Abstract
Background:
Most patients in Guatemala are diagnosed with advanced HIV disease. Patients that present late in the disease process may miss the morbidity and mortality benefits associated with early treatment and may unknowingly spread HIV to others.
Research Questions:
We examined trends in HIV detection and levels of immunosuppression at diagnosis from 2005 -2012 to determine if expanded HIV testing was associated with earlier detection of HIV.
Setting:
This study was conducted at the Clínica Familiar Luis Ángel García (CFLAG), a major HIV center associated with one of Guatemala’s two national hospitals. HIV testing expanded rapidly after 2007 due to grants from the Global Fund which allowed for routine prenatal testing.
Methods:
This study examined existing hospital and clinic databases from to evaluate results from HIV tests performed, and baseline CD4 cells/mm3 on all patients newly diagnosed with HIV infection from 2005 to 2012.
Results:
We found a decline in the number of HIV positive tests over the study period despite an increase in the total number of tests performed. Sixty-two percent of HIV infected individuals had AIDS at diagnosis. We observed a decrease in median CD4 cells/mm3 among the prenatal testees and no change in non-prenatal testees.
Discussion:
Expanded HIV counseling and testing services in our clinic did not result in earlier HIV diagnosis.
Introduction
According to estimates by the Guatemalan Ministry of Public Health and Social Assistance, HIV/AIDS is a growing public health issue in Guatemala. 1 With a population of approximately 14 million, the estimated prevalence of HIV hovers around 0.8%. 1 –3 The most recent data report 20 484 cases of AIDS since 1984, though estimates exceed 60 000. 2,4 Seventy-eight percent of reported cases were found in 7 of the 22 regions of Guatemala. Men who have sex with men (MSM), sex workers, prisoners, high-risk youth, and patients with tuberculosis are the populations principally infected with HIV. 2 HIV is transmitted primarily through sexual contact (94% of cases) and via mother to child (5% of cases). 1,2
Given that two-thirds of potential HIV infections remain undiagnosed in Guatemala, infected individuals may unknowingly facilitate the spread of HIV. The Clínica Familiar Luis Ángel García (CFLAG), a major HIV center in Guatemala City, attempted to address this issue by expanding HIV-related services in 2007. It adopted a universal screening program for prenatal patients and expanded testing and counseling services to the general population. We undertook this study to examine the trends in HIV detection and levels of immunosuppression at diagnosis and to determine if expanded testing was associated with earlier detection of HIV from January 2005 to September 2012.
Methods
Setting
This investigation was conducted at CFLAG, an HIV specialty clinic in Guatemala City, Guatemala. The clinic was founded in 1988 in affiliation with the Hospital General San Juan de Dios (HGSJDD), 1 of the 2 national hospitals in Guatemala. The CFLAG receives logistical and financial support from a nonprofit organization, the Asociación de Salud Integral (ASI). It also received a grant from the Global Fund in 2007 in order to expand HIV-related services in the inpatient and outpatient settings.
The CFLAG provides HIV testing and counseling, education, medical services, and treatment to adults and children living with HIV/AIDS. It also provides postexposure prophylaxis to hospital personnel. Services are rendered in the CFLAG outpatient clinic and San Juan de Dios Hospital. The CFLAG currently serves more than 3000 individuals living with HIV/AIDS and performs more than 7000 hospital consults annually. Patients receive services free of charge.
Study Design and Data Sources
We conducted a retrospective chart review to evaluate rates of HIV testing and newly diagnosed HIV cases from January 2005 through September 2012. Information was collected on adult patients (individuals over the age of 13) and pregnant women. HIV testing was offered in the labor and delivery service, outpatient obstetric clinic, and hospital wards. Prenatal patients were universally offered HIV testing. All other patients self-selected for HIV testing or were referred by a physician managing their care. Patient information and HIV test results were obtained from medical records and an HIV testing database. HIV test results, CD4 counts, pregnancy status, and basal CD4 counts were extracted for this analysis.
The testing service at CFLAG has been described in detail previously. 5,6 Patients were initially screened with a rapid test; positive results were confirmed by an enzyme-linked immunosorbent assay. If the initial 2 tests were discordant (1 positive and 1 negative), a third assay using Western blot was conducted. All confirmatory tests were conducted by individuals who are blinded to the results of the initial rapid test.
Data Analysis
The data were analyzed in 2 cohorts based on pregnancy status and testing modality. The prenatal patients (group 1) included antepartum or intrapartum pregnant women who were universally screened for HIV during the study period. The referred patients (group 2) included all other male and female HIV testees.
Median basal CD4 counts were collected and used as a proxy for levels of immunosuppression. Basal CD4 counts were considered the first documented CD4 value either prior to or within 6 months of initiation of antiretroviral therapy. Lower-median CD4 counts signified higher levels of immunosuppression at diagnosis among the respective populations. The World Health Organization (WHO) classification for HIV immunosuppression was used to stratify patients based on CD4 counts: greater than 350, 200 to 349, and less than 200.
We conducted 3 separate analyses. The first analysis looked at the number of HIV-positive and HIV-negative tests performed over the study period. The second analysis compared median basal CD4 counts in each year and in each population of testees (prenatal and referred). The third examined the proportion of individuals presenting with immunologic AIDS based on the baseline CD4 count.
Statistical Analysis
Medians with interquartile ranges (25-75) and frequencies were used to compare the changes in the basal CD4 count over time. We also used Mann-Whitney U test to examine the differences in the median CD4 counts, and the chi-square test was employed for comparison of categories (α = .05).
Ethical Approval
The study was approved by the research committee at the HGSJDD and the Committee on Clinical Investigation at Albert Einstein School of Medicine.
Results
Did HIV Detection Increase at CFLAG over the Study Period?
Between January 2005 and September 2012, a total of 90 577 and 36 972 tests were carried out in the prenatal and referred patients, respectively (Table 1). A 5-fold increase in HIV testing was observed in the prenatal group over this period, and a 1.6-fold increase was observed in the referred group. Despite the increased number of tests performed, there was a statistically significant downward trend in the absolute number of detected cases in both groups (P < .001).
HIV Tests and Positive Tests (2005-2012).a
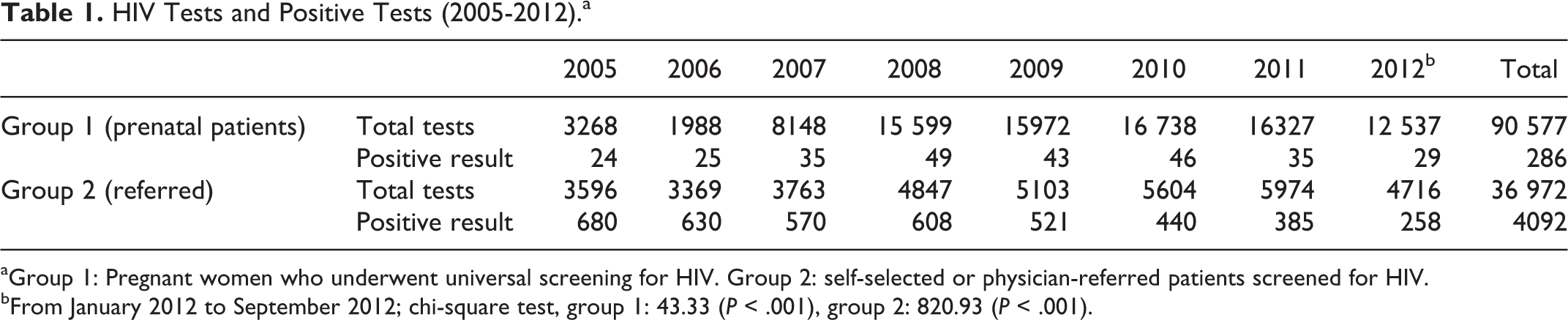
aGroup 1: Pregnant women who underwent universal screening for HIV. Group 2: self-selected or physician-referred patients screened for HIV.
bFrom January 2012 to September 2012; chi-square test, group 1: 43.33 (P < .001), group 2: 820.93 (P < .001).
Was There a Change in Median Levels of Immunosuppression at Diagnosis of HIV?
Median basal CD4 counts in the 2 study groups from 2005 to 2012 are shown in the Figure 1. CD4 data were available for 258 (90%) of the 286 prenatal patients, and 3054 (74%) of the 4092 referred patients. The median basal CD4 count for these groups was 267 and 124, respectively, a statistically significant difference. Despite initially higher median CD4 counts in the prenatal group, a pronounced downward trend in median CD4 counts was observed over the study period (P < .001).
What Proportion of Patients Initially Presented with AIDS over the Study Period?
We had baseline CD4 data available for 3312 patients; 2053 (62%) presented with CD4 counts less than 200, 640 (19.3%) patients presented with CD4 counts 200 to 350, and 619 (18.7%) presented with CD4 greater than 350. Of those patients with CD4 counts less than 200 (2053), 862 had CD4 counts less than 100 (26% of the total population) and 1191 had CD4 counts between 100 and 199 (36% of the total population). With respect to gender and pregnancy status, 1369 (66.7%) men, 592 (28.8%) women, and 92 (4.5%) prenatal patients presented with CD4 counts less than 200.
Discussion
The largest absolute number of HIV-positive cases was detected in 2005 compared to the pregnant patients referred in 2008 among. We also observed a general downward trend in HIV detection in both groups over the study period. This trend was observed in the prenatal group, despite a surge in HIV testing efforts after 2007 when universal screening was implemented (P < .001). The decline in HIV incidence at CFLAG parallels a worldwide decline in HIV incidence. Global trends in incidence have resulted from increased antiretroviral access, changes in sexual behavior practice, and greater public awareness of HIV transmission. 7
Pregnant women had lower levels of immunosuppression. They presented with much higher basal CD4 counts (264 cells/mm3) compared to the referred population (124 cells/mm3). Although the baseline CD4 counts in pregnant women appear higher, it is still remarkably low and represents moderate immunosuppression according to WHO classifications. Universal HIV screening during pregnancy, age and general good health may explain the differences in CD4 counts between the 2 groups. Universal screening may also consider pregnant women for testing before they would ordinarily seek it out themselves and confer protection against late diagnosis. A study carried out in Argentina from 1997 to 2006 also reported low levels of immunosuppression in the prenatal population. Median CD4 counts were 416 for pregnant women universally screened for HIV. 8 Similar findings were presented in this study, though we observed worsening immune status in this group over the study period. It is unclear what is driving this phenomenon.
The majority of patients (62%) had AIDS at diagnosis, and over a quarter of those with AIDS had very severe immunodeficiency. It is possible that undiagnosed HIV-infected individuals are not being captured in CFLAG’s catchment area of Guatemala City and may be missed for HIV screening. Studies have named indigenous and rural populations as underserved and underdiagnosed groups. 9 Fifty-one percent of Guatemalans live in rural areas and often have limited access to health services. 9 Far proximity to a testing center has been identified as a risk factor for late HIV diagnosis and likely represents a barrier to early diagnosis in this group. 4 Studies have also identified community and internalized stigma and low awareness of health services as risk factors for late presentation to care. 10 More studies are needed to elucidate barriers to early presentation in Guatemala.
The strengths and limitations to this study design/analysis are as follows: the large number of patients in a stable clinic setting was a major strength of the study. However, 10% of the CD4 data from the clinic population was not available. Additionally, some cases of new infections were referrals from small centers, though referral patterns have been stable over time.
In this study, expanded testing did not result in increased HIV diagnoses and failed to find people with earlier stage of infection. Thus, expanded testing did not appear to encourage at-risk asymptomatic individuals to get tested. This research was important in highlighting the need for ongoing efforts targeted at early identification of HIV-infected individuals in Guatemala and of motivators and barriers to testing. Enhanced efforts are also needed to reach the population who are cutoff from HIV testing facilities and to examine the experience of other HIV testing sites in Guatemala. A cost–benefit analysis may be helpful in ascertaining the feasibility of further expansion of programs or implementation of universal screening programs for all national hospitals.

Median basal CD4 counts 2005 to 2012.
Footnotes
Acknowledgment
The authors thank Dr Peter Selwyn for his support in this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was carried out with the financial support of the Department of Family and Social Medicine at Montefiore Hospital in Bronx, New York and the ASI.
