Abstract
Background:
In India, a zidovudine-based regimen is preferred as the first-line drug treatment for HIV, despite high rates of drug toxicity. This study estimates the treatment costs for HIV.
Methods:
Eligible patients were enrolled from Antiretroviral Therapy Center, Christian Medical College, India. Baseline demographic and clinical characteristics, medical and nonmedical expenditure, and lost income were collected.
Results:
Of 41 patients enrolled and followed for 6 months, HIV treatment toxicity and opportunistic infections were reported by 12 (29%) and 13 (31.7%) patients, respectively. The median total costs, direct costs, and out-of-pocket expenditure were Indian rupees (INR) 9418 (US$181), 8727 (US$168), and 7157 (US$138), respectively. Diagnostic tests accounted for 58% of the expenses. HIV treatment accounted for 34% of the median income earned INR 21 000 (US$404). Expenditure for treatment with toxicity was 44% higher than without toxicity.
Conclusion:
Current treatment is associated with toxicity, increasing treatment costs and imposing a significant economic burden.
Introduction
India has the third-highest HIV burden in the world, with at least 2.5 million people living with HIV. The national prevalence of HIV in India is 0.36%. 1,2 A free national antiretroviral therapy (ART) program was initiated by the Government of India in 2004, and ART has been provided free of costs by 200 ART centers throughout India. 1 Antiretroviral therapy is being rolled out to HIV-infected individuals in the resource-limited settings of the public sector health care system as a combination ART (cART) regimen containing 2 nucleoside antiretroviral agents (nucleoside reverse transcriptase inhibitor [NRTI]) and either a nonnucleoside reverse transcriptase inhibitor (NNRTI) or a protease inhibitor (PI). The regimen used in the ART centers in India includes stavudine (d4T) or zidovudine (ZDV; depending on the hemoglobin level) and lamivudine (3TC) as the NRTI with the NNRTI, nevirapine (NVP).
Available data suggest that the toxicities of the ZDV or d4T backbone surpass the toxicities of other available nucleoside backbones. Approximately 20% to 30% of the patients started on d4T or ZDV regimen are reported to experience drug toxicity within the first 6 months of treatment. 3 –5 This toxicity necessitates additional clinic visits and laboratory tests to monitor toxicity, thus increasing costs.
A tenofovir (TDF)-based regimen is considered to be one of the least toxic regimens for HIV and appears to maintain high CD4 levels, suppress viral load, and support a good quality of life as compared to patients on d4T or ZDV regimens. 6 In resource-limited settings, a TDF-based regimen is typically not considered to be the first choice for ART because of its expense.
A cost-effectiveness analysis from both the government and patient perspective will compare the incremental costs and health benefits of a TDF regimen to the typical first-line d4T- or ZDV-based regimens. The analysis will determine whether the net health benefits of TDF are sufficiently large to justify net added costs or whether the toxicity avoided costs associated with TDF use offset its higher purchase price. As a preliminary step, this pilot study estimated the costs associated with the treatment of HIV for 6 months and compared the HIV care costs between patients with and those without drug-related toxicity.
Methods
Study Population
This study was conducted at the Government of India sponsored ART center at Christian Medical College (CMC), Vellore, Tamil Nadu, India. Adults more than 18 years of age with a confirmed diagnosis of HIV, eligible for initiation of cART based on the National AIDS Control Organisation of India guidelines 2009, willing to participate and follow-up for 6 months, and who provided written informed consent were enrolled in the study. Participants requiring hospitalization at the time of initiation of cART, patients with active tuberculosis (TB) and those who were on treatment for TB, patients with diabetes or neurological impairment, pregnant and breast-feeding women, and children were excluded from the study. Patients were followed for 6 months and interviewed monthly to elicit incurred costs and other study-related information. Treatment adherence was assessed using pill counts and patient interviews. The study was approved by the institutional review board and ethics committee at CMC, Vellore (IRB Min No. 7354, December 08, 2010).
Definition of Toxicity
Drug-related toxicity was assessed during all scheduled and unscheduled visits. Toxicity assessed in this study includes lactic acidosis (based on the levels of lactic acid >2 mmol/L), peripheral neuropathy, severe anemia (defined as hemoglobin levels <8 g/dL), and neutropenia (defined as absolute neutrophil count <1000 cells/ µL).
Cost Calculation
Cost data were collected from patient interviews and medical bills. Collection drew on established methods for economic analysis in clinical practice. 7 Direct medical costs included costs attributable to prehospital visits, hospital visits, medication, and diagnostics. Prehospital visit costs are costs incurred during visits (direct and indirect costs) to any medical facility other than CMC for treatment between the scheduled visits. If patients had any nonhospitalized visit to other hospitals for opportunistic infections, they were encouraged to save the bills; data were extracted from the bills during patients' next scheduled visit to CMC. Hospital visit costs include professional charges, nursing charges, bed costs, and any additional special costs associated with treatment. Drug costs include costs for cART (government procurement costs) and drugs for treating opportunistic infection and toxicity. Expenditures on radiological procedures and laboratory investigations were captured as diagnostic costs. Indirect costs for food, accommodation, and transport for the patient and caregiver were collected for every hospital visit. All costs were collected for inpatient, outpatient, and emergency visits. Cost data were collected in Indian rupees (INR) from patient interviews and cash receipts. The unit costs for every intervention, all diagnostics, and all hospital charges are predetermined annually by the hospital administration, depending on local, geographical, and economic factors.
Adherence to Treatment
Participants were requested to return the drug containers during the follow-up visit, and drug counts were verified to assess treatment compliance. Patients were also interviewed for the drug adherence.
Statistical Analysis
The pilot study included 50 participants to allow estimation of the mean cost with a 95% confidence interval of ±0.28 standard deviations (SDs). Analysis was done using statistical software SAS v9.2 (32; English). Means and proportions were computed to describe the baseline characteristics of individuals enrolled in the study. Socioeconomic status was assessed using the updated Kuppuswamy socioeconomic scale. 8 For overall costs and individual cost components, the median and interquartile ranges were computed. An exploratory analysis compared the baseline characteristics and costs for patients with and without toxicity.
Results
The study was conducted from February 2011 to November 2011. A total of 50 participants were enrolled into the study. Two participants were lost to follow-up. Among the remaining 48 participants, 7 had other complications requiring hospitalization (pregnancy, 1; adenocarcinoma, 1; and tuberculosis within the first month of enrollment, 5) and hence were withdrawn from the study. In all, 41 participants successfully completed the 6-month follow-up and were included in the analysis. The mean age of enrolled participants was 37 years (SD ± 9 years), with 21 (51%) males and 20 (49%) females. Of the enrolled patients, 35% were illiterate while the remaining 65% had formal education (Table 1). The population unemployment rate was 32%, and 76% of the employed people were unskilled laborers (Table 1). The mean body mass index at enrollment was 26 kg/m2 (SD, ±9.9), with a baseline CD4 of 176 cells/µL (SD, ±105 cells/mm3). At the time of enrollment, 22 (54%) patients were diagnosed with stage 1 disease (Table 1). Based on the baseline hemoglobin levels, ZDV-based regimen (ZDV 300 mg + 3TC 150 mg + NVP 200 mg) was started in 32 (78%) patients, while 9 (22%) were started on d4T regimen (d4T 30 mg + 3TC 150 mg + NVP 200 mg). Treatment adherence exceeded 90%.
Baseline Demographic and Clinical Characteristics of Participants Enrolled in the Study.

Abbreviations: BMI, body mass index; SD, standard deviation; WHO, World Health Organization.
a Comparison between patients with and without toxicity.
The median for total direct costs for overall care and treatment of HIV over 6 months was INR 8727 (US$168.40, US$1 = INR 52). Approximately 58% of the expenses were attributable to diagnostic tests, including radiological and laboratory investigations, followed by 33% of expenses attributable to drug costs and 6% to hospital visit/admission costs. The median indirect cost over 6 months was INR 720 (US$14). Total costs for treatment were approximately INR 9418 (US$181). Patient out-of-pocket expenditures for treatment were INR 7157 (US$138). Total income earned during this 6-month period was INR 21 000 (US$404). Approximately 34% of the money earned during this 6-month period was spent on HIV treatment (Table 2).
Median Costs with IQR for Treatment of HIV for a Period of 6 Months.
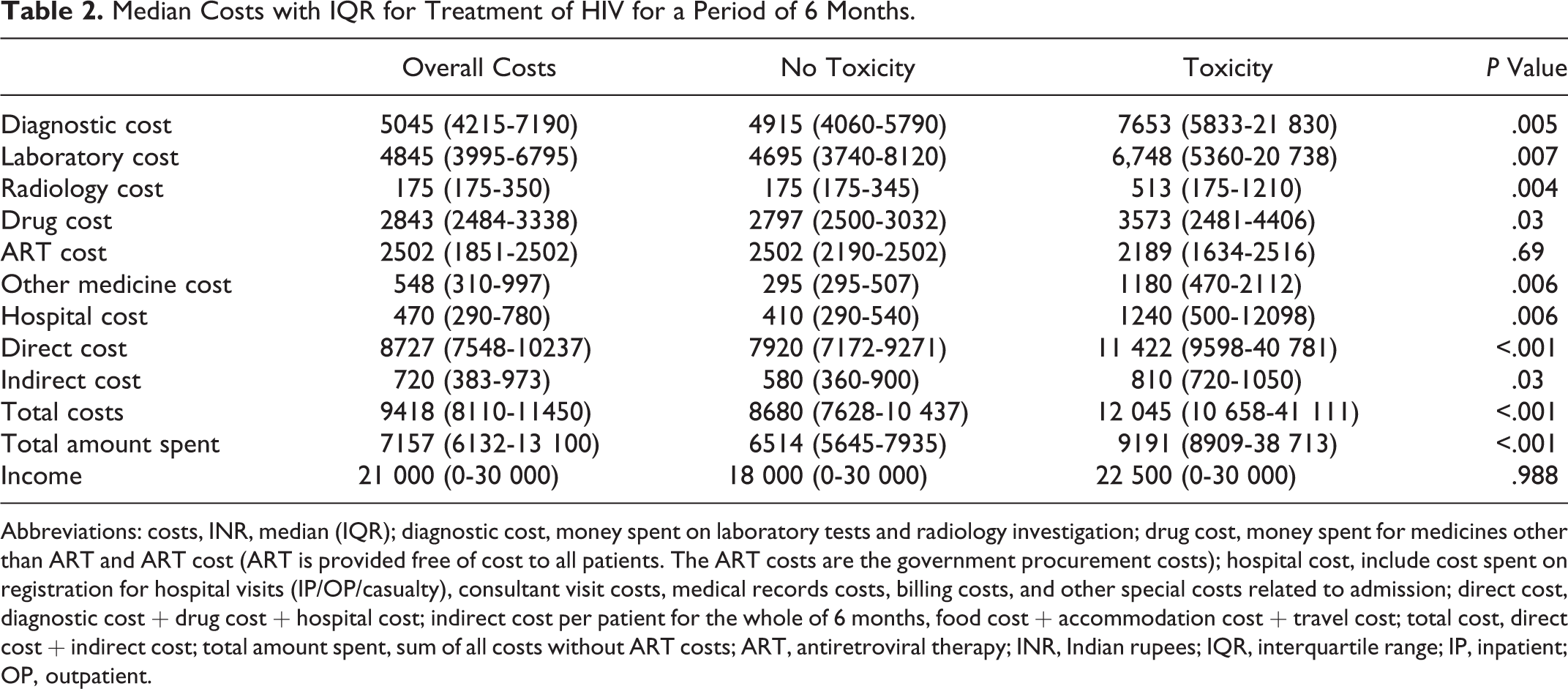
Abbreviations: costs, INR, median (IQR); diagnostic cost, money spent on laboratory tests and radiology investigation; drug cost, money spent for medicines other than ART and ART cost (ART is provided free of cost to all patients. The ART costs are the government procurement costs); hospital cost, include cost spent on registration for hospital visits (IP/OP/casualty), consultant visit costs, medical records costs, billing costs, and other special costs related to admission; direct cost, diagnostic cost + drug cost + hospital cost; indirect cost per patient for the whole of 6 months, food cost + accommodation cost + travel cost; total cost, direct cost + indirect cost; total amount spent, sum of all costs without ART costs; ART, antiretroviral therapy; INR, Indian rupees; IQR, interquartile range; IP, inpatient; OP, outpatient.
Of the 41 patients followed, 12 (29.3%) had drug-related side effects and required additional diagnostic tests for treatment. In all, 11 patients in the ZDV regimen and 1 patient in the d4T regimen had drug-related toxicity; 5 (12%) patients had severe anemia requiring hospitalization and blood transfusion, 4 (10%) patients had anemia treated with oral iron supplementation, 2 patients had skin rashes (5%), and 1 patient had both anemia and skin rashes. All patients were switched from current regimen to less toxic regimens. None of the patients had lactic acidosis, neutropenia, or peripheral neuropathy. Opportunistic infections were observed in 13 (31.7%) patients. Tuberculosis, pleural effusion, cryptosporidium infection, eczema, diarrhoea, herpes infection, and tuberculosis with lymphadenopathy each were diagnosed in 1 patient, while 3 patients had oral candida infection and 3 had lymphadenopathy.
Exploratory analysis comparing patients with and without toxicity showed that the 2 groups were similar with respect to all baseline characteristics (Table 1). Individual costs differed significantly between the 2 groups (Table 2). Patients with toxicity had to spend 44% more for toxicity treatment than did those without toxicity and 56% more on laboratory and radiology tests. Patients with toxicity averaged 8.2 visits (SD ± 2.0), approximately 50% more than the corresponding average of 5.5 visits for patients without toxicity (SD ± 2.0). The number of outpatient referral visits to the infectious disease clinic was 40% more in patients with toxicity (7.6 ± 2.6 days) than for those without toxicity (5.4 ± 2.3 days).
With treatment, the baseline mean CD4 level of 176 cells/µL (SD ± 105 cells/mm3) increased significantly (P < .0001) to 362 cells/mm3 (SD ± 213 cells/mm3) at the end of follow-up. This trend was similar in patients with and without toxicity.
Conclusion
Early and regular HIV treatment with the best possible regimen will help reduce the progression and transmission of disease. According to World Health Organization (WHO) recommendations, most countries provide TDF-based regimen as the first-line drug for HIV, since it has a very low drug-related toxicity rate compared to d4T- or ZDV-based regimen. 6 But in developing countries like India and other Asian and African countries, TDF is not considered to be part of the first-line regimen because of its expense. Despite the high rates of drug toxicity, ZDV is the drug of choice in these countries. This pilot study provides data supporting the contention that the ZDV-based regimen has a substantial rate of drug toxicity. This study also provided cost estimates for treatment of HIV among patients receiving the current regimen.
In this study, 29.3% of patients had drug-related toxicity, a result that is consistent with a previous study in this setting, which reported a toxicity rate of 27%. 5 This result is also consistent with other studies from other parts of the world, where toxicity rates range from 20% to 30%. 3,4 During the study period, the Indian guidelines recommended offering highly active antiretroviral therapy to all patients living with HIV with a CD4 count of less than 200 cells/mm3. Studies 9 have clearly demonstrated that initiating treatment with a CD4 count of less than 200 cells/mm3 increases the adverse drug reaction risk compared to patients with a CD4 count ≥200 cells/mm3. Currently the CD4 count cutoff for treatment in India is <350 cells/mm3. Anemia was the most commonly observed side effect in this study. Other defined side effects were not identified possibly due to the limited follow-up period.
The comparison between patients with and without toxicity is exploratory and has limited numbers. Despite this limitation, the study found that as expected, patients with toxicity paid more for treatment, and much of their expenditures were attributable to diagnostic tests. In addition, all other cost components for patients with toxicity were higher than the corresponding costs for patients without toxicity. Although the drugs are provided free of cost, the expenditures on treatment of drug-related toxicity and opportunistic infections are mostly paid by the patients. These costs are paid out of their income or by borrowing from others. This study estimated the proportion of income that patients spend on care related to HIV, excluding ART costs, and found this to be 34% of their earnings. As a result, these expenditures impose an additional significant economic burden not only on the patient but also on the community. Providing less toxic medications can lessen this burden and likely improve patient health and quality of life. Patients with less drug-related toxicity will have fewer hospital visits, decreasing indirect costs not only for the patient but also for caregivers accompanying the patient. To our knowledge, this study is the first study from India to estimate HIV care costs using individual-level itemized costs.
Studies have shown that good adherence to ART helps to improve CD4 levels and to decrease the viral load in patients with HIV. 10 –13 Our study also showed that CD4 levels improved during the 6 months of treatment. This improvement reflects high treatment adherence rates. Although similar outcomes were observed in patients with and without toxicity, it is very difficult to confirm this with the small sample size and the limited follow-up of only 6 months. This study focused on toxicity associated with ZDV and d4T. We were not able to capture other toxicities that arise after 6 months of treatment. Future studies with larger samples and a longer follow-up will provide a clearer longitudinal toxicity profile.
In resource-sufficient countries, WHO recommendations specify that the TDF-based regimens are the first-line treatment for patients with HIV. Despite the high toxicity levels (20%-30%), ZDV-based regimens are still preferred to the TDF-based regimen (less than 5%) in India since the ZDV-based regimens are presumed to be less expensive. However, compared to patients on the TDF regimen, patients on ZDV regimen spend more money for additional hospital visits and admissions, laboratory investigations, and other medications due to ART-induced toxicity. A proposed cost-effectiveness analysis that compares TDF-based regimen with ZDV will help inform deliberations regarding the need for a policy recommendation to switch first-line regimens from ZDV to TDF.
Footnotes
Authors’ Note
TVS, RK, CAW, OCA, and GK were involved in the conception and design of the study; TVS, RK, and DM were involved in the capturing of data; TVS, NT, MM, JTC, and JPM carried out data analysis and interpretation of the study data; TVS prepared the manuscript; and CAW, OCA, GK, JPM, JTC, MM, and NT helped in critical revision of the manuscript.
Acknowledgments
We thank ACTFID (ACC-CMC Trust for Infectious Diseases) ART Centre, Christian Medical College, Vellore, India, for conducting the study at the ART center. We also thank all the staff at the ART center and IDTRC, CMC, Vellore, India, for their help and cooperation in conducting this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
