Abstract
Registrational studies and observational cohorts clearly suggest sustained virologic response (SVR) rates in HIV-/hepatitis C–coinfected patients are similar to monoinfected patients when utilizing interferon-free regimens, and this can be accomplished with agents that are well tolerated with minimal adverse events. These randomized trials that led to the approval of several of our new direct-acting antiviral agents, however, specifically excluded patients who had significant comorbidities and none to our knowledge accepted patients with a history of cancer. Therefore, the effect of treatment of active hepatitis C in such patients and the effect on preexisting neoplasia are relatively unknown. We prospectively followed a 62-year-old male coinfected with HIV/hepatitis C who had a history of anal squamous cell carcinoma, prostate carcinoma, renal cell carcinoma all clinically cured and a myelodysplastic syndrome that was in remission. The patient achieved an SVR of hepatitis C with simeprevir/sofosbuvir without ribavirin and died shortly thereafter of a fatal relapse of his previously clinically controlled myelodysplastic syndrome.
Introduction
A significant percentage of patients infected with HIV are also coinfected with hepatitis C due to the similar route of transmission. It is estimated that about 185 million people in the world are infected with chronic hepatitis C infection and approximately 5 million of these are coinfected with HIV. 1 Since the advent of highly active antiretroviral therapy (HAART), liver disease has emerged as a leading cause of morbidity and mortality in patients coinfected with hepatitis C virus (HCV)/HIV 2,3 and has now exceeded the death rate of AIDS diagnosis in the United States. Hence, the treatment of hepatitis C cannot be overemphasized. The majority of studies today suggest that sustained virologic response (SVR-12) rates in coinfected patients are similar to monoinfected patients when utilizing the newly available interferon-free regimens, 4 -8 and this can be achieved in as little as 8 weeks with antiviral agents that are well tolerated with a positive significant adverse event. These studies however specifically excluded patients with active comorbidities and a majority excluded patients with a history of neoplasia even when these cancers were thought to be in remission. Our case report raises the scientific question about the potential reactivation of a myelodysplastic syndrome with fatal outcome in conjunction with SVR-12 of hepatitis C.
Case Report
We prospectively followed a 62-year-old male with a medical history as listed in Table 1.
Medical History.

He was entirely asymptomatic when simeprevir/sofosbuvir became available for the treatment of his hepatitis C. His virologic response and patient baseline data are summarized in Table 2. Despite virologic response similar to published data, 1 week after SVR-12, his myelodysplastic syndrome relapsed with 80% blast cells and hypoplasia of all cell lines. This occurred 300 days after bone marrow transplant, which was the documented time of his remission. He died of overwhelming systemic inflammatory response syndrome and severe aspergillus pneumonia 13 weeks after SVR.
Virologic Response and Baseline Data.
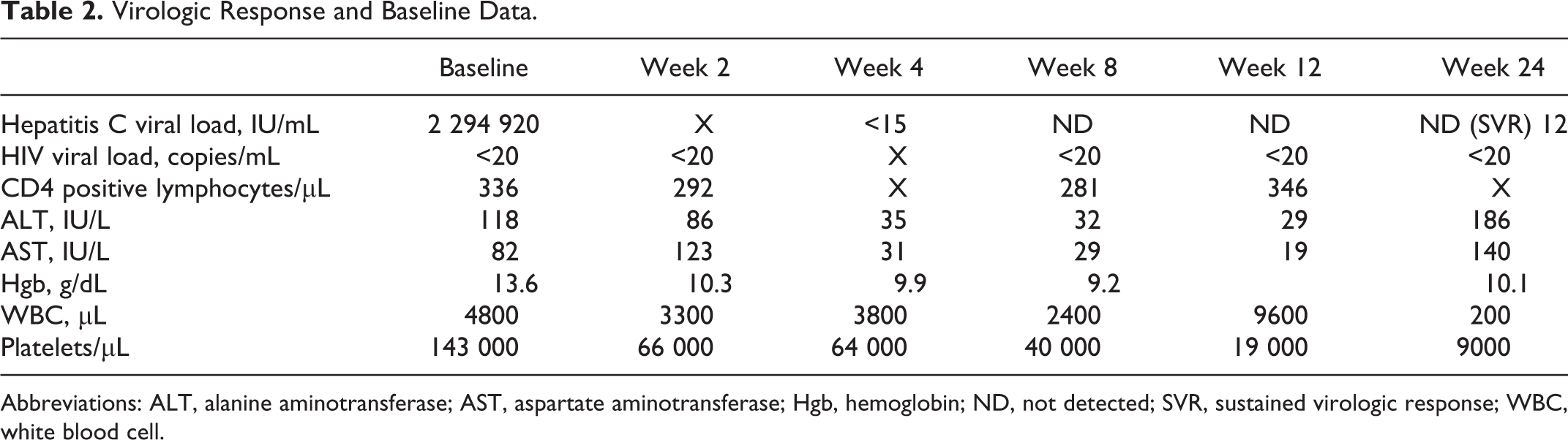
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; Hgb, hemoglobin; ND, not detected; SVR, sustained virologic response; WBC, white blood cell.
Discussion
Simeprevir/sofosbuvir without ribavirin achieved SVR-12 in this patient with long-standing HIV/HCV coinfection despite multiple prior comorbidities. Unfortunately, contemporaneously with virologic control of the hepatitis C, patient reactivated his myelodysplastic syndrome, which led to a fatal outcome. Although this may be simply coincidental and association does not prove causation, we are concerned that this reactivation may not be coincidental but may have something to do with the potential immunologic consequences of rapid virologic control in a patient who had the hepatitis C infection for decades. Of note, if you look at Table 2, the patient at week 4 in retrospect began to show bone marrow abnormalities, namely, a fall in his platelet count in the first 4 weeks of therapy, which would not be anticipated by the agent chosen to treat nor was there any evidence of hepatic decompensation over this time. Why we do not necessarily compare the science of what may have happened to the immune reconstitution syndrome seen in patient with advanced HIV disease receiving HAART, 9 that syndrome was not recognized in registrational studies as patients with advanced AIDS then were specifically excluded from those studies and it was not until clinicians began to use HAART that the syndrome came to light. Our group strongly suggests that studies should be undertaken in both monoinfected and coinfected patients with hepatitis C to monitor effects on comorbidities that were specifically excluded in registrational studies. We find of interest the presentations at the latest European Association for the Study of the Liver (EASL) conference at Barcelona of the development of hepatocellular carcinoma shortly after successful treatment of hepatitis C that has led to significant morbidity and mortality in those at risk. 10 We welcome input from our immunologic colleagues as to the effect of rapid control of hepatitis C and its effect on the immune system that may help to explain and identify those most at risk for reactivation of previously quiescent morbidities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joseph C. Gathe and Carl C. Mayberry have been consultants or speakers in conferences supported by AbbVie, Bristol-Myers Squibb, GlaxoSmithKline, ViiV Healthcare, Janssen, Merck, and Gilead and are members of institutions that received research grants from AbbVie, Bristol-Myers Squibb, GlaxoSmithKline, ViiV Healthcare, Boehringer Ingelheim, Pfizer, Janssen, Merck Frost, Gilead, and Tibotec (Janssen) Therapeutics; Joseph C. Gathe has served as an investigator for Abbott, Avexa Ltd, Boehringer Ingelheim Pharmaceuticals (BI), Inc, Gilead Sciences, GlaxoSmithKline, Merck & Co, Inc, Pfizer Inc, Roche Laboratories, Parexel, Hiesped, and Tibotec Therapeutics, and his institution has received honoraria for speaking or chairing engagements from Abbvie, Bristol-Myers Squibb, GlaxoSmithKline, Gilead, Merck & Co, Inc, Pfizer Inc, and Tibotec Therapeutics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
