Abstract
Cost-effective HIV prevention programs should target persons at high risk of HIV acquisition. We conducted an observational HIV incidence cohort study in Kisumu, Kenya, where HIV prevalence is triple that of the national rate. We used referral and venue-sampling approaches to enroll HIV-negative persons for a 12-month observational cohort, August 2010 to September 2011, collected data using computer-assisted interviews, and performed HIV testing quarterly. Among 1292 eligible persons, 648 (50%) were excluded for HIV positivity and other reasons. Of the 644 enrollees, 52% were women who were significantly older than men (P < .01). In all, 7 persons seroconverted (incidence rate [IR] per 100 person-years = 1.11; 95% confidence interval [CI] 0.45-2.30), 6 were women; 5 (IR = 3.14; 95% CI 1.02-7.34) of whom were ≤25 years. Most new infections occurred in young women, an observation consistent with other findings in sub-Saharan Africa that women aged ≤25 years are an important population for HIV intervention trials in Africa.
Introduction
Successful HIV prevention trials, similar to successful programmatic strategies to address HIV acquisition, require the identification and engagement of persons at risk for HIV infection. 1,2 To detect intervention effectiveness without cost-prohibitive large sample sizes or very lengthy studies, clinical prevention trials are usually conducted in populations with high incidence of diseases targeted by the biomedical intervention. Recent HIV prevention trials, for example, have been done in settings with HIV incidence rates (IRs) of 3% to 6%. 3 –6 Organizing such trials in the United States, where the overall HIV incidence is 0.02%, 7 has required finding and enrolling subpopulations at markedly increased risk, some of whom may be socially marginalized, stigmatized, and/or challenging to reach and retain. 8 The US domestic HIV prevention trials can therefore be labor intensive and expensive. 9 In Africa, where two-thirds of persons living with HIV reside, 10 the identification of populations with high HIV IRs should theoretically be easier. But several trials have found unexpectedly low incidence in a context of high prevalence, 11 –13 the reasons for which have not always been satisfactorily explained. 1
Despite these challenges, several biomedical modalities using antiretroviral drugs, including 1 vaccine, have been shown in recent years to be partially effective in reducing HIV infection. 3,4,14 –16 When and if such modalities are adopted as national standards of care, however, the ethical imperative to offer them to control group participants in future trials will likely lower the HIV incidence, this in turn will increase the study sample size needed to detect the efficacy of new interventions. 17 In the current global economic environment where resources for HIV programs are already constrained, 18 the adoption of partially effective interventions may unavoidably create concomitant fiscal and logistic challenges to improving these interventions. In this context, the need to identify high-incidence populations is crucial for both HIV prevention trials and HIV prevention programs focused on interrupting HIV transmission.
Nyanza province in northwest Kenya has an overall HIV prevalence of 15%, the highest in the country. 19 A recent serosurvey among adults in its largest city, Kisumu, reported a prevalence of 26%, 3 times that of the national HIV prevalence estimate. 20 In preparation for future HIV prevention trials, the Kenya Medical Research Institute (KEMRI)/Centers for Disease Control and Prevention (CDC) enrolled 625 sexually active residents of Kisumu aged 18 to 34 years in the Kisumu Incidence Cohort Study 21 (KICoS) 1, beginning in January 2007, and 206 adolescents aged 16 to 17 years in KICoS2, beginning in April 2009. 22 As the overall HIV incidence in KICoS1 (1.41% overall; 2.59% among women and 0.59% among men) 23 was unexpectedly low relative to the high prevalence, we initiated KICoS3 to better target individuals at higher risk for acquiring HIV than KICoS1—which had recruited using mobilization strategies targeting the general community (eg, discussions and/or presentations at market centers, churches, women’s groups, community groups, formal and informal work-based groups, colleges and schools, and voluntary counseling and testing centers).
Several studies in sub-Saharan Africa have shown commercial sex work, 24 age, gender, marital status, number of sex partners, 25 –28 genital herpes (Herpes simplex virus [HSV-2]) seropositivity, 25,28 reported sexually transmitted infections (STIs), 25 lack of condom use, 25,27,29 and alcohol use during sex 27 to be among behavioral risk factors associated with acquiring HIV. We used this knowledge, including information gained from a formative study conducted to identify strategies to recruit persons at high risk for HIV infection 2 in western Kenya to guide our recruitment for KICoS3. We sought to (1) identify HIV prevalence among screened persons, (2) determine HIV incidence among enrolled participants, (3) assess differences in the incidence by sex and age among enrolled persons, and (4) examine demographic and behavioral differences between our initial “seed” recruits and the persons enrolled into the study (“offspring” recruits).
Methods
Design
Similar to KICoS1, KICoS3 was an observational, prospective cohort study following individual participants for 12 months 23 but using modified eligibility criteria and recruitment methods described subsequently. Kisumu Incidence Cohort Study 3 accrual occurred during March 2010 to August 2010 with a 12-month routine study follow-up ending in September 2011. Based on a Poisson distribution, an enrollment sample size of 625 high-risk cohort participants in Kisumu, projected an incidence of 3.0% and retention of 500 (80%); we expected at least 15 HIV seroconverters and a statistically significant result around the incidence.
Setting
Kisumu is a city of approximately 504 000 people located in Nyanza Province, Kenya. 30
Ethical Review
The study protocol, consent forms, and data collection instruments for this study were reviewed and approved by the KEMRI Scientific Steering Committee Ethical Review Committee as well as by the US CDC Institutional Review Board. Written informed consent was completed by persons meeting the prescreening eligibility criteria before taking part in data and specimen collections. Options were provided to complete the informed consent process and data collection in Dholuo, English, or Kiswahili.
Recruitment
A participant referral approach, a modification of respondent-driven sampling, 31 was employed to amplify recruitment strategies identified in the formative study 2 : initial study recruits (referred to as seed recruits) were asked to refer those they knew, who in turn referred those they knew (referred to as offspring recruits). Seed enrollees were recruited from a variety of different venues, including bars, night clubs, market centers, truck stops, fish landing beaches, churches, women’s groups and community groups, formal and informal work-based groups, colleges and schools, and HIV voluntary counseling and testing centers.
Prescreening, Screening, and Enrollment
Prescreening, informed consent, screening, and enrollment procedures were conducted at the study facility, the KEMRI-CDC Clinical Research Center on the campus of the New Nyanza Provincial General Hospital. Prescreening assessment was conducted using interviewer-administered computer-assisted personal interview (CAPI). Persons who met the prescreening criteria received detailed information about the study prior to providing written informed consent to proceed with screening procedures. At screening, a baseline behavioral questionnaire was administered using audio computer-assisted self-interview (ACASI); medical history and clinical evaluation were documented using CAPI; and blood, urine, and vaginal swabs were obtained. Participants were tested for hemoglobin and platelet levels, liver and kidney function, STI serologies (syphilis and genital herpes [HSV-2]), and pregnancy for women. Rapid HIV testing following the Kenyan national algorithm was conducted with pre- and posttest counseling. Male circumcision was verified by physical examination. Laboratory results were used to make the final study eligibility determination. Persons were informed of their enrollment eligibility status 2 weeks following the initial (screening) visit.
Enrollment Criteria
Study enrollment was limited to male and nonpregnant female residents of the Kisumu catchment area, aged 15 to 64 years. This expanded age range from 18 to 34 years in KICoS1was used to capture data on the prevalence and incidence of HIV seroconversion among Kenyans aged 50 to 64 years, an understudied, suspected high-risk population identified through the Kenya AIDS Indicator Survey 2007. 19 This group requires more focus on HIV research studies and intervention trials as do minors who are pregnant, married, or have children. Enrollees needed to report being sexually active within the 3 months prior to enrollment and be HIV negative but belonging to a “high-risk group” for HIV acquisition based on the following criteria (derived from previous HIV prevalence and qualitative studies) 2 : earned a living as a commercial sex worker (CSW), truck driver, car washer, police officer, or in fishing-related activities; had a regular sexual partner who was HIV infected; was a widow or widower; was a man who reported having sex with men (MSM); or was a homeless youth living on the streets. To be included in enrollment, participants also needed to answer “yes” in ACASI to acknowledge at least 1 occasion of “high-risk behavior” in the previous 12 months: Did you have a sexual partner who is infected with HIV? Did you have vaginal or anal sexual intercourse with an anonymous partner who could not be contacted again? Did you have vaginal or anal sex in exchange for money, goods, or services? Did you have vaginal or anal sex with 2 or more partners? Did you have a STI? Additionally, participants needed to be (1) willing to undergo an HIV test and receive results, (2) not to be in another HIV intervention study, (3) willing to give locator information, and (4) not planning to relocate outside the catchment area within the following 12 months. Offspring recruits could only be enrolled in the study if they had a referral card received from someone else in the referral chain. For those eligible for cohort study follow-up, a study photoidentification card with a unique barcode was issued, and detailed locator information was obtained and later verified by a home visit by study staff.
Study Incentives
All participants received provision of allowable incentives (transport reimbursement, bar soap, insecticide-treated mosquito net, and exercise book) to minimize loss to follow-up. Participants received HIV risk-reduction counseling and condom provision, treatment for common ailments (eg, diarrhea, respiratory and skin infections), and referrals for other services (including medical male circumcision) at all visits.
Follow-Up Procedures
Follow-up visits were scheduled every 3 months for a period of 12 months. At each follow-up visit, multiple activities were completed, including an ACASI behavioral questionnaire, rapid HIV testing, and a blood draw for laboratory testing.
Laboratory Procedures
All blood, urine, and vaginal specimens were processed and tested at the KEMRI/CDC ISO-certified laboratory located at the KEMRI/CDC facilities in Kisumu. 32 Hemoglobin and platelet counts were done using the Becton, Dickinson and Company (BD; Franklin Lakes, New Jersey) coulter counter complete blood count from whole blood, while the liver and kidney functions were analyzed using the ROCHE biochemistry analyzer from serum (ROCHE Diagnostics, Indianapolis, Indiana). Rapid HIV testing was conducted using Uni-Gold HIV-1/2, (Trinity Biotech, Wicklow, Ireland) and Determine HIV-1/2 (Abbott Labs, Tokyo, Japan) with the Bioline test (Meridian Life Science Company, Cincinnati, Ohio) as a tiebreaker. Testing for syphilis was done using BD Micro-Vue Rapid Plasma Reagin cardtest with all reactive tests confirmed by Serodia TP-PA Syphilis Test. Herpes simplex virus 2 was detected using KALON HSV-2 Immunoglobulin G enzyme-linked immunoassay (Kalon Biologicals Ltd, Surrey, United Kingdom), and urine pregnancy test was done using First Sign HCG One Step (UNIMED International, Inc, South San Francisco, California).
Procedures for HIV-Positive Persons
At screening, individuals who tested HIV-positive were provided with CD4 count results and referred to HIV care and treatment clinics, as they were ineligible for enrollment. Enrolled persons who seroconverted during the course of the study were provided with CD4 count and viral load results, follow-up visits, counseling, and referrals for HIV care and treatment and were retained in the study.
Data Analysis
For those screened but not eligible for cohort study enrollment, HIV prevalence by sex and age was examined. The rate of increase in prevalence by age for each gender was calculated using the median age. For those enrolled, demographic and behavioral characteristics were examined, and age distributions were compared by sex. Participants who acquired HIV during the course of the study were characterized by demographics and risk factors. HIV IRs were calculated based on 1 year of follow-up.
We calculated disease incidence using person-days of observation in the at-risk population. For enrolled participants, person-time was computed separately for those who did not seroconvert during the course of the study and those who did. Person-days for those who did not serconvert were calculated as the number of days from the first negative HIV test (ie, baseline) to the last negative HIV test. For participants who seroconverted, person-days of observation were calculated as the number of days from the baseline test date to the date of the last negative HIV test plus half the number of days between the last HIV-negative test and the first HIV-positive test. We chose this midpoint as a crude estimate of the actual time of seroconversion. Person-days were then converted to person-years (py) by dividing total person days by the number of days in a year (365.25 days). Participants who dropped out of the study were accounted for in our calculations of person-time. Subgroup and overall IRs per 100 py were calculated as the number of new cases of disease identified within the population (or subgroup) divided by the total number of py contributed by all patients within the population (or subgroup).
Demographic and behavioral differences between seed and offspring recruits were examined. To assess the significance of these differences, we used the chi-square test for categorical variables and the Wilcoxon test for continuous variables. A linear regression analysis was used to test for linear trend in HIV prevalence by age. SAS version 9.1.3 (SAS Institute, Cary, North Carolina) was used for all analyses.
Results
Screening and Enrollment
Among the 2205 persons (966 men, 1239 women) presenting for study consideration, 1347 (61.1%) met study prescreening eligibility criteria. Of the 1292 of 1347 (96%) who completed screening, 644 persons were enrolled in the study. About three-quarters (73%) of the women who presented for study consideration were ineligible (for reasons such as being HIV-positive at baseline, sexual inactivity, pregnancy, or not meeting the high-risk criteria), reducing the proportion of female enrollees to 51.7%. Very few (n = 83, 6.4%) persons prescreened were less than 20 years of age, and approximately one-third (n = 29, 35%) of persons aged <20 years were not enrolled, resulting in an 8.4% (n = 54) final proportion of enrollees. Of the enrolled persons, 127 (20.0%) were initially recruited (seed recruits) and 517 (80.0%) were referrals (offspring recruits) identified by the seed recruits and by other offspring recruits (Figure 1).
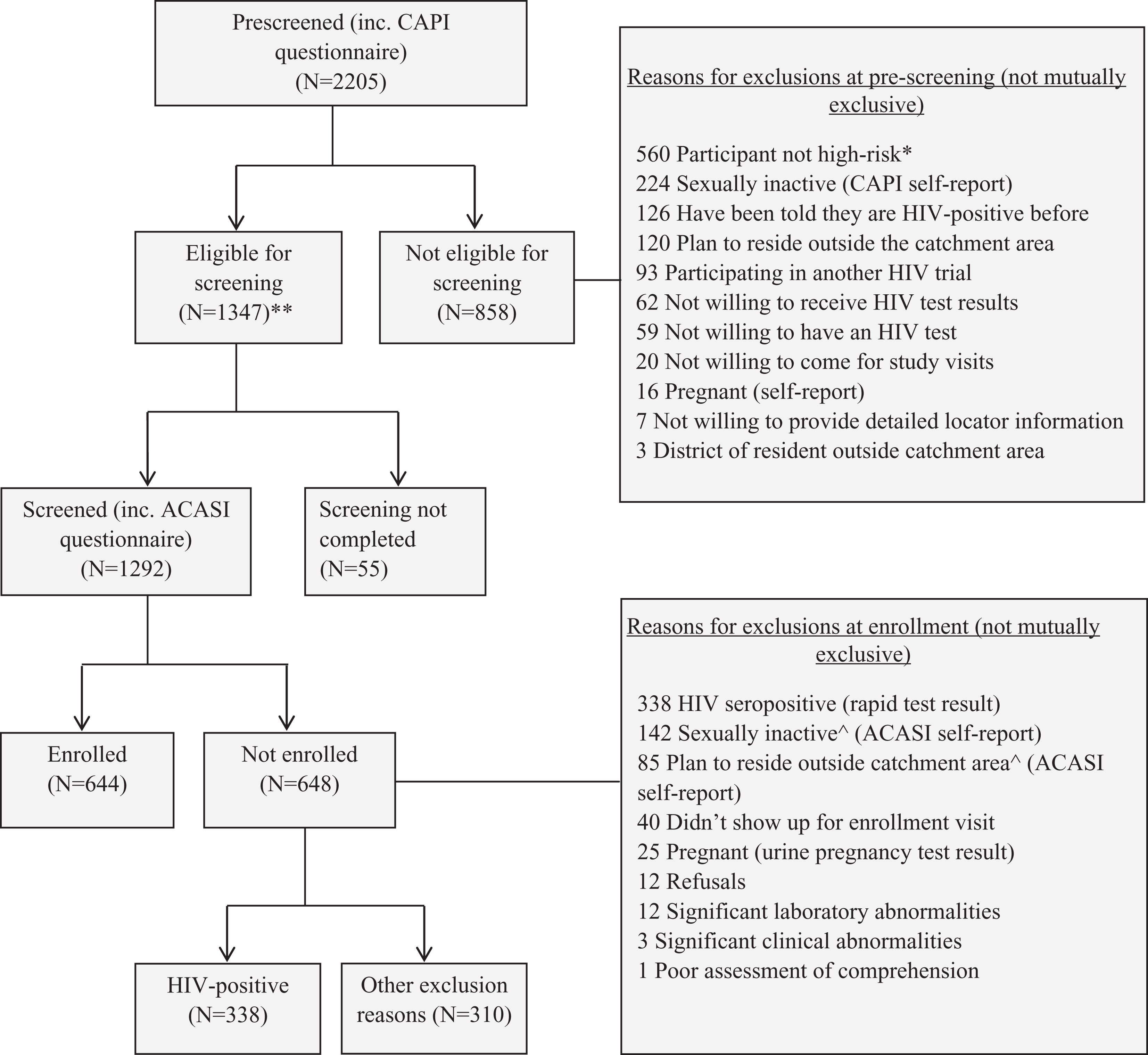
KICOS3 study profile at baseline screening and enrollment. *, not member of a “high-risk group” or did not acknowledge at least 1 “high-risk behavior” as defined in methods section. ⁁, participants were asked twice, once during prescreening CAPI, another time during screening ACASI. **, written informed consent sought prior to proceeding with screening procedures. ACASI, Audio Computer-Assisted Self Interview; CAPI, Computer Assisted Personal Interview; KICOS3, Kisumu Incidence Cohort Study 3.
Baseline Characteristics of Enrollees
Age and sex (Figure 2): while the proportion of the men and women was approximately equal (48.3% versus 51.7%), enrolled women were significantly older than men statistically: median age 25 versus 23 years (P < .01). Women were twice as likely to be aged 30 to 64 years (P < .01) and half as likely to be <20 years (P < .01). The largest single group (42.5%, n = 274) of the enrollees was men aged 20 to 24 years. We also recruited 77 (12%) persons aged 35 to 64 years.
Behavioral risks (Table 1): about half (51%) of the enrollees reported being male and female CSW in the 12 months prior to enrollment, and the remaining half had a variety of other risk factors. Among the 272 female CSW, 153 (56%) were aged ≤25 years. In all, 56 (18%) men reported being CSW and 97 (31%) men were in the MSM category.

Age distribution of enrolled participants by sex, KICOS3 2010 to 2011 (N = 644). The horizontal lines at the top show the age distribution of the study participants. The arrow indicates the median age (years); the numbers at the ends of the horizontal line are the 25th (left) and 75th (right) percentiles; and the length of the line is the interquartile distance around the median. KICOS3, Kisumu Incidence Cohort Study 3.
Baseline Characteristics of Enrolled Participants, KICOS3 2010 to 2011.a

Abbreviations: IQR, interquartile range; KICOS3, Kisumu Incidence Cohort Study 3; STI, sexually transmitted infection; HSV-2, Herpes simplex virus 2.
a Fractions are included to show missing values.
About half of the enrolled men (48%) were not circumcised. Of the enrollees, 79% reported having one or more occurrences of unprotected vaginal or anal sex and 35% reported treatment for an STI in the previous 3 months. The most common STI syndrome for which treatment had been received was genital ulcer disease (42.6%). Frequencies of reporting unprotected sex (P = .77) and treatment for an STI (P = .99) were not significantly different between the seed and the offspring recruits.
Baseline HIV Prevalence among Screened Persons
The overall HIV prevalence among individuals screened was 26.1% (338 of 1292). Prevalence was nearly 3 times as high among women as among men: 35.6% (272/765) versus 12.5% (66 of 527; P < .01) and increased by sex with age (P for trend = .04). The rate of the rise in prevalence per year of age for a person aged 15 to 29 years was approximately double for women compared to men: 1.6% versus 0.8% (P < .01). Baseline prevalence was 33.1% (236 of 712) in male and female CSW and 6.9% (9 of 130) in MSM. HIV prevalence among the seed recruits was similar compared to offspring recruits: 22.8% (52 of 228) versus 26.9% (286 of 1064; P = .21; Figure 3).

Baseline HIV prevalence by sex and age group among screened participants, KICOS3 2010 to 2011 (N = 338/1292). KICOS3, Kisumu Incidence Cohort Study 3.
HIV Incidence among the Enrollees
Of the 644 persons who were enrolled and followed for a period of 1 year, 61 (9.5%) voluntarily withdrew from the study, resulting in 627 py of follow-up overall (women, 318 py; men, 309 py). Seven of the enrollees seroconverted during the course of their study participation (IR per 100 py, 1.1; 95% CI: 0.45-2.30). Of the 7 incident cases, 6 (86%) occurred among women, 5 of whom were aged 20 to 25 years. CSWs accounted for 5 (83%) of the 6 cases among women. The single male seroconverter was a 26-year-old MSM CSW who was not circumcised. HIV-seroconversion among female CSW aged ≤25 years was significantly higher (5 of 153, 3.3% versus 0 of 21, 0%, P < .01) compared to female nonsex workers aged ≤25 years. The distribution of seed and offspring recruits among cases (seed:offspring ratio = 1:3.5) was similar to their relative proportions in the study population (seed:offspring ratio = 1:4). The HIV IR was 1.88 (95% CI: 0.69-4.10) per 100 py among women overall, 3.14 (95% CI: 1.02-7.34) per 100 py among women aged ≤25, 4.21 (95% CI: 1.36-9.82) per 100 py among women aged 20 to 24 years, 1.90 (95% CI: 0.69-4.11) per 100 py among male and female CSW, 1.03 per 100 py (95% CI 0.03-5.79) among MSM, and 0.32 (95% CI: 0.01-1.80 per 100 py among men (Table 2).
HIV Incident Casesa among Enrolled Participants, KICOS3 2010 to 2011.b

Abbreviations: KICOS3, Kisumu Incidence Cohort Study 3; STI, sexually transmitted infection; HSV-2, Herpes simplex virus 2.
a N = 7.
b N = 644.
Discussion
This study cast a broad net attempting to identify an urban population with a high HIV incidence suitable for HIV prevention trials in western Kenya. As a result of the effort to focus on those at the highest likelihood of prevalent or incident HIV by expanding the recruitment age from 15 to 34 years (in KICOS1) to 15 to 64 years and changing the targeted risk groups, more than 50% of the persons screened did not meet enrollment criteria. Baseline HIV prevalence among the study participants was high (26% overall, rising to nearly 50% among women aged >29 years), suggesting that we were successful in identifying persons from communities with a high burden of HIV. Furthermore, the ACASI results confirmed that the enrolled participants reported multiple risk factors (eg, treatment for an STI in the previous 3 months, unprotected sex, multiple sex partners, sex with known/suspected HIV infected persons, and sex with partners of unknown HIV status). Despite this, the overall HIV cumulative incidence in the cohort after 1 year follow-up was 1.1% (or IR = 1.1 per 100 py), considerably lower than the 3% to 6% levels commonly targeted for successful HIV intervention trials. Review of HIV positivity patterns in the screened group suggests, however, that the observed incidence should not have been entirely surprising. The rate of rise in HIV prevalence per year of age in the screened group at baseline was quite similar to the observed incidence during the 1 year follow-up: 1.6% versus 1.9% for women and 0.8% versus 0.3% for men, respectively. Of the 7 persons, 6 who seroconverted during the study were women, 5 of whom were aged ≤25 years, a pattern of risk consistent with prevalence patterns among women in the screened group. These 5 young women were also engaged in CSW, a factor identified as a risk factor for HIV acquisition in low- and middle-income countries. 24
These results suggest that the apparently paradoxical finding of high prevalence and comparatively low incidence may have a relatively simple explanation. In our study, prevalence was nearly 3 times as high among women as among men, confirming similar findings previously reported in Kenya. 28,33 Among both men and women, prevalence steadily rose with age, consistent with other studies in the same locality. These prevalence data suggest that demographically the group with highest incidence would be younger women. Most of our incident cases occurred among women ≤25 years of age, among which the IR was 3.1 per 100 py. Nevertheless, this group only constituted 23% of our study population. Women had constituted >60% of the initial recruits, but two-thirds of them were excluded (for reasons, at baseline, such as being pregnant, HIV-seropositive, sexually inactive, or not being considered high risk), so that the proportions of men and women among enrollees ended up approximately equal. In addition, study women were significantly older than the men: less than 10% of our population was aged <20 years, and women were half as likely as men to be in this age group. The largest single group constituting almost half the enrolled population was men aged 20 to 24 years, who would be expected to have low incidence and, in fact, had no seroconversions. Expanding our upper age eligibility to 64 years to study incidence in older age groups may have also contributed to the low yield.
Our study has a number of limitations. We depended upon the self-report for our behavioral indicators, and the validity of self-report about sexual behavior has been called into question. 5 Study participation can be very attractive to potential participants for multiple social, financial, and associated reasons, resulting perhaps in exaggeration or even impersonation of high-risk status. 34 Participants involved in snowball or respondent-referral sampling studies (like KICoS3) may also be inclined to refer persons who are not actually from the same risk groups as they are, either for their own benefit or for the benefit of the offspring participants. 30 We offered study participants a standard package of counseling and condoms, so that incidence observed in the study may have been lower than it might have been otherwise, a common occurrence in HIV incidence studies that offer HIV prevention services. 13 We followed a small study cohort for a relatively short duration (1 year) and had a 10% dropout rate, which may also have affected results. The small number of our incident cases makes any comparison of the incident to the nonincident cases difficult. Finally, due to the types of rapid HIV testing used in this study, the exact timing of seroconversion (which may have ranged within a month of testing positive) for each of the 7 cases could not be accurately determined. We did not ascertain how many of the individuals who were ineligible due to being HIV seropositive were recently infected. Therefore, the number of high-risk individuals who were screened but not enrolled could include recent incident cases which would have increased the number of incident HIV infections.
Despite our population’s self-reported behavioral risks for HIV acquisition, the overall rate of rise in age-specific prevalence in our study is fairly similar to that found by other studies in the same province. 28,33 Unlike in the United States, HIV transmission in Africa is generalized; the risk for HIV infection in high-prevalence areas like Kisumu, as indicated by our results, may most easily be captured by simply targeting the factors of sex, age, and CSW. Our findings, consistent with suggestions from other population-based studies in sub-Saharan Africa, suggest that young women aged ≤25 years attending venues similar to those we sampled from are prime candidates for prevention programs and intervention trials aimed at interrupting HIV transmission in western Kenya, and indeed in other parts of Africa with high HIV prevalence. Future prevention trials in urban centers should target this vulnerable group.
Footnotes
Acknowledgments
We would like to thank all study participants as well as CDC and KEMRI staff who were involved in the study for their assistance with study design and protocol development.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded through an Interagency Agreement Y1-AI-7278-01/ ST07015 between CDC and the National Institutes of Health (NIH).
