respectively; however, this rate was low in the Philippines between 2008 and 2016 (2.7%) as shown here in the present study. Low HIV prevalence and its location as an island country may have contributed to this low rate. The spread of “transmitted drug resistance” strains may still be in the early phase, but is becoming more widespread in the Philippines.
The rate of “acquired drug resistance” was estimated to be 9.2% in 2015 to 2016. As described above, a few patients harbored “transmitted drug resistance” strains (2.7%) in the Philippines. Therefore, most of the patients harbored drug-sensitive strains as the majority and drug-resistant ones as minor quasispecies. These minor quasi-species may have emerged under drug-selective pressure in the treatment-experienced individuals in this study. 17 Other countries showed higher acquisition rates (Henan in China, >15% in 12 months; Hubei in China, 57.1% in 9 to12 months 18 ; 40%, Washington DC 16 ) than that (9.2%) in the present Philippine study. As described above, “acquired drug resistance” strains commonly emerge from the preexisting viral quasi-species pool under the selective pressure of antiretroviral drugs. 17 This principle suggests that the rate of drug-resistant strains in the quasispecies pool is markedly lower among HIV carriers in the Philippines. However, a recent study reported that a larger percentage of individuals harbored minority drug-resistant HIV quasi-species in the Philippines. 19 These findings suggest that the “acquired drug resistance” rate will soon become obvious.
The attained growth level of the drug-resistant variants in vitro correlated with the values of the plasma viral load in vivo. Intrinsic replication competence appears to reflect the viral load for drug-resistant strains. Therefore, the identification of replication competence at this reemergence of variants with drug resistance mutations has the advantage of providing an early alert for reaching a higher plasma viral load. Previous studies also indicated that HIV has an intrinsic replicative capacity and the capacity reflects set point HIV load at an acute phase by 33%, 20 which is supported by the close relationship between the viral load of the donor and seroconverting partner. 21 -23
The present study showed that high replication competence was retained by drug-resistant HIV isolates. Some of these strains propagated in in vitro primary cultures and reached levels of more than 109 HIV-RNA copies/mL. These attained levels of viral growth in vitro were more than the values of the plasma viral load in vivo without any exception. The plasma HIV load has been used in clinical settings in order to predict a patient’s prognosis and its upper limit is typically less than 1 × 107 HIV-RNA copies/mL. 20,24,25 Consistent with these findings, the plasma HIV load ranged between 1.3 × 103 and 3.4 × 106 copies/mL (mean: 4.1 × 105 copies/mL) in the present study. The higher HIV yield in vitro than in vivo is presumably attributed to in vitro conditions, in which immunity does not function well. 3 These results imply that HIV-infected cells produce progeny viruses that are markedly larger than the levels of the plasma viral load in an immune deficiency. 26,27 A previous study speculated that high replicative strains may be preferentially excluded by immune responses and may not become the majority in the viral population. 20 Even if this is the case, retained high replicative strains must be one of the highest risk factors for the development of AIDS. Therefore, these high replicative strains that reemerge under drug-induced selective pressure need to be effectively detected and preferentially extinguished in order to prevent the development of AIDS. However, intrinsic replication competence cannot not be estimated precisely by the plasma viral load of current clinical marker. Diagnostic tools need to be developed to estimate intrinsic replication competence in the near future.
Conclusion
Replication competence of strains is one of the indispensable parameters for the selection of a new drug regimen at the increasing phase of the plasma viral load during ART. The new drug regimen needs to eliminate the highest replicative strains preferentially in order to avoid serious illnesses and minimize secondary transmission.
Footnotes
Acknowledgments
The authors thank Ms Naohara for her technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Japan Society for the Promotion of Science (KAKENHI-15K08734) and a Grant-in-Aid for Scientific Research on Infection Control and Prevention by the International Platform for Dryland Research and Education, Tottori University.
