Abstract
As part of the HPTN 065 study in the Bronx, New York and Washington, the authors, we surveyed clinicians to assess for shifts in their practices and attitudes around HIV treatment and prevention. Antiretroviral therapy (ART)-prescribing clinicians at 39 HIV care sites were offered an anonymous Web-based survey at baseline (2010-2011) and at follow-up (2013). The 165 respondents at baseline and 141 respondents at follow-up had similar characteristics—almost 60% were female, median age was 47 years, two-thirds were physicians, and nearly 80% were HIV specialists. The percentage who reported recommending ART irrespective of CD4 count was higher at follow-up (15% versus 68%), as was the percentage who would initiate ART earlier for patients having unprotected sex with partners of unknown HIV status (64% versus 82%), and for those in HIV-discordant partnerships (75% versus 87%). In line with changing HIV treatment guidelines during 2010 to 2013, clinicians increasingly supported early ART for treatment and prevention.
Introduction
There are over 1.2 million persons living with HIV 1,2 and approximately 50 000 3,4 new HIV infections annually in the United States, motivating efforts for expanding access to HIV care and prevention interventions 5 to achieve national strategy goals. 6 Based on recent scientific evidence regarding the benefits of antiretroviral therapy (ART), 7 -9 there have been significant changes in US guidelines for the use of ART for the treatment of infection and for the prevention of HIV transmission, 10 as well as continued emphasis on routine opt-out HIV testing in clinical settings to increase timely HIV diagnosis. 11 Starting ART as soon as possible after HIV diagnosis, regardless of CD4 count, is now recommended as it reduces AIDS-related and non-AIDS-related morbidity and mortality. 10 Virologic suppression also reduces the risk of HIV transmission from HIV-infected persons to their sexual and needle-sharing partners. 8,12,13 To describe and gauge potential shifts in the attitudes and practices of HIV care providers regarding HIV treatment and testing during the conduct of the HPTN 065 (“Test and Linkage to Care Plus Treat” or TLC-Plus) 14,15 study in the Bronx, New York and Washington, DC, we surveyed the ART-prescribing clinicians at participating sites twice and analyzed their responses in 4 key areas—assessment of sexual risk behaviors, initiation of ART, use of ART to prevent HIV transmission, and routine HIV testing.
Methods
Study Design and Participants
The clinician survey was administered at baseline and follow-up as part of the HPTN 065 (TLC-Plus) study, which evaluated the feasibility of community-level expanded testing, linkage to care, and virologic suppression on ART, as a strategy for HIV prevention in the Bronx, New York and Washington, DC. 14,15 The baseline survey was conducted from September 2010 to May 2011 and a follow-up survey from May 2013 to December 2013. Antiretroviral therapy–prescribing clinicians at 39 participating HIV care sites were asked to complete an anonymous Internet-based survey to assess attitudes and practices regarding ART use and other elements of the HIV “test, link to care plus treat” interventions. Sites provided e-mail addresses of all their ART-prescribing clinicians (ie, physicians, nurse practitioners, physician assistants, residents, and fellows). The same sites were included in both surveys and select baseline results were described previously. 15 Clinicians received an introductory e-mail with survey instructions and up to 4 automated e-mail reminders during the next 3 weeks. Site investigators were also asked to encourage staff to complete the survey. Clinicians confirmed their consent for participation online prior to answering any questions; no identifying information was collected. Clinicians who accessed (but did not necessarily complete) the survey received a US$35 electronic gift certificate. The study protocol was approved by institutional review boards (IRBs) prior to sites’ participation. The protocol was exempt from IRB at the US Centers for Disease Control and Prevention (CDC) because the CDC staff did not interact with study participants or handle identifiable data.
Data Analysis
We assessed survey response rates, in aggregate and for each jurisdiction, at both baseline and follow-up. Baseline and follow-up survey results could not be linked by respondent, due to anonymous survey design. We summarized the characteristics of respondents and their clinical sites as well as clinicians’ attitudes and practices related to asking patients about HIV-transmission behaviors, prescribing ART for treatment and prevention, and routine HIV testing. Unless otherwise indicated, survey items were collected on 4-point Likert scales and analyzed in 4 separate strata or grouped for analysis into either 1 of the 2 dichotomous variables, depending on the distribution of responses—(1) agree/strongly agree versus disagree/strongly disagree or (2) strongly agree versus all other responses. We used Wilcoxon 2-sample test for continuous variables and the χ2 or Fisher exact tests for categorical variables to assess for statistical differences in distribution of responses across the 2 surveys. 16 We first presented the overall P values for differences in distribution of disaggregated responses and then P values for comparisons of select dichotomized variables. We could not account statistically for the correlation of survey responses for clinicians who completed the survey twice, and multiple comparisons may lead to type I error; therefore, in addition to statistical significance testing, our descriptive tables highlight in boldface the categories of variables for which there was a meaningful shift in response of at least 10%.
Using follow-up survey data, we conducted univariate and multivariable logistic regression analyses 16 to explore factors associated with select key outcome variables of interest—prescribing ART for HIV-infected patients regardless of CD4 count, prescribing ART for prevention, and support for 1-time testing and routine HIV testing in clinical settings. We evaluated the associations of several clinician-related variables (ie, age, sex, race/ethnicity, occupation, number of years caring for HIV-infected patients) and type of HIV practice against the aforementioned outcomes. In multivariable logistic regression models, we included all predictor variables from univariate analyses with χ2 P values less than .20. The final multivariable models retained all variables regardless of statistical significance, except for either age or years caring for HIV-infected patients, whichever had a lower P value, to avoid aberrant results due to multicollinearity.
Results
At baseline, 174 (60%) of 288 clinicians accessed the survey—94 (53%) of 177 from the Bronx, New York and 80 (72%) of 111 from Washington, DC. At follow-up, 150 (57%) of 264 clinicians accessed the survey—88 (56%) of 156 from the Bronx and 62 (57%) of 108 from DC. Nine surveys were excluded from both baseline and follow-up analyses because no questions were answered, resulting in 165 baseline and 141 follow-up surveys for analyses. Fifty-three (38%) of 141 respondents indicated on follow-up survey that they had also participated in the baseline survey.
Clinician Characteristics
In both surveys, almost 60% of respondents were female, the median age was 47 years, over 60% were white, two-thirds were physicians, and three-quarters reported caring for HIV-infected patients for >6 years (Table 1). Clinicians had a median of approximately 100 HIV-infected patients under their direct care and over half completed ART-related training in the past 3 months. Respondents at baseline and follow-up had a broadly similar patient mix, with almost 60% of patients being African American, about one-third women, and about one-third gay, bisexual, or other men who have sex with men (MSM).
Characteristics of the Clinicians Surveyed in the Bronx and Washington, DC, HPTN065, 2010 to 2013.a,b
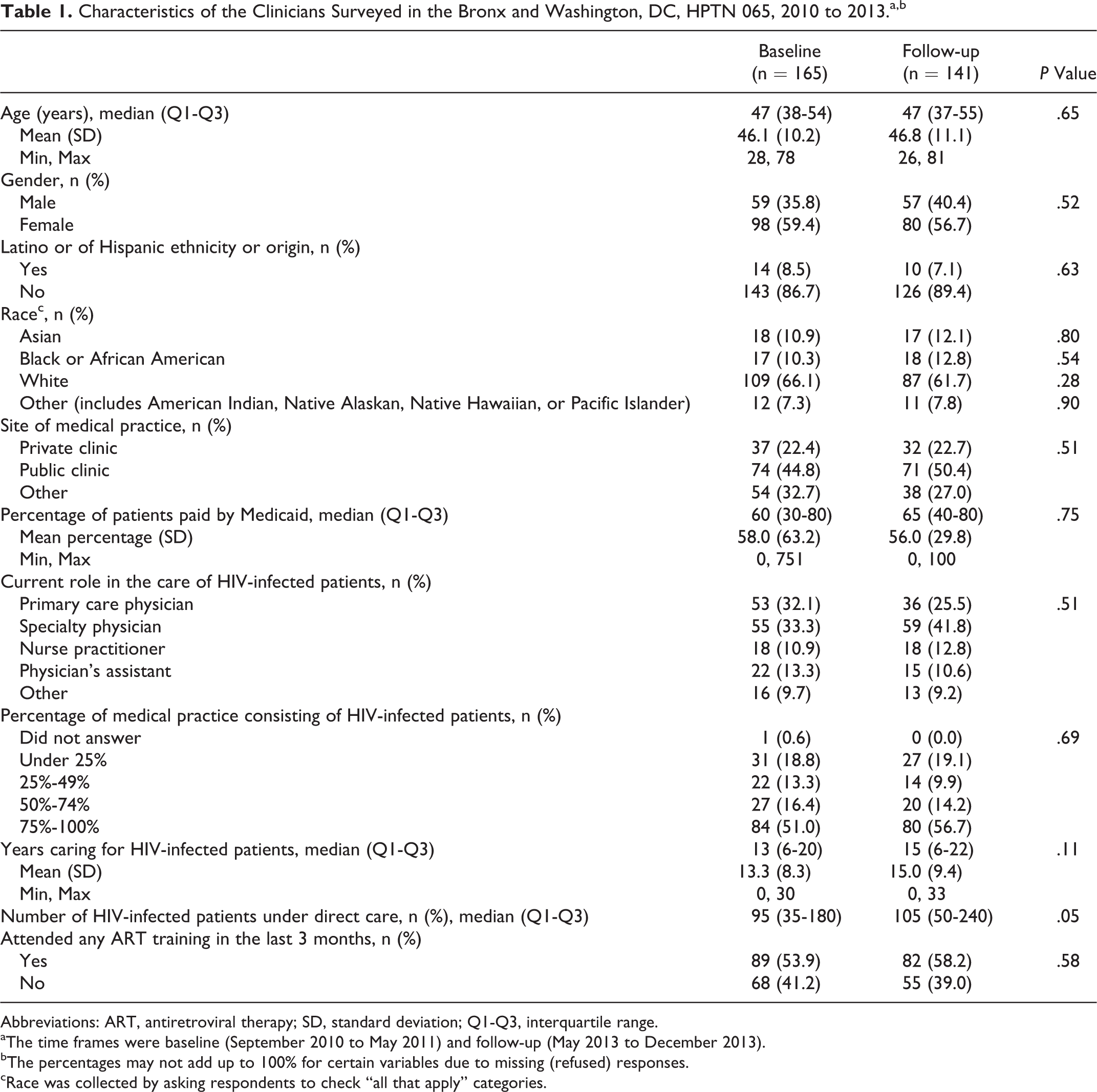
Abbreviations: ART, antiretroviral therapy; SD, standard deviation; Q1-Q3, interquartile range.
aThe time frames were baseline (September 2010 to May 2011) and follow-up (May 2013 to December 2013).
bThe percentages may not add up to 100% for certain variables due to missing (refused) responses.
cRace was collected by asking respondents to check “all that apply” categories.
Transmission Risk Behaviors
There were no appreciable differences between baseline and follow-up survey in the percentages of ART-prescribing clinicians who indicated always asking their HIV-infected patients about sexual partners (49% versus 53%; P = .44), sexual partners’ HIV status (37% versus 46%; P = .16), or use of condoms (58% versus 56%; P = .72), Table 2 (items 1-3).
Clinicians’ Practices and Attitudes Related to HIV Prevention and Treatment, HPTN065, 2010 to 2013.a,b

Abbreviations: ART, antiretroviral therapy; N/A, not applicable; SD, standard deviation.
aThe time frames were baseline (September 2010 to May 2011) and follow-up (May 2013 to December 2013).
bThe percentages may not add up to 100% for certain variables due to missing (refused) responses.
cAt baseline, this question was phrased: “In which of the following scenarios would you recommend that ART be initiated for HIV-infected patients in any circumstance?” and there was no option d.
Use of ART for Treatment
The percentage of clinicians who reported recommending ART initiation “irrespective of CD4 count” was substantially lower at baseline when compared to follow-up survey data (15% versus 68%, P < .001). Of note, the survey question phrasing and answer options were somewhat different at follow-up, as a result of changing recommendations on CD4 count threshold for ART initiation 10 (see Table 2, item 5 in note). At follow-up, 50% of respondents recommended ART initiation in general at CD4 count >500 cells/mm3 (at baseline, this response option was not disaggregated). Consequently, in sum, at follow-up, a total of 76% of clinicians supported ART initiation either at CD4 count > 500 cells/mm3 or irrespective of CD4 count level. Compared with baseline findings, we noted at follow-up that markedly smaller percentage of clinicians agreed or strongly agreed with a concern that patients will develop ART-resistant viral strains (43% versus 18%; P < .001) or will develop side effects, toxicity, or complications when ART is initiated too early (48% versus 21%; P < .001; Table 2, items 8 and 10).
Use of ART for Prevention
When comparing baseline and follow-up results, markedly higher percentages of clinicians at follow-up reported support for earlier initiation of ART for multiple patient groups—for patients having unprotected sex with partners of unknown HIV status (64% versus 82%; P < .001), for those in HIV-discordant sexual partnerships (75% versus 87%; P = .009), and for those newly diagnosed with a sexually transmitted infection (40% versus 65%; P < 0.001; Table 2, item 6). The percentage of clinicians who agreed or strongly agreed with the statement (item 12) “If a patient tells me that he or she is engaging in high-risk behaviors, I am more likely to recommend ART, irrespective of their CD4 count” was 71% at baseline versus 85% at follow-up (P < .001). The percentage who agreed or strongly agreed with the statement (item 7) “Early initiation of ART can slow the spread of HIV in a community by making patients less infectious to others” was consistently high (95% and 97%; P < .01). Clinicians estimated having initiated more patients on ART in the past year (item 16, a median of 10 at baseline versus 20 at follow-up; P = .04) and also reported initiating more patients on ART “with the main goal of making it less likely that these patients would transmit HIV to their sexual partners” (item 17, median of 0 versus 3 patients; P < .01). Of note, in the follow-up survey, 62% of clinicians strongly agreed and a further 29% agreed with this statement, “Because ART reduces the risk of HIV transmission, I routinely recommend ART to my HIV-infected patients” (Table 2, item 15).
Attitudes Regarding HIV Testing
HIV testing was offered at nearly all HIV care sites participating in HPTN 065, and almost 40% of respondents at both baseline and follow-up indicated that they personally administered HIV testing at the clinical sites where they worked (Table 3, item 1). The majority of respondents agreed or strongly agreed with a statement that “All persons in the United States should get tested for HIV at least once” (87% at baseline and 90% at follow-up, respectively; P = .37), and most also agreed or strongly agreed with routinely offering HIV testing in hospital emergency departments (EDs; 92% and 96%, respectively; P = .17) and for inpatients admissions (95% and 96%; P = .74; Table 3, items 5-7). There were meaningful differences in the percentages of clinicians who agreed or strongly agreed that a person should be able to get an HIV test kit at a drugstore without prescription to self-test for infection (61% versus 77%; P < .001) or to ask their partners to test (56% versus 76%; P < .001; items 8 and 9). On both surveys, about 97% of clinicians agreed or strongly agreed with a statement that “people who have unprotected sex or who use drugs should be offered an HIV test at least once per year” and over 93% agreed or strongly agreed with a statement that “people with an STI or TB should be tested for HIV no matter when their most recent HIV test was,” items 10 and 11. In analyses restricted to clinicians who cared for any HIV-uninfected patients, 115 (93%) of 124 at baseline versus 106 (97%) of 109 at follow-up reported offering HIV testing to all of their patients aged 13 to 64 years (P = .20), consistent with prevailing CDC recommendations (item 12).
Clinicians’ Practices and Attitudes Related to HIV Testing, HPTN065, 2011 to 2013.a,b
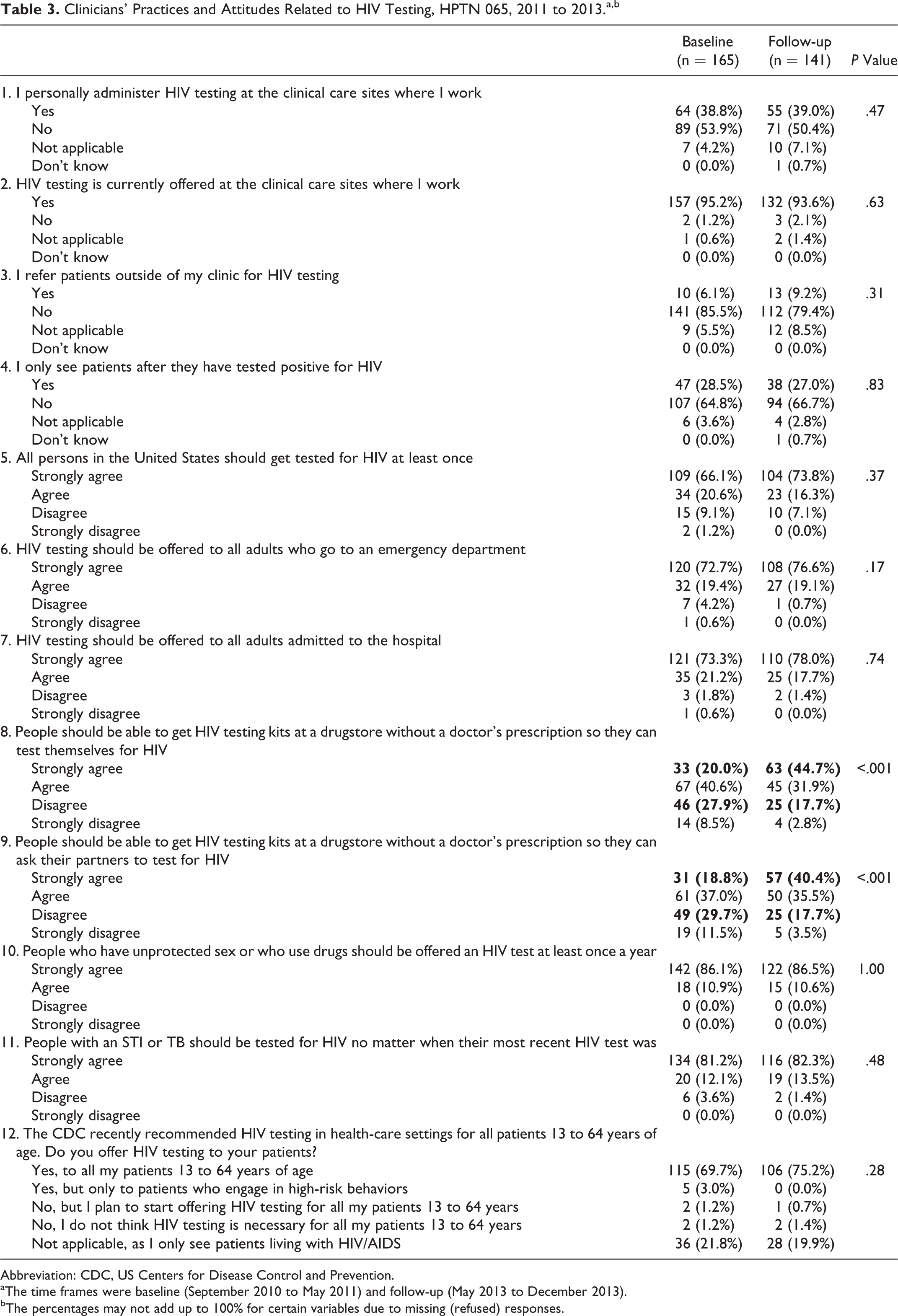
Abbreviation: CDC, US Centers for Disease Control and Prevention.
aThe time frames were baseline (September 2010 to May 2011) and follow-up (May 2013 to December 2013).
bThe percentages may not add up to 100% for certain variables due to missing (refused) responses.
Clinician Characteristics Associated with Select Outcomes at Follow-up
At follow-up, 68% of clinicians indicated that they would generally recommend that ART be initiated for all patients irrespective of CD4 count, and 88% strongly agreed that early ART can slow the spread of HIV in a community by making patients less infectious to others (Table 2). For both outcomes, the only univariate correlates (P < .20) were younger age (positively associated) and years caring for HIV-infected patients (negatively associated). The only statistically significant result was that compared with clinicians aged >49 years, those aged ≤40 years were more likely (odds ratio [OR]: 1.3, 95% confidence interval [CI]: 1.1-6.7) to prescribe ART regardless of CD4 count (clinicians aged 41-49 years were as likely as those aged >49 years). At follow-up, 62% of clinicians strongly agreed with the statement that “Because ART reduces the risk of HIV transmission, I routinely recommend ART to my HIV-infected patients” (29% agreed and 6% disagreed/strongly disagreed; Table 2). In univariate analyses, younger age and fewer years caring for HIV-infected patients were significantly associated with strong agreement with this statement; in the multivariable analyses, only age ≤40 years versus >49 years was associated with strong agreement with routinely recommending starting ART to reduce HIV-transmission risk (OR: 3.7, 95% CI: 1.2-11.3).
At follow-up, 74% of clinicians strongly agreed that “All persons in the United States should get tested for HIV at least once”; 16% agreed with this statement and 7% disagreed. In univariate analyses, women and younger clinicians were more likely to strongly agree, whereas clinicians of “other” race/ethnicity (compared with white), and those caring for HIV-infected patients longer were less likely to strongly agree. In multivariable models, the only independent correlates, both of which were negatively associated with strong agreement with universal lifetime testing, were “other” race/ethnicity (OR: 0.1, 95% CI: 0.04-0.4) and longer years caring for HIV-infected patients (OR: 0.4, 95% CI: 0.2-0.7). Similarly, strong agreement with offering HIV testing to all adults who go to the ED or to all adults admitted to the hospital (Table 3) was less frequent among older clinicians and those caring for HIV-infected patients longer. In multivariable analyses, 10 additional years of caring for HIV-infected patients was associated with about 70% reduction in strong agreement with routine offering of HIV testing in the ED (OR: 0.3, 95% CI: 0.1-0.9) and about 50% reduction in strong agreement with routine offering of HIV testing to inpatients (OR: 0.5, 95% CI: 0.3-0.8).
Discussion
A follow-up survey of ART-prescribing clinicians found that, compared with baseline findings more than 2 years previously, a higher percentage of clinicians in the Bronx, New York and Washington, DC supported the use of early ART for treatment and prevention and that support for routine HIV testing in medical settings was consistently high. Respondents of the follow-up survey voiced more support for prescribing ART to HIV-infected patients irrespective of CD4 count, for patients reporting unprotected sex, and for those in an HIV-discordant relationship. These shifts in practices are consistent with (1) the scientific evidence that emerged between baseline and follow-up surveys about the effectiveness of ART for reducing HIV transmission in discordant couples (the HPTN 052 results were published in July 2011) 8 and (2) the updated US Department of Health and Human Services HIV treatment guidelines (first released in February 2013) recommending ART initiation regardless of CD4 count based on individual clinical benefits in observational studies and START randomized clinical trial. 9,10 Early ART for treatment and prevention and routine HIV testing are critical to realizing the benefits of ART for individual health and reduction in HIV transmission. 5
Our follow-up results regarding the percentage of providers who reported prescribing ART regardless of CD4 count (68%) were similar to the analogous population-based results from the Medical Monitoring Project surveying HIV clinicians from June 2013 to January 2014 (71%), 17 and those from a regional survey among infectious disease physicians in late 2013 (69%). 18 However, our result (68%) was lower than the estimate from the September 2014 survey of infectious disease physicians in the United States and Canadian Emerging Infections Network (87%). 19 The waning concerns about emergence of resistance and about side effects noted in our follow-up survey likely reflect accumulating evidence that newer ART regimens are more potent, better tolerated, and more forgiving of nonadherence, 10 thus resulting in fewer patients experiencing virologic failure 1,20 and developing antiretroviral drug resistance. These increasingly more effective ART regimens have contributed to improved life expectancy over time. 21
Most clinicians who completed the survey supported using ART for prevention at baseline in 2010-2011; the level of support was even higher among participants at follow-up survey. Their attitudes were also in line with the growing evidence from clinical trials and observational studies regarding reduced infectiousness with ART 8,12 and population-level epidemiologic and modeling studies (reviewed by Wilson) 22 which, to varying degree, suggest public health benefit of ART for limiting the continued spread of HIV in high- and low-income settings. 13,23 -26 Although the practice of always asking HIV-infected patients about the HIV status of their sexual partners was also more common at follow-up than baseline, there was a substantial minority of clinicians (20% to 25%, depending on the survey question) who indicated they “never”, “rarely,” or only “occasionally” asked patients about sexual partners, partners’ HIV status, and condom use. These results reinforce the need for continued efforts and structural interventions (eg, computer prompts) with clinicians to enhance HIV prevention efforts with positive persons. 5 Ascertaining the HIV status of sexual partners by clinicians can open the door to discussions about not only the use of ART by the HIV-infected partner to reduce transmission risk but also the use of condoms, preexposure prophylaxis, or postexposure prophylaxis by HIV-uninfected partners, 27 to further limit HIV transmission in HIV-discordant partnerships. 13,28,29
In July 2013, during implementation of the study, the US Preventive Services Task Force released the recommendation that clinicians screen persons aged 15 to 65 years (younger adolescents and older adults who are at increased risk should also be screened) for HIV infection at least once, with at least annual rescreening of individuals at very high risk, such as sexually active MSM. 11 In our study, ART-prescribing clinicians expressed consistently high levels of support for routine HIV testing in clinical settings, including EDs and inpatient wards, both in the baseline and in the follow-up survey. This enthusiasm, however, may not translate into hospital practice. In another component of HPTN 065, aimed at scale-up of HIV screening in EDs and inpatient settings at participating hospitals, the observed scale-up was modest over the 3-year study period and limited by institutional and logistical obstacles, 30 echoing findings from prior US-based studies. 31 Finally, use of commercially available HIV-test kits for self-testing or offering testing to partners was also favored by the majority of clinicians at baseline; support was even higher during follow-up, after the US Food and Drug Administration (FDA) approved the first in-home HIV test in July 2012. Acceptability and demand for such testing options was documented in prior studies in New York 32 and in a survey in Philadelphia 33 and may increase as more affordable and accurate HIV rapid test kits enter the market. 34
The study has notable strengths, including that it targeted a large panel of HIV providers in 2 urban areas with a high burden of HIV infection. These were providers from the 20 largest local HIV clinics (based on HIV surveillance data) per jurisdiction, representing the majority of HIV providers in each area. Our repeat survey permitted us to infer secular changes in attitudes, beliefs, and practices in the same centers over a 2-year time span. The Web-based, anonymous design was chosen to maximize participation and honest responses.
The findings from this research are also subject to some caveats. Despite repeat reminders, a nominal incentive for accessing the survey, and its anonymous design, the participation rates in our Web-based survey were under 60%, thus response bias is possible. However, these response rates are within the range reported for Web-based clinician surveys. 35 Because of the survey design, we were unable to compare the characteristics of the respondents to nonrespondents to assess how nonresponse may have affected our results; we also could not track changes in responses for the same individuals over time. We focused on highlighting meaningful (at least 10%) differences in survey responses, because statistical contrasts could not account for potential correlation in responses of repeat anonymous participants, and because approximately 5% of results could be statistically significant by chance alone due to multiple comparisons. Although survey response rate could be tracked for each jurisdiction (Bronx, New York versus Washington, DC), we were unable to analyze results on both surveys by jurisdiction. Finally, clinicians surveyed were, on average, highly experienced and had sizable patient loads; thus, our results may not be applicable to clinicians with lesser experience or to those who treat few patients with HIV.
In summary, we observed that ART-prescribing clinicians were increasingly supportive of offering ART to all HIV-infected patients and there was high and increasing enthusiasm for use of ART for prevention. Respondents also favored offering HIV testing in clinical settings and access for patients to self HIV testing. Our study findings can be used to inform continuing medical education for clinicians as well as to inform health-care policies and institutional environments that maximize voluntary opt-out HIV screening and enable prompt ART initiation with ongoing support for those diagnosed with HIV.
Footnotes
Authors’ Note
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention.
Acknowledgments
The authors wish to thank participating clinicians and study sites as well as the study protocol team members.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Institutes of Health (NIH) grants #UM1 AI 068619-05 and UM1 AI 068617 and the National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, Centers for Disease Control and Prevention.
