Abstract
Hemophagocytic lymphohistiocytosis (HLH) is traditionally regarded as a rapidly progressive and often fatal illness. In patients with AIDS, HLH usually occurs secondary to opportunistic infections. Although popular guidelines exist for the diagnosis and management of HLH in general, no formal study has evaluated their applicability among adult patients who develop HLH in the setting of AIDS and opportunistic infections. The study reports on a case of HLH in a patient with AIDS and disseminated histoplasmosis. Eighteen other previously reported cases of HLH in the setting of AIDS and histoplasmosis were reviewed. Majority of the cases occurred in patients with a CD4 count of less than 70 cells/mm3. Overall mortality was 44%. Not getting antifungal treatment and having Histoplasma in blood were the 2 main risk factors for death. Among the patients who had a timely diagnosis of histoplasmosis and were initiated on antifungal therapy, the survival rates were significantly better, especially in the post-2000
Introduction
Hemophagocytic lymphohistiocytosis (HLH) is a syndrome characterized by nonmalignant proliferation and activation of lymphocytes and macrophages (histiocytes); hemophagocytosis in the reticuloendothelial system; and the clinical findings of fever, hepatosplenomegaly, cytopenias, and hyperferritinemia. 1 This syndrome presents as the final common pathway for certain familial syndromes with specific genetic mutations, resulting in impaired function of cytotoxic T cells and natural killer cells (primary HLH) as well as acquired conditions such as autoimmune disorders and infections (secondary HLH). 2
Infections contribute to the initiation of hemophagocytosis in 2 types of scenarios. 3 They have been often identified as the triggers for the onset of hemophagocytic syndrome in genetic disorders or autoimmune diseases, with increased propensity for HLH. They can also act as the primary drivers for the onset of HLH in patients without those underlying disorders. A wide variety of bacterial, viral, fungal, and parasitic infections have been associated with HLH. 3 Among them, HIV infection has been associated with HLH in several different ways. In the majority of such cases, AIDS acts only as the underlying disease setting in which an opportunistic infection triggers the onset of HLH. 4 However, HIV itself, without opportunistic infections, has been attributed to be the cause of HLH in several reported cases. 4 Hemophagocytic lymphohistiocytosis has also been described in the settings of immune reconstitution inflammatory syndrome (IRIS) and acute HIV infection. 5,6 Here, we present a case report of HLH in a patient with AIDS and disseminated histoplasmosis and also present a review of literature on this topic.
Methods
The case reports and case series of HLH with HIV and histoplasmosis were identified in Pubmed® database through the use of search criteria (“HIV Infections”[Mesh] OR “HIV” [Mesh] OR “HIV-1”[Mesh] OR “HIV-2”[Mesh] OR “Acquired Immunodeficiency Syndrome”[Mesh] OR “AIDS-Related Opportunistic Infections”[Mesh])
Case
A 42-year-old woman with a history of AIDS presented to the emergency department with fever and night sweats for a few weeks and malaise, generalized weakness, vague, abdominal discomfort and weight loss for several months. She denied the history of dysphagia or odynophagia, vomiting, diarrhea, cough or sputum production, or skin rash. She had been diagnosed with HIV infection and AIDS prior to 2 years but did not have a history of opportunistic infections. She had been nonadherent to both antiretroviral medications and prophylaxis therapy with trimethoprim–sulfamethoxazole and azithromycin. In the past, she had unprotected sex with different partners. She had been living in New Orleans, Louisiana, for many years and denied any foreign travels. She was a chronic smoker and had used cocaine in the past but denied abuse of alcohol or intravenous drugs. Examination revealed an emaciated and frail patient who was febrile with a temperature of 101.4°F and a heart rate of 115/min. Her blood pressure was 110/70 mm Hg and oxygen saturation was 98% in room air. Icterus and hepatosplenomegaly were absent. Regional lymph node examination was positive for enlarged lymph nodes palpable in bilateral inguinal regions only. Blood tests revealed white blood cell count of 2.9 × 109/L, hemoglobin 9.1 g/dL, platelets 356 × 109/L, and normal renal and liver functions. Her last CD4 count was 40 cells/mm3 (6.2%). Serum lactate dehydrogenase was slightly elevated at 485 U/L (normal range <201 U/L). Abdominal computed tomography showed diffuse mesenteric and retroperitoneal lymphadenopathy. Routine blood culture showed no bacterial or fungal growth. Mycobacterial blood culture was also sent and remained negative. Interferon gamma release assay was negative. While the workup had been in progress, the patient continued to have high-grade fevers. There was an increase in serum lactate dehydrogenase to 2556 U/L and serum ferritin was elevated at >15 000 nG/mL. Inguinal lymph node biopsy showed focal aneutrophilic necrosis with narrow-based budding yeasts visible on fungal stain. Histoplasma urine antigen was reported as positive. Bone marrow biopsy revealed focal granulomatous inflammation with budding yeasts and hemophagocytes. Epstein-Barr virus DNA polymerase chain reaction (PCR) was reported as negative (lowest threshold of detection 100 copies/mL). The level of interleukin 2 (IL-2) receptor alpha was elevated at 1020 U/mL (normal range 223-710 U/mL). The natural killer cell count was 22/μL (11%; reference range 24-406/μL, 1.4%-19.4%). The patient was started on intravenous infusions of liposomal amphotericin B after which she had rapid resolution of fevers and other symptoms within days. She was discharged home in a few days. After completing a 2-week course of liposomal amphotericin B infusions, she was started on oral itraconazole as well as antiretroviral therapy. On follow-up at 4 months since discharge from the hospital, her serum ferritin level had decreased to normal range, and the patient had gained 20 pounds of weight and was symptom free.
Results
A total of 18 cases with HLH in the setting of histoplasmosis and HIV infection, reported in 11 articles, were identified from Pubmed search. 7 –17 The features of the 18 cases as well as our case are summarized in Tables 1 and 2.
Clinical, Microbiological, and Pathological Data and Outcome on Reported Cases of HLH with HIV and Histoplasmosis.

Abbreviations: AB, amphotericin B; ABD, amphotericin B deoxycholate; ABLC, amphotericin B lipid complex; Bld, blood; BAL, bronchoalveolar lavage; BI, bilateral infiltrates; BM, bone marrow; CNS, central nervous system; CSF, cerebrospinal fluid; F, female; Fl, fluconazole; HLH, ; IRIS, immune reconstitution inflammatory syndrome; KS, Kaposi sarcoma; LAB, liposomal amphotericin B; LN, lymph node; LRT, lower respiratory tract; M, male; MRSA, methicillin-resistant Staphylococcus aureus; NR, not reported; PCP, Pneumocystis jiroveci pneumonia; PR, present report.
aNot further specified.
bPeriurethral ulcer.
cHemophagocytosis seen on lymph node aspirate smear.
Hematological and Biochemical Data on Reported Cases of HLH Associated with HIV and Disseminated Histoplasmosis.
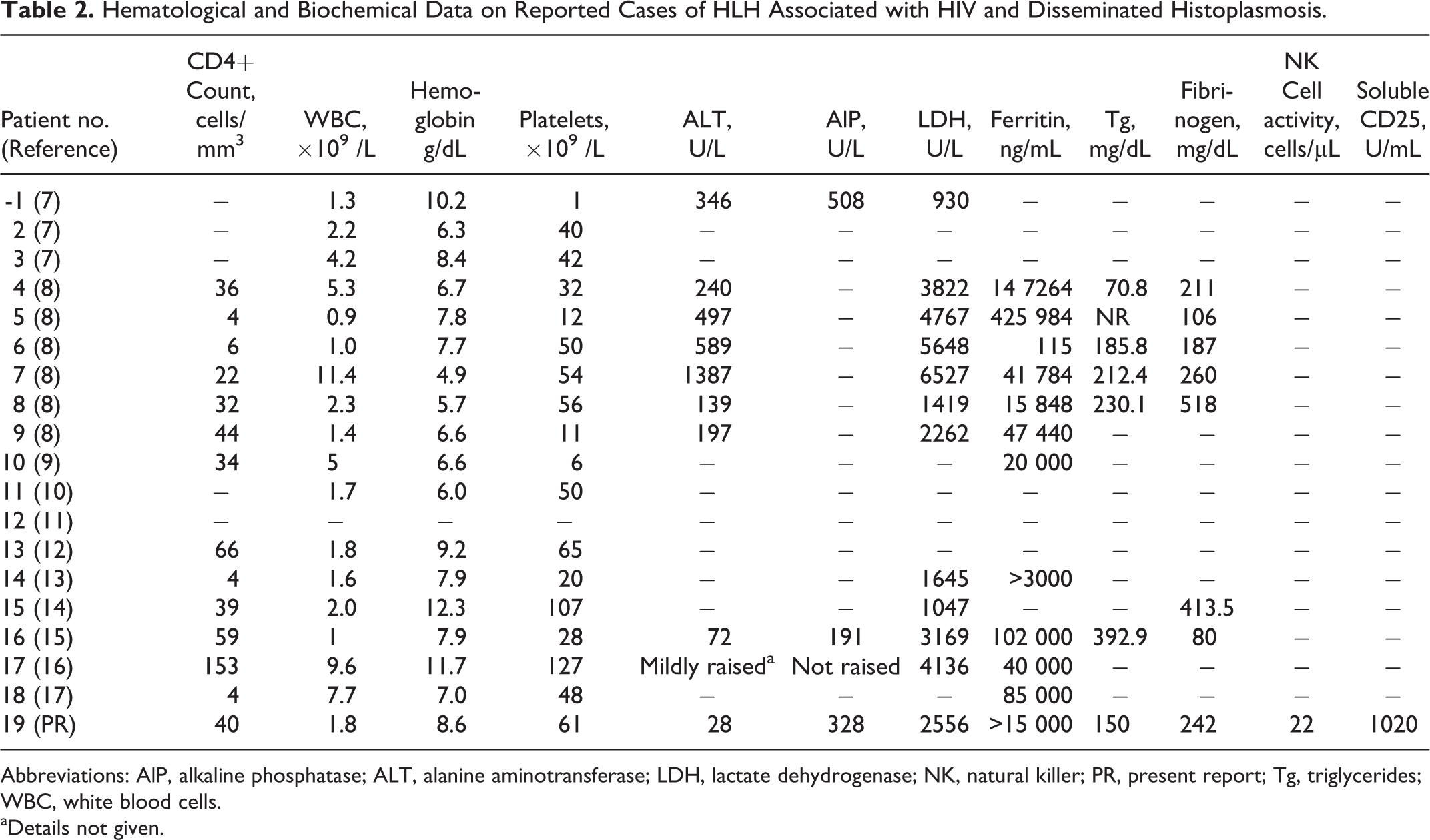
Abbreviations: AlP, alkaline phosphatase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; NK, natural killer; PR, present report; Tg, triglycerides; WBC, white blood cells.
aDetails not given.
Among the 13 cases whose sex was mentioned, all were male patients, except our patient. Another comorbidity was reported in 9 patients, which was another opportunistic infection, acute infection, complication of histoplasmosis, or, in one case, IRIS. Fever was a presenting symptom in all patients. Rash, which was a manifestation either of histoplasmosis or HLH, was reported only in some patients. Hepatomegaly or splenomegaly was only reported in some patients. Among the 14 patients whose last CD4 count at presentation was mentioned, 13 had a CD4 count <70 cells/mm3. Majority of the patients had significant pancytopenia or cytopenias involving at least 2 cell lines. Among laboratory tests reported, transaminitis, elevated lactate dehydrogenase, and hyperferritinemia (which was absent only in 1 case) were the rule. The diagnosis of HLH associated with disseminated histoplasmosis was confirmed by visualization of narrow-based yeast and hemophagocytic macrophages in bone marrow biopsy in all cases except 1 where they were seen in the aspirate of an enlarged lymph node. In some cases, histoplasmosis was further confirmed by positive antigenemia in the urine or blood, by positive PCR in blood, or by culture and morphological identification.
None of the patients were treated with chemotherapy regimens often used in the treatment of HLH in other settings such as familial HLH syndromes or Ebstein Barr virus associated HLH. Neither was treatment with monoclonal antibodies or steroids reported. Five patients were treated with intravenous immunoglobulin infusions along with antifungal treatment. Where described, antifungal treatment was with a form of amphotericin B infusion, except in 3 cases, 1 (case 1) treated with fluconazole, and the remaining 2 cases (cases 17 and 18) where the antifungal medication used was not named. In 3 cases, (cases 3, 11, and 15) the antifungal treatment was not given clearly. For 2 cases (cases 10 and 12), no information was given regarding whether antifungal treatment was provided or not.
For analysis of outcomes, 1 case without information on final outcome (case 10) was excluded from analysis. Of the remaining 18 patients, 8 (44.4%) did not survive. Higher mortality was associated with fungemia with Histoplasma and lack of antifungal treatment. Of the 9 patients in whom Histoplasma was identified in blood smear or culture, 5 died. Among the 14 cases in which antifungal therapy was given clearly, 10 cases survived. Of the patients reported after the year 2000 (cases 12-19), 5 of the 6 patients who clearly received antifungal therapy survived.
Discussion
We presented a case report of a female patient with AIDS who developed HLH during the course of disseminated histoplasmosis. We also presented a review of all case reports of HLH in patients with AIDS and histoplasmosis that we could identify on Pubmed search.
Except for her sex, our patient met the description of the typical patient described earlier in literature for HIV infection, histoplasmosis, and HLH, especially relatively young patients with advanced AIDS and very low CD4 count. For unclear reasons, this report is the first to describe a female patient with HIV infection, histoplasmosis, and HLH. Male predominance (82%) was also noted in a review of previously published 39 patients with HLH and underlying HIV infection, with or without opportunistic infections. 18
For most of the cases reviewed here, the typical presentation was of “fever of unknown origin,” although in several cases there were other coexistent causes for fever such as other opportunistic infections or acute infections. Fever in an AIDS patient has a very long list of differential diagnoses. Identification of disseminated histoplasmosis, which is often overshadowed by other considerations such as tuberculosis, is therefore likely to be delayed. Diagnosis of HLH, which itself is a very rare presentation, in the setting of disseminated histoplasmosis in AIDS is therefore naturally delayed. Another reason for the delay, as observed in our patient’s case, is that the identification of both of these entities requires either evaluation of tissue specimens or tests (histoplasma antigen in urine or serum or nucleic acid amplification in blood) that are not available even in many tertiary hospitals in the United States. 19 Recent data have suggested that Histoplasma capsulatum is more widely spread in different parts of the world than previously thought. 20 Recent literature also suggests that the global burden of disseminated histoplasmosis and associated mortality, especially among HIV-positive patients, has also been hugely underestimated. 21,22 Therefore, it would not be inappropriate to assume that cases of HLH in patients with disseminated histoplasmosis in the setting of HIV infection are also grossly underdiagnosed and underreported. Since early diagnosis and initiation of antifungal therapy appear to have significant impact in outcome, as discussed later, physicians need to be mindful of local epidemiology patterns and consider early the possibilities of both histoplasmosis and HLH in cases of fever of unknown origin in patients with AIDS so that serum ferritin level, microbiological tests for histoplasmosis, or biopsies looking for yeasts or hemophagocytosis can be ordered sooner.
Traditionally HLH used to be regarded as an often rapidly progressive and, if untreated, fatal disorder. Based on the data largely drawn from pediatric population with certain familial disorders, autoimmune syndromes, and malignancies, the Histiocyte Society has released guidelines that outline specific clinical and laboratory criteria (Table 3) for the diagnosis of HLH and specific algorithms for its management. 1,23,24 A study evaluating how applicable the diagnostic criteria and treatment algorithm laid out in these guidelines are to adult HIV-infected patients has not been reported so far.
Diagnostic Criteria for HLH.

Abbreviations: HLH, hemophagocytic lymphohistiocytosis; NK, natural killer.
Most of the features listed as the diagnostic criteria for HLH such as fever, splenomegaly, bicytopenias, hypertriglyrecidemia, and hyperferritinemia are often present in patients with advanced HIV with or without opportunistic infections such as histoplasmosis and with or without antiretroviral therapy. 4,25 –29 Hemophagocytosis itself may not be an uncommon finding in AIDS either, as demonstrated in an autopsy case series of 56 patients, of whom 54 consecutive cases had AIDS and 11 had hemophagocytosis. 30 Another study reported hemophagocytosis in bone marrow biopsy in around 10% of patients with HIV prior to the initiation of antiretroviral therapy. 31 Presence of hemophagocytosis has also been described in nonselected populations of patients with severe sepsis and multiple organ dysfunction syndrome. 32 Of 107 autopsies of critically ill medical patients, 64.5% had hemophagocytosis in bone marrows. 33 This overlap of clinical and laboratory features between AIDS and HLH as well as between sepsis syndromes and HLH has given rise to significant diagnostic challenges in identifying HLH in patients with AIDS and has probably led to HLH being significantly underdiagnosed in AIDS population. At the same time, the overlap also means that the cases of HLH identified among patients with AIDS using the Histiocyte Society guidelines are probably more diverse than the uniformly critically ill HLH patients who are diagnosed using these guidelines, in other populations such as familial HLH syndromes, Ebstein-Barr virus associated HLH in non-HIV patients or macrophage activation syndromes among juvenile connective tissue disorders.
It is not clear from previously reported data whether a particular HIV-associated clinical setting or opportunistic infection is more likely to lead to the development of HLH. In a review of previously reported 39 cases of HLH in patients with HIV, 9 (23%) cases had no significant comorbidity except HIV infection; 6 (15%) had disseminated histoplasmosis; 5 (13%) each had Kaposi sarcoma or lymphoma (B cell, T cell, or primary effusion lymphoma); and the rest had toxoplasmosis, CMV disease, mycobacterial infections, or other opportunistic infections. 18 This review did not include cases of HLH reported in the setting of acute HIV infection or IRIS; however, multiple cases of HLH occurring during primary HIV infection or IRIS have been reported. 5,34
It is similarly difficult to ascertain if outcomes of HLH occurring in the setting of disseminated histoplasmosis are different from those of HLH occurring in other clinical settings in HIV-infected patients. In the aforementioned review of HLH in 39 patients with HIV, overall only 28% recovered, and 50% of those who died did so within the first month. 18 On breaking down the numbers even further, of the 6 patients with disseminated histoplasmosis, half survived, while among those with HIV infection alone or with HIV and a nonhistoplasmosis opportunistic infection or comorbidity, only a quarter survived. Information regarding treatment was available only for 15 patients. Intravenous immunoglobulin was used in 6 patients, steroids were used in 5 patients, and non-HLH protocol chemotherapy was used in 2 patients; and only 1 patient survived. In a review of acute HIV-infection-associated HLH, however, all of the reported 7 cases survived, despite only 1 of the patients receiving antiretroviral therapy. Because of enormous variabilities in baseline CD4 counts, the pathophysiological attributes of specific types of comorbidities, opportunistic infections, or the stage of HIV infection involved, the thoroughness of workup done for each case to identify all possible associated comorbidities, or the appropriateness and promptness of disease-specific treatment provided, specific conclusions regarding differences in outcomes between HLH in setting of disseminated histoplasmosis and other clinical settings in HIV-infected patients would be premature. Previous literature has been more unanimous in indicating poorer survival rate among HIV-infected patients with HLH compared to the survival rate of 60% to 70% in HIV-negative patients with reactive HLH. 4,35
In the present review, we also noticed a high overall mortality rate of 44.4% among the 18 patients reviewed here for whom the outcome data were available. Not getting antifungal treatment and having Histoplasma in blood were the 2 main risk factors for death. Of the 14 patients who received antifungal therapy, 10 survived HLH. More significantly, among the 6 cases reported after the year 2000 who clearly received antifungal therapy, 5 survived the illness. The patients who did not get antifungal therapy, probably due to the delay in diagnosis, uniformly died in the acute stage. The majority of patients who had Histoplasma identified in blood smear or culture died. The only patient (case 17) who did not have either of the above 2 risk factors but still died had a severe comorbidity (Streptococcus pneumoniae bacteremia) as well as widely disseminated histoplasmosis involving the lungs, skin, and oral mucosa. This suggests that despite the presence of HLH features and disseminated histoplasmosis, these patients may have a window of opportunity before fungemia and more severe stages of HLH set in, when the course of illness is still reversible with appropriate antifungal and supportive treatment alone. It is important to note that the standard of care for HLH, as recommended in the Histiocyte Society guidelines, is to start cytotoxic chemotherapy and plan for possible bone marrow transplant in cases of familial or severe or relapsing HLH. 1 However, none of the survivors among these reviewed cases were treated with chemotherapy or bone marrow transplant, and these treatments are probably not needed at least for most of such cases as long as early diagnosis and timely initiation of antifungal therapy have occurred.
To summarize, we reported a case of HLH associated with advanced HIV infection and disseminated histoplasmosis and reviewed a total of 19 such cases so far published in literature. Not getting antifungal therapy and fungemia with Histoplasma were associated with higher mortality among these cases. Despite the significantly high mortality rates reported for HLH in general without chemotherapy and even worse outcomes reported for patients with HIV and HLH, we noted that among the patients who received timely diagnosis of histoplasmosis and were initiated on antifungal therapy, the survival rates were significantly better, especially in the post-2000
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
