Abstract
The risk for occupational exposure to HIV is a serious public health problem that is well characterized in the developed world, but less so in the developing countries such as Ghana. This study was undertaken to examine the characteristics of occupational exposure to HIV and the utilization of a risk assessment system (RAS)–based postexposure prophylaxis (PEP) among health care workers (HCWs) and health care students (HCSs) in the Korle-Bu Teaching Hospital (KBTH). During the study period (January 2005–December 2010), a total of 260 and 35 exposures were reported by HCWs and HCSs, respectively. Ward attendants reported the highest incidence rate of 6.46 of 100 person-years (P-Y). The incidence of high-risk exposures was 0.33 of 100 P-Y (n = 65); 60.0% occurred during a procedure of disposing of a needle and 24.6% during a cannula insertion. A total of 289 of the 295 individuals were administered PEP, of which 181 (62.6%) completed the 6-month follow-up testing schedule and none sero-converted. This shows that with a good RAS in place, it is possible to deploy an effective PEP program in a typical African teaching hospital like the KBTH in Accra, Ghana.
Health care workers (HCWs) represent the human resource capacity of any health care delivery system in any country. However, in their activities and duties, they are continuously exposed to all types of risks including exposure to blood and other body fluids, which may pose a risk of infection by the HIV and other blood pathogens. 1,2 The World Health Organization (WHO) estimates that 3 million percutaneous exposures occur annually among 35 million HCWs worldwide, which represents 12% of the working population. 3 It is worth noting that while 90% of the occupational exposures occur in the developing world, 90% of the reports of occupational infections occur in the United States and Europe. 4 There is clearly underreporting of occupational exposures in developing countries, which could be attributed to several factors including ignorance about occupational exposures and their management, lack of avenues for easy reporting, lack of protection for workers, and absence of postexposure prophylaxis (PEP) programs. 4
Postexposure prophylaxis, which involves the administration of antiretroviral (ARV) drugs, has been estimated to reduce HIV infection by about 81%, 5 but this is most effective within 1 to 2 hours of exposure and not more than 72 hours after exposure. 1 Postexposure prophylaxis is hence a very important requirement in health care settings where workers are often exposed to body fluids. While published data exist on the outcomes of PEP programs in a few countries in the West-Africa subregion including Nigeria 6–8 and La Cote d’Ivoire, 9 no publication is available on Ghana, although PEP services in Ghana started in 2004.
In December 2003, Ghana, with a population of approximately 25 million and an estimated HIV adult sero-prevalence of 1.7% to 2.2% (425 000 patients living with HIV), 10 started providing antiretroviral treatment (ART) to persons living with HIV/AIDS (PLWHA). 11 Although data specific to HIV prevalence among hospitalized patients are limited, it is estimated to be higher than that of the general population. Ghana, being a resource-constrained country, is faced with numerous economic challenges and thus the provision of requisite universal precautions necessary to protect HCWs comprehensively remains a goal being pursued. As part of a scaling up exercise to extend ART to PLWHA in all parts of the country by the National AIDS/STI Control Program (NACP) and in conformity with the “3 by 5” policy of the WHO, guidelines on the use of PEP were developed to ensure the provision of PEP in all sites that offer clinical care to patients living with HIV. 11 The present study was therefore undertaken to provide information on the provision of PEP in Ghana using information from 1 of the 4 pilot sites for ART delivery in Ghana—the Korle Bu Teaching Hospital (KBTH), an urban government premier teaching hospital in Accra. Data collected between January 2005 and December 2010 were analyzed to give the Ghana example of a PEP service in a resource-constrained setting and the effectives of an implemented risk assessment system (RAS) in reducing the rate of HIV sero-conversion.
Methods
The study was a retrospective cohort study. The cohort was assembled from historical records on HCWs and health care students (HCSs) who reported exposure and requested for PEP at the Pharmacy Department of the KBTH between the period of January 2005 and December 2010. The study involved giving each reported exposed HCW/HCS a unique study ID based on the hospital’s staff registry or student’s identification list, which ensures that any multiple reported exposure is detected and captured appropriately. The study protocol was approved by the Ethical and Protocol Review Committee of the University of Ghana Medical School (MS-Et/M.6-P.5.3/2009-10).
Setting
The Korle Bu Teaching Hospital started providing care to HIV-infected patients in December 2003. Software developed by Family Health International was used to capture data on patients and to monitor the use of the ARV drugs. Guidelines on PEP service was developed by the institution based on the recommendations of the US Centers for Disease Control and Prevention (CDC). 1 A team of health care professionals made up of medical and pharmacy personnel were charged with the responsibility of providing the PEP service and capturing data on HCWs/HCSs who report for PEP. Posters advertising the service and listing the mobile phone numbers of physicians and pharmacists in the PEP team were posted at all clinical departments of the hospital. Approximately 1930 HCWs and 1400 HCSs per year provide services at the hospital, and an initial sensitization exercise was done to ensure awareness among the staff. Subsequently all incoming students and interns were orientated on the PEP service.
Exposure Management
The procedure for managing PEP at the KBTH is as follows: any exposed HCW/HCS reports the incident to the immediate supervisor who directs the staff to a physician listed in the PEP team. The physician examines the exposure, determines the HIV status of the staff and the source patient, and then directs the exposed staff to a listed pharmacist who provides counseling and PEP medications, if considered necessary. Figure 1 shows a flowchart of the management procedures.
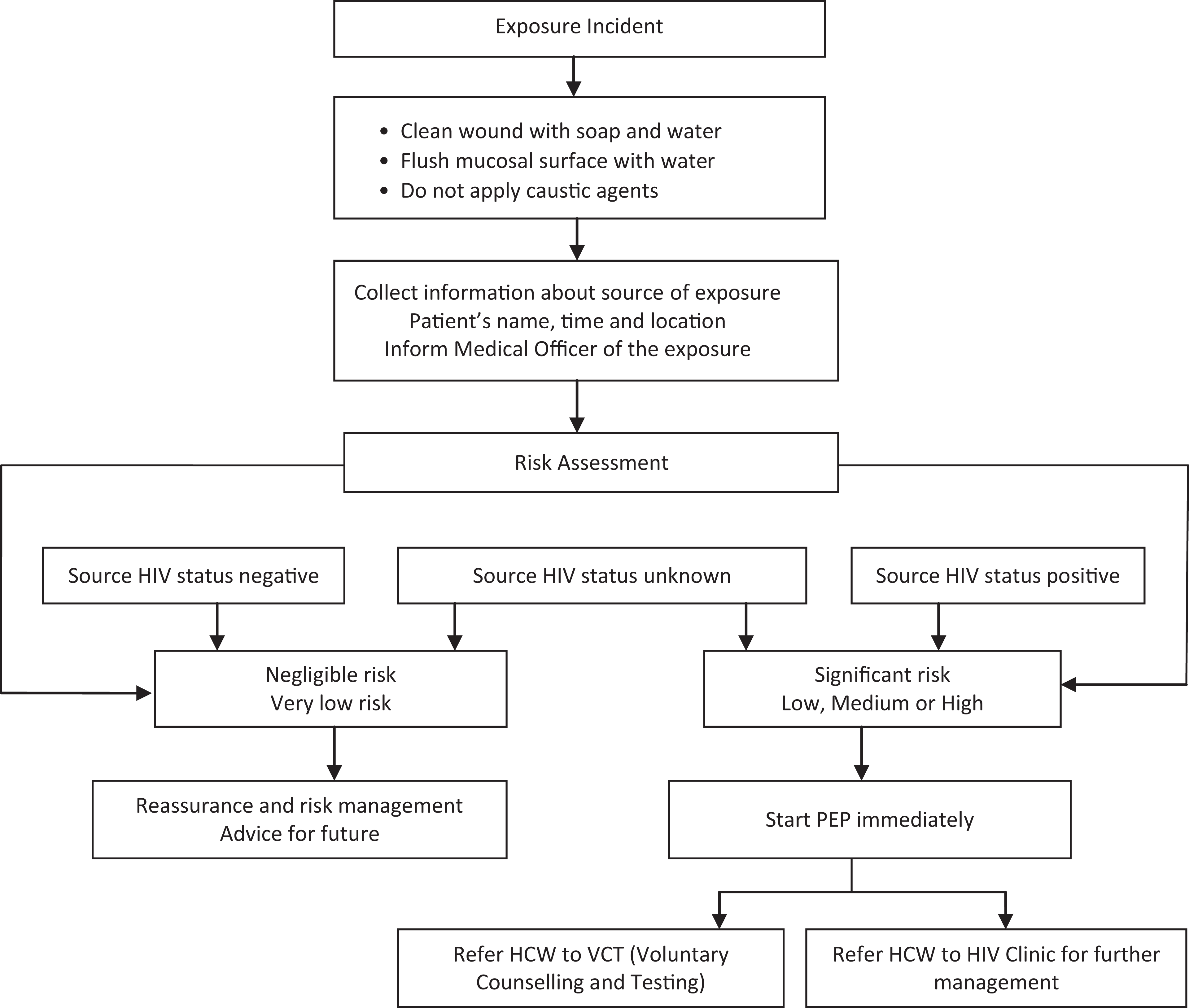
Occupational postexposure prophylaxis flowchart.
Risk Assessment System
In determining whether to administer PEP, a risk assessment of the exposure is carried out using both the 2001 CDC occupational exposure guidelines 1 and the local policy in place at the KBTH. 11 This system considers the risk versus benefit analysis for every individual presenting following an exposure, and the decision to initiate PEP is made on a case-to-case basis. Exposures classified as high risk were administered 28 days of expanded PEP regimen, moderate risk exposures were also administered 28 days of expanded PEP regimen, and low risks were administered 3 days of stat dose until the source patient’s HIV status is determined. The stat dose given in all cases was a 3-day course of zidovudine (ZDV) + lamivudine (3TC) as dual therapy or ZDV + 3TC + lopinavir/ritonavir (LPV/r) as triple therapy. The same medications were continued up to 28 days as expanded PEP (in both dual and triple therapy as the case may be), based on the confirmation of the source patient’s HIV status being positive or on one-to-one case decision by the risk assessment team in charge of using the RAS.
Data Collection
The data analyzed were collected from January 2005 to December 2010, a period of 72 months. A data collection instrument was developed in-house to capture the details of exposed HCW/HCS, the source of exposure, laboratory findings, and risk assessment, which is based on the details of the event, time of event, reporting time, and type of exposure. Additional information collected included the type of PEP medication regimen received by the exposed HCW/HCS in addition to the recording of adverse events (AEs) and the level of adherence to the treatment regimen over a total of 3 days/28 days. Data on HIV status as an outcome of PEP provision were also collected during follow-up testing schedules of 6 weeks, 3 months, and 6 months after PEP administration. During follow-up visits, data were collected on reported AEs and adherence to treatment regimen on exposed HCWs/HCSs but will be discussed in a subsequent article.
Statistical Analysis
All statistical analyses were performed using SPSS Version 19.0 (IBM, Armonk, New York). Incidence rates were determined for the various categories of HCWs and HCSs. Person time as 100 person-years (100 P-Y) was calculated using the assumption that each HCW and HCS was available all-year round over the study period. Risk of exposure to injury warranting PEP intervention was calculated for each category of HCW, with nurses as the reference profession. One-way analysis of variance was also performed to assess trends in proportions of exposures and behavioral types over the study period of January 2005 to December 2010.
Results
Characteristics and Rates of HCWs/HCSs Reporting Exposures
During the study period, 260 HCWs and 35 HCSs reported a total of 295 occupational exposures without any multiple report (a total of 1930 HCWs and 1400 HCSs works in the KBTH yearly). The median age of exposed HCWs and HCSs was 28 years and 21.5 years, respectively. Of the exposed HCWs/HCSs, 175 (59.3%) were female (Table 1). Five departments, namely, child health, obstetrics and gynecology, surgery, medicine, and surgical/Medical Emergency and Trauma accounted for 71.5% of reported exposures (Table 1). Department of surgery recorded the highest reported number of exposures (53 HCWs, 18.0%) of which the majority (48 reports, 90.1%) were due to needlestick injuries. In all, the 5 departments also recorded the highest number of needlestick injuries (183 reports, 62.0%).
Characteristics of 295 HCWs/HCSs Reporting Occupational Exposures at KBTH in Accra, Ghana.

Abbreviations: HCWs, health care workers; HCSs, health care students; KBTH, Korle-Bu Teaching Hospital; SME, surgical/medical emergency; OPD, outpatient department.
aColumn percentages within rows.
bRow percentages within supercolumns.
The greatest number of exposures was reported among nurses (116 reports, 39.3% of all reports) followed by physicians (95 reports, 32.2%). However, ward attendants reported the highest incidence rate of 6.46 of 100 P-Y (Table 2). Ward attendants were 4 times more likely to be exposed than nurses (relative risk [RR], 4.01, 95% confidence interval CI 2.90-5.55;
Incidence of Exposures Per 100 P-Y of HCWs/HCSs at KBTH in Accra, Ghana.
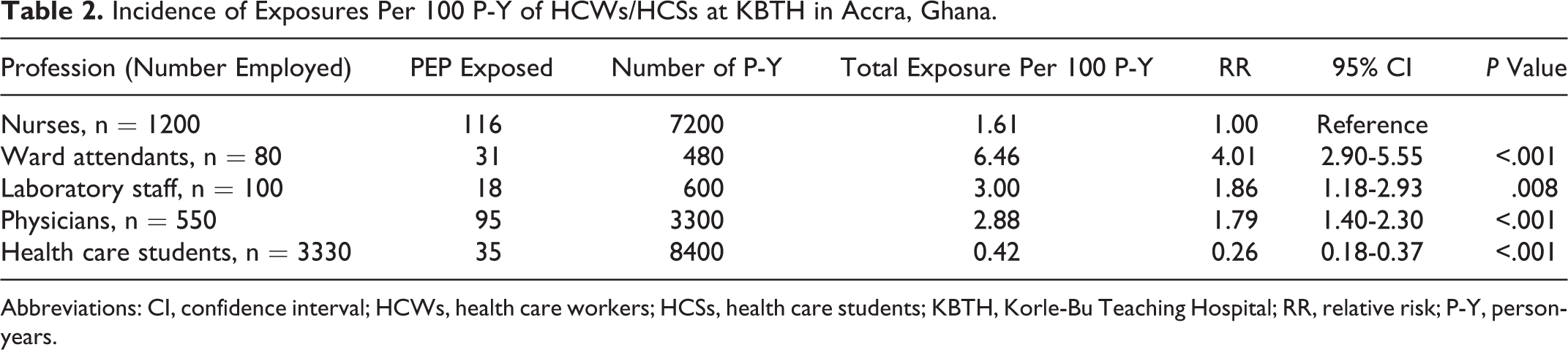
Abbreviations: CI, confidence interval; HCWs, health care workers; HCSs, health care students; KBTH, Korle-Bu Teaching Hospital; RR, relative risk; P-Y, person-years.
Follow-up telephone calls to all exposed HCWs/HCSs in the cohort administered PEP (289 individuals) during the 3 days/28 days treatment schedule showed a complete adherence to medication of 77.2% (n = 183), with 37 lost to follow-up.
Exposure and Exposure Source Description
Of the total 295 exposures, 271 (91.9%) were percutaneous and 24 (8.1%) were mucocutaneous (Table 1). A total of 277 (93.9%) exposures were due to handling/after a procedure with a sharp object and 10 (3.9%) exposures occurred from blood splash. Percutaneous exposures were the most common exposure type across all job categories. Of the 271 percutaneous exposures, 270 (99.6%) were injuries from either needlesticks (246 reports) or cannulae (24 reports). Trend analysis showed that over the 6-year period of the study (2005-2010), percutaneous exposure decreased significantly (

Trends over time in exposure types and source of HIV status at KBTH in Accra, Ghana. KBTH indicates Korle-Bu Teaching Hospital.
Of the 295 exposure source patients, it was possible to conduct HIV tests on 247. Forty-eight exposure source patients were unavailable or the data on their HIV status could not be ascertained. Of those tested (n = 247), 88 were HIV positive, giving the HIV-sero-positivity among the exposure source tested in this study as 35.6%. Trend analysis showed that the proportion of exposure sources with HIV-positive status did not change significantly (
Risk Assessment System and Exposure Management
Using the RAS, the clinical assessment team graded 65 (22.0%) cases of the 295 reported exposures as being of high-risk exposure, 65 (22.0%) as medium-risk exposures, and 165 (55.9%) as low-risk exposures (Figure 3). Laboratory investigations later confirmed that as many as 57 (87.7%) of the graded 65 high-risk cases were cases with exposure source being HIV positive, 1 (1.5%) exposure source was HIV negative, and 7 (10.8%) were of unknown HIV status because of unknown exposure sources (Figure 3). A majority (55 reports, 84.6%) of the high-risk exposures were percutaneous (Table 1). The incidence of high-risk exposures was 0.33 of 100 P-Y (n = 65); 60.0% occurred during a procedure of disposing of a needle and 24.6% occurred during a cannula insertion. The incidence of high-risk exposures did not change significantly (

The postexposure prophylaxis (PEP) expanded regimen use: clinical decisions.
A total of 289 exposures were administered PEP, with 224 exposures receiving dual therapy and 64 receiving triple therapy. Of the 65 exposures who received triple therapy, 64 belonged to the “high”-risk category, while 64 of 65 and 160 of 165 exposures belonged to the “medium”- and “low”-risk categories, respectively. Six exposures (2.0%) were not administered any medication because the reportage was more than 72 hours after the incident. The RAS, however, categorized 5 of these exposures as low risk and 1 case as medium risk.
Adherence to Follow-Up Testing
Adherence to follow-up testing for HIV status in the 289 patients administered PEP was 87.2% (252 exposures) at the first follow-up testing schedule of 6 weeks and 75.1% (217 exposures) during the second follow-up testing schedule of 3 months. The last follow-up testing schedule of 6 months recorded 62.6% (181) of the exposed HCWs/HCSs. Further analysis indicated that there was no statistical difference (
Discussion
In this study, 295 occupational exposures to HIV occurred in 260 HCWs and 35 HCSs with each exposed HCW/HCS being recorded as reporting once for the PEP service during the study period. It is, however, unknown whether after the 6-month period each person in the cohort had any further occupational exposure to HIV or sought PEP elsewhere. This information is clearly beyond the scope of this study.
Despite the 62.6% level of follow-up testing adherence after the 6th month, which compares with other similar studies, 12,13 results from this study indicate that the PEP service provided in the KBTH as a policy intervention over the period under review was beneficial to HCWs and students who were exposed occupationally as none of the exposed followed-up HCW/HCS sero-converted to HIV positive. Additionally, it offered the platform for exposed HCWs/HCSs to gain confidence in the availability of adequate, effective, and efficient means of addressing occupational exposures to HIV, which otherwise would have lead to tremendous fear, anxiety, and stress, which are likely to have negative effect on them and their families. The RAS deployed as part of PEP in the teaching hospital seems to have led to the effectiveness of the service. This finding, while is consistent with similar studies in Nigeria, 7 India, 14 and Kenya, 15 also reemphasizes the use of dual/triple triple therapy over monotherapy (ZDV) as the latter has being implicated in 21 instances of PEP failure to prevent HIV sero-conversion in the United States. 16 The results of this study also indicated that of the 65 cases classified as high risk, 57 (87.9%) of the source patients turned out to be HIV positive, with 7 (10.8%) of unknown source, further confirming the real-life usefulness of the triaging system. This RAS is a necessary complementary tool in resource-constrained settings where there is limited availability of HIV rapid diagnostic test kit at all service delivery points meaning that at the site of injury there may be no rapid diagnostic test to tell whether the source patient was HIV positive or not. This, coupled with the time lapse between injury and the availability of the results of laboratory investigation of the HIV status of the source patient (usually more than 72 hours—the time limit for PEP initiation), makes it imperative to have a tool, like the RAS, to be used to complement the decision of exposed HCWs/HCSs eligible for PEP and also to decide on those who receive short-course versus expanded PEP.
Using the RAS being implemented in the KBTH, 6 (2.03%) of the exposures reported outside the 72-hour window during which PEP is known to be effective and were not therefore administered PEP. These 6 HCWs were, however, counseled and advised to check their status and that of the source patient as appropriate and feasible. Although no sero-conversion has been reported by these exposed individuals to the hospital authorities, this study being a retrospective one was unable to definitely rule out any sequelae in these due to confidentiality and ethical considerations and issues. This observation is worrying and calls for increased education and advocacy efforts of all HCWs and HCSs to ensure that every single one of them is aware of the provision of PEP in the teaching hospital and to seek the intervention within the stipulated time.
Although results from other studies 17–19 have shown that interns and students are more at risk than senior staff members, results from this study indicate that HCSs appeared to be the group with the least risk. This information is, however, limited by not capturing the various grades of the HCWs. However, one of the most interesting findings of this study is the relative risk for PEP among the different categories of HCWs/HCSs. Although nurses, by simple count, reported more exposures than the other HCWs, ward attendants recorded the highest incidence rate of 6.46 of 100 P-Y, which was far higher than the overall incidence rate of 1.48 of 100 P-Y for all HCWs/HCSs. In addition, ward attendants were found to be 4 times at greater risk of getting an injury requiring PEP than nurses. Although reasons which can be advanced for this observation include lack of tools and facilities, huge numbers of patients, pressure from other health workers, naivety, inadequate education, and poor adherence to procedures and safety practices which are counterproductive to the delivery of an effective health service, this delicate group of HCWs should in future be targeted for specific education on universal requirements for protection. Future studies must explore the reasons for this observation and corrected.
The observation that 5 departments, namely, departments of surgery, child health, surgical/medical emergency and trauma, obstetrics and gynaecology, and internal medicine in the reported order recorded more than 70.0% of the total reported exposures needs to be studied further and the reasons for departmental differences need to be ascertained and addressed.
Results from this study indicated that as many as 84.6% of the high-risk graded exposures were due to injuries from needlestick or cannula, This result, although similar to other PEP studies, 17,18,20 underlines the need for the use of safer medical devices (eg, needleless systems and sharps with engineered sharps-injury protections) to reduce the occurrence of especially high risk to contaminated sharps. In fact, certain clinical practices such as recapping used needles have been documented to be related more to the likelihood of needlestick injuries and thus the practices have been condemned. 21–23 This preventive measure, however, poses a challenge in terms of cost on the already resource-constrained health care delivery system in Ghana, which nevertheless must be addressed to reduce health care occupational injuries.
Results from the study also show that a vast majority (91.9%) of the reported exposures during the entire study period were percutaneous. However, trend analysis revealed that the proportion of percutaneous injuries reduced significantly over the 6-year period, while mucocutaneous injuries rose steadily although the relative numbers remained below 20% of all exposures. This may be due to awareness by HCWs/HCSs in reporting all types of exposures including mucocutaneous exposures, which were previously overlooked because of intact skin. Similar trend analysis did not point to any decreasing number of retropositive source patients or high-risk exposures, which could have reflected the steady decrease in the HIV prevalence rate from an earlier 3.7% in 2000 to 1.9 in 2010 but which has moved up to 2.1 in the last sentinel survey report on Ghana. 10
Notwithstanding the significant findings and the particular usefulness of the retrospective type of study design for occupational exposures, in terms of less time and cost-effectiveness, a possible limitation is the absence of data on the number of years of experience and grade of the various categories of the exposed HCWs, which may be a potential confounding factor. Additional possible limitation of the current study is the number or proportion of HCW who did not seek care after PEP. Although the present study did not actually examine that, it will be reasonable to conduct such a survey as soon as possible to complement the outcome of the present study. A simple survey to assess the knowledge and utilization of PEP among HCW at the KBTH should suffice. In addition, the inability to capture the viral hepatitis status of exposed HCW/HCS and that of source patients limited the provision of PEP for occupational exposure to anti-HIV medicines only. A possible reason for this is the challenge of paying out of pocket for hepatitis screening. However, all exposed HCWs and HCSs were encouraged to check their hepatitis status, and if negative, hepatitis vaccine was recommended. Furthermore, the study was based in one, albeit in major, institution in Ghana, and the findings cannot be generalized to the whole country. Although the KBTH, which is a tertiary referral hospital, has the largest number of HCWs/HCSs in Ghana and also receives referrals for PEP from other hospital that do not offer PEP services, a more specific, purposive, and well-funded survey is required to obtain the national picture. Data available, which is collated by the National AIDS and STI Control Program of Ghana (NACP), included only the number of exposures and types of ART used. An overview of PEP service nationwide would provide useful findings that could be used to guide a national policy on the management of occupational and nonoccupational exposures to body fluids.
Conclusions
The present study shows that the RAS as a complement to the standard HIV rapid diagnostic kit at the KBTH for providing PEP is effective in identifying and classifying high-risk exposures for appropriate management. Of the 289 exposures of HCWs/HCSs who were provided PEP, only 181 (62.6%) adhered completely to the follow-up testing schedule, and none sero-converted at the last test after 6 months. The 289 exposed HCWs/HCSs reported for PEP within 72 hours of exposure and only 6 (2%) reported after more than 72 hours of exposure. The study also suggests that despite resource constrains in Ghana, PEP services should continue to be encouraged for all injured HCWs/HCSs. Efforts should be made to reduce underreporting, and targeted education should be implemented for ward attendants and other HCWs especially on the avoidance of percutaneous type injuries. The introduction of safer medical devices for procedures could also help avoid some of the injuries requiring a PEP service.
Footnotes
Authors’ Note
The authors RAT, ETN, ML, and PAN worked in the conception, study design, and the final article composition. ML, ETN, AMT, HGML, and ANOD contributed to the methods, results, and its continuous critical review. RAT, ETN, AMT, PAN, and ANOD worked on the data analysis, discussion, and revising it critically. RAT and ETN are the cofirst authors. All the authors read and approved the final manuscript.
Acknowledgments
Francisca Zigah-Hama and Henry Obuobi helped in the data collection and database capture. Charles Ofei Palm assisted in editing and research advice. Dr Nii Akwei Addo, the Country Manager of the National Aids Control Program supported the establishment of the PEP service. The PEP Team worked together to generate the information on the utilization of the PEP service, and exposed health care workers and students availed themselves willingly when contacted for information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Bert Leufkens and Aukje Mantel-Teeuwisse receive no direct funding or donations from private parties, including pharma industry. Research funding from public–private partnerships, that is, IMI and TI Pharma (![]() ), has been accepted under the condition that no company-specific product or company-related study is conducted. They have received unrestricted research funding from public sources, ie the Netherlands Organisation for Health Research and Development (ZonMW), the Dutch Health Care Insurance Board (CVZ), the EU 7th Framework Program (FP7), the Dutch Medicines Evaluation Board (MEB), and the Dutch Ministry of Health.
), has been accepted under the condition that no company-specific product or company-related study is conducted. They have received unrestricted research funding from public sources, ie the Netherlands Organisation for Health Research and Development (ZonMW), the Dutch Health Care Insurance Board (CVZ), the EU 7th Framework Program (FP7), the Dutch Medicines Evaluation Board (MEB), and the Dutch Ministry of Health.
