Abstract
Objectives:
HIV-infected patients have higher mortality when coinfected with hepatitis B virus (HBV). With potent highly active antiretroviral therapy (HAART) and the use of tenofovir (TDF), outcomes may improve. Our objective was to determine the clinical and virological outcomes of a HIV/HBV-Coinfected cohort at our center since TDF became available.
Methods:
We retrospectively studied all HIV/HBV-Coinfected adults followed between 2002 and 2012 for ≥3 months. Outcome measurements included HBV DNA suppression, HBV e-antigen (HBeAg) and HBV surface antigen (HBsAg) clearance, cirrhosis diagnosis, development of liver complications, and overall and liver-related mortality. Predicting factors were assessed with log-rank test and logistic regression.
Results:
Median time to follow-up of the 99 patients included was 5 years. Undetectable HBV DNA and HBsAg loss were achieved by 65% and 18%, respectively. Overall and liver-related mortality rates were 4.58 and 0.91 per 100 person-years, respectively. Most patients died of causes unrelated to the liver. Four patients died from hepatocellular carcinoma (HCC) and one, hepatitis C virus (HCV) coinfected, from liver failure. Higher CD4 counts at last follow-up were associated with HBV suppression (odds ratio [OR] 1.004, 95% confidence interval [CI] 1.001-1.006, P = .007), HBeAg loss (OR 1.003, 95% CI 1-1.005, P = .02), HBsAg loss (CD4 count >700 cells/mm3, OR 3.80, 95% CI 1.06-13.58, P = .04), and survival (OR .994, 95% CI 0.990-0.997, P < .0001). HCV coinfection was associated with higher overall mortality (OR 7.74, 95% CI 1.47-40.81, P = .02).
Conclusion:
Mortality was high and most often unrelated to liver disease in this HIV/HBV-Coinfected cohort treated predominantly with TDF-containing HAART. Optimal CD4 counts predicted survival and the achievement of HBV virological end points. Tenofovir prevented liver decompensation but not HCC, which was the predominant cause of liver death.
Introduction
Soon after the AIDS epidemics began, it became apparent that HIV-infected patients coinfected with hepatitis B virus (HBV) had a higher mortality risk. 1 A negative effect of HIV on HBV-related liver disease was also identified. Cirrhosis was more common, and mortality was higher in HIV/HBV-Coinfected patients compared to HBV-monoinfected patients. 1,2 Lamivudine (3TC), which was the first antiretroviral drug with activity also against HBV, provided a mild reduction in liver-related mortality in HIV/HBV-Coinfected patients, although it still remained high. 3,4
With the widespread use of tenofovir (TDF) as part of HIV treatment, a new era began in the management of HBV, as it proved to be a more potent HBV inhibitor. 5 Tenofovir provides much more effective suppression of HBV replication without selecting resistance mutations. 6 –13 However, complete suppression of HBV may take a long time or even never be achieved in patients with high levels of replication. The duration and degree of HBV suppression needed to prevent liver disease progression and liver complications in HIV-HBV coinfection have not been well established. Short-term data from Africa suggest a positive effect of TDF on mortality in HIV/HBV-Coinfected patients. 14 However, there are limited data in other settings. The objective of the present study was to determine the clinical and virological outcomes of a HIV/HBV-Coinfected cohort at a tertiary center since TDF became available. The ultimate goal is to identify areas for potential intervention to increase survival in this population.
Methods
We performed a retrospective and longitudinal cohort study based on a chart review of adult patients with HIV and HBV coinfection seen in the HIV Clinic at Wake Forest University Baptist Medical Center (WFUBMC) between 2002 and 2012. Data from this cohort have been previously published, with information obtained up to 2008. 10 To be included in the study, patients had to have chronic HBV infection defined as HBV surface antigen (HBsAg) or detectable HBV DNA for a span of at least 6 months, HIV infection defined as positive HIV antibody confirmed by Western blot, and a minimum follow-up of 3 months.
Data collected included (1) demographics: age, sex, ethnicity, and primary HIV risk factor; (2) laboratory data: serum HBV DNA, HBV e-antigen (HBeAg), serum HIV RNA, alanine aminotransferase (ALT) levels, and CD4 counts at baseline and during follow-up, hepatitis C virus (HCV) antibody, with HCV RNA if antibody was positive; (3) clinical data: time to follow-up, HBV treatment history, comorbidities, self-reported alcohol use (none/ moderate [<50 g/d]/severe [>50 g/d]), adherence to highly active antiretroviral therapy (HAART) as recorded in the medical record and based on self-report and refill information obtained from the pharmacy (suboptimal defined as <90% of doses), compliance with follow-up (“poor” defined as <2 visits/year), imaging liver studies (liver ultrasound, computed tomography scan, or magnetic resonance imaging), history of liver decompensation (defined as ascites, encephalopathy, jaundice, or gastrointestinal bleeding), diagnosis of cirrhosis and of hepatocellular carcinoma (HCC), and death occurrence with its cause. The cause of death was obtained from reports in the chart, with verification of each case by reviewing the medical records). The diagnosis of cirrhosis was made through clinical findings, which include liver image studies and presence of thrombocytopenia, portal hypertension, poor liver synthetic function, or ascites.
Time to follow-up was counted since 2002 or since the first visit to our center if it occurred after 2002. Hepatitis B virus baseline data refer to the time when patients initiated HBV-active antiretroviral treatment. HIV RNA, CD4 counts, and ALT levels were obtained at approximately 3 months’ intervals coinciding with clinic visits. However, HBV data were not obtained at predetermined time points but as dictated by HIV care providers. CD4 counts were measured by flow cytometry. HIV RNA levels were measured by polymerase chain reaction (PCR; lower limit of detection 400 copies/mL until June 2000 and 50 copies/mL thereafter), and HBV DNA levels were measured by PCR (measured in pg/mL until 2004, lower limit of detection <0.01 pg/mL, 1 pg = approximately 150 000 copies; and in IU/mL since 2005, lower limit of detection <100 IU/mL until 2011 and <20 IU/mL currently). Serum HCV RNA was measured by quantitative PCR.
Statistical Analyses
Outcome measurements included DNA suppression, HBeAg and HBsAg clearance, diagnosis of cirrhosis, development of liver complications, and overall and liver-related mortality. Continuous variables are expressed as median (interquartile range [IQR]) and categorical variables as absolute numbers (percentage). Chi-square and t tests were used to compare categorical and continuous variables among groups, respectively, except when the distribution of the variables was not normal, in which case we used nonparametric tests. Logistic regression was used to identify factors associated with the outcomes. Variables analyzed for association with virological outcomes include age, sex, baseline parameters (HBV DNA levels, HBeAg status, ALT levels, and CD4 counts), time to follow-up, HIV suppression on HAART (well-controlled HIV was defined as undetectable HIV RNA levels for >80% time to follow-up), CD4 counts at last visit, and changes during follow-up, duration of TDF treatment, self-reported adherence to therapy, and HCV coinfection. Besides the above-mentioned measurements, variables analyzed for association with clinical outcomes include prior use of 3TC without TDF, achievement of undetectable HBV DNA during follow-up, HBeAg or HBsAg loss during follow-up, ALT levels during follow-up, and history of alcohol use. Survival estimates are expressed as cumulative proportion of surviving at the specific time point and as incidence rate. For the survival analysis, data were censored at the date of the respective end point, when the last information about survival and clinical status was available. The bivariate survival analysis was performed using the method of Kaplan-Meier. Curves were compared by the log-rank test. Differences were considered to be significant with P values <.05. Data were analyzed using IBM SPSS Statistics version 21.0 software package (IBM, Armonk, New York, USA).
Results
Ninety-nine HIV/HBV-Coinfected patients met the specified criteria and were included in the study. Of them, 52 had positive HBeAg, 17 had negative HBeAg, and 30 had an unknown HBeAg status prior to initiation of HBV-active HAART. The characteristics of the patients are summarized in Table 1. Baseline HBV DNA was not available for 38 (38%) patients. Table 2 records the clinical and virological outcomes of the patients. They were followed up for a median time of 5 years and received HBV-active HAART for a median duration of 3 years. Ninety patients were treated with a TDF-based therapy. Of the 9 patients who did not receive TDF, 3 received 3TC as part of their HAART (2 of them had chronic kidney disease) and 6 had not initiated HAART at the last follow-up.
Baseline Patients’ Characteristics.a ,b

Abbreviations: ALT, alanine aminotransferase; IV, intravenous; HBV, hepatitis B virus; HBeAg, HBV e-antigen.
a n = 99.
bResults expressed as median (interquartile range) unless otherwise stated.
cCopies/mL were reported because IUs were not universally available prior to 2005.
Clinical and Virological Outcomes.a,b
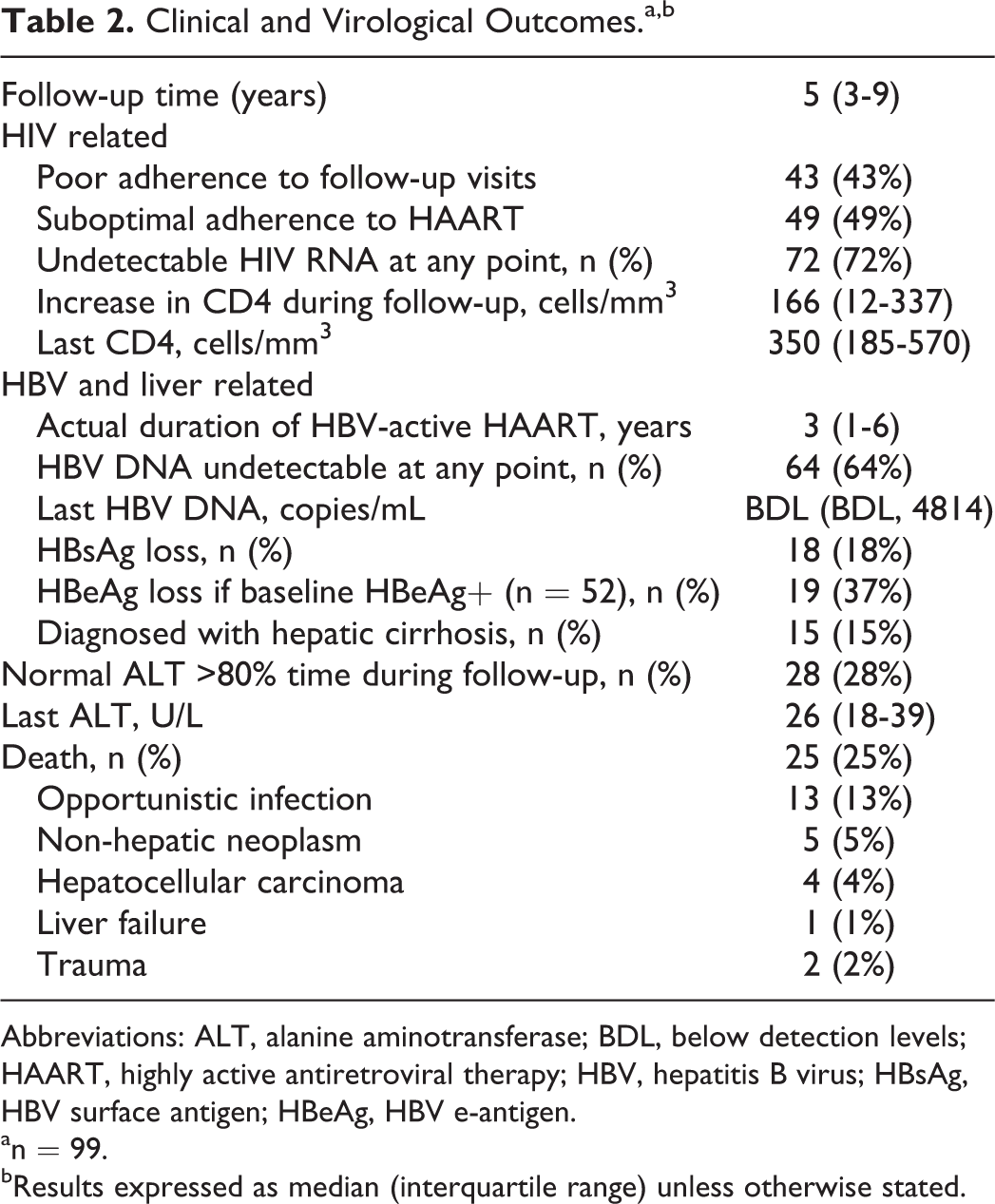
Abbreviations: ALT, alanine aminotransferase; BDL, below detection levels; HAART, highly active antiretroviral therapy; HBV, hepatitis B virus; HBsAg, HBV surface antigen; HBeAg, HBV e-antigen.
an = 99.
bResults expressed as median (interquartile range) unless otherwise stated.
Virological Outcome
HBV DNA Suppression
All patients
Undetectable HBV DNA was achieved at least once during follow-up in 65% of patients. As depicted in Figure 1, CD4 count ≥400 cells/mm3 at last follow-up (Figure 1A; log-rank test, P = .001), negative HBeAg at baseline (Figure 1B; log-rank test, P < .0001), and self-reported optimal adherence to HAART (Figure 1C; log-rank test, P = .02) favored the achievement of HBV suppression. Other factors associated with undetectable HBV DNA were age >45 (odds ratio [OR] 2.89, 95% confidence interval [CI] 1.25-6.68, P = .01), well-controlled HIV (OR 3.33, 95% CI 1.28-8.70, P = .01), higher nadir CD4 counts (OR 1.004, 95% CI 1.001-1.007, P = .009), and greater CD4 gains on HAART (OR 1.004; 95% CI 1.001-1.007; P = .003). In the final multivariate analysis model, only 70 patients were included as HBeAg status was unknown in the remaining, baseline HBeAg status tended to prevent HBV DNA suppression (OR 0.20, 95% CI 0.04-1.03, P = .05), while higher CD4 counts at last follow-up were associated with undetectable HBV DNA (OR 1.004, 95% CI 1.001-1.006, P = .007). Of note, while patients with well-controlled HIV replication during follow-up were more likely to achieve undetectable HBV DNA (OR 6.87, 95% CI 2.53-18.71, P < .0001), it was not a guarantee of HBV suppression. Thus, of 37 patients who had undetectable HIV for at least 3 years, 8 (22%) never achieved HBV DNA suppression. All of them had positive HBeAg or high HBV DNA levels at baseline.

A, Patients achieving undetectable hepatitis B virus (HBV) DNA according to CD4 count at last follow-up. B, Patients achieving undetectable HBV DNA according to the baseline HBV e-antigen (HbeAg) status. C, Patients achieving undetectable HBV DNA according to the self-reported adherence to highly active antiretroviral therapy (HAART).
HBeAg-positive patients
Of the 52 HIV/HBV-Coinfected patients with known positive HBeAg at baseline, 51 received TDF with or without emtricitabine and one 3TC. Thirty-one (61%) achieved undetectable HBV titers at least once during follow-up after TDF-based treatment was initiated. In univariate analysis, higher CD4 counts as measured by absolute count at last follow-up and by cell increases increase during HAART, well-controlled HIV, and self-reported optimal adherence to HAART were associated with the achievement of undetectable HBVDNA. In multivariate analysis, only self-reported optimal adherence to HAART (OR 4.3, 95% CI 1.01-18.22, P = .04) predicted HBV DNA suppression.
HBeAg clearance
HBeAg loss occurred in 19 (36.5%) of 52 HIV/HBV-Coinfected patients with known positive HBeAg at baseline. In logistic regression analysis, higher CD4 counts at last follow-up (OR 1.003, 95% CI 1-1.005, P = .02) and CD4 gains on HAART (OR 1.004, 95% CI 1.001-1.007, P = .02) were associated with HBeAg clearance.
HBsAg clearance
Eighteen (18%) patients lost the HBsAg during follow-up. However, 3 of them had reverse HBsAg seroconversion thereafter. HBsAg loss was comparable between patients with positive and negative HBeAg at baseline. Having CD4 count >700 cells/mm3 at last visit was associated with negative HBsAg (OR 4.15, 95% CI 1.15-14.98, P = .03).
Liver-Related Morbidity
During follow-up, 15 (15%) patients were diagnosed with cirrhosis. Two of them were also HCV coinfected, and they died. Univariate and multivariate analyses identified ALT elevation during follow-up (having ALT level greater than 2 times the upper normal limit [>60 U/L for men and >38 U/L for women] for at least 4 years) as only factor associated with a clinical diagnosis of cirrhosis (OR 6.4, 95% CI 1.73-23.47, P = .005), while HBsAg loss was the only protective factor (OR 4.81, 95% CI 1.28-18.002, P = 0.02).
Liver-related complications in patients diagnosed with cirrhosis occurred in 6 patients. One patient with a long history of noncompliance had recurrent variceal hemorrhage and is currently alive. Another patient, HCV coinfected, developed liver failure and died. The remaining 4 patients were diagnosed with HCC and died.
Mortality
Overall mortality
A total of 25 (25%) patients died during follow-up. The mortality rate was 4.58 deaths per 100 patient-years. Figure 2A and B depicts all-cause and liver-related mortality curves, respectively. Most deaths occurred within the first 5 years of follow-up. The survival was 75% at 5 years and 61% at 10 years. The causes of death are summarized in Table 2. Most patients died of causes unrelated to the liver, mostly due to opportunistic infections followed by malignancies (esophagus, rectosigmoid, head/face/neck, pancreas, and squamous carcinoma of undetermined origin, one case each). Of the 15 patients diagnosed with cirrhosis, 7 died during follow-up, but in 2 cases it was not due to liver causes.

A, All-cause mortality. B, Liver-related mortality.
Hepatitis C virus coinfection was associated with significantly worse survival (P = .002; Figure 3A). Thus, 7 of the 11 patients with hepatitis C coinfection died during follow-up, 3 from HIV-related causes, 2 from nonhepatic neoplasms, and 2 from liver-related causes (1 from HCC and 1 from liver failure). Besides HCV coinfection, univariate analysis also identified lower CD4 counts measured at baseline and last follow-up, smaller CD4 gains on HAART, higher HIV RNA levels at baseline, and lack of HBV DNA suppression during follow-up as factors associated with overall mortality. Having a CD4 count >400 cells/mm3 at last follow-up (P < .001) predicted survival (Figure 3B). In multivariate analysis, HCV coinfection was the only predictor of mortality (OR 7.74, 95% CI 1.47-40.81, P = .02), and higher CD4 counts at the last visit was the only predictor of survival (OR .994, 95% CI 0.990-0.997, P < .0001.

A, Overall mortality according to hepatitis C virus (HCV)-coinfection status. B, Overall mortality according to CD4 count at last follow-up.
Liver-related mortality
Survival curve from liver-related causes is depicted in Figure 2B. Five (5%) patients died from liver-related causes during follow-up. The liver-related mortality rate was 0.91 per 100 patient-years. The most frequent cause of liver-related death was HCC, which occurred in 4 cases, while only 1 patient died from liver failure. Two patients were also infected with HCV: 1 died from HCC and the other from liver failure. The 4 patients who died from HCC were men and had HCC diagnosed at ages ranging from 50 to 58. Two of them were diagnosed with HCC in 2007, 1 in 2008, and 1 in 2011. CD4 counts ranged between 240 and 720 cells/mm3. All of them had received prolonged treatment with TDF (3-6 years) and had achieved undetectable HBV DNA during follow-up (for 1-3 years). One had negative HBeAg at baseline and 3 positive but 1 lost HBeAg during follow-up. However, transaminases remained elevated throughout follow-up in all cases. All patients had a history of alcohol use.
Discussion
In this cohort of HIV/HBV-Coinfected patients with advanced HIV from a single institution, predominantly treated with TDF-containing HAART and with one of the longest follow-up published to date, the overall mortality remained high and was most often attributable to immunosuppression. This suggests that, as for any HIV-infected patient, avoiding CD4 lymphopenia through HIV control is paramount to improve survival in this population. In our study, maintaining the optimal CD4 counts did also help to achieve HBV virological end points. In addition, we showed that HBV suppression requires a higher level of adherence to treatment compared to HIV, especially with very high baseline HBV DNA levels. However, TDF appears to confer some benefit over liver disease even with more modest HBV responses, as liver decompensation was rare despite more than one-third of patients never achieving undetectable HBV DNA. On the contrary, HCC was not fully prevented as it was the predominant cause of liver-related mortality and it occurred despite having achieved HBV suppression. Of note, HCV and/or alcohol history were present in all liver-related deaths. This suggests that besides treating HBV, other factors contributing to liver-related mortality in this population need to be addressed.
Consistent with other reports, HBV suppression was achieved more often and earlier in HBeAg-negative patients compared to those with positive antigen. 12,13,15,16 It was among those HbeAg-positive patients that optimal compliance with treatment, but not HIV suppression, was identified as factor predicting HBV DNA suppression, in support of a prior report. 16 This suggests that HBV is less forgiving than HIV, requiring higher levels of adherence to treatment. Most importantly, our data suggest an important role for immune restoration in the achievement of all virological end points. The importance of optimal CD4 counts to attain undetectable HBV DNA and to achieve greater declines in quantitative serum HBsAg has been previously reported. 6,16,17 We also showed that higher CD4 counts increase the likelihood of eliminating the HBeAg and HBsAg. Significant CD4 gains with HAART in a population with advanced HIV disease at the time of HAART initiation might explain the high incidence of HBsAg loss in our cohort compared to prior reports in HIV/HBV-Coinfected and HBV-monoinfected patients. 11,12,16 –22
The proportion of patients diagnosed with cirrhosis during follow-up was within the range of prior reports. 2,8,11,12,18,23,24 The incidence of cirrhosis was likely underestimated, given the retrospective mature of our study and inconsistent ultrasound surveillance. Supporting results from a prior study, elevated ALT was associated with a diagnosis of cirrhosis during follow-up. 23 HBsAg loss was identified as factor protecting from being diagnosed with cirrhosis in our study. Liver decompensation was rare in our cohort, which is in line with the results from another long-term study on TDF use among HIV/HBV-Coinfected patients. 12 Interestingly, less patients achieved undetectable HBV DNA in our cohort compared to the aforementioned study, suggesting that TDF may provide histological benefit even without complete HBV suppression.
The overall mortality rate in this study is among the highest reported in HIV/HBV-Coinfected patients. 1,23,25 –27 Deaths were most often due to nonhepatic causes, primarily due to opportunistic infections, and CD4 counts at follow-up were the strongest predictor of mortality despite a majority of patients being on HAART. A predominance of AIDS-related mortality among HBV-coinfected patients has been reported by 2 multicenter cohorts, from Europe and United States. 25,26 This is despite the consistent finding in multiple studies that HBV coinfection does not compromise the virological and immunological response to HAART. 25 –29 The high AIDS-related death rate in our study could be explained by the advanced HIV disease and the often poor adherence to HAART and to follow-up. However, a possible effect of HBV on HIV disease cannot be completely ruled out. Our findings suggest that emphasis should be placed on the importance of HAART and of preserving or achieving optimal CD4 counts to prevent opportunistic infections and to improve the virological response to HBV treatment.
The other factor that greatly affected mortality in our study was HCV coinfection. Nevertheless, HCV-coinfected patients most often died from nonhepatic causes. Confounding factors, such as smoking, illicit drug use, and psychiatric disease, often present in patients with HCV infection might have contributed to the high mortality in this subset of patients. Therefore, these patients with triple infection constitute the group at highest risk of death and require multidisciplinary care.
Information on mortality attributed to liver disease among HIV/HBV-Coinfected patients is scarce. Limited data suggest that liver-related mortality rates were higher during the early HAART era, with less frequent or shorter exposure to TDF (1.4-2.2 per 100 patient-years). 1,24,26 Two other studies which reported lower liver-related mortality rate (0.7 per 100 patient-years) were not comparable, given the younger patient’s age, which averaged mid-30s, when liver complications are still infrequent. 23,25 Liver-related mortality in our cohort was low and comparable to a recent report with similar time to follow-up. 12 All in all, these data suggest that current HAART regimens relying on TDF are more effective than early HAART combinations in preventing liver complications as previous reports from underdeveloped countries had indicated. 14,30
Combining HCC and nonhepatic cancers, 36% of all deaths (9 of 25) were due to non-AIDS-related cancers. This supports prior reports of increasing the proportion of lethal non-AIDS-related cancers in an aging HIV population. 31 While TDF appears to have prevented liver decompensation to a great extent in our population, it did not fully protect from the development of HCC, which was the predominant cause of liver-related deaths in our cohort. There were no survivors among patients diagnosed with HCC, supporting prior data which suggest worse outcome in HIV-infected patients with HCC compared to their HIV-negative counterparts. 32 This is likely due to a number of reasons including too advanced disease at presentation, curative therapy offered less frequently, and no widespread use of liver transplantation. 33 –35 The risk of HCC in patients with hepatitis B correlates with HBV DNA levels, and anti-HBV therapy appears to decrease the incidence of HCC in HBV monoinfection, although HCC still occurs in some patients with virologic suppression. 36 –39 Early treatment and more prolonged HBV suppression may be needed to prevent HCC development. As HCC occurs more frequently in patients with cirrhosis and this can be prevented and partially reversed by prolonged antiviral therapy, with potent first-line HBV therapy, a major contributory factor for the development of HCC can be acted upon. 22,39,40 –42
Limitations of our study include incomplete data, given the retrospective design and the frequent nonadherence to follow-up of our patients. While the demographics of our cohort are very similar to HIV/HBV-Coinfected individuals of US metropolitan areas, 43 it differs from the world’s HIV/HBV-Coinfected population, and therefore our results may not be applicable to other settings.
In summary, mortality was high and most often unrelated to liver disease in this HIV/HBV-Coinfected cohort with advanced HIV disease treated predominantly with TDF-containing HAART. Optimal CD4 counts predicted not only survival but also the achievement of HBV virological end points. Liver-related mortality rate was low, confirming the positive effect of TDF on liver disease. Nevertheless, TDF use prevented liver decompensation but not HCC, which was the predominant cause of liver-related death in this cohort.
Footnotes
Authors’ Note
The authors contributed to the study’s design, literature search, statistical analysis, writing of the manuscript, and approved the final draft.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been possible because of the 2012 Medical student summer Research training Program, which was supported through grants from the National Institutes of Health, Wake Forest University School of Medicine and private gifts.
