Abstract
The impact of highly active antiretroviral therapy (HAART) on the epidemiology of AIDS-associated histoplasmosis in the past decade is poorly defined. Among 100 patients with histoplasmosis in an endemic region between 2000 and 2009, 42 patients were immunocompetent, 32 were infected with HIV, and 26 were non-HIV-immunocompromised patients. The percentage with HIV decreased 67% in 2000-2001 to 18% in 2008-2009 (P = .004), while the proportion of non-HIV immunocompromised patients increased, 8% to 41% (P = .14). Histoplasma antigen was the most sensitive test (73%), whereas potassium hydroxide examination of clinical specimens was the least sensitive test (33%) in all 3 groups. Bronchoalveloar fluid culture (74%) had the highest yield among the cultures. The relapse rate was higher in HIV-infected patients compared to the other 2 groups (P = .04). The epidemiology of histoplasmosis in our endemic area has changed during the era of HAART. Organ transplantation and increasing use of immunosuppressive agents for chronic inflammatory conditions in non-HIV patients now account for most of the cases of histoplasmosis.
Introduction
Histoplasmosis is an endemic dimorphic fungus that is prevalent in the Ohio and Mississippi river valleys including Kentucky. 1,2 The incidence rate of histoplasmosis in Kentucky has ranged from 1.0 to 1.7 per 100 000 person-years from 2000 to 2008. 3,4 There were no documented outbreaks of histoplasmosis in Kentucky within the study period. Early in the HIV epidemic before the availability of highly active antiretroviral therapy (HAART), histoplasmosis occurred in 5% to 27% of the patients with AIDS living in endemic areas of the United States. 1 In this study, we examined the impact on the epidemiology of histoplasmosis by HAART. Clinical data from individuals with histoplasmosis and treated at the University of Kentucky Medical Center from 2000 to 2009 were compiled. The data were used to compare clinical and outcome parameters among patients with HIV infection, non-HIV-immunocompromised conditions, and those considered immunocompetent.
Patients and Methods
The study was a retrospective chart review conducted at the University of Kentucky Medical Center, a tertiary care center serving patients from Central and Eastern Kentucky and was approved by the University of Kentucky institutional review board. All patients diagnosed with pulmonary or disseminated histoplasmosis from January 1, 2000, to December 31, 2009, were included in this study. Information collected included demographic patient characteristics, comorbidities, immune status, clinical manifestations, date and duration of hospital stay, intensive care unit admission, laboratory results, radiographic studies, microbiology results, histopathology results, antifungal therapy, mortality, and recurrence.
Cases were categorized as immunocompetent, HIV, and non-HIV immunocompromised. Progressive disseminated histoplasmosis (PDH) was defined as the presence of clinical, laboratory (positive culture or histopathology), or imaging evidence of extrapulmonary involvement. The diagnosis of pulmonary histoplasmosis required respiratory symptoms and pulmonary radiographs or computerized tomography that demonstrated infiltrates or mediastinal lymphadenopathy in the absence of PDH. Positive urinary Histoplasma antigen (UHA; preformed by Histoplasmosis Reference Laboratory, Indiana University in 2000-2001 and MiraVista Diagnosis Laboratory from 2002 to 2009) was defined as detectable antigen. Positive serology was defined as either H or M precipitin bands by immunodiffusion with complement fixation antibodies titers of ≥1:8.
Descriptive statistics included medians and interquartile range (IQR) for continuous variables, counts, and percentages for categorical variables. The chi-square test was used to evaluate associations between categorical variables and the Kruskal-Wallis tests for differences in medians across continuous variables. Logistic regression was used to test for a linear change in the proportion of patients with histoplasmosis depending on their immune status group throughout the study period. The 10-year study period was divided into five 2-year intervals (2000-2001, 2002-2003, 2004-2005, 2006-2007, and 2008-2009). Prism 5 (GraphPad Software Inc, La Jolla, California) and JMP (version 9.0, SAS Institute Inc, Cary, North Carolina) were used for statistical analysis. The level of significance for statistical testing was defined as P < .05 (2 sided) unless otherwise specified.
Results
One hundred patients with newly diagnosed symptomatic histoplasmosis from 2000 to 2009 were identified. The incidence rate of histoplasmosis ranged from 0.26/1000 hospital admissions in the year 2000-2001 to 0.32/1000 hospital admissions in 2008-2009. In all, 42 patients were immunocompetent, 32 were infected with HIV, and 26 were non-HIV immunocompromised (Table 1). There was a linear decrease in the proportion of HIV-infected patients with histoplasmosis from 67% to 18% (P = .004) and a linear increase in non-HIV-immunocompromised patients from 8% to 41% (P = .14) between 2000 to 2001 and 2008 to 2009 intervals of the study.
Number and Percentage of Cases of Newly Diagnosed Symptomatic Histoplasmosis by Immune Status.

a P value denotes a 1 degree of freedom test for linear trend using logistic regression.
The median age of all study patients was 43 years, 74% of the patients were men, and 79% were white (Table 2). Patients with HIV infection were more likely to be men as compared to the other 2 groups (P = .03). White was the predominant race in all 3 groups, with African American and Hispanic representing 22% each in the HIV group.
Demographic, Clinical, and Laboratory Characteristics and Outcomes of Patients with Histoplasmosis by Immune Grouping.

Abbreviations: Non-HIV, non-HIV immunocompromised; IQR, interquartile range; COPD, chronic obstructive pulmonary disease; CNS, central nervous system; WBC, white blood cells; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; CF, complement fixation, positive titer 1:8 or higher to yeast or mycelia antigen; ID, immunodiffusion, positive H, M, or both bands; BAL, bronchoalveolar lavarge; GMS, Grocotts methenamine silver; KOH, potassium hydroxide.
a Data are collected when available, shown as number (%).
b Source for other cultures included pleural fluid, peritoneal fluid, synovial fluid, CSF, skin, sinus, vocal cord, tongue, pharyngeal, intestine, liver, lymph node, kidney, bone marrow, brain.
c P value comparing immuncompetent and non-HIV.
The 26 non-HIV-immunocompromised patients included kidney transplant (7), rheumatoid arthritis (6), solid organ malignancy (4), leukemia (3), lymphoma (2), autoimmune disease (2), heart transplantation (1), and hypogammaglobulinemia (1). In all, 16 (62%) patients were on T-cell suppressive medication and 12 (44%) were receiving at least 20 mg of prednisone daily. Three patients had at least 2 doses of tumor necrosis factor-α (TNF-α) inhibitor. Immunocompetent patients with histoplasmosis were more likely to have chronic obstructive pulmonary disease compared to HIV and non-HIV-immunocompromised patients (P = .01).
Immunocompetent patients were more likely to have pulmonary histoplasmosis (P = .0002), and HIV-infected patients were more likely to have disseminated histoplasmosis (P = <.001). Overall, respiratory symptoms were the most common presentation (80%) followed by fever (67%) and gastrointestinal symptoms (34%). Of all the patients, 13% (12 of 96) had central nervous system (CNS) symptoms, but only 4 immunocompromised patients (2 with HIV and 2 without HIV) met the criteria for probable CNS involvement (ie, CNS symptoms, positive UHA, abnormal cerebrospinal fluid [CSF] analysis, and imaging studies but negative CSF mycological culture). Gastrointestinal symptoms, leukopenia, thrombocytopenia, and abnormal liver function tests were more common in HIV-infected patients, whereas weight loss of more than 5 lbs and anemia (hemoglobin less than 10 g/dL) were more common in non-HIV-immunocompromised patients.
Urine Histoplasma antigen was the most ordered diagnostic test, followed by serum fungal serology and blood isolator culture. The UHA was positive in 73% (63 of 86) of all the study patients. No blood or bronchoalveolar lavarge (BAL) Histoplasma antigen tests were ordered. The UHA was more likely to be positive in HIV-infected patients (97%) than in non-HIV-immunocompromised patients (70%) and immunocompetent patients (56%; P = .001). Anti-Histoplasma antibodies were detected in 67% of the study patients with no apparent difference among the 3 groups (P = .69).
Positive results were reported from pathology (64%), culture (52%), Grocotts methenamine silver stain (46%), and potassium hydroxide (KOH) smear (33%). The BAL fluid analysis was performed in 43% of the study patients and was diagnostic in 74% (32 of 43). Histoplasma fungemia was diagnosed in 48% of the study patients, including 25% of immunocompetent patients, 50% of HIV-infected patients, and 67% of non-HIV-immunocompromised patients (P = .12). Among 25 patients with fungemia, 96% (22 of 23) had positive UHA and 62% (8 of 13) had positive serology.
Of the 32, 29 (94%) HIV-infected patients had AIDS at the time of diagnosis of histoplasmosis, with 26 (84%) having a CD4 count <150 cells/mm3. In all, 10 (31%) were diagnosed with HIV infection and histoplasmosis at the same time. The median CD4 count at the time of diagnosis of histoplasmosis was 33 cells/mm3 and the median viral load was 61 000 copies/mm3. No patients were identified who developed immune reconstitution inflammatory syndrome.
Of the 42 immunocompetent patients (34 had pulmonary histoplasmosis and 8 had disseminated histoplasmosis), 9 received amphotericin B deoxycholate or lipid formulations (AB) followed by itraconazole; 17 received itraconazole alone; and 16 did not receive antifungal treatment. The median treatment duration was 4.5 months. Four immunocompetent patients died in the hospital (Table 3). One patient relapsed 16 months after stopping the therapy. Among 32 HIV-infected patients (12 with pulmonary histoplasmosis and 20 with disseminated histoplasmosis), 18 received AB followed by itraconazole and 12 received itraconazole alone. Two patients did not get antifungal treatment and were discharged with hospice care. One HIV-infected patient died with sepsis due to disseminated histoplasmosis in the hospital (Table 3). Of the 16 who were followed for at least 1 year, 5 relapsed. Of the 26 non-HIV-immunocompromised patients with histoplasmosis (13 with pulmonary and 13 with disseminated histoplasmosis), 14 were treated with AB followed by itraconazole and 10 received itraconazole alone. Two patients received other triazoles; 1 patient received posaconazole due to allergic reaction with itraconazole; and the remaining 1 received voriconazole for CNS histoplamosis. The median treatment duration was 8.7 months. Three died in the hospital from histoplasmosis (Table 3), and 1 patient relapsed 18 months after the initial diagnosis, which was likely due to nonadherence to itraconazole.
Characteristics of Patients with In-House Mortality.
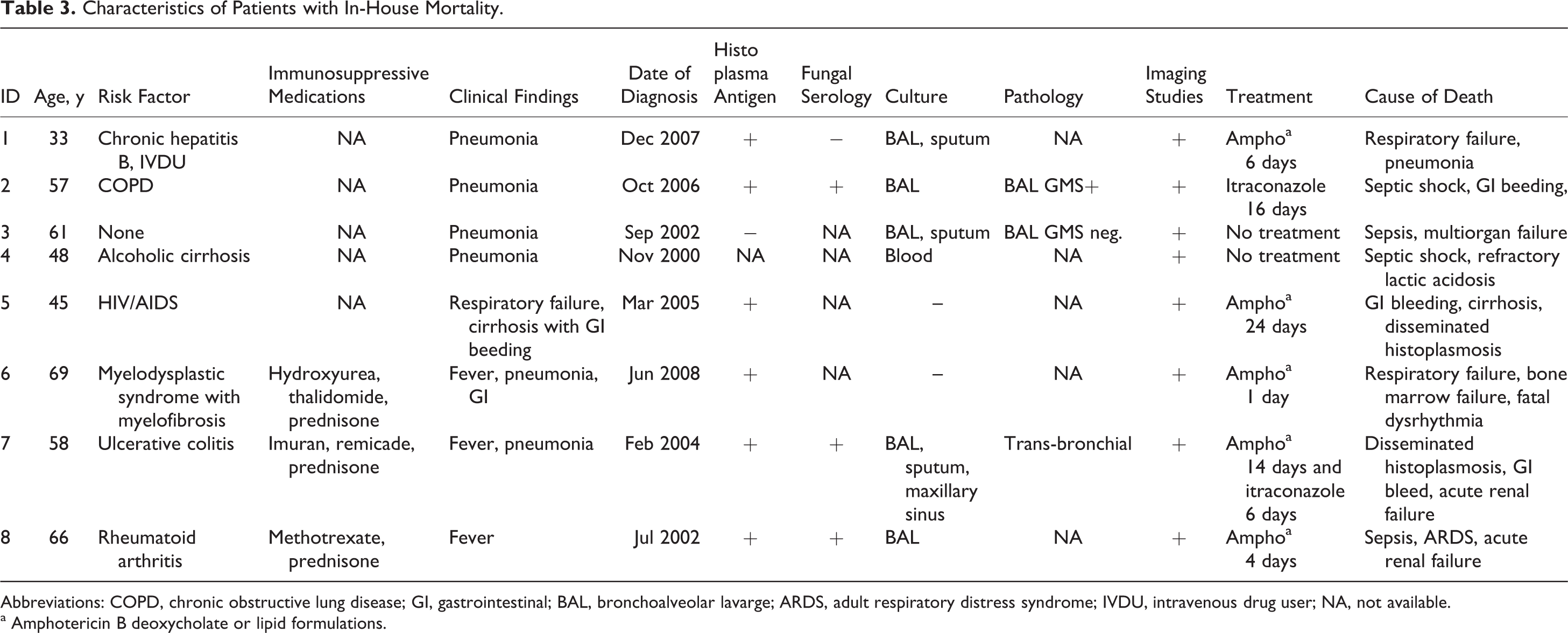
Abbreviations: COPD, chronic obstructive lung disease; GI, gastrointestinal; BAL, bronchoalveolar lavarge; ARDS, adult respiratory distress syndrome; IVDU, intravenous drug user; NA, not available.
a Amphotericin B deoxycholate or lipid formulations.
Discussion
The data demonstrate a decrease in the proportion of patients with HIV infection among all new cases of histoplasmosis in an endemic region over the past decade, while the incidence rate has been stable between 0.26 and 0.32/1000 hospital admissions in 2 consecutive years in our institution. The incidence rate of AIDS in Kentucky has also remained stable between 4.3 and 6.0 cases/100 000 person-years. 3,4 The decline is likely related to HAART and is consistent with declining incidence rates of opportunistic infections in patients with HIV infection. 5
On the other hand, it appears that there is a trend toward an increase in the proportion of non-HIV-immunocompromised patients among new cases of histoplasmosis. During the past decade, there have been 588 kidney transplants, 76 kidney–pancreas transplants, and 88 heart transplants at our institution; 7 kidney transplant recipients and 1 heart transplant recipient had histoplasmosis. In endemic areas, histoplasmosis has been the most common fungal infection in the patients receiving TNF-α inhibitor agents, 6 and in this study 5 patients with long-standing rheumatoid arthritis were identified, with 3 of them having received such agents.
Health care providers in our area were more likely to use noninvasive diagnostic tests such as UHA, fungal serology, or blood isolator compared to invasive tests such as tissue culture or biopsy. This led to many disseminated histoplasmosis infections being diagnosed by noninvasive diagnostic tests. Histoplasma urinary antigen results are usually available within 3 to 5 working days. In previous studies, the sensitivity of urine antigen test in patients with AIDS and disseminated histoplasmosis was reported as high as 94.6%. 7 In our study, Histoplasma urinary antigen was sensitive in detecting disease in immunocompromised patients (73% overall, 97% in HIV, and 70% of non-HIV-immunocompromised yielding positive urinary antigen test results) but was not as effective in detecting disease in immunocompetent hosts, where only 56% had positive results.
Serolology can be negative in up to 50% of immunosuppressed patients, especially those with HIV/AIDS. 8 In our study, it was positive in 67% to 72% of HIV-infected patients, 59% in non-HIV-compromised patients, and 68% to 71% in immunocompetent patients. It is comparable to other series of study, positive in 69% of immunocompromised patients, 85% in immunocompetent patients. 9
Among invasive tests, BAL and KOH smear and culture were the most ordered tests and positive for 46% and 74%, respectively. The yield for KOH smear appeared to depend on the source of the specimen, as BAL fluid yield was the best at 46%. Bone marrow culture was the least performed invasive procedure.
In all, 63% of HIV-infected patients and 50% of non-HIV-immunocompromised patients had disseminated histoplasmosis; 2 HIV patients and 2 non-HIV immunocompromised patients had probable CNS histoplasmosis. The median CD4 count was 33 cells/mm3 at the time of histoplasmosis, which coincided with the ranges from 28 to 47 cells/mm3 in previous studies. 10
The relapse rate was more common in HIV-infected patients compared to the other 2 groups. It was associated with noncompliance of antifungal and HAART medication. Of the 7 patients who relapsed, 1 died of histoplasmosis, emphasizing the importance of adherence to therapy, careful follow-up with symptoms, and monitoring for increase in Histoplasma antigen.
Limitations in our study include it being a single-center retrospective study at a tertiary care referral center and the results may have been influenced by referral bias. In conclusion, the epidemiology of histoplasmosis in an endemic area has evolved in the era of HAART. Organ transplantation and increasing use of immunosuppressive agents for chronic inflammatory conditions in non-HIV patients now appear to be the underlying risk factor for most cases of histoplasmosis.
Footnotes
Acknowledgments
The authors thank Dr L. Joseph Wheat for his suggestions and review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
