Abstract
Repeat HIV testing after receiving a positive result has never been studied systematically and may give insight into reasons for delayed linkage to care. Among 831 adults in 6 secondary facilities in Oromia, Ethiopia, who completed an interviewer-administered structured questionnaire within 2 weeks of initiating antiretroviral therapy in 2012 to 2013, 110 (13.2%) reported having retested after an HIV-positive result. The odds of repeat (versus single) HIV-positive testing were higher among those who had doubted their HIV status (adjusted odds ratio [AOR]ref=nodoubt = 6.5; 95% confidence interval [CI]: 3.7-11.4) and those who initially tested at another facility, whether another secondary facility (AOR ref=studyfacility = 22.7; 95% CI: 11.0-46.9) or a lower-level facility (AORref=studyfacility = 19.1; 95% CI: 10.5-34.5). The odds of repeat (versus single) HIV-positive testing were lower among those who initially tested because of symptoms (AORref=not a reason = 0.40; 95% CI: 0.24-0.66). Median time between initial diagnosis and enrollment in care was 12.3 versus 1.0 month for repeat and single HIV-positive testers, respectively (P < .001). Repeat HIV-positive testing—not a rare occurrence—appears to stem from doubt, seeking care at a facility other than where diagnosed, and testing for a reason other than having symptoms. Because repeat HIV-positive testing is associated with delay in linkage to care, providers should be aware of this potential when counseling those who test HIV positive.
Introduction
Increasing the number of people who are tested regularly for HIV—and therefore diagnosed early—and linking them promptly to care are critical for realizing the individual and population benefits of antiretroviral therapy (ART). 1 –5 Studies of individuals’ testing histories have shown a wide range of testing behaviors: some people, including those at high risk, never get tested, and others get tested regularly, sometimes as a “prevention” strategy. 6 A few studies have also suggested that HIV-positive people may retest after their initial HIV-positive result, 7,8 but this behavior has not been studied systematically; we are aware of only 1 study that reported the frequency of a previous HIV-positive test (3.3% among a sample of men attending a voluntary counseling and testing [VCT] clinic in Tanzania). 8 Estimating the prevalence of repeat HIV-positive testing, characterizing individuals who undergo testing after an HIV-positive result and their reasons for doing so, and examining the relationship of repeat HIV-positive testing with subsequent care outcomes may offer important insights for health systems and providers to enable HIV-positive individuals to rapidly link to care after diagnosis.
We used data from the Late ART Initiation (LSTART) study conducted in Oromia National Regional State, Ethiopia, in 2012 to 2013 to address these questions. LSTART was designed to examine a range of factors associated with late antiretroviral therapy (ART) initiation and included both individual interview and clinical data. 9 In Ethiopia, only 450 000 (37%) of the estimated 1.2 million people living with HIV are enrolled in care, despite efforts by the Ethiopian government to increase this number. 10 Given this context, a study examining testing behavior prior to enrollment has immediate relevance for local providers and policy makers as well as for efforts elsewhere in sub-Saharan Africa to decrease late enrollment in care. 1,2
Methods
Participants and Data Sources
HIV-positive adults (≥18 years old) newly initiating ART between June 2012 and April 2013 at 1 of 6 study sites in Oromia, Ethiopia, were eligible for inclusion. All 6 sites are secondary health facilities in urban areas and were receiving support from the International Center for AIDS Care and Treatment Program (ICAP) at Columbia University with funding from the US President’s Emergency Plan for AIDS Relief. All sites offer physical escort from diagnosis location to HIV care and treatment enrollment, on-site CD4+ testing, ART pharmacies, VCT and provider-initiated counseling and testing (PICT), adherence support, and peer educator programs.
Clinical providers referred eligible patients to study staff on the day they were to initiate ART. Interested participants provided written informed consent and completed an interviewer-administered structured questionnaire within 2 weeks of ART initiation; participants were reimbursed 20 birr (∼US$1) upon interview completion. Questionnaire data were linked to an existing electronic medical record database that included medical history, visit dates, and test results. Ethical approval was obtained from the Oromia Regional Health Bureau, Columbia University Medical Center, and the City University of New York.
Variables and Definitions
HIV-testing history
Participants were asked about the total number of HIV tests they had ever received, regardless of the outcome. They were then asked the date of and where they had tested HIV positive and their reasons for testing. Next, they were asked whether there was another time they had tested HIV positive. Those who responded affirmatively were classified as repeat HIV-positive testers and were asked the date of testing and where the other test occurred and their reasons for retesting. We only asked about 2 HIV-positive tests to avoid confusion. Based on reported testing dates, we characterized the earliest HIV-positive test as the initial test and the later HIV-positive test as the subsequent test. We excluded participants if it was not possible to determine which test occurred first or if they reported a test date after their HIV clinic enrollment date.
Location of HIV-positive testing
For each HIV-positive test, participants were asked where the testing occurred: (1) at the study facility, a lower-level facility (eg, health center without specialized departments or services), or another secondary health facility (eg, hospital with multiple departments and wards) and (2) whether the test occurred in a VCT setting or a PITC setting.
Reasons for testing
Participants were first asked an open-ended question and responses were coded. Participants were then read a list of common reasons for testing and asked which ones applied to them. Therefore, it was possible for participants to provide multiple reasons for testing, all of which were coded as a reason or not a reason.
HIV care behaviors
Date of enrollment in care was obtained from the clinical record. Participants were asked whether they had transferred from another facility, whether they had questioned their HIV-positive status since being diagnosed (dichotomized as “not at all” versus “somewhat or a lot”), and whether they had ever used Holy Water for their HIV; Holy Water is a popular remedy for HIV and other conditions in Ethiopia.
Sociodemographic and clinical characteristics
Sociodemographic characteristics collected included age, religion, highest education level obtained, area of residence (urban or rural), relationship status, and whether they had been widowed. The CD4 count at enrollment in care, obtained from clinical records, was defined as the earliest available measurement within 6 months of enrollment into HIV care but prior to ART initiation.
Statistical Analyses
We used chi-square statistics to compare individuals who had repeat versus single HIV-positive tests. For testing events, we compared testing location and reasons for testing between the initial test of repeat HIV-positive testers and the only test of single HIV-positive testers using Fisher exact test, and the initial and subsequent HIV-positive test of repeat testers using test for matched data: McNemar chi-square test for matched pairs and Bowker symmetry test.
To identify the correlates of having repeat HIV-positive tests, we used conditional logistic regression models with site as the stratifying variable and examined the crude odds ratios associating repeat HIV-positive testing with variables that might motivate individuals to seek testing after an HIV-positive result. Multivariable models initially included all variables with P values <.5 in bivariate analyses. Covariates with the highest P values were eliminated iteratively until all variables had P values <.05. Variables excluded from the final model were individually added back one by one to rule out the possibility that they confounded other associations.
Multiple imputation was conducted for participants (n = 99) who were missing data on using Holy Water for HIV due to a change in the questionnaire design early in the study and to account for missing data on urban/rural residence, children, widowhood, and education (all ns ≤ 5). For the final models, sensitivity analyses were conducted by excluding these individuals and including them with a missing category.
To examine whether reporting repeat HIV-positive tests was associated with longer time from diagnosis to enrollment in care or lower CD4 count at enrollment, we used Wilcoxon rank sum tests. All analyses were completed using SAS version 9.3 (Cary, North Carolina, SAS Institute Inc.).
Results
Participant Characteristics and Comparisons between Single and Repeat HIV-Positive Testers
Between June 2012 and April 2013, a total of 1245 adults who were newly initiating ART at the 6 study facilities were eligible for the study, and 1180 agreed to participate and completed the interview (5.2% refusal). After excluding participants who could not remember when they were tested, who reported an HIV-positive test after the date of HIV care enrollment, or who reported 2 HIV-positive tests in the same month and year (N = 349, 30%; 249 single HIV-positive testers and 100 repeat HIV-positive testers), the final sample was 831 participants. The mean age was 35.0 years, 58.0% were female, and 69.2% were Ethiopian Orthodox.
When asked about previous HIV tests, 67.5% (n = 561) of participants reported that their only HIV test was the one that led to their diagnosis; 160 (19.3%) reported a previous HIV-negative test, and 110 (13.2%) reported receiving more than 1 HIV-positive test.
Characteristics of participants with single versus repeat HIV-positive tests are shown in Table 1. Compared to single HIV-positive testers, repeat HIV-positive testers were more likely to have attended university (7.9% of single testers versus 16.4% of repeat testers; P = .012), report doubting their HIV status (22.2% of single testers versus 58.2% of repeat testers; P < .001), report using Holy Water (11.5% of single testers versus 25.5% of repeat testers; P < .001), and report having transferred from another facility prior to ART initiation (1.5% of single testers and 9.2% of repeat testers; P < .001). Also, repeat HIV-positive testers were less likely to have children (79.1% of single testers versus 66.4% of repeat testers; P = .007) and identify as Muslims (10.4% of single versus 2.7% of repeat, P = .036).
Characteristics of Repeat versus Single HIV-Positive Testers Initiating ART at 6 Ethiopian Hospital-Based Clinics.a

Abbreviation: ART, antiretroviral therapy.
aStudy period June 2012-April 2013. N = 831.
bChi-square test.
Characteristics of Tests: Single and Repeat HIV-Positive Tests
Characteristics of testing events, including testing location and reasons for testing, are shown in Table 2. With respect to location, 81.4% of single HIV-positive tests occurred at the same facility in which ART was initiated (versus another hospital or a lower-level facility), notably different from the respective percentage of initial tests of repeat testers (19.1%; P < .001). In all, 42% of single HIV-positive tests occurred in PITC versus VCT settings, similar to the percentage of initial HIV-positive tests among repeat testers (41.8%; P = .083) and markedly different from the percentage of subsequent tests of repeat testers (70.0%; P < .001).
Testing Locations and Reasons for Testing among Single and Repeat HIV-Positive Testers.a

Abbreviations: PITC, provider-initiated testing and counseling VCT, voluntary counseling and testing.
aN = 831.
bFisher exact test.
cMcNemar and Bowker tests of symmetry.
dParticipants could report multiple reasons for testing.
The most common reason reported for getting HIV tested by single HIV-positive testers was feeling sick (66.0%). This percentage was significantly different from that reported for this reason of initial (but not subsequent) tests of repeat testers (P < .001). The second most common reason reported by single HIV-positive testers was a providers’ recommendation (41.1%), which was also significantly different from that reported for this reason of initial but not subsequent tests of repeat testers (P = .01). Among repeat HIV-positive testers, the most common reason for the initial HIV-positive test was feeling sick (43.6%), followed by a providers’ recommendation (30.9%). For this group’s subsequent HIV-positive test, the most common reason was wanting to be sure they were HIV positive (60.0%; P < .001), which was significantly different from the percentage of initial tests reported for this reason, followed by feeling sick (40.9%) and being encouraged to test by a health care provider (20.9%).
Factors Associated with Reporting Repeat HIV-Positive Tests
In the multiple logistic regression model (Table 3), participants who had doubted their HIV status since diagnosis had higher odds of being repeat HIV-positive testers (adjusted odds ratio [AOR]ref=no doubt = 6.5; 95% confidence interval [CI]: 3.7-11.4) as did participants who were initially tested at another secondary health facility (AORref=study facility = 22.7; 95%CI: 11.0-46.9) or at a lower-level facility relative to those initially tested at the facility where ART was initiated (AORref=study facility = 19.1; 95%CI: 10.5-34.5). Participants who indicated that feeling sick was a reason for testing (relative to those who did not cite this reason) had lower odds of repeat HIV-positive testing (AORref=not a reason = 0.40; 95%CI: 0.24-0.66) as did participants who were Muslims (AORref=Ethiopian Orthodox = 0.26; 95%CI: 0.07-0.99).
Factors Associated with Repeat HIV-Positive Tests.a

Abbreviations: AOR, adjusted odds ratio; ART, antiretroviral therapy; CI, confidence interval; OR odds ratio.
aN = 831.
bRegression models initially included all variables with P values <.5 in bivariate analyses. Covariates with the highest P values were eliminated by hand iteratively until all variables had P values <.05.
Clinical Characteristics of Individuals Reporting Repeat versus Single HIV-Positive Tests
The median time between the initial and subsequent HIV-positive test for repeat HIV-positive testers was 6.0 months (interquartile range [IQR]: 1.0-26.0 months). The median time between initial HIV positive test and enrollment in care at the facility where ART was initiated was longer for those reporting repeat than single HIV-positive tests (repeat testers: 12.3 months, IQR: 2.4-35.4; single testers: 1.0 months, IQR: 0.6-2.2; P < .001; Table 4). The median time between the initial and subsequent HIV-positive test was shorter among patients who reported their first HIV-positive test was conducted at another health facility (4.9 months) compared to those reporting their first HIV-positive test at the facility of ART initiation (22.9 months; P < .001; data not shown). The median CD4 count at enrollment in care did not differ between repeat and single HIV-positive testers (247 cells/mm3; IQR: 105-353 versus 216 cells/mm3; IQR: 112-334; P = .230). The median time between enrollment in HIV care and ART initiation was longer for repeat than for single HIV-positive testers (1.2 months, IQR: 0.5-9.1 months versus 0.7 months IQR: 0.5-9.2 months, respectively; P = .034).
Care Experience After Enrollment of Single HIV Testers versus Repeat HIV Testers.a
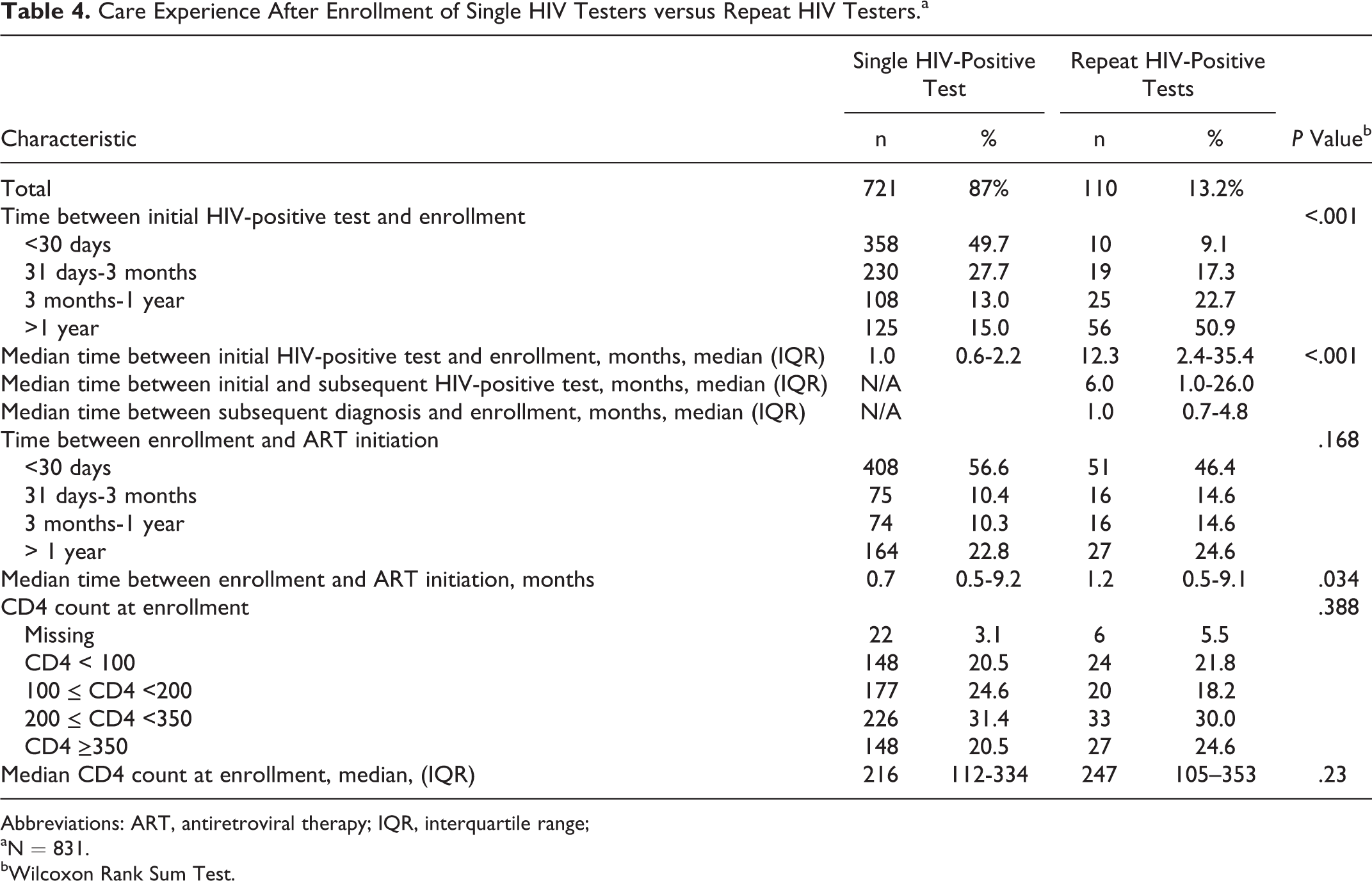
Abbreviations: ART, antiretroviral therapy; IQR, interquartile range;
aN = 831.
bWilcoxon Rank Sum Test.
Discussion
To our knowledge, no published studies have examined the prevalence of or motivation for repeated HIV testing by individuals who had an HIV-positive test result in either resource-limited or resource-rich locations, making the findings from this study of particular value. We found that repeat HIV-positive testing, reported by more than 10% of participants, was not a rare occurrence in this setting and was associated with substantially delayed enrollment in care, during which time individuals were not receiving medical or psychosocial services and were potentially at risk of transmitting HIV to others. Our estimate of the frequency of repeat HIV-positive testing may be low because participants may have been reluctant to acknowledge they had previously tested HIV-positive but not enrolled in care and because the interview was conducted at the time of ART initiation, thus excluding anyone who died or was lost to follow-up before initiating ART.
We identified several correlates of repeat HIV-positive testing in multivariable analyses. Participants who were initially tested at a facility other than the one where they started ART were more likely to be repeat HIV-positive testers than those who were tested in the same facility, regardless of the type of facility. Indeed, almost 56% of repeat HIV-positive testers were initially tested at either mobile, stand-alone VCT clinics or lower-level health posts compared to 12% of single HIV-positive testers. This suggests that some repeat HIV-positive testers transferred to another facility because their testing site did not offer ART as is the case with sites only offering HIV testing. Repeat testing in such circumstances is likely due to facility procedures, as facilities usually retest any patient presenting for enrollment in HIV care without an official referral form documenting their initial HIV-positive test. 11 However, in addition to transferring due to a lack of access to ART, patients may also transfer from one facility to another for other reasons, including those related to stigma (individual does not want to receive HIV care near home) or after delaying care enrollment (individual was ashamed to go back to the same clinic after delaying). 12 In addition to stigma-related factors, 12,13 reasons for changing clinics are likely related to other documented barriers to care, including doubt about diagnosis 14 and transportation issues. 15,16 Based on the available data, we were unable to determine the reasons for transfer among those individuals whose initial and subsequent HIV-positive tests were done at different sites, but a stratified analysis comparing those who changed clinics after initial testing versus those who did not change did not reveal any additional findings.
Almost 30% of participants indicated that they doubted their HIV-positive result, and those who doubted had a 6-fold higher odds of getting another HIV test compared to those who had not doubted, demonstrating that doubt is a common and significant predictor of repeat testing and may have led to delay in enrolling in care. Previous studies have also found that doubt about HIV status is an impediment to engagement in care and treatment initiation. 17 Finally, participants who reported (in relation to their initial or only HIV-positive test) that feeling sick was a reason for getting tested had lower odds of repeat HIV-positive testing than those who did not report symptoms as a reason for testing. This finding suggests that those without symptoms and at earlier stages of HIV disease were less likely to link to care after an HIV-positive test result, a finding consistent with reports from the United States, 18,19 sub-Saharan Africa, and Asia. 20
Our finding that Muslim participants were less likely to receive repeat HIV-positive tests is less intrinsically understandable, but one possibility is that they had fewer connections to the health care system. The same Muslim participants included in this analysis were found in another analysis to be less likely to have accurate beliefs about the benefit of ART clinical care than their Christian counterparts. 21 More generally in Ethiopia, Muslims were found to have lower health-seeking behavior. 22 While it is possible that these findings reflect lower penetration of HIV knowledge and concepts within the Muslim community in Ethiopia, the association between repeat HIV-positive testing and either beliefs about ART clinical care or health-seeking behavior remains unclear.
We found that there was no significant difference in CD4 count at enrollment in care among those who reported versus did not report repeat HIV-positive testing. Therefore, although the duration from initial HIV-positive test to enrollment in care was longer for repeat than single HIV-positive testers, this did not influence the stage of disease at enrollment in care. This may reflect the fact that those who had a prior HIV-positive test and opted not to engage in care may have been asymptomatic or had fewer symptoms at the time of the first HIV-positive test and only returned for another HIV test when symptoms appeared or became more severe. This hypothesis is consistent with that proposed by Hallett and Eaton—that some patients may not follow a direct path through the care and treatment cascade but rather may enter care through a “side door.” They suggest, for example, that compared to undiagnosed individuals, those who previously tested HIV positive but did not enroll in care may enroll more quickly after a motivating event. 7 Given that the CD4 count at enrollment was <353 for 75% of repeat HIV-positive testers and that the median time to enrollment in care after the subsequent test was 1 month, it is possible that these patients were beginning to experience symptoms.
This study has a number of strengths, including a large sample size and detailed information on testing history and reasons for testing. Additionally, we linked interview data with medical record data, enabling us to describe clinical outcomes of repeat versus single HIV-positive testers. The major limitation is that we had to exclude a substantial number of individuals who either could not remember when they were tested or reported an HIV-positive test date after enrollment in care. This may have biased the results, but it is difficult to determine in what direction. Another limitation is that the data were based on self-report; recall bias could have influenced the reasons provided for the initial and subsequent HIV tests, with recall being more accurate for more recent events. Another limitation is the absence of information on HIV disease stage at the time of the HIV-positive test.
Despite these limitations, this study demonstrates that repeat testing after an HIV-positive result, regardless of the reason for testing, is associated with a significant delay in enrolling in care. Although it may not be realistic nor desirable to discourage people who want confirmation of their diagnosis by seeking a second test, individuals should be advised during posttest counseling that if they doubt the results of the test, they should repeat it promptly even if they are asymptomatic, and immediate repeat HIV testing can be offered at the testing site. The findings from this study also highlight the importance of active linkage of HIV-positive individuals to HIV care with direct navigation, particularly when HIV test and care sites are colocated. Additionally, posttest counseling must ensure that individuals understand the benefits of enrolling in care, regardless of symptom status, especially in the era of earlier ART initiation. Finally, it behooves programs to prioritize active tracking of all HIV-positive individuals who have failed to enroll in HIV care to ensure that such individuals reap the benefits from treatment and to prevent further transmission to their communities.
In conclusion, we found that repeat testing after an HIV-positive result is not a rare occurrence and that it is associated with significant delay in linking to care. Some repeat testing may arise from administrative requirements when patients need (or choose) to enroll in care at a location different from where they were initially tested, but in other cases, repeat testing seems to be an indication of an individual’s doubt of their status, particularly if they are asymptomatic.
Footnotes
Acknowledgements
We thank all study participants at the HIV care and treatment facilities and our in-country data collectors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by PEPFAR and by a grant from the National Institute of Mental Health (grant number R01MH089831). All clinics included in this analysis received support from ICAP through funding from PEPFAR.
