Abstract
Introduction:
Rhodococcus equi, previously called Corynebacterium equi, is known to cause pneumonia in foals and swine. Although it was known to cause infection rarely in humans, R equi infection in humans has increased with the advent of HIV and increased use of immunosuppressants.
Case:
We report a case of a 48-year-old male patient with newly diagnosed HIV/AIDS presenting with recurrent R equi bacteremia, pericardial effusion, and pericardial cyst. The infection was treated with drainage of the pericardial effusion and cyst and 2 weeks of intravenous vancomycin and 6 months of oral azithromycin and levofloxacin.
Discussion:
Rhodococcus equi causes pericarditis and pericardial effusion. It can be effectively treated with debridement, drainage, and a prolonged course of antibiotics. In vitro antibiotic susceptibility should be checked as resistance to antibiotics can develop, especially if drainage is inadequate.
Keywords
Introduction
Rhodococcus equi is a gram-positive coryneform bacterium that can cause zoonotic infections mainly in horses, cattle, swine, sheep, goats, dogs, cats, and wild birds. Rhodococcus equi is an aerobic organism that is found in soil and water worldwide as well as in the manure of herbivores. Infection in animals and humans is thought to occur by inhalation of organisms. Outbreaks of Rhodococcus pneumonia among foals has been reported in breeding farms and seemed to be associated with a high concentration of Rhodococcus in the soil. 1 Immunocompromised patients with defective cell-mediated immunity such as HIV-infected patients and transplant patients appear to be more susceptible to R equi infection. 2,3
Rhodococcus equi can cause pneumonia, bacteremia, and abscesses. 4 As it is a gram-positive rod, it might be confused as a contaminant in cultured specimens and discarded without proper identification. Knowing that HIV-infected patients and transplant patients have defective cell-mediated immunity, gram-positive rods in their blood or abscess cultures should not be discarded but identified. In an environment where horse and cattle farms are a major source of employment, this is especially true due to the possibility of R equi infection.
Case Report
A 48-year-old man presented to an emergency department with a 2-month history of shortness of breath, cough productive of yellow sputum, night sweats, myalgia, and 20-pound weight loss. The patient had failed to respond to levofloxacin and ciprofloxacin. The patient had lived with a single male partner for 16 years. He raised horses and chickens. On presentation, he was markedly cachectic, with oral thrush and mild respiratory distress. He was found to have AIDS with an HIV viral load of 143 000 copies/mL and CD4 count of 10 cells/mm3 (5%). Diagnostic studies found R equi bacteremia, disseminated histoplasmosis, and pneumocystis pneumonia (PCP). The latter 2 were found in bronchoalveolar lavage fluid. Community-acquired pneumonia was also a consideration. He was intubated and treated with sulfamethoxazole/trimethoprim, levofloxacin, vancomycin, piperacillin–tazobactam, and an intravenous amphotericin B lipid formulation. The patient also had a right-side thoracentesis with chest tube placement for empyema. Pleural fluid KOH staining revealed yeast, which was later identified as Histoplasma capsulatum. The R equi initially was sensitive to vancomycin, clarithromycin, minocycline, sulfamethoxazole/trimethoprim, tobramycin, and rifampin but intermediate to linezolid and resistant to amoxicillin/clavulanic acid, ceftriaxone, and imipenem. Piperacillin–tazobactam was stopped, and azithromycin and doxycycline were substituted as guided by the susceptibility results. As his condition improved, vancomycin was discontinued after 2 weeks of therapy, and HIV treatment was started with emtricitabine, tenofovir (TDF), and raltegravir.
After completion of 21 days of sulfamethoxazole/trimethoprim for PCP, the patient developed significant leucopenia, and secondary prophylaxis treatment for PCP was changed to atovaquone. After 23 days of his stay, patient’s blood culture grew R equi again. At this time, its susceptibility pattern revealed resistance to sulfamethoxazole/trimethoprim and tobramycin but intermediate susceptibility to rifampin and linezolid. The patient complained of chest pain, and an electrocardiogram revealed diffuse ST-segment elevation (Figure 1). Transthoracic echocardiogram demonstrated a large pericardial effusion, without cardiac tamponade and a pericardial cyst (Figure 2). A repeat computed tomography (CT) of the chest without contrast performed 4 weeks after admission revealed a pericardial effusion and cyst; both were also seen on an echocardiogram (Figure 3). Pericardiocentesis with drainage of pericardial effusion and cyst was performed, and the pericardial fluid and pericardial cyst fluid both grew R equi. One week after drainage, a pericardial drain was placed. Pericardial fluid again grew R equi. The pericardial drain was removed after 5 days. The patient was discharged after 2 months of hospitalization to an acute rehabilitation facility and continued to receive azithromycin, levofloxacin, itraconazole, emtricitabine, TDF, raltegravir, and atovaquone. The patient’s symptoms improved and no recurrence of pericardial effusion was identified after 6 months of treatment with azithromycin and levofloxacin.

Electrocardiogram—arrows show ST-segment elevation.
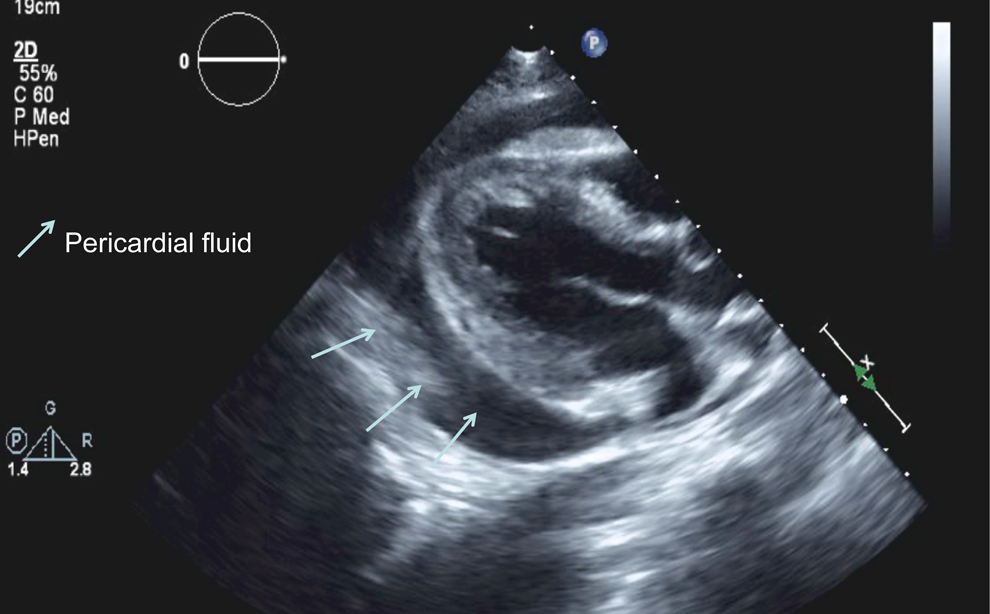
Transthoracic echocardiogram—arrows show pericardial effusion.

Computed tomography chest with contrast—arrows show pericardial effusion and cyst.
Discussion
There have been 3 reported cases of R equi pericarditis: 1 renal transplant recipient, 1 failed renal transplant, and 1 HIV-infected patient. 5 –7 To our knowledge, this is the second reported case in the literature (only in English) of R equi pericarditis in an HIV-infected patient. The presentation, treatment, and outcome of the 3 reported patients with Rhodococcus pericarditis are shown in Table 1. In this case, the recurrence of R equi bacteremia and a follow-up CT of the chest led to the diagnosis of pericarditis.
Characteristics of 3 Patients and the Case Report Diagnosed with Rhodococcus equi Pericarditis.

Abbreviations: AZM, azithromycin; AMK, amikacin; CLR, clarithromycin; ERY, erythromycin; ETH, ethambutol; GEN, gentamycin; IPM, imipenem; LVX, levofloxacin; PIP/TZB, piperacillin–tazobactam; RIF, rifampin/rifampicin; SMZ/TMP, sulfamethoxazole/trimethoprim; VAN, vancomycin; INH, isoniazid; F, female; M, male; Meds, medications.
The majority of R equi infections have occurred in immunocompromised patients. Eighty-six percent of cases occurred in immunocompromised hosts, with half of those occurring in patients with HIV infections. 8 In 1 review of 72 cases, pneumonia occurred in 76% of patients, and the lung was the only site of infection in 82%. Pneumonia was accompanied by extrapulmonary infection in 18% of cases, while infection at extrapulmonary sites occurred without evidence of pulmonary involvement in 24%. The most common extrapulmonary sites of infection complicating pulmonary infection in immunocompromised hosts have been the brain and skin (presenting as subcutaneous abscesses). 8
In a multicenter, retrospective analysis of 67 cases of R equi infection in HIV-infected patients, pulmonary disease was the predominant presentation (64 [96%] patients), and 40 (60%) patients had R equi bacteremia. Twenty-three (34%) patients died due to R equi-related causes. All deaths were in patients not receiving highly active antiretroviral therapy. 9
Monotherapy for R equi infection is not recommended, as it is often ineffective. A 2-drug regimen often including agents such as macrolide, rifampin, or fluoroquinolone is the treatment of choice for R equi infections. Selection of antimicrobials should include in vitro susceptibility data. R equi is usually susceptible to erythromycin and extended spectrum macrolides, rifampin, fluoroquinolones, aminoglycosides, glycopeptides, and imipenem. 10
Initial therapy in immunocompromised persons is 2 to 6 weeks of an active intravenous antibiotic such as vancomycin (or a carbapenem or an aminoglycoside) in combination with an oral drug such as azithromycin or rifampicin. 3 The duration of therapy depends on the site(s) and extent of infection, underlying immunocompetence of the host, and the clinical response to therapy. A minimum of 6 months of antibiotic therapy is typically required for immunocompromised patients with pulmonary, bone and joint, or central nervous system infections. 4 Any improvement in the immunocompetence of the host should be pursued as a therapeutic adjunct in treating R equi infections, either through curtailment of immunosuppressive medications or through aggressive antiretroviral therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
