Abstract
Background:
Without antiretroviral therapy (ART), approximately one-half of HIV-infected infants will die by two years. In 2010, the World Health Organization (WHO) recommended that all HIV-infected infants < 24 months be initiated on ART regardless of their clinical/immunologic status. However, there remains little published data detailing cohorts of infants on ART in Sub-Saharan Africa. This study describes baseline characteristics and 12 month outcomes of a cohort of HIV-infected children < 24 months of age at pediatric HIV centers in Mwanza and Mbeya, Tanzania.
Materials and Methods:
Retrospective chart review. Inclusion criteria: children < 24 months of age, initiated on ART at Baylor Children s Foundation Tanzania clinics, between March–December 2011.
Results:
Conclusion:
Our cohort of HIV infected children < 24 months initiated on ART did well clinically at 12 month outcomes despite being severely immunocompromised and malnourished at baseline. Nevirapine based regimens had good 12 month clinical outcomes, regardless of maternal exposure. Loss to follow-up rate was high for our cohort, demonstrating the need to develop strong mechanisms to counteract this.
Introduction
Despite improved global coverage in the prevention of mother-to-child HIV transmission (PMTCT), daily more than 700 infants are infected with HIV, mostly in the resource-constrained settings in Africa. 1 In the absence of antiretroviral therapy (ART), these children have a high risk of death, that is, more than one-third by 1 year of age, 2 one-half by 2 years of age, 3 and nearly two-thirds before the age of 5 years. 4 Important questions in addressing pediatric HIV in Africa revolve around when and what ART regimen to initiate in children. 2,5 –7 In 2010, on the basis of both observational and clinical trial data demonstrating the survival benefit imparted by beginning ART in infancy, 5,8 –11 the World Health Organization (WHO) recommended initiating ART in all HIV-infected children younger than 24 months, irrespective of their clinical or immunological status. 12 In June 2013, the WHO extended its guidance to initiate ART on all children <5 years of age, but it has yet to be implemented by most partnering countries. 13 The 2013 guideline will expand ART coverage and should reduce lost to follow up (LTFU), which is a factor in HIV morbidity and mortality. 13 In Tanzania, where our study was conducted, of the children who qualify for ART only 26% are on treatment, 14 and there is growing evidence that while patients wait to become eligible for ART there is a high rate of LTFU. 15
The WHO 2013 guidelines also recommend initiating a ritonavir-boosted lopinavir (LPV/r)-based regimen in treatment for all HIV-infected children <3 years of age, regardless of nevirapine (NVP) exposure during PMTCT. The 2013 guideline was made based on new clinical evidence that young children acquire resistance to NVP even without exposure to PMTCT and that children on LPV/r-based regimen have better virologic suppression. 16 However, in many African settings such as ours in Tanzania, early treatment and use of LPV/r-based regimens have several barriers to implementation including LPV/r liquid formulation, which has poor palatability and requires cold chain transport, an LPV/r tablet formulation that cannot be crushed due to the bioavailability effects, and funding limitations due to its expense. 5,17 It is therefore of great interest to know how nonnucleoside reverse transcriptase inhibitor (NNRTI)-based ART regimens perform in resource-constrained settings where initiation with LPV/r-based regimen is still not possible.
As the ART programs in Africa attempt to adapt new WHO guidance for their particular settings, evidence from routine clinical settings outside clinical trials is of substantial utility, especially in resource-limited settings such as ours with limited access to LPV/r and virologic testing. To date, there are limited published data on clinical outcomes at African sites that have followed the 2010 WHO guidelines of initiating ARTs in all HIV-infected children <24 months of age. Our study objective is to describe 12-month clinical outcomes and survival in a cohort of HIV-infected children in Tanzania in whom ART was initiated before the age of 24 months.
Materials and Methods
Opened in 2011, the Baylor Children’s Foundation Tanzania Clinical Centres of Excellence (COEs) is Tanzania government’s principal partner in pediatric HIV care and treatment. Baylor Children’s Foundation Tanzania is part of a larger consortium of pediatric HIV clinics within the Baylor International Pediatric AIDS Initiative (BIPAI) at Texas Children’s Hospital. Within Tanzania, there are 2 COEs, one located in Mwanza and the other in Mbeya. Both COEs are funded by the US Agency for International Development (USAID). As of 2013, Tanzanian COEs combined have >2500 HIV-infected or HIV-exposed children in care, of whom more than 1800 are on ART. The COEs have clinicians specialized in pediatric HIV care, including nutritionists, counselors, and social workers. Patients receive care at the COEs every 1 to 3 months depending on the health of the child and receive enough ART to last until their next scheduled appointment.
Retrospective chart review was performed to collect and analyze relevant information. All children <24 months of age registered at either Tanzania COEs, who commenced ART between March 2011 and December 2011 were included in this study. The clinics became operational in March 2011. Patients were excluded from the study if they were ≥2 years of age at the time of ART initiation or were started on ART by an outside facility. Laboratory and demographic data were obtained from the patients’ medical records and analyzed for the following characteristics: ART initiation date, age at initiation, ART regimen, gender, PMTCT exposure, LTFU, mortality, CD4%, WHO clinical or treatment stage; WHO immunosuppression classification; and nutritional status.
Patients who were LTFU, transferred, or died before the outcome period of 12 months were not considered for analysis comparing baseline and outcome groups. Lost to follow up was defined as a patient who did not return for appointment >3 months during the study period, this is because all patients are routinely seen every 1 to 3 months and are given only enough ARTs for that time period. Patients who were not seen for >3 months but later returned for care were still considered LTFU due to the ART nonadherence and failure of our team to track these patients and return them into care in an appropriate time frame. Data used for outcome analysis were based on laboratory and anthropometric measurements closest to 12 months from ART initiation but not <9 months or >15months post-ART initiation. All children <24 months were initiated on ART per the criteria stated in the 2010 WHO and Tanzanian Guidelines. 12,18 At the time of the study, first-line regimen for children was zidovudine–lamivudine–nevirapine (ZDV-3TC-NVP). The use of LPV/r for NVP-exposed infants was not routinely practiced and none of our patients were started on this regimen. At the time of study, Tanzania was practicing option A for PMTCT as set by the 2010 WHO PMTCT Guidelines, 19 that is, daily NVP for infant until 1 week after exposure to breast milk has ended and for the mother twice daily (BID) ZDV after 14 weeks of gestation, with single-dose NVP at delivery and initiation of ZDV-3TC BID until 1 week postpartum. Treatment stage is defined as the clinical stage after 6 months of ART represented by T1, T2, T3, and T4. 12 Immunosuppression classification was based on WHO immunological classification established for HIV infection. 20 Nutritional status was based on edema, weight-for-length z scores, or mid-upper arm circumference definitions set by the WHO. 21 Chronic malnutrition (stunting) was considered for patients’ WHO staging but not for assessment of acute malnutrition.
Comparison of nominal data between cohorts was done using Fisher exact test and matched pair outcome analysis was done with Stuart-Maxwell test for homogeneity using STATA 14.0 (College Station, TX). Statistical analysis comparing 2 groups with numerical data was done by 2-sample t test in Microsoft Excel 2013.
Ethical Approval
This study was approved by the Tanzanian National Institutes of Medical Research (NIMR), Mbeya Medical Ethics and Research Committee, Catholic University Health and Allied Sciences Research Committee in Mwanza, Tanzania, and the Institutional Review Board, Baylor College of Medicine, Houston, Texas.
Results
A total of 93 patients met the inclusion and exclusion criteria, 53.8% (50/93) were male. Median age at ART initiation was 13.4 months (range 2.8-23.9 months). See Table 1 for baseline demographics.
Baseline Demographic and Clinical Data for 93 Infants Initiated on ART at <24 Months of Age, at the Baylor Children’s Foundation—Clinical Centers of Excellence in Tanzania.

Abbreviations: ART, antiretroviral therapy; ZDV, zidovudine; d4T, stavudine; NVP, nevirapine; PMTCT, prevention of mother-to-child transmission; 3TC, lamivudine.
Data on baseline WHO clinical stage, WHO immunosuppression category, and nutrition status show that majority of patients were severely ill at ART initiation (Figure 1). Average CD4% at baseline was 19.6 (range 3-48).

Baseline and 12-month outcome characteristics of infants initiated on ART at <24 months of age, at the Baylor Children’s Foundation—Clinical Centers of Excellence in Tanzania. P < .001. P < .001. P < .001. Malnutrition classification based on WHO definitions of weight–height z scores and mid upper arm circumference. ART indicates antiretroviral therapy; WHO, World Health Organization.
Within 12 months of starting ART, there were 4 (4.3%) deaths, 5 (5.4%) patients transferred to other clinics, and 11 (11.8%) LTFU. No information is available on those patients transferred to other facilities or those LTFU, as at the time of study we had limited capabilities in tracking patients. The 73 patients who remained alive and in care at 12 months after initiating ART showed considerable improvement (Figure 1). Ninety percent of patients were in WHO treatment stage 1 at 12-month outcome (P < .001). Average CD4% at outcome was 32.7 (P < .001), which was an absolute rise of 13% from the baseline. Eighty-one percent (58/72) of patients had ≥5 rise in CD4%. Sixty-four percent (47/73) of patients had no significant WHO immunosuppression at outcome (P < .001). Nutritional status of our patients also improved greatly, 86.3% (63/73) were within normal limits and had no malnutrition (P < .001). Of those with malnutrition, most cases were mild, 8.2% (6/73), with only 1 patient having severe malnutrition.
Of the patients who had no infant and/or maternal PMTCT exposure, average CD4% at outcome was 33.6 (N = 30), with 86.5% (32/37) reaching normal nutritional status. Of the patients who did have exposure to infant and/or maternal PMTCT, average CD4% at outcome was 32.7 (N = 18, P = .74), with 86.4% (19/22, P = 1) reaching normal nutrition status. Average rise of CD4% for those with no infant and/or maternal PMTCT was 12.5 (N = 30), and average rise of CD4% of those with infant and/or maternal PMTCT was 16.9 (N = 18, P = .15). This suggests that in our 12-month follow-up time frame, PMTCT exposure did not statistically alter CD4% or nutritional outcomes (Table 2, Figure 2).
Outcome Characteristics at 12 Months Comparing Infant and/or Maternal PMTCT Exposure of Infants Started on ART at <24 Months of Age at the Baylor Children’s Foundation—Clinical Centers of Excellence in Tanzania
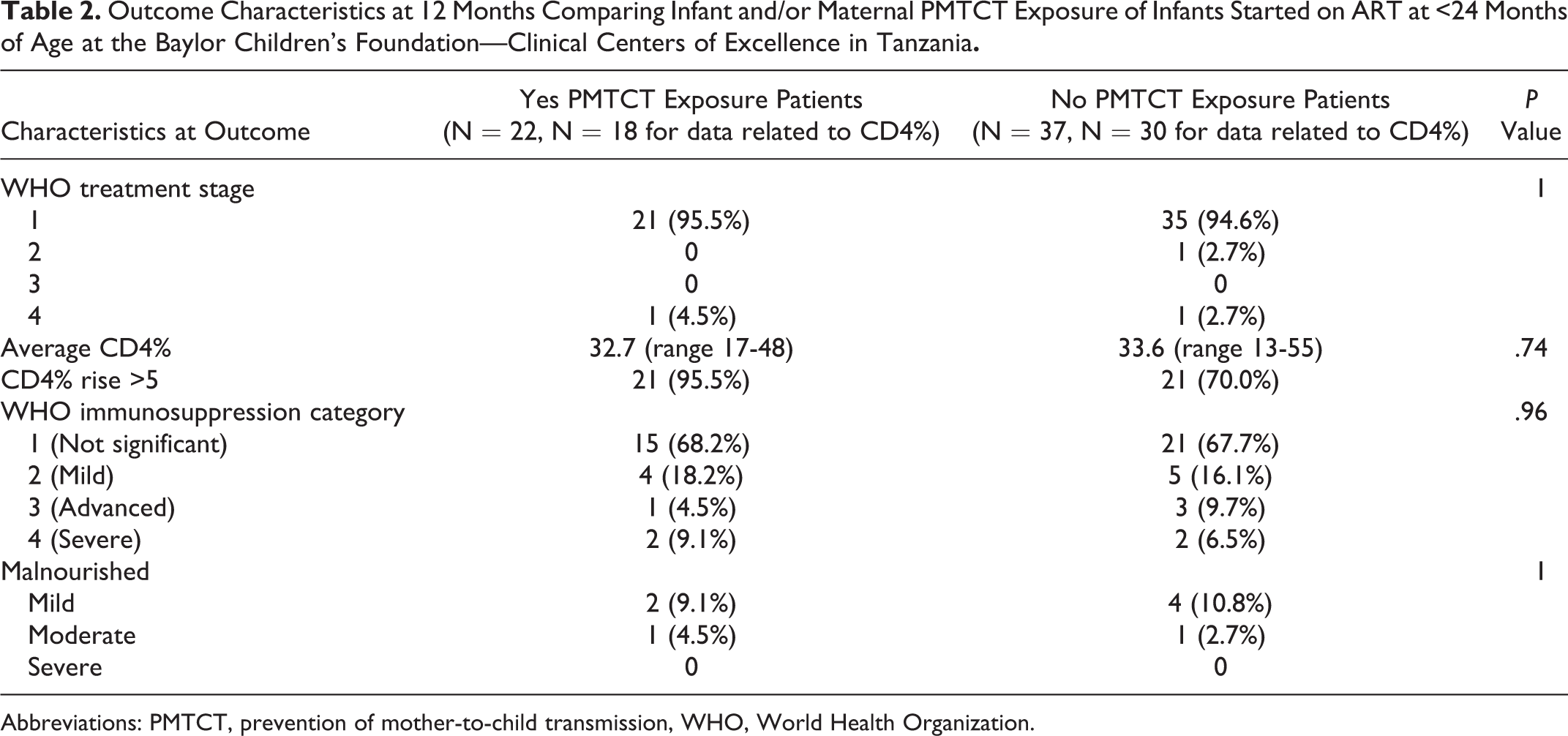
Abbreviations: PMTCT, prevention of mother-to-child transmission, WHO, World Health Organization.
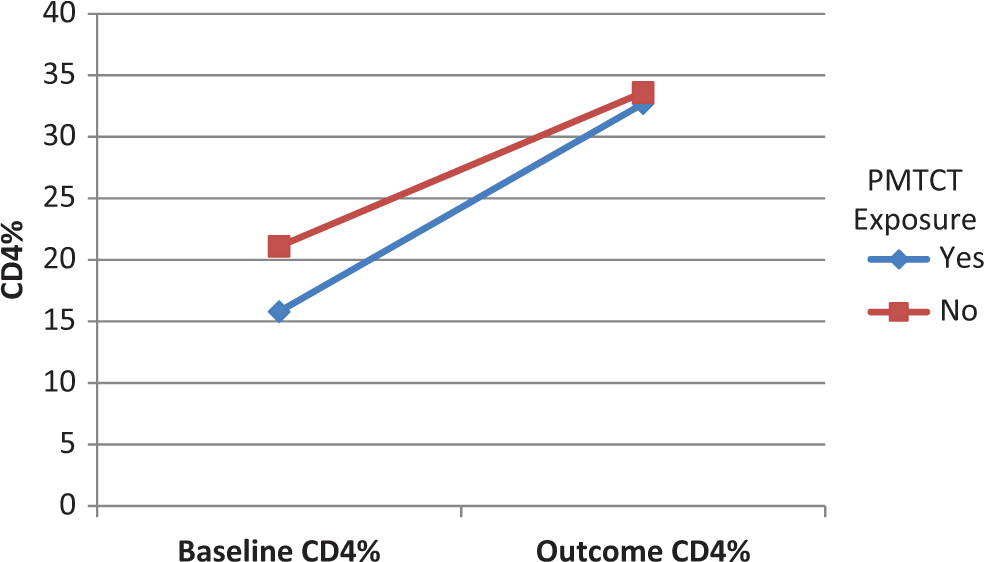
Average CD4% at baseline and 12-month outcome, comparing infant and/or maternal PMTCT exposure of infants started on ART at <24 months of age, at the Baylor Children’s Foundation—Clinical Centers of Excellence in Tanzania. Yes PMTCT exposure N = 18, no PMTCT exposure N = 30. Outcome CD4% P = .74, CD4% ▵ P = .15. PMTCT indicates prevention of mother-to-child transmission; ART, antiretroviral therapy.
Of the 4 patients who died (Table 3), average age of ART initiation was 12 months (range 2.8-21.9 months). Fifty percent (2/4) were initiated on stavudine(d4T)-3TC-NVP. Average hemoglobin at baseline was 10.4 g/dL. Average CD4% was 16.0 (range 9%-23%) which is lower than the baseline average of 19.3% for the rest of the patient population. All patients who died had severe WHO immunologic suppression at baseline. Fifty percent (2/4) of patients who died were exposed to maternal and/or infant PMTCT, suggesting that lack of PMTCT and late ART initiation did not lead to the patient deaths.
Baseline Characteristics of Patients Who Died from Cohort of Infants <24 Months of Age Started on ART at the Baylor Children’s Foundation—Clinical Centers of Excellence in Tanzania.

Abbreviations: ART, antiretroviral treatment; ZDV, zidovudine; d4T, stavudine; NVP, nevirapine; PMTCT, prevention of mother-to-child transmission; 3TC, lamivudine; WHO World Health Organization.
Eleven patients (11.8%) were LTFU (Table 4). Eighty-two percent (9/11) of LTFU patients were lost within 6 months of ART initiation, with majority, 72.7% (8/11), lost within 1 month of ART initiation. Of the patients LTFU, 63.6% (7/11) were WHO stage 4, 72.3% (8/11) were malnourished, and 45.5% (5/11) were severely malnourished at baseline, which is higher than patients who survived at 12-month outcome, though not statistically significant. The average age at ART initiation for LTFU patients was 18.0 months, which is higher than patients who survived, 12.6 months (P = .02). Forty-six percent (5/11) of LTFU patients had exposure to infant and/or maternal PMTCT.
Baseline Characteristics of Patients LTFU and Those Alive in Care from Cohort of Infants Started on ART at <24 Months of Age, at the Baylor Children’s Foundation—Clinical Centers of Excellence in Tanzania.
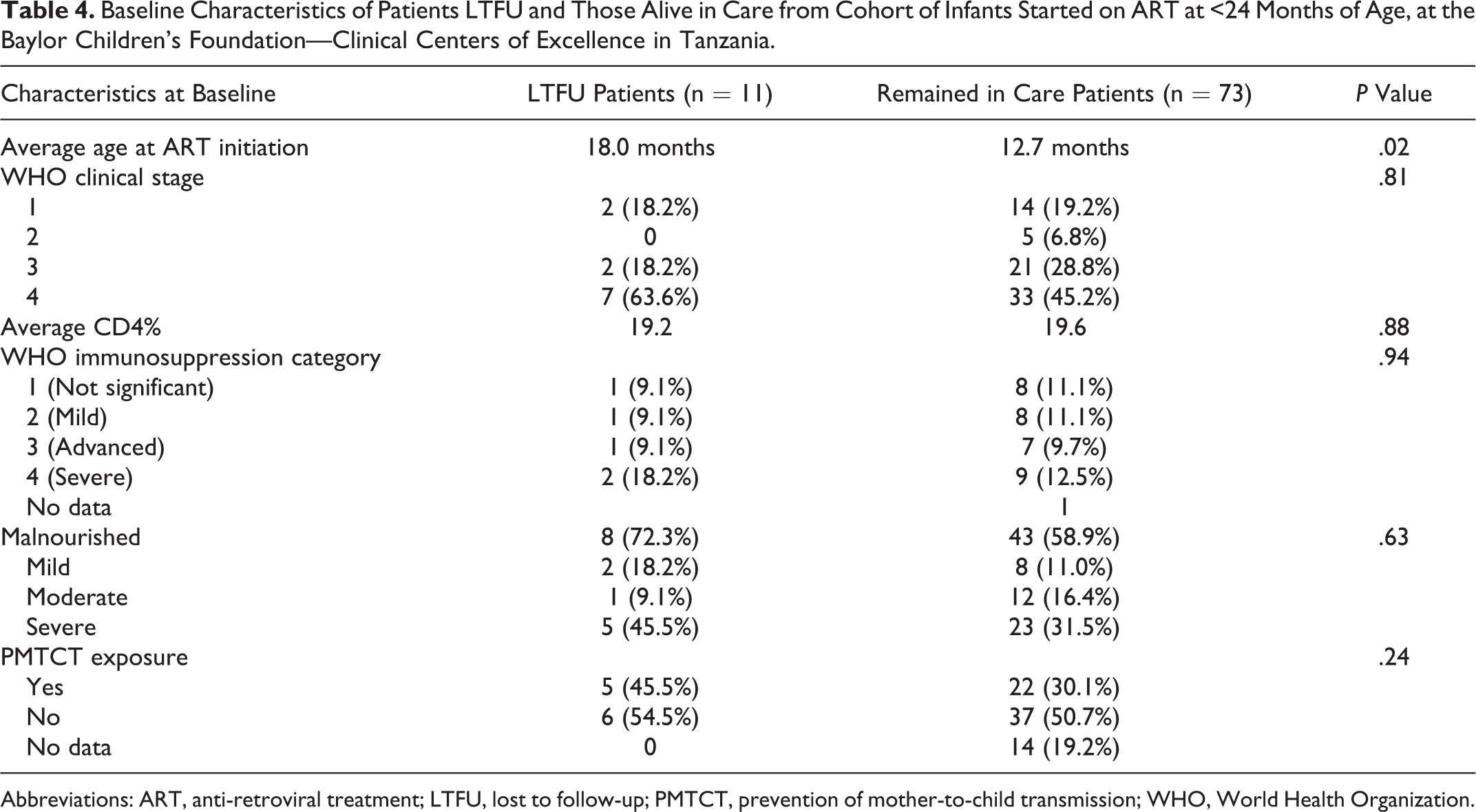
Abbreviations: ART, anti-retroviral treatment; LTFU, lost to follow-up; PMTCT, prevention of mother-to-child transmission; WHO, World Health Organization.
Discussion
In our cohort of HIV-infected children <24 months of age in Tanzania, good clinical outcomes were achieved with early initiation of ART even with severely ill patients at baseline. The majority of our patients presented extremely sick with WHO stage 4, WHO severe immunosuppression, and severe malnutrition, yet the overwhelming majority of patients were doing very well at 12 months of outcome. Similar findings, of good clinical outcomes despite very sick patients at baseline, have been reported in multiple African studies following children initiated on ART. 11,22 –30 More striking in our cohort is the low death rate of 4.3%, especially since multiple studies in resource-limited settings have found higher death rates among children <3 years when ART is initiated. 23,24,27,29 –34 This has been particularly noted from cohorts such as ours without access to virologic monitoring 23,24,31,33,34 and in countries with high incidence of tuberculosis. 29 –33 This has greater importance when accumulating evidence suggests that early ART initiation is particularly crucial for subsequent neurodevelopmental outcomes. 11,35
A recent multicenter study from southern African countries showed a 1-year mortality rate of 16% for children <24 months initiated on ART, which is considerably higher than our cohort. 29 Similarly high mortality statistics for young children initiated on ART ranging from 10% to 30% have been confirmed in other studies as well. 25,31,33 Although all these studies show a higher mortality rate than our cohort, the studies include data from clinics in rural parts of countries, whereas our 2 Tanzanian clinics are urban centers with access to referral hospitals and expert care.
In addition to benefits of an urban setting, the Baylor College of Medicine Children’s Foundation Tanzania COEs are funded by the USAID and have many resources that are unfortunately not found in any local government HIV clinics throughout southern Africa. Our Baylor COEs have highly trained clinicians specialized in pediatric HIV care, including nutritionists, counselors, and social workers. Our ancillary staff plays a crucial role in tracing LTFU patients and intervening with noncompliant patients. We have a lower patient to clinician ratio than many government clinics and, unlike many government clinics, have access to specialized therapies such as ready-to-use therapeutic foods for malnourished children. The COEs also draw on the support and experience from the BIPAI network of clinics in sub-Saharan Africa and Romania dedicated to the treatment of pediatric HIV. Unfortunately, most African countries lack these types of resources or investments. Good outcomes such as ours will be attainable in African countries via a global effort through governments and nongovernmental organizations investing in specialized pediatric HIV centers and increased outreach into rural areas.
A high rate of LTFU—which may confound the low mortality rate observed—was seen in our patient cohort, which has been noted in many African studies. 30,31,36 –38 Studies from adult populations in sub-Saharan Africa estimate the mortality rate of LTFU patients at 30% to 40%. 39 –41 Our LTFU rate was 11.8%, and we can therefore reason that many of our deaths may not be recorded because they became LTFU. However, if 40% of our LTFU patients were hypothetically considered dead, our corrected mortality rate would then equal 9.0% (9.4/93). This is more in line with other published studies but still very respectable.
Studies have shown LTFU is associated with young age, recent ART initiation, malnutrition, and advanced disease. 30,36 –38 Therefore, special attention should be given to young patients like our cohort especially due to their subsequent increased risk of morbidity and mortality. 31 –33,36,39 –41 Obstacles that contribute to high rates of LTFU at our clinics include frequent relocation of families due to employment, poor records of contact information, and no physical addresses of residences, making tracing of LTFU patients difficult. Similar findings were noted in a Kenyan study which interviewed mothers of children LTFU. 42 They found that socioeconomic factors were the root cause of majority of LTFU patients, including long distance to clinic, inability to pay for transport costs, lack of time due to work, preference for work in order to earn income for food, false belief in patient’s improving health, stigma from family and society, and being persuaded to try traditional treatments over modern medicine. A meta-analysis of LTFU patients with HIV also found that 83% of patients were missing or had incorrect phone number and physical address on file, making tracking of LTFU patients almost impossible. 39 At our clinics, we are currently undertaking several measures to reduce LTFU including updating contact information at every visit and attempting to contact all patients who miss their appointment either by phone or by home visit. We are also providing early education to caregivers about the implications of lifelong treatment, because our study shows the majority of LTFU occurs within 1 month of ART initiation. Future cohort data analysis will be needed to evaluate the efficacy of these approaches. More research needs to be done to develop innovative approaches to reduce LTFU in resource-limited settings, as many of our patients lack working mobile phones and most Tanzanian cities lack physical addresses making home visits difficult. Our LTFU patients were started on ART at a statistically older age than those who remained in care, suggesting that prior to becoming LTFU they were a vulnerable population who presented to care late. Novel approaches, such as mother–child linkages through option B+, will be needed to bring these vulnerable patients into care earlier.
Although our study lacked virologic testing, our patients did equally well clinically regardless of whether they had NNRTI exposure or not. This is important because although the P1060 trial indicated LPV/r-based regimens are superior to NVP-based regimens even in the absence of NNRTI exposure, 16 and the new WHO 2013 recommends LPV/r-based regimens as the first-line for children <3 years, 13 the expansion of LPV/r formulations presents a distinct challenge for resource-constrained settings in Africa. Some of the challenges to implementation of LPV/r include poor linkages between PMTCT, early infant diagnosis and infant ART services, as previously mentioned poor palatability, need for cold chain transport, LPV/r tablet formulation that cannot be crushed and its expensive nature. 5,17 Some countries such as Tanzania have recently reported difficulties in ensuring reliable distribution of liquid LPV/r formulation beyond a few specialized centers (Pediatric Technical Working Group, National AIDS Coordinating Programme, personal communication, 2011). Even though studies have shown early virologic failure among infants who started on NNRTI-based regimen, 16,43,44 our study exemplifies how clinical failure is not evident immediately. Therefore, we hope our cohort continues to do well as we wait for LPVr-based regimens to become more available in resource-limited settings. In settings such as ours, the results of the CHAPAS-2 evaluating a sprinkle formulation of LPV/r which could replace LPV/r syrup will be received with great interest and may allow countries that are currently unable to implement wider use of LPV/r to move ahead. 45
Although our cohort showed good outcomes using the 2010 WHO guidelines in a Tanzanian setting, further evaluation of infant ART cohorts is needed, particularly prospective studies to evaluate the effectiveness of continued NVP use because of lack of LPV/r, LTFU rates after implementation of new interventions, and overall outcomes after roll out of new WHO ART initiation guidelines, and option B+ in Tanzania.
Conclusion
Our cohort of HIV-infected children <24 months, initiated on ART at specialized pediatric HIV centers in Tanzania, did well clinically at the 12-month outcomes despite many of them being severely immunocompromised and severely malnourished at baseline. Low death rates may be attributed to urban settings of our clinics and to the holistic specialized care provided at our clinics, which unfortunately is not available in most African settings. LTFU rate was high for our cohort, as it is for many HIV pediatric centers in developing countries. Most LTFU occurred within 1 month of ART initiation and a number of mechanisms to counteract LTFU have been undertaken at our clinics. An area of interest for future studies is to evaluate novel methods of mother–child linkages and retaining enrollment in resource-limited settings such as ours, where many patients lack cell phones and homes with physical addresses. The lack of availability of LPV/r is a continuing problem for infants initiated on ART in resource-limited settings, however as we wait for increased availability, our data show that infants started on NVP-based regimens have good short-term clinical outcomes, even if they have exposure to NNRTI during PMTCT.
Footnotes
Authors’ Note
All data composed in Excel (Microsoft), and statistical analysis conducted in Excel and STATA (StataCorp) are available for sharing if needed.
Acknowledgments
The authors are grateful for USAID’s continued support of our endeavors; the prevention, treatment, and advancement of care for children affected by HIV in Tanzania. The authors would also like to thank Bridgette Naik for her help in proofreading and editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No specific research funding was used for this study. However, the operations of Baylor College of Medicine Children’s Foundation Tanzania are generously funded by the US Agency for International Development (USAID).
