Abstract
Pityriasis rubra pilaris (PRP) is a poorly understood dermatologic condition usually accompanied by keratoderma and intense erythroderma with islands of unaffected skin. The PRP categories include HIV-associated PRP VI. A 23-year-old HIV-infected, dark-skinned woman in the Dominican Republic developed an extremely severe, disfiguring process characterized first by a dry scaly rash involving her face, trunk, and extremities with hyperpigmentation and islands of spared skin and minimal erythroderma, followed by alopecia and development of a thick horny layer on the scalp and face. The condition, histologically proven to be PRP, was accompanied by fever, wasting, and decline in CD4 count. Initiation of combination antiretroviral therapy (cART) was followed by rapid and sustained resolution of PRP. Nine years after ART initiation, she remains well, with viral suppression and immune recovery, without PRP recurrence but with sparse hair regrowth and facial scarring. In some dark-skinned patients, severe PRP may not feature characteristic erythroderma but will respond to combination ART.
Introduction
Pityriasis rubra pilaris (PRP) is a poorly understood chronic dermatologic disease characterized by palmoplantar keratoderma and eruption of follicular hyperkeratotic papules, which may coalesce to form salmon-colored plaques with adjacent areas of unaffected skin (islands of sparing). 1,2 The process is typically accompanied by intense erythema that can progress to erythroderma. Previously thought to be secondary to vitamin A deficiency and defective synthesis of retinol-binding proteins, it is at present understood to be of unclear etiology and generally acquired after birth, although Mendelian inheritance has been described in some cases. 3,4 Most often confused with psoriasis, PRP can be definitively diagnosed by correlation of the clinical presentation and characteristic histopathologic findings including alternating epidermal orthokeratosis and parakeratosis, psoriasiform acanthosis, and follicular plugging. 4
In 1980, Griffiths first described several categories of PRP based on age of onset, rate of progression, and characteristic pattern of the lesions. 5 These categories include the classic and atypical adult types and classic, circumscribed, and atypical juvenile types. A sixth category was recognized in the 1990s related to HIV infection. 6 Pityriasis rubra pilaris may represent an abnormal immune response to an antigenic trigger, as it has reportedly followed some acute infectious processes. 7 –9 Combination antiretroviral therapy (cART) reportedly favorably affects the clinical course of patients with PRP having HIV infection. 10 We describe in this report what, to our knowledge and that of consultants at HIV Online Provider Education who provided support in the management of this case, may be a particularly severe case of PRP. 11 In this Dominican HIV-infected woman, PRP followed varicella, coincided with clinical AIDS, and responded to cART.
Case Report
The patient was a 23-year-old woman diagnosed with HIV infection at the age of 20 during her first pregnancy. She received zidovudine (ZDV) during the last 2 months of pregnancy for prevention of vertical transmission, but the treatment was discontinued after the birth of the child in 2003. She was not considered a candidate for cART in 2004 because she remained asymptomatic, and her CD4 count was above 500 cells/mm3, well above the threshold for cART initiation in asymptomatic HIV-infected persons in the Dominican Republic at the time (less than 200 cells/mm3). She remained well until February 2006, when she developed varicella, with numerous lesions and high fevers. Weeks into her recovery, when most varicella lesions had cleared, she developed a severe scaling skin disease accompanied by severe crusting, darkening, and thickening of the skin, with particularly severe involvement of the face. She lost weight rapidly, becoming more and more debilitated and moved in with her mother approximately 6 weeks after the resolution of varicella. The mother requested a home visit from the HIV clinic 4 months after symptom onset.
On the day of the first home visit, she weighed 84 pounds (38.2 kg; height 160 cm, body mass index [BMI] = 14.9 kg/m2) and had a body temperature of 38.2°C. Her facial skin was severely crusted and thickened, with reduced mobility related to the severity of the thickening (Figure 1). The axillary and neck skin was thickened, cracked, emanating purulent material. She had genital and oral candidiasis, accompanied by ulcerating genital lesions, which she described as painful and present for over a month. Although there were islands of relatively spared skin, there were also multiple severely thickened, darkened crusted lesions from which blood, purulent discharge, and fragments of skin fell (Figure 2). Her physical examination revealed mild pallor, but cardiac and pulmonary auscultation and abdominal examination were otherwise normal. An unpleasant odor permeated the house, most intense near her torso. Her nails were thickened and dystrophic. She could not stand for more than a few minutes because of severe pain, numbness, and tingling of her feet, accompanied by reduced sensation of the lateral aspect of both feet, where the skin condition was relatively mild.
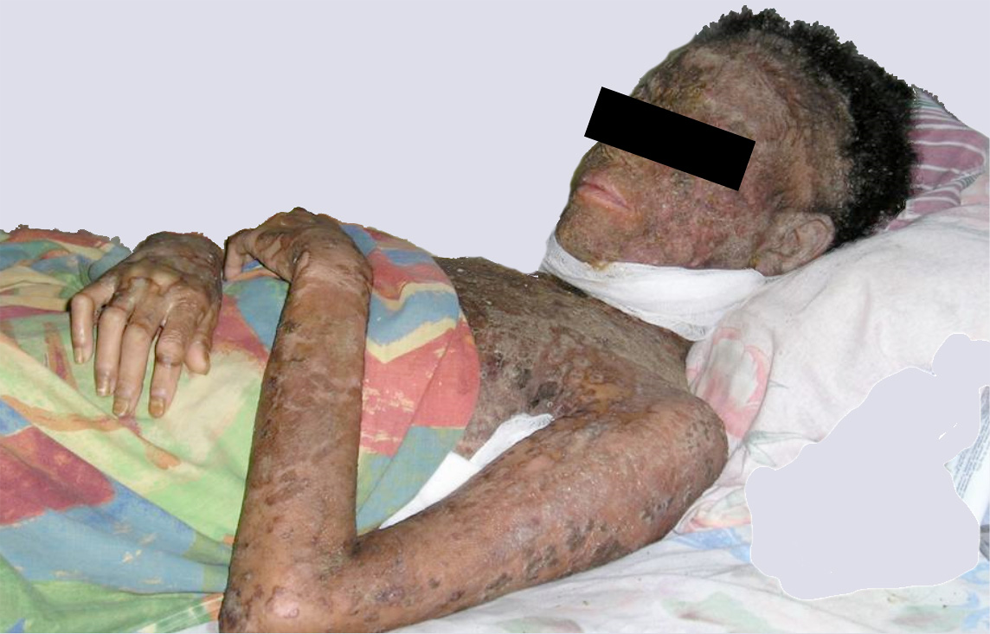
Crusting and discoloration of skin of face and arms, with lips, periorbital skin, and conjunctivae spared.

Areas of relatively spared skin and multiple severely thickened, darkened crusted lesions from where blood, purulent discharge, and fragments of skin have fallen.
Specimens for microscopic examination and culture for bacteria, mycobacteria, and fungi were collected. All showed no growth except for a heavy growth of Acinetobacter baumanii, a common gram-negative water contaminant, sensitive to aminoglycosides. No organisms that could explain this presentation were isolated. Specimens obtained for microscopic evaluation for Sarcoptes scabeii showed no evidence of scabies. Blood specimens collected during the visit showed a hemoglobin of 8.1 g/dL (hematocrit = 25.2%; white blood cell count 9.9 × 109/L). Platelet count was 334 000 × 109/L. Her CD4 count had declined from over 500 cells/mm3 prior to the varicella to 320 cells/mm3. A serologic test for antibody to HTLV-1 was nonreactive. Serum transaminases, bilirubin, urea, and creatinine were within normal limits. Presumptive treatment for scabies, fungus, and other causes of severe crusting diseases had no effect.
Over the next few weeks, the patient lost her hair, and the skin of her scalp and face developed a thick, horny layer (Figure 3). Horny protuberances were visible from each of which erupted 3 to 4 hairs.

Infiltration of facial skin and scalp with thick, hardened leathery tissue accompanying alopecia.
A plastic surgeon visiting the Dominican Republic volunteered to perform the biopsy in the patient’s home, and, with the patient’s consent, a full-thickness skin sample was taken from her upper arm. The specimen was sent to the Emory University School of Medicine Department of Pathology, Atlanta, Georgia. Combination antiretroviral therapy was started after the biopsy was obtained based on wasting and fever consistent with clinical AIDS, using stavudine (d4T) 30 mg (because of the anemia, ZDV was deferred) and lamivudine (3TC) 150 mg every 12 hours. Efavirenz (EFV) 600 mg was used at bedtime. Cotrimoxazole 3 times per week was initiated for Pneumocystis jirovecii pneumonia prophylaxis, and she was provided high-calorie nutritional supplements.
Results of the biopsy, received after cART initiation, showed histologic features consistent with the diagnosis of pityriasis rubra pilaris including psoriasiform acanthosis, hyperkeratosis, with alternating horizontal and vertical orthokeratosis and parakeratosis as well as follicular plugging. In the dermis, there was a mild lymphocytic inflammatory cell infiltrate. Special stains for acid–fast bacilli and fungal organisms (Grocott-Gomori methenamine silver stain and periodic acid–Schiff stain) were negative. There was only superficial bacterial colonization in the epidermis.
Three months after cART initiation, she left her mother’s home for the first time in over a year to visit the clinic. She weighed 50 kg (110 pounds), and her CD4 count had risen to 483 cells/mm3. Because her hemoglobin had risen to 11.5 g, the d4T was replaced by ZDV, and she continued the other antiretroviral medications. The lesions on her torso and extremities had receded almost completely. The facial and scalp lesions resolved, leaving severe facial scarring but with restoration of normal facial movement and slight erythema (Figure 4). Very sparse hair regrew.

Remission of facial lesions with recovery of facial motion, weight gain, and resolution of wasting.
Nine years after the PRP episode, she continues first-line cART (ZDV, 3TC, and EFV). She delivered a second HIV-uninfected, full-term child in 2009 and remains clinically well. Her BMI (27 kg/m2) is normal. Severe, noninflammatory, normally pigmented facial scarring remains on her face, although facial motion is now normal. She has minimal hair regrowth and wears a wig. She denies symptoms of neuropathy or any complaint other than almost complete alopecia and facial scarring. Her most recent quantitative HIV RNA and CD4 count were less than 40 copies/mL and 408 cells/mm3, respectively, in 2015.
Discussion
Response to treatment is variable in PRP unrelated to HIV. Most patients have reportedly had complete remission in 3 to 5 years, and partial and full responses to systemic retinoids, immunosuppressive therapy, and phototherapy have been reported. 12,13 Antitumor necrosis factor treatment has been shown to be associated with rapid resolution of redness and scaling in weeks. HIV-related PRP remains rare and may be underrecognized in high HIV prevalence, low- and middle-income countries. As was the case in our patient, a dark complexion may attenuate PRP’s characteristic redness. 14 It should be noted that cART, to which our patient responded within weeks, was started on the basis of evidence of clinical AIDS, with wasting, fever, and presumably herpetic lesions of prolonged duration and on the basis of a CD4 count less than 350 cells/mm3 which was an indication for cART initiation in industrialized countries in 2006 and in low- and middle-income countries in 2010. 15 The nature of her dermatologic disease, its relatedness to HIV, and the potential for response to cART were only recognized later, when the biopsy results were provided by one of us (D.P.). Accompanying lesions, notably the hidradenitis suppurativa 16 previously described in HIV-related PRP, responded promptly as well.
Dermatologic lesions, often severely disfiguring, frequently accompany HIV infection. 14 Their disfiguring quality may add to the stigma of an HIV diagnosis and may compel patients to avoid public appearances, including clinic visits, delaying initiation of cART, as in this case. Combination ART can reverse these processes without specific treatment, but a specific diagnosis may be important to patients and their caregivers who fear that some of these conditions may be contagious, further isolating them. This case underscores the need for home- and community-based care, cART, and accompaniment for HIV-infected homebound patients in low- and middle-income countries. It also illustrates the value of international distance collaboration in cases with challenging diagnostic features in low- and middle-income countries.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
