Abstract
Keywords
Introduction
HIV prevention priorities have shifted over the past decade, from reducing the risks of uninfected individuals contracting HIV, to treating persons already infected. 1 The rationale for targeting prevention to HIV-positive persons is in part based on the potential for reducing HIV infectiousness with antiretroviral therapy (ART). 2,3 Mathematical models project that implementation of universal HIV testing and treatment could have a significant impact on HIV incidence at the population level. 3 –5 Although using HIV treatments for prevention poses significant challenges for generalized HIV epidemics in resource-constrained countries, 6 –8 treatment for prevention is being implemented in the United States and Canada. For example, the San Francisco Health Department currently offers ART to all persons who test HIV positive. Similar initiatives are underway in the Bronx, New York, Washington, DC, and Vancouver, British Columbia. The potential for ART to reduce infectiousness and prevent new HIV infections led the Swiss Federal AIDS Commission to state that people living with HIV/AIDS who have effectively suppressed HIV replication, as demonstrated by repeated undetectable viral load test results, can be considered noninfectious; alleviating concerns about HIV transmission. 9,10 Research suggests that this policy shift has indeed resulted in less protected sexual behavior among men and women who are familiar with the Swiss policy and have undetectable viral loads. 11
Although the potential for ART to prevent HIV infections is theoretically compelling, there are at least 2 behavioral factors that will undermine the use of HIV treatments for prevention: poor adherence to ART regimens and co-occurring sexually transmitted infections (STIs). 12 Specifically, poor treatment adherence results in nonsuppressive therapeutic levels of ART and therefore unchanged infectiousness. Even worse, nonadherence can lead to resistant virus that can subsequently be transmitted to others. 13,14
Less obvious is the role of co-occurring STI in the infectiousness of genital secretions. Sexually transmitted coinfections increase HIV viral shedding in the genital tract, resulting in significant increases in HIV infectiousness. 15 Local inflammation activates HIV replication in the genital immune compartment independent of HIV in peripheral blood, such that a person can have a clinically monitored undetectable blood viral load while they are highly infectious in their genital tract. 15,16 Thus, people living with HIV and sexually transmitted coinfections are more infectious than they can possibly know from routine monitoring of blood plasma viral load. 17 A recent review of research on the correspondence between HIV concentrations in blood and semen found only moderate concordance; the mean correlation between blood plasma viral load and semen viral load was r = .44, accounted for at least in part by sexually transmitted coinfections. 17
The overall median point-prevalence of confirmed STI in people living with HIV/AIDS is 12.4%, and the most common STI in people with HIV are those that cause HIV shedding, specifically syphilis (median 9.5% prevalence), gonorrhea (9.5%), chlamydia (5%), and trichamoniasis (18.8%). 18 Although STIs are frequently detected at the time of HIV diagnosis, reflecting the role of STI in HIV transmission, exposure to sexually transmitted pathogens persists long after HIV diagnosis. In addition, STI prevalence among individuals receiving HIV treatment is not appreciably different from their untreated counterparts. 19 Sexually transmitted coinfections have significant implications for people receiving ART, especially when ART is administered with the intent of preventing HIV transmission.
The purpose of the current research was to examine the history of co-occurring STI in a community sample of people living with HIV/AIDS who are receiving ART. We examined STI diagnoses during the time since testing HIV positive and STI diagnoses in the 12 months prior to assessment. To determine factors associated with sexually transmitted coinfections, we compared persons who had been diagnosed with an STI since testing HIV positive to those who had not been diagnosed with an STI on demographic, behavioral, and health characteristics. We hypothesized that people living with HIV/AIDS and receiving ART, who have a history of STI diagnoses would demonstrate a pattern of continued substance use and sexual behaviors that maintain risks of new STI, greater infectiousness, and HIV transmission.
Methods
Participants
People living with HIV/AIDS (N = 713) were recruited through community sampling to participate in a single-session survey. We used targeted venue and snowball sampling techniques to identify individuals in and out of care. Recruitment relied on responses to brochures placed in waiting rooms of HIV service providers and infectious disease clinics throughout Atlanta, Georgia, as well as an explicit systematic approach to word-of-mouth chain recruitment. Specifically, participants were given recruitment brochures and encouraged to refer their HIV-infected friends to the study. These procedures were designed to extend recruitment beyond any one service setting in order to achieve a broad community sample.
Interested persons contacted our research program to schedule an assessment appointment. The study entry criteria were age 18 years or older and proof of positive HIV status using a photo ID with either a matching ART prescription bottle, HIV clinic card, positive HIV test result, laboratory report, or any other proof of positive HIV status. Participants received $25 for completing the computerized interview (approximately 1 hour). Data were collected between December 2009 and March 2011. All study procedures were approved by the university institutional review board.
Measures
For the purposes of the current study, measures included STI history, demographic characteristics, substance use, STI knowledge, and sexual behavior. Measures were administered using audio-computer-assisted structured interviews (ACASIs) to reduce demand characteristics and socially evoked response biases. 20,21
STI history
Participants reported whether they had been diagnosed with gonorrhea, chlamydia, syphilis, genital herpes, or trichomoniasis in 2 separate time frames. First, we asked the participants whether they had ever been diagnosed with any one of the STIs. Participants who had been diagnosed with an STI were asked for the approximate date of their last diagnosis. We used the date of STI diagnoses and date of their HIV-positive diagnosis to determine whether each STI had been diagnosed since testing HIV-positive. Specifically, we defined having been diagnosed with an STI after testing HIV positive if the difference in dates was 1 month or greater. We also used the dates to determine whether the diagnosis had occurred within the previous 12 months of the assessment session. We used the same format to assess the occurrence of genital ulcers, genital pain, and unexplained genital discharge to detect potentially undiagnosed STI symptoms. We did not, however, include these nonspecific symptoms in our definition of having contracted an STI.
Demographic and health characteristics
Participant characteristics including gender (including whether they were transgender), age, years of education, income, ethnicity, and employment status were collected. We assessed HIV-related symptoms using a previously developed and validated measure concerning the experience of 14 common symptoms of HIV disease. 22 Participants also indicated their most recent CD4 count and viral load. We asked the participants whether they were currently being treated with ART and those who were receiving treatment indicated whether they had missed any of their ART in the past week. Participants also responded to a single-item rating scale for assessing medication adherence. The adherence rating scale asked the individuals to indicate the point along a continuum showing how much of ARTs they have taken in the past month. 23,24 For the computerized administration, we adapted the response format by using a 100-point slide bar tool anchored by 0%, 50%, and 100%. The specific instructions read as follows “We would be surprised if most people take 100% of their medications. Below, 0% means you have taken no HIV medications the past month, 50% means you have taken half of your HIV medications the past month, and 100% means you have taken every single dose this past month. What percentage of your HIV medications did you take?” Participants indicated the percentage of medications taken by clicking their mouse anywhere on the 100-point slide bar continuum. The adherence rating scale has been found reliable and valid. 23,24
STI knowledge
We administered 14 items from a previously developed test of STI knowledge, reflecting a broad range of information about STI transmission, symptoms, and disease manifestations. 25 The specific items used in this study are shown in the results. Items were responded True/False and Do Not Know, with Do Not Know responses scored incorrect. The total score was obtained by calculating the percentage correct responses, Kuder Richardson-20 coefficient = .71.
Sexual risk and protective behaviors
Participants responded to items assessing their number of male and female sex partners and frequency of sexual behaviors in the previous 4 months. Specifically vaginal and anal intercourse with and without condoms were assessed within HIV-seroconcordant and -serodiscordant partnerships. A 4-month retrospective period was selected because previous research has shown reliable reports for numbers of sex partners and sexual events over this time period. 26 Participants were instructed to think back over the past 4 months and estimate the number of sex partners and number of sexual occasions in which they practiced each behavior. The instructions included cues for recollecting behavioral events. Data were analyzed within seroconcordant and serodiscordant relationships with individual behaviors examined as well as behaviors collapsed across unprotected and protected aggregates. In addition, we calculated the percentage of intercourse occasions in which condoms were used by taking the ratio (condom-protected vaginal + condom-protected anal intercourse/total vaginal + total anal intercourse).
Data Analyses
All of the main analyses for this study focused on the participants who were currently taking ART. We first conducted descriptive analyses to determine the prevalence of sexually transmitted coinfections in the sample. We report STI diagnoses and symptoms for the time since testing HIV positive and in the past year, separately for men, women, and transgender persons. Based on this initial analysis, we identified participants who had and had not contracted an STI since testing HIV positive. Groups were compared using bivariate logistic regressions on demographic, health, substance use, STI knowledge, and sexual behaviors. Predictors found significant in bivariable models were selected for inclusion in the multivariable model. For all analyses, we performed logistic regressions, reporting odds ratios and 95% confidence intervals, with statistical significance defined as P < .05.
Results
Among the 713 persons screened for the study, 570 (79%) were currently taking ART. The final sample for analyses consisted of 415 men and 155 women, of which 26 men and 19 women identified as transgender. Table 1 shows the rates of STI since testing HIV positive and rates for having been diagnosed with an STI in the past year. Overall, 53% of participants had been diagnosed with at least 1 STI since testing HIV positive, 29% were diagnosed once, and 24% had 2 or more diagnoses. Genital pain and discharge since testing HIV positive were also reported. In terms of STI diagnoses in the past year, 26% women, 19% men, and 12% transgender persons had been diagnosed with an STI. The most common new STI diagnosis was genital herpes (7%), followed by gonorrhea (6%) and the least common was trichomoniasis (1%).
Sexually Transmitted Coinfections Reported since HIV Diagnosis and in the Previous Year among Men, Women, and Transgender Persons Receiving ART a
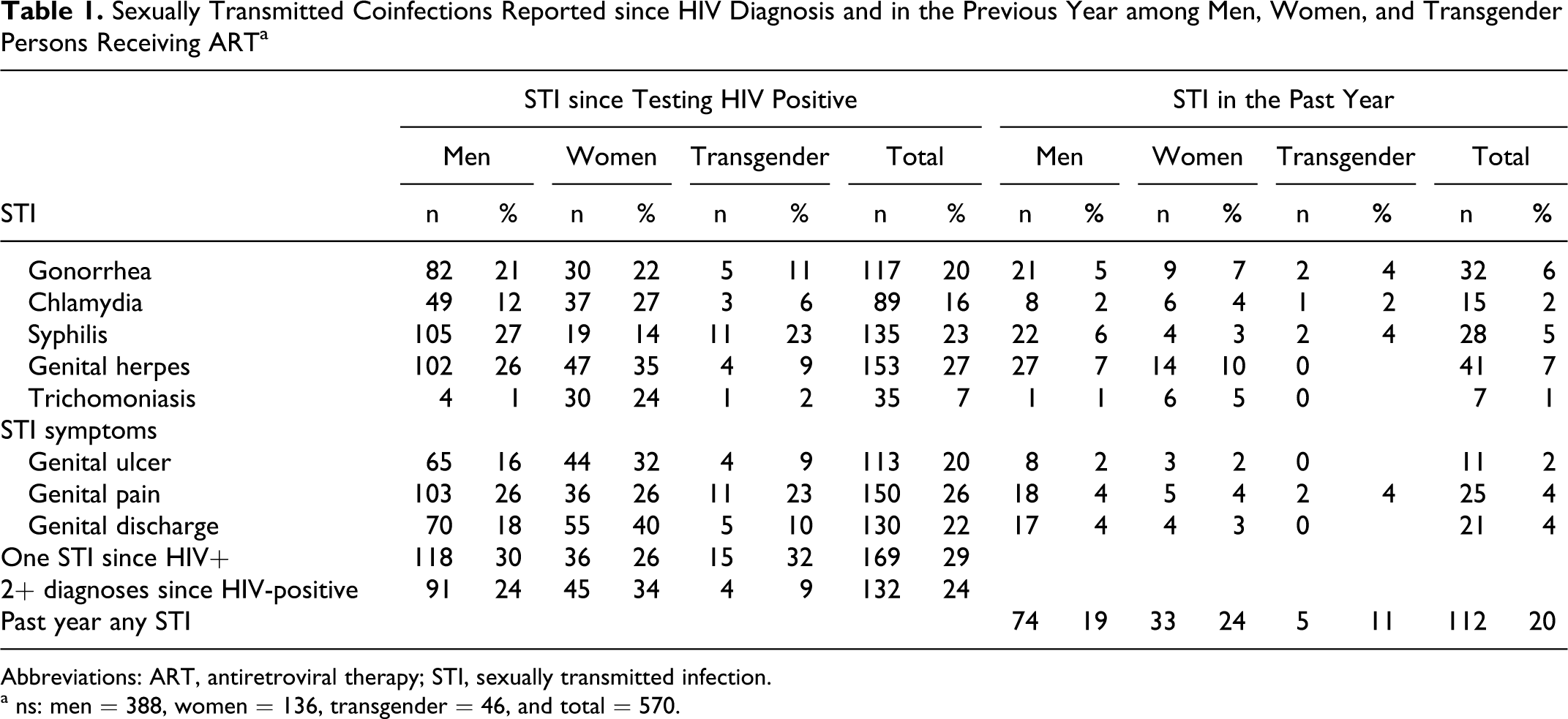
Abbreviations: ART, antiretroviral therapy; STI, sexually transmitted infection.
a ns: men = 388, women = 136, transgender = 46, and total = 570.
Demographic and Health Characteristics
Analyses showed that participant age differed between people living with HIV who were and were not diagnosed with an STI; persons diagnosed with a post-HIV STI were significantly younger. In addition, participants who had been diagnosed with an STI were significantly more likely to have a history of incarceration. The history of STI was not associated with gender, income, race, or employment status. In terms of health-related factors, individuals who had been diagnosed with an STI were significantly more likely to have missed their HIV medications in the previous week and were more likely to have taken less than 85% of their medications in the previous month (see Table 2 ).
Demographic, Health Characteristics and Substance Use among People with HIV Who Had Not and Who Had Been Diagnosed with an STI since Testing HIV Positive

Abbreviations: ART, antiretroviral therapy; CI, confidence interval; OR, odds ratio; STI, sexually transmitted infection; VAS, visual analogue scale.
a P < .05.
b P < .01.
Substance Use
Participants with a history of STI since testing HIV positive indicated significantly more alcohol and other drug use in the previous 4 months. Nearly 2 of 3 persons with STI diagnoses drank alcohol, half had used at least 1 illicit drug, and 30% were poly-drug users. In addition, having had an STI was associated with using drugs before sex in the past 4 months (see Table 2).
STI Knowledge
Results showed that most participants, regardless of whether they had an STI, did not have high levels of accurate information about STI transmission, symptoms, and treatment (see Table 3 ). Less than half of the participants were aware that genital herpes can be transmitted in the absence of genital ulcers and 1 in 3 believed that having gonorrhea resulted in an immunity against future infection. In addition, only 37% of participants knew that sexually transmitted coinfections increase HIV infectiousness in genial fluids. Results also showed that individuals who had been diagnosed with an STI answered more STI knowledge items correctly compared to participants who had not been diagnosed with an STI.
Knowledge about STI (Percentage Correct) among People with HIV Who Had Not and Who Had Been Diagnosed with an STI since Testing HIV Positive

Abbreviations: CI, confidence interval; OR, odds ratio; STD, sexually transmitted disease; STI, sexually transmitted infection.
a P < .05.
b P < .01.
Current Sexual Behaviors
The associations between having been diagnosed with an STI since testing HIV positive and recent sexual behaviors are shown in Table 4 . A total of 248 (43%) participants reported HIV-positive (seroconcordant) sex partners in the past 4 months and 193 (33%) had sex partners in that time period whose HIV status was negative or unknown (serodiscordant). Among the individuals with serodiscordant partners, 107 (55%) had engaged in unprotected vaginal or anal intercourse. Comparisons between groups demonstrated that individuals with a history of STI since their HIV diagnosis were significantly more likely to have HIV-positive (seroconcordant) sex partners in the past 4 months. Participants with a history of sexually transmitted coinfection were more likely to have engaged in unprotected anal intercourse and total unprotected intercourse, as well as significantly more occurrences of these behaviors.
Sexual Risk and Protective Behaviors among People with HIV Who Had Not and Who Had Been Diagnosed with an STI since Testing HIV Positive

Abbreviations: CI, confidence interval; M, mean; OR, odds ratio; STI, sexually transmitted infection.
a P < .01.
b P < .08.
c P < .05.
Multivariable Model
To identify factors independently associated with history of STI since testing HIV positive, we tested a multiple logistic regression model predicting having been diagnosed with an STI since testing HIV positive (See Table 5). Results showed that younger age, more accurate knowledge about STI, drug use, and having missed ART in the past week were significantly associated with having had an STI.
Multivariable Logistic Regression Model Predicting History of Post-HIV STI

Abbreviations: CI, confidence interval; OR, odds ratio; STI, sexually transmitted infection.
a P < .01.
b P < .05.
Discussion
The current study demonstrates a history of post-HIV diagnosis of STI is common among people receiving ART. We found that more than half of the participants had been diagnosed with at least 1 STI since testing HIV positive. Among those who had been diagnosed with an STI, 24% had 2 or more STI diagnoses. More than 1 in 4 women and nearly 1 in 5 men receiving ART had been diagnosed with an STI in the previous year. The symptoms of STI were also common in the previous year. These STI rates are consistent with a recent review that found a median STI point prevalence of 12% among people living with HIV. 18 We confirmed our study hypothesis that people living with HIV who had a history of STI were at continued high risk of contracting new STI, increased infectiousness, and transmitting HIV. These results have implications for the use of ART to lower HIV infectiousness.
Local inflammation of the genital tract caused by sexually transmitted coinfections increases HIV viral shedding and therefore HIV infectiousness. 8 Although several ART regimens effectively penetrate the genital immune compartment, activation of HIV-infected CD4 cells spike HIV concentrations in genital secretions, 27,28 increasing infectiousness beyond what can be estimated from peripheral blood viral loads. We found that having contracted an STI was associated with poor ART adherence, posing an added challenge to using ART for HIV prevention. Multiple factors that can act as mediating variables may explain the association between sexually transmitted coinfections and ART nonadherence including the use of alcohol and other substances, health consciousness, and quality of health care. 29 Individuals who contract STI and are nonadherent to ART will likely remain highly infectious and undermine the preventive effects of using ART for HIV prevention.
We found surprisingly high levels of misinformation about STI in this sample of people living with HIV/AIDS. Similar to studies of other populations, 25 people living with HIV only knew about half of the STI knowledge items. Contrary to expectations, individuals who had been diagnosed with an STI since testing HIV positive had slightly better STI knowledge, possibly reflecting educational and counseling experiences related to their previous STI. Despite their greater STI knowledge, those who had been diagnosed engaged in substantially more risk behaviors. This finding suggests a need for STI education among people living with HIV but education alone will not be sufficient to prevent new infections.
The findings from this study should be interpreted in light of its methodological limitations. Our methods relied on self-reported health status and sexual behaviors which may have been affected by self-report biases, tendencies to underreport sexual behaviors and substance use, 30 as well as overreport medication adherence, 31 suggesting that the behaviors reported here should be considered lower-bound estimates. Our cross-sectional study design also precludes any causal or directional conclusions. Our measures may have excluded important covariates that could have helped explain the results, such as quality of health care, stigmas associated with STI, and perceptions of infectiousness. Finally, our results are based on a convenience sample that is predominantly African American people living with HIV/AIDS in 1 southern US city. Although our results converge with other studies, caution is warranted before generalizing these findings to other populations of people living with HIV/AIDS. Acknowledging these limitations, we believe that our findings have implications for programs that seek to test and treat people for HIV prevention.
Co-occurring STIs are a significant threat to the potential for HIV treatments to reduce HIV infections. Indeed, mathematical models of infections averted by population scale-up of ART are unrealistically optimistic when they do not include estimates of sexually transmitted coinfections. 3,32 Although viral load in the genital tract is typically lower than viral load in blood plasma, this association is inverted when there are co-occurring STIs. 33 Implementing ART as a preventive strategy therefore requires aggressive STI detection and treatment. Patients taking ART should receive comprehensive information about STI as well as instruction in symptom detection, self-examination, and ability to attain sexual health services. Sexual history taking, STI screening, and risk-reduction counseling should be fully integrated with the routine care for people with HIV/AIDS. Failure to allocate adequate resources to stemming STIs and monitoring adherence in people receiving ART could render HIV treatment ineffective in preventing HIV transmission.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Institute of Mental Health R01-MH082633 and the National Institute on Alcohol Abuse and Alcoholism RC1-AA018983.
